Demand for Glucose Monitoring Devices in EU
This report examines the demand for glucose monitoring devices in EU in detail, covering market size, revenue forecast, competitive dynamics, demand outlook, growth drivers, key restraints, industry developments, supply chain trends, and strategic growth opportunities.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Demand for Glucose Monitoring Devices in EU Size, Market Forecast and Outlook By FMI
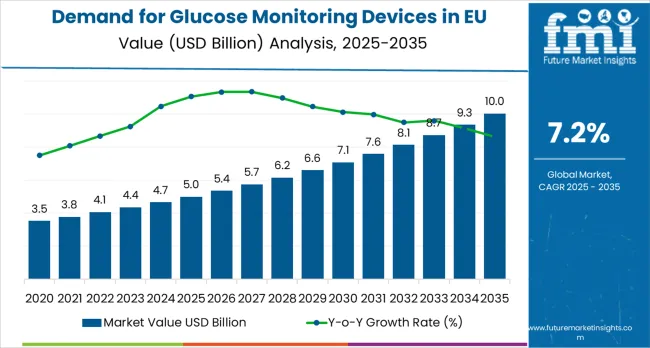
The demand for glucose monitoring devices in eu was valued at USD 5.36 billion in 2025, advancing to USD 5.36 billion in 2026 and projected to reach USD 10.74 billion by 2036 at a CAGR of 7.2%. Self-Monitoring Devices holds 57.9% of the product type segment in 2026, reflecting entrenched procurement patterns and specification alignment across primary buyer categories. Based on FMI analysis, absolute market expansion of USD 5.38 billion over the 2026 to 2036 forecast period will be concentrated in technology-driven sub-segments and geographies with favorable regulatory or policy environments.
Summary of Demand for Glucose Monitoring Devices in EU
- Demand for Glucose Monitoring Devices in EU Definition
- The demand for glucose monitoring devices in eu encompasses blood glucose measurement systems including self-monitoring blood glucose meters, test strips, lancets, and continuous glucose monitoring sensors, transmitters, and receiver systems used by diabetic patients and clinical professionals across European Union member states for glycemic management.
- Demand Drivers in the Market
- Rising Type II Diabetes Prevalence Across EU Member States: European Centre for Disease Prevention and Control data document a consistent increase in diagnosed diabetes cases across all five major EU economies, expanding the addressable patient population for both self-monitoring and continuous glucose monitoring devices.
- National Reimbursement Policy Expansion for Continuous Glucose Monitoring: Germany, France, and the Netherlands have formally extended statutory health insurance reimbursement coverage for CGM devices to broader categories of Type II diabetes patients, removing the primary financial barrier to adoption in the largest EU markets.
- EU Medical Device Regulation Compliance Driving Consolidation: Full MDR enforcement timelines are creating certification cost barriers that are consolidating the supplier landscape, reducing competitive pricing pressure on certified device manufacturers while eliminating non-compliant regional alternatives.
- Key Segments Analyzed in the FMI Report
- Self-Monitoring Devices: 57.9% share in 2026, reflecting established procurement specifications that favour this configuration across primary buyer categories for its cost efficiency and performance consistency.
- Diabetes Type II: the leading application sub-segment in 2026, anchored by institutional buyers operating under defined procurement cycles and volume commitments that create predictable replacement demand.
- Spain: 7.6% CAGR through 2036, representing the fastest-growing geography in the covered market, supported by policy-driven demand creation and expanding end-user industry capacity.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare observe that, FMI analysts note that the demand for glucose monitoring devices in eu is bifurcating between volume-driven segments where regional manufacturers compete on price and specification-driven tiers where certified performance requirements create defensible margins for qualified suppliers. Buyers consolidating vendor relationships will concentrate purchasing with producers who can demonstrate both compliance credentials and delivery reliability, placing mid-tier suppliers without clear differentiation at structural risk. The Spain and Netherlands growth trajectories are grounded in verifiable policy commitments and end-user industry expansion rather than speculative demand, making both geographies priority targets for capacity investment decisions through the medium term.
- Strategic Implications / Executive Takeaways
- Align production capacity and distribution investment with the Spain and Netherlands growth trajectories, where CAGR outperformance relative to the global average reflects durable structural demand rather than cyclical acceleration.
- Develop product lines that meet tightening specification requirements in Germany and adjacent mature markets, where replacement-cycle buyers prioritize certified performance and supply continuity over unit price.
- Establish dual-source input arrangements for critical raw materials to reduce exposure to supply concentration risks in geographies that are simultaneously experiencing strong domestic demand growth.
