Air Shower System Market
Air Shower System Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Air Shower System Market Forecast and Outlook By FMI
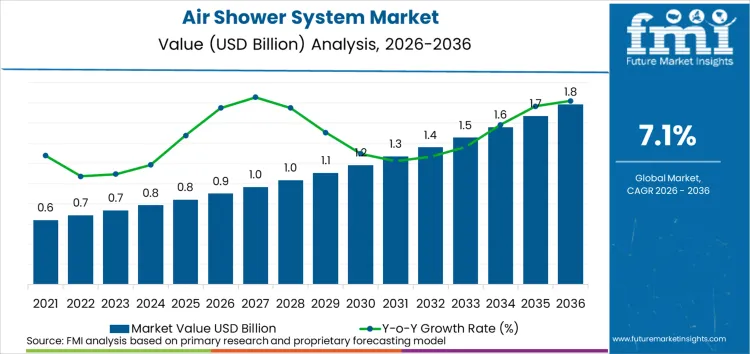
The global air shower system market is forecasted to reach USD 0.9 billion in 2026 and expand to USD 1.8 billion by 2036, advancing at a CAGR of 7.1%. As per FMI, this outlook shows a transformation in contamination control technology toward precision-engineered airflow systems, where traditional entry methods are evolving toward multi-functional decontamination platforms. Expansion is driven by the rising demand for controlled environment access, with pharmaceutical manufacturers and semiconductor facilities embracing specialized air shower solutions across cleanroom applications globally.
By leveraging advanced filtration engineering and precision airflow techniques, producers can now develop systems that offer controlled particle removal and enhanced decontamination effectiveness through improved velocity mechanisms and filtration retention systems. This technological advancement allows for the creation of energy-efficient installations with extended operational capabilities and reduced maintenance variability, effectively addressing facility demands for consistent contamination control while maintaining environmental standards throughout extended operational cycles.
Summary of Air Shower System Market
- The air shower system market comprises single-person air showers, two-person air showers, tunnel/high-throughput systems, and specialty decontamination units used for controlled personnel and material decontamination across pharmaceutical facilities, semiconductor manufacturing, food processing, and laboratory applications.
- The defined scope is structured under FMI taxonomy with configuration categorization across single-person, two-person, tunnel, and other specialized formats, excluding portable cleaning units, static air filters, laminar flow installations, and custom fabrication services.
- The air shower system market is projected to grow at a CAGR of 7.1% from 2026 to 2036, expanding from USD 0.9 billion in 2026 to USD 1.8 billion by 2036, based on FMI proprietary bottom-up equipment revenue modeling, installation cost analysis, facility requirement assessment, and contamination control performance evaluation.
- The sector is transitioning toward modular contamination control innovations, automated decontamination technology integration, and regulatory compliance advancement, supported by primary interviews with manufacturers and facility operators and scenario modeling aligned with cleanroom expansion and protocol enhancement momentum.
- Competition is concentrated among major players such as Terra Universal, AES Clean Technology, and Angstrom Technology, with strategic partnerships and acquisitions strengthening portfolios, expanding comprehensive contamination control solutions, and reinforcing market leadership across pharmaceutical manufacturing and cleanroom facility channels.
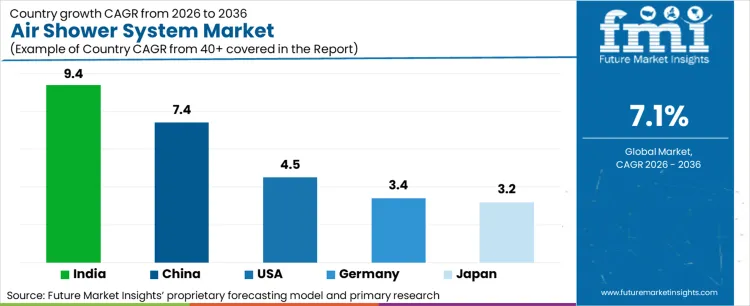
- India is projected to record the fastest growth at 9.4% CAGR through 2036, while the United States retains significant market presence supported by extensive pharmaceutical manufacturing and high demand for standardized contamination control systems.
In February 2025, Camfil, a global leader in air filtration solutions, rolled out a new video series exploring the role of molecular air filtration in protecting data centers from airborne contaminants. “This series is designed to help facility managers and data center engineers understand the risks associated with airborne contaminants and how molecular filtration solutions can mitigate them,” said Adam Wiggins, Data Center Segment Sales Manager at Camfil USA. “By providing educational content, we aim to support professionals in extending the life of critical equipment and improving overall system reliability.”
The industry's value proposition is shifting from basic air cleaning functionality toward comprehensive contamination control solutions that address specific operational requirements. Advanced manufacturers are integrating velocity optimization systems, filtration enhancement elements, and customizable airflow options to reduce contamination limitations while improving facility cleanliness. Specialized equipment providers are gaining preference among facilities, enabling direct supply partnerships and customized air shower solutions across pharmaceutical, semiconductor, and food processing channels.
Summary of Air Shower System Market
| Metric | Value |
|---|---|
| Market Value (2026) | USD 0.9 billion |
| Market Forecast Value (2036) | USD 1.8 billion |
| Forecast CAGR (2026 to 2036) | 7.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
What is Driving the Demand for Air Shower Systems?
Demand for air shower systems is being propelled by the broad industrial shift toward standardized contamination control methods and the escalating complexity of cleanroom protocols requiring controlled personnel decontamination mechanisms. The increasing preference for consistent contamination control outcomes in pharmaceutical facilities, semiconductor manufacturing, and food processing operations has amplified demand for precision-engineered air shower solutions that minimize contamination transfer and enhance operational safety.
The regulatory momentum toward enhanced cleanroom requirements and air fresheners, exemplified by updated FDA guidelines for pharmaceutical manufacturing environments and ISO cleanroom standards, is driving widespread adoption of automated air shower systems and protocol-compliant decontamination installations in commercial production workflows. Manufacturers are responding to these requirements through specialized system features that integrate compliance measures without compromising functionality. The expanding pharmaceutical market in emerging economies is shifting production toward effective contamination control solutions, underscoring a sustained demand trajectory rooted in facility safety and efficiency-driven purchasing patterns.
How is the Air Shower System Market Segmented?
The air shower system market has been segmented based on configuration, end use, cleanroom class target, and sales channel. In terms of configuration, the market is divided into single-person air showers, two-person air showers, tunnel/high-throughput systems, and other specialized units. By end use, the market includes pharmaceutical & biotechnology, semiconductor & electronics, food & beverage, and hospitals & laboratories. By cleanroom class target, the market is segmented into ISO 5-6, ISO 7, ISO 8, and other classifications. By sales channel, the market includes cleanroom integrators, direct OEM sales, HVAC/MEP contractors, and other distribution methods. Regionally, the market spans North America, Europe, East Asia, South Asia, Latin America, and Middle East and Africa.
Why Do Single-Person Air Showers Dominate the Air Shower System Market?
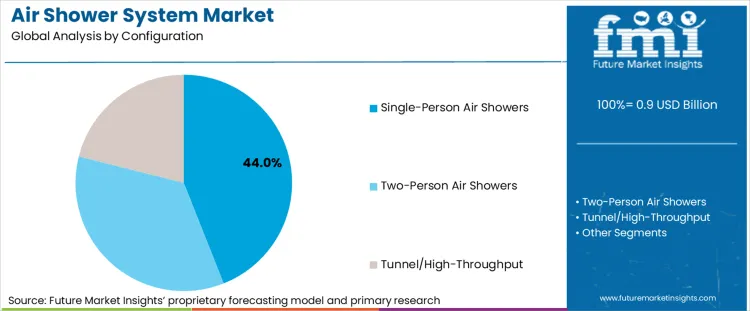
Single-person air showers continue to lead the market, accounting for 44.0% of the share, driven by their versatility in facility applications, cost-effectiveness, and adaptability to various cleanroom requirements. This configuration type offers the flexibility needed for standard personnel decontamination while maintaining effectiveness across different operational conditions, making it essential for pharmaceutical facilities, semiconductor manufacturing, and laboratory applications. Single-person systems provide facilities with operational efficiency for personnel entry protocols and scaling capabilities while supporting standard cleanroom requirements.