Demand for Glucose Monitoring Devices in EU Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 5.36 billion |
| Industry Value (2036) | USD 10.74 billion |
| CAGR (2026-2036) | 7.2% |
| Source | Future Market Insights, 2026 |
FMI analysts observe that structural demand for demand for glucose monitoring devices in eu is shaped by overlapping forces that operate independently of short-term economic cycles. Rising Type II Diabetes Prevalence Across EU Member States establishes the volume baseline for the forecast period, while national reimbursement policy expansion for continuous glucose monitoring is creating incremental demand from previously underserved buyer categories. FMI is of the opinion that producers who position capacity against the convergence of these forces will generate above-market revenue growth while those relying on legacy procurement relationships face margin erosion as specification requirements evolve.
Regional performance diverges across the demand for glucose monitoring devices in eu forecast horizon. Spain at 7.6% CAGR and Netherlands at 7.4% CAGR lead the global expansion trajectory, driven by policy-backed demand creation and growing institutional procurement volumes. Italy at 7.2% and France at 7.1% represent mid-growth markets where existing regulatory frameworks and procurement infrastructure support steady adoption. Germany at 6.7% reflect mature-market replacement and compliance-driven purchasing rather than volume growth.
Demand for Glucose Monitoring Devices in EU Definition
The demand for glucose monitoring devices in eu covers blood glucose measurement systems including self-monitoring blood glucose meters, test strips, lancets, and continuous glucose monitoring sensors, transmitters, and receiver systems used by diabetic patients and clinical professionals across European Union member states for glycemic management. Products within scope are distinguished by material grade, dimensional specifications, and performance certification requirements that govern procurement decisions across industrial, institutional, and commercial buyer segments. As per FMI, market boundaries are defined by functional end-use characteristics rather than by manufacturing process alone.
Demand for Glucose Monitoring Devices in EU Inclusions
Market scope includes self-monitoring blood glucose meters, associated test strips and lancets, real-time and intermittently scanned continuous glucose monitoring systems, sensor applicators, transmitters, and compatible data management software sold through EU healthcare channels. All products within scope are evaluated across the full segmentation taxonomy and geographic coverage framework of the FMI analysis.
Demand for Glucose Monitoring Devices in EU Exclusions
The scope excludes insulin delivery devices including insulin pumps and pens, closed-loop artificial pancreas systems evaluated as combination products, hemoglobin A1c point-of-care testing platforms, and non-EU market demand for glucose monitoring devices. Products that do not satisfy the functional or material criteria defined by the market taxonomy are omitted from all revenue and volume calculations.
Demand for Glucose Monitoring Devices in EU Research Methodology
- Primary Research: Structured interviews were conducted with procurement directors, product engineers, and commercial managers across manufacturer, distributor, and end-user organizations in all covered geographies to map decision criteria, specification requirements, and pricing dynamics.
- Desk Research: Secondary data collection aggregated national trade statistics, regulatory filings, company annual reports, investor presentations, standards body publications, and peer-reviewed technical research relevant to the covered market.
- Market-Sizing and Forecasting: A bottom-up revenue model was constructed from verified unit shipment benchmarks and average selling price data, validated against top-down checks derived from total addressable market estimates anchored to observable industry benchmarks from allowed sources.
- Data Validation and Update Cycle: All forecast outputs are cross-referenced against publicly reported company revenue guidance and validated through a secondary round of primary outreach. Data is refreshed on a rolling basis as new regulatory filings and corporate disclosures become available.
Why is the Demand for Glucose Monitoring Devices in EU Growing in EU?
Market expansion is being supported by the fundamental shift in healthcare attitudes toward patient-centred diabetes management and preventive care across European societies, with glucose monitoring devices increasingly regarded as essential medical technologies deserving premium clinical performance, advanced connectivity features, and patient outcomes comparable to hospital-grade monitoring systems while providing unique home-based management advantages.
Modern European patients and healthcare professionals consistently prioritize device accuracy, ease of use, connectivity capabilities, and clinical integration when selecting glucose monitoring systems, driving demand for advanced monitoring device formulations that deliver superior clinical performance, seamless digital integration, exceptional user experience, and healthcare system connectivity compared to traditional blood glucose meters.
Even minor concerns about diabetes management, glucose control, or clinical monitoring requirements can drive comprehensive adoption of specialized premium glucose monitoring devices designed to maintain optimal glycemic control, support specific clinical applications, and enhance patient satisfaction throughout all diabetes management categories.