Two-person components capture 28.0% of the market share, supporting high-throughput applications where enhanced capacity and operational efficiency are critical factors. Tunnel/high-throughput formulations maintain 22.0% market presence for specific applications requiring material decontamination compatibility and specialized operational profiles, particularly in manufacturing and processing segments where continuous operation and high-volume processing justify specialized configuration costs.
How Do End Use Requirements Influence Air Shower System Selection?
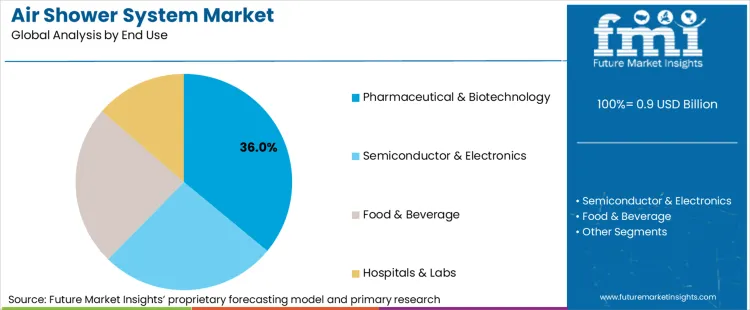
Pharmaceutical & biotechnology applications stand as the dominant segment, commanding 36.0% of the market share. Air shower systems remain centered on pharmaceutical applications due to the exceptional contamination control these installations offer in regulatory compliance and product safety. Pharmaceutical operations often require precise decontamination protocols and consistent contamination removal outcomes, making specialized air showers ideal for controlled access while maintaining facility cleanliness throughout extended operational cycles.
The market is shaped by the development of pharmaceutical-grade installations and enhanced filtration technologies that require rigorous testing to establish performance consistency and extended operational life. Semiconductor & electronics applications demand specialized systems that integrate precision contamination control features and operational reliability to prevent product contamination while ensuring consistent facility performance. Companies are expanding their capabilities to address these specialized requirements. Advanced testing protocols help developers optimize system performance and ensure consistent operation across different facility environments, making these validation services essential for regulatory approval and market acceptance.
How Is Technological Evolution Reshaping Air Shower Systems?
The air shower system sector is shifting from traditional contamination control equipment to integrated decontamination platforms that combine controlled airflow, particle filtration, and monitoring capabilities. Strategic partnerships between equipment suppliers and facility operators are accelerating this transition toward more sophisticated contamination control solutions. This evolution brings air shower systems closer to pharmaceutical-grade standards by enabling more predictable contamination control and enhanced operational consistency using advanced filtration and airflow technology innovations.
Improved system design reduces energy consumption during operational cycles and enhances facility performance through more efficient airflow processing and consistent decontamination delivery. The ability to customize operational parameters and incorporate monitoring technologies is reshaping facility design decisions, allowing operators to address specific contamination control requirements while maintaining regulatory compliance and operational efficiency.
How Is the Air Shower System Market Evolving Globally?
Global demand for air shower systems is increasing as facilities expand contamination control capabilities while addressing regulatory requirements, operational standardization specifications, and protocol compliance across pharmaceutical, semiconductor, food processing, and laboratory applications. Growth demonstrates rising adoption of automated decontamination systems, application-specific configuration positioning, and performance-optimized air shower solutions across facility operators, equipment suppliers, and regulatory environments. System selection focuses on contamination control reliability, operational efficiency, and regulatory compliance under various facility scenarios.
India records 9.4% CAGR, China records 7.4% CAGR, United States records 4.5% CAGR, Germany records 3.4% CAGR, and Japan records 3.2% CAGR. Adoption remains driven by regulatory requirements and contamination control effectiveness rather than capacity expansion alone.
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 9.4% |
| China | 7.4% |
| United States | 4.5% |
| Germany | 3.4% |
| Japan | 3.2% |
Source: FMI historical analysis and forecast data
United States: How is Regulatory Compliance Driving Market Growth?
The primary driver in the United States is the significant expansion of pharmaceutical manufacturing capabilities and regulatory compliance infrastructure. With the growth of biotechnology and semiconductor industries, American production facilities are experiencing substantial increases in contamination control demand, making traditional decontamination methods insufficient for current regulatory standards. This has promoted advanced air shower technology adoption for pharmaceutical manufacturing to ensure product safety and operational compliance.