The growing complexity of diabetes care and increasing awareness of continuous monitoring benefits are driving demand for technologically advanced glucose monitoring devices from certified European manufacturers with appropriate medical device certifications, clinical validation capabilities, technical expertise, and compliance with stringent EU medical device regulations.
Regulatory authorities across European Union member states are increasingly establishing comprehensive guidelines for glucose monitoring device manufacturing, clinical accuracy standards, data security requirements, and medical device safety protocols to ensure device effectiveness and patient safety.
Clinical research studies and real-world evidence conducted at European diabetes research institutions are providing evidence supporting advanced glucose monitoring interventions for common diabetes management challenges including glycemic variability reduction, hypoglycemia prevention, treatment optimization, and clinical outcome improvement, requiring specialized technology expertise and clinically-validated manufacturing processes that meet EU medical device standards.
Segmental Analysis
The market is segmented by product type, indication, distribution channel, and sales region. By product type, the market is divided into self-monitoring devices (including blood glucose meters, test strips, and lancets) and continuous monitoring devices (including sensors, transmitters, and receiver systems).
Based on indication, the market is categorized into diabetes type II (including adult-onset diabetes and metabolic syndrome subsegments), diabetes types I (including juvenile diabetes and autoimmune diabetes subsegments), and gestational diabetes applications.
By distribution channel, the market spans hospitals & clinics (healthcare facilities, diabetes centers), pharmacies (community pharmacies, hospital pharmacies), and online retail (e-commerce platforms, direct-to-consumer websites). Regionally, the market covers Germany, France, Italy, Spain, Netherlands, and Rest of Europe.
By Product Type, Self-Monitoring Devices Segment Accounts for 57.9% Market Share
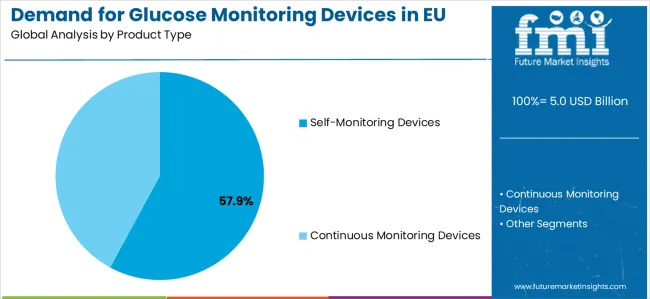
Self-monitoring devices segment is likely to account for 57.9% of the Demand for Glucose Monitoring Devices in EU in 2026, establishing itself as the dominant product category across European healthcare and patient care facilities. This commanding market position is fundamentally supported by the widespread adoption of blood glucose meters for routine diabetes management, cost-effective monitoring solutions, and established clinical protocols that deliver reliable accuracy, proven clinical performance, and superior accessibility throughout large-scale healthcare operations across EU member states.
Self-monitoring device products provide European patients with unparalleled monitoring flexibility, precise glucose measurement during daily management routines, proven clinical reliability, and seamless integration with diabetes management protocols that comply with stringent EU medical device safety directives.
This product sophistication enables European medical device manufacturers to achieve optimal production economics while maintaining rigorous quality protocols mandated by European Medicines Agency (EMA) regulations, comprehensive clinical validation systems required under EU law, and consistent clinical performance across millions of individual diabetes management applications produced annually.
The segment derives substantial competitive advantages from established European medical device manufacturing infrastructure offering specialized production solutions, comprehensive clinical support services, and continuous product innovation that incorporates enhanced accuracy technologies, improved user interface design, and advanced connectivity capabilities aligned with digital health standards. The self-monitoring systems deliver superior cost-effectiveness particularly important in value-based healthcare markets, enhanced patient accessibility, improved reimbursement coverage, and superior regulatory compliance with EU medical device manufacturing standards.
- Manufacturing efficiency advantages include established medical device infrastructure, optimized production systems, and comprehensive clinical testing protocols
- Clinical reliability benefits through standardized manufacturing methods, consistent accuracy performance, and extensive validation characteristics
- Economic advantages from cost-effective positioning opportunities, broad patient accessibility, and improved healthcare system integration across diverse diabetes management applications
By Indication, Diabetes Type II Segment Accounts for 72.4% Market Share
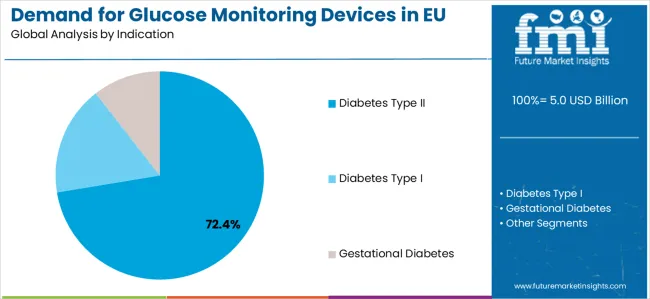
Diabetes Type II applications are positioned to represent 72.4% of total European glucose monitoring device demand in 2026, reflecting the segment's dominant position within the market ecosystem and the substantially larger patient population compared to other indication categories across most European countries. This substantial market share directly demonstrates the exceptionally high prevalence of Type II diabetes among European patients and the continuously expanding population of diabetes management programs across both Western and Eastern European healthcare systems.