Driven by this national scale of regulatory enhancement, the American air shower system market is set to grow at a 4.5% CAGR during the study period, led by massive demand in pharmaceutical and semiconductor sectors. Over the next five years, the most significant opportunity lies in the integration of automated decontamination systems with monitoring capabilities. As the United States targets leadership in pharmaceutical manufacturing, there is growing demand for effective solutions that support complex regulatory requirements and high-performance contamination control capabilities.
Germany: Will Engineering Excellence Drive Precision Air Shower Adoption?
Germany's air shower system market is shaped by precision manufacturing capabilities and quality-focused operational standards. German manufacturers are adopting advanced contamination control technologies that integrate reliability assurance with innovative filtration features for premium facility positioning. The German air shower system market is expected to grow at a 3.4% CAGR during the study period.
Over the next five years, opportunities exist in developing specialized systems for pharmaceutical and precision manufacturing applications. As German companies maintain leadership in precision manufacturing, there is significant demand for air showers that integrate advanced contamination control features, regulatory compliance, and premium functionality to serve both domestic and export markets effectively.
China: Is Manufacturing Infrastructure Expansion Bolstering Contamination Control Standards?
In China, the market is dominated by the evolution toward advanced contamination control standards and efficiency-positioned commercial applications. This quality-first approach is essential for Chinese manufacturers competing with international standards in domestic pharmaceutical and semiconductor applications. The Chinese air shower system market is projected to grow at a 7.4% CAGR during the study period.
Over the next five years, the rise of pharmaceutical manufacturing and expanding semiconductor production will necessitate high-performance air shower solutions for contamination control applications. China's growing role as a manufacturing hub for global pharmaceutical and electronics brands creates opportunities for cost-effective decontamination solutions that meet international contamination control standards while addressing local facility requirements and operational efficiency demands.
India: How is Pharmaceutical Growth Reinforcing the Demand for Air Shower Systems?
The growth of the air shower system market in India is primarily driven by a convergence of pharmaceutical manufacturing expansion and the rising adoption of standardized contamination control methods, regulatory-positioned facility operations. As Indian pharmaceutical operators increasingly adopt automated systems, facilities are shifting toward specialized configurations, such as single-person units, high-throughput systems, and precision decontamination installations, which rely on controlled airflow to offer consistent contamination control, efficient operation without the need for traditional cleaning procedures. This trend is accelerated by stringent Indian pharmaceutical regulations, pushing manufacturers to innovate with performance-controlled air shower designs made from validated components, standardized systems, or enhanced filtration formulations, thereby combining effectiveness with regulatory compliance.
The market is being propelled by the mainstream adoption of pharmaceutical manufacturing, a sector no longer limited to traditional production but now expanding into biotechnology manufacturing, specialized pharmaceutical production, and commercial research facilities. High-profile pharmaceutical success stories have normalized the contamination control revolution, proving that operators are willing to invest in precision decontamination systems that eliminate contamination variability entirely. This shift is further supported by a demand for facility-ready and scalable formats, where consistent, specialized air shower systems offer an efficient alternative to manual decontamination procedures, effectively merging regulatory compliance with high-performance operational convenience.
Japan: How is Precision Manufacturing Shaping Advanced Air Shower Development?
The Japan market focuses on precision manufacturing and specialized applications for pharmaceutical and semiconductor production facilities. This approach emphasizes technological advancement and operational efficiency while maintaining competitive positioning in global markets. The Japan air shower system market is set to grow at a 3.2% CAGR during the study period.
Over the next five years, opportunities lie in developing specialized systems for precision applications and advanced contamination control formulations. As regulatory requirements become more stringent for pharmaceutical and semiconductor manufacturing applications, there is growing demand for air showers that integrate automated control features, precise operational mechanisms, and advanced functionality while maintaining facility convenience and operational efficiency.
Who are the Major Players Active in the Air Shower System Market?
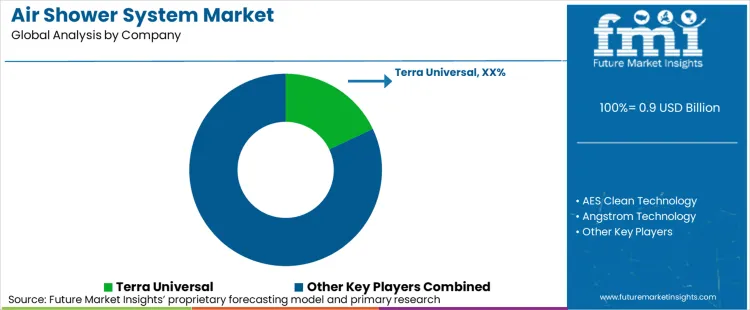
With contamination control requirements becoming more sophisticated and regulatory standards being more stringent, major players like Terra Universal, AES Clean Technology, and Angstrom Technology are competing on their ability to offer comprehensive decontamination solutions that integrate system precision with regulatory compliance. These industry leaders are investing in advanced filtration technologies and system innovations to provide seamless integration capabilities, allowing facility operators to focus on production while outsourcing specialized contamination control requirements.
The market is observing distinct regional specialization in competitive strategy. While North American firms like Terra Universal maintain leadership through system expertise and proximity to major pharmaceutical manufacturers, European players like Camfil are advancing precision filtration engineering and automated operational capabilities. This has created an innovation cycle where equipment providers are diversifying into specialized applications such as high-efficiency particulate air filtration and automated monitoring systems to address the complex requirements of pharmaceutical and regulated facility environments.
Key Industry Developments:
- In December 2025, Camfil acquired F.C.R., an Italian company specializing in air filtration solutions. The acquisition expands Camfil’s presence in Italy and strengthens its ability to serve customers with reliable, energy‑efficient solutions for cleaner air.
Key Players in the Air Shower System Market
- Terra Universal
- AES Clean Technology
- Angstrom Technology
- Camfil
- Abtech
- Berkshire Corporation
- TMI International
- Cleanroom World
- Kleanlabs
Market Definition
The air shower system market refers to the global equipment applications of specialized contamination control systems designed to support controlled personnel and material decontamination through standardized airflow processes. It includes both conventional and advanced system offerings, with segments that cater to various facility requirements such as pharmaceutical facilities, semiconductor manufacturing, food processing, and laboratory operations. The market size is measured in USD billion and analyzed over the 2026 to 2036 period.
The air shower system market includes contamination control systems categorized by configuration, end use, and sales channel, utilized through facility operations and manufacturing applications. The market is expanding due to growing demand for standardized contamination control methods, regulatory compliance requirements, and evolving facility preferences toward consistent decontamination and reduced contamination transfer capabilities.
Market Inclusion
Included in the air shower system market scope are contamination control systems categorized by configuration (single-person air showers, two-person air showers, tunnel/high-throughput, other), end use (pharmaceutical & biotechnology, semiconductor & electronics, food & beverage, hospitals & laboratories), and sales channel (cleanroom technologies, direct OEM sales, HVAC/MEP contractors, other). The scope includes air shower systems with focus on contamination control standardization, operational consistency, and regulatory compliance, particularly those marketed through equipment suppliers, facility operators, and commercial installation companies.
Geographically, the market encompasses key regions such as North America, Europe, East Asia, South Asia, Latin America, and the Middle East and Africa, with country-level analysis for significant markets like United States, Germany, China, India, Japan, United Kingdom, and Canada.
Market Exclusion
Excluded from the air shower system market scope are non-contamination control equipment solutions and products not designed for standardized decontamination applications, such as basic ventilation systems, portable air filtration units, and static cleaning installations. Additionally, systems not intended for commercial contamination control applications are not included.