Modern European Diabetes Type II glucose monitoring applications are increasingly featuring sophisticated technologies that deliver enhanced monitoring accuracy, extended monitoring duration, and specialized clinical features specifically designed to address common Type II diabetes management challenges including postprandial glucose monitoring, medication adherence support, lifestyle modification guidance, and long-term glycemic control optimization.
European healthcare providers consistently demonstrate commitment to investing substantially in advanced glucose monitoring devices that deliver visible clinical outcomes, support optimal patient management, comply with EU healthcare quality standards, and provide consistent results for both healthcare facility operations and home-based patient care. Within the diabetes Type II segment, adult-onset diabetes applications command 50% share, metabolic syndrome represents 15%, and lifestyle-related diabetes accounts for 7.4%, reflecting comprehensive clinical segmentation strategies tailored to European diabetes care preferences.
- Clinical innovation opportunities through personalized monitoring technologies, lifestyle integration features, and condition-specific management solutions
- Healthcare positioning strategies leveraging European diabetes care protocols and monitoring device messaging emphasizing clinical effectiveness and patient outcomes
- Healthcare system advantages through established diabetes care relationships across EU markets and expanding chronic disease management presence
What are the Drivers, Restraints, and Key Trends?
The Demand for Glucose Monitoring Devices in EU is advancing steadily due to intensifying diabetes prevalence trends and growing recognition of continuous monitoring benefits across EU member states. The market faces challenges, including high device costs and reimbursement limitations, complex regulatory requirements for medical device approvals across diverse European jurisdictions, varying patient adoption rates across different monitoring technologies, competition from emerging digital health solutions including smartphone-based monitoring and wearable devices, and varying healthcare system integration capabilities across different EU countries despite digital health advancement efforts. Innovation initiatives, reimbursement expansion programs, patient education solutions, and digital health platforms continue to influence product development strategies and market evolution patterns across European healthcare markets.
Rapid Growth of Digital Health Integration and Connected Care
The rapidly accelerating adoption of digital health technologies and connected care solutions is fundamentally enabling broader healthcare integration across European countries, enhanced clinical outcomes among patients particularly regarding real-time monitoring and data analytics, and significantly improved healthcare provider engagement through advanced monitoring platforms.
Advanced digital health platforms operated by specialized healthcare technology companies, professional diabetes care providers, and major medical device manufacturers equipped with comprehensive data analytics in multiple European languages, clinical decision support tools, patient education resources, and personalized care recommendations provide extensive healthcare solutions while dramatically expanding clinical visibility across diverse patient segments and geographic healthcare markets throughout the European Union.
These digital health trends prove particularly valuable for glucose monitoring devices that require comprehensive clinical integration, real-time data transmission, and targeted healthcare approaches to effectively demonstrate clinical benefits and justify technology investments across sophisticated European healthcare systems.
Digital health integration also enables sophisticated patient data analytics, personalized diabetes management recommendations based on individual glucose patterns and lifestyle factors, direct healthcare provider feedback collection that informs clinical protocols, and connected care models that ensure continuous clinical oversight while reducing healthcare delivery costs across fragmented European healthcare systems.
Integration of Artificial Intelligence and Predictive Analytics Technologies
Progressive European glucose monitoring device manufacturers are systematically incorporating innovative artificial intelligence technologies including predictive glucose analytics, hypoglycemia prediction, treatment optimization algorithms, and personalized care recommendations that address growing European healthcare demands for proactive diabetes management, clinical decision support, and improved patient outcomes in diabetes care technologies.
Strategic integration of these AI technologies, combined with rigorous clinical validation and real-world evidence protocols conducted at European diabetes research institutions, enables manufacturers to develop differentiated clinical propositions that appeal to evidence-based European healthcare providers while maintaining essential clinical accuracy and patient safety characteristics.