The market excludes non-decontamination equipment products, as well as contamination control systems that are not marketed as standardized air shower solutions, such as individual filtration components or custom fabrication services. Products with non-compliant operational specifications or those that do not meet regional safety standards for commercial applications are outside the market's defined scope.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD 0.9 billion |
| Configuration | Single-Person Air Showers; Two-Person Air Showers; Tunnel/High-Throughput; Other |
| End Use | Pharmaceutical & Biotechnology; Semiconductor & Electronics; Food & Beverage; Hospitals & Labs |
| Sales Channel | Cleanroom Integrators; Direct OEM Sales; HVAC/MEP Contractors; Other |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | United States, Germany, China, India, Japan, United Kingdom, Canada, and 40+ countries |
| Key Companies Profiled | Terra Universal; AES Clean Technology; Angstrom Technology; Camfil; Abtech; Others |
| Additional Attributes | Revenue analysis by configuration and end use; performance in facility applications and contamination control capabilities across pharmaceutical manufacturers, semiconductor facilities, and commercial installation providers; system efficiency enhancement, contamination control optimization, and operational reliability under various facility scenarios; impact on facility performance, operational efficiency, and contamination reduction during installation processes; compatibility with regulatory requirements and facility preferences; procurement dynamics driven by contamination control positioning, regulatory compliance programs, and long-term supply partnerships. |
Air Shower System Market by Segment
Configuration:
- Single-Person Air Showers
- Two-Person Air Showers
- Tunnel/High-Throughput
- Other
End Use:
- Pharmaceutical & Biotechnology
- Semiconductor & Electronics
- Food & Beverage
- Hospitals & Labs
Sales Channel:
- Cleanroom Integrators
- Direct OEM Sales
- HVAC/MEP Contractors
- Other
Region:
- East Asia
- China
- Japan
- South Korea
- Taiwan
- Rest of East Asia
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- BENELUX
- Nordic
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- South Asia
- India
- Australia & New Zealand
- Southeast Asia
- Rest of South Asia
- Latin America
- Brazil
- Chile
- Colombia
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- Contamination Control Association. (2023). Global air shower system trends and facility applications: Recommendations from performance assessments. Commercial Facility Initiative.
- Martinez, A. (2024). Fostering contamination control equipment industry: Global trends and efficient decontamination systems. International Journal of Facility Management.
- Thompson, R., & co-authors. (2023). Advanced air shower technologies and commercial facility operations: A review on recent developments. Contamination Control Journal, 18(2), 1456.
Frequently Asked Questions
How big is the air shower system market in 2026?
The global air shower system market is estimated to be valued at USD 0.9 billion in 2026.
What will be the size of the air shower system market in 2036?
The market size for the air shower system market is projected to reach USD 1.8 billion by 2036.
How much will the air shower system market grow between 2026 and 2036?
The air shower system market is expected to grow at a 7.1% CAGR between 2026 and 2036.
What are the key configurations in the air shower system market?
The key configurations in the air shower system market include single-person air showers, two-person air showers, tunnel/high-throughput systems, and other specialized units.
Which end use will contribute a significant share in the air shower system market in 2026?
In terms of end use, the pharmaceutical & biotechnology segment is set to command a 36.0% share in the air shower system market in 2026.
Table of Content
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Desk Research Programme (Secondary Evidence)
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Configuration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Configuration , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Configuration , 2026 to 2036
- Single-Person Air Showers
- Two-Person Air Showers
- Tunnel/High-Throughput
- Single-Person Air Showers
- Y to o to Y Growth Trend Analysis By Configuration , 2021 to 2025
- Absolute $ Opportunity Analysis By Configuration , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceutical & Biotechnology
- Semiconductor & Electronics
- Food & Beverage
- Hospitals & Labs
- Pharmaceutical & Biotechnology
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Configuration
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Configuration
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Configuration
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Configuration
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Configuration
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Configuration
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Configuration
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Configuration
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Configuration
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Configuration
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Configuration
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Configuration
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Configuration
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Configuration
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Configuration
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Configuration
- By End Use
- Competition Analysis
- Competition Deep Dive
- Terra Universal
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- AES Clean Technology
- Angstrom Technology
- Camfil
- Abtech
- Berkshire Corporation
- TMI International
- Cleanroom World
- Kleanlabs
- Terra Universal
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Configuration , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Configuration , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Configuration , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Configuration , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Configuration , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Configuration , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Configuration , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Configuration , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Configuration , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Configuration , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Configuration
- Figure 6: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Configuration , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Configuration , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Configuration
- Figure 23: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End Use
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Configuration , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Configuration , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Configuration
- Figure 30: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End Use
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Configuration , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Configuration , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Configuration
- Figure 37: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End Use
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Configuration , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Configuration , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Configuration
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Configuration , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Configuration , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Configuration
- Figure 51: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End Use
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Configuration , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Configuration , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Configuration
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Configuration , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Configuration , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Configuration
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