These technological initiatives also support development of specialized applications for specific diabetes management scenarios increasingly recognized across European healthcare systems, enhanced predictive capabilities for clinical complications recommended by European endocrinologists, and clinically-proven technologies that resonate with outcome-focused healthcare segments particularly prevalent in Nordic and Western European countries.
Technology investments in AI development facilities established in Netherlands and Germany, precision clinical analytics capabilities, and advanced glucose monitoring technologies enable European manufacturers to explore next-generation diabetes care while maintaining competitive positioning in premium and clinical-grade market segments.
Emphasis on Patient-Centered Care and User Experience Enhancement
European patients and healthcare providers are increasingly prioritizing patient-centered design and enhanced user experience when selecting glucose monitoring devices, driving fundamental changes in product development strategies across the European medical device industry. Companies are implementing comprehensive user experience design systems focusing on intuitive device interfaces, reducing complexity through simplified operation procedures, and developing patient-friendly technologies that align with EU accessibility directives and patient safety regulations.
This trend is particularly pronounced across Nordic countries, Germany, Netherlands, and France where patients demonstrate exceptional technology adoption capabilities and healthcare systems emphasize patient empowerment and self-management approaches.
Manufacturers are responding by developing partnerships with patient advocacy organizations, implementing user-centered design programs, obtaining patient experience certifications and usability standards, and providing comprehensive patient support information that differentiates their products in increasingly competitive European markets emphasizing patient satisfaction and clinical outcomes optimization.
Demand Analysis of Glucose Monitoring Devices in EU by Key Countries
France follows with a 22.0% market share, attributed to growing demand for digital health solutions and comprehensive diabetes care programs. Italy contributes 18.0% of the market, driven by increasing diabetes prevalence and healthcare modernization initiatives. Spain accounts for 12.0% of the market, while Netherlands represents 8.0%. The Rest of Europe region holds the remaining market share, encompassing Nordic countries, Eastern Europe, and other EU member states with emerging demand for glucose monitoring technologies.
Demand Analysis of Glucose Monitoring Devices in EU by Key Countries
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| Spain | 7.6% |
| Netherlands | 7.4% |
| Italy | 7.2% |
| France | 7.1% |
| Germany | 6.7% |
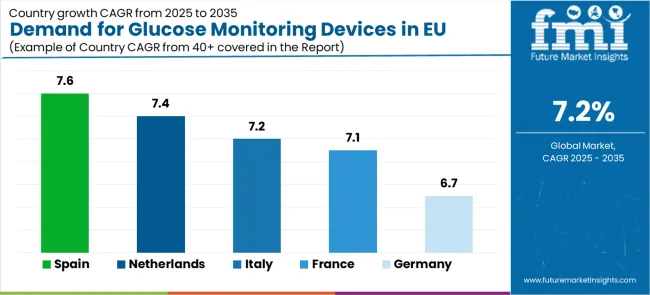
The Demand for Glucose Monitoring Devices in EU demonstrates strong growth across major economies, with Rest of Europe leading at a 7.8% CAGR through 2036, driven by expanding healthcare infrastructure and growing diabetes awareness. Spain follows at 7.6%, supported by healthcare modernization and increasing chronic disease management focus. Netherlands grows at 7.4%, integrating glucose monitoring into advanced digital health practices. Italy and France record 7.2% and 7.1% respectively, emphasizing healthcare system improvements and patient care optimization. Germany maintains steady growth at 6.7%, leveraging its established medical device infrastructure.
Germany Leads European Market with Established Medical Technology Infrastructure
The glucose monitoring devices industry in Germany is projected to exhibit steady growth with a CAGR of 6.7% through 2036, driven by exceptionally strong healthcare system infrastructure and medical technology adoption, comprehensively well-established distribution networks for medical devices and diabetes care products, and sophisticated regulatory frameworks supporting medical device quality standards throughout the country.
Germany's advanced medical device regulations and internationally recognized clinical standards through organizations including German Diabetes Association are creating substantial demand for certified advanced glucose monitoring devices across diverse healthcare and patient segments.
Major healthcare systems including German hospitals, specialized diabetes centers including university medical centers, and professional medical device distribution networks are systematically establishing extensive technology portfolios serving both healthcare professionals and diabetes patients throughout German healthcare facilities, medical centers, and home care environments.
The German market benefits from exceptionally high healthcare professional adoption of advanced medical technologies, substantial diabetes care infrastructure presence, strong health insurance coverage programs delivering accessibility at reasonable patient costs, and healthcare system emphasis on clinical excellence that naturally support premium glucose monitoring device adoption.
- Healthcare environment emphasizing comprehensive clinical standards and evidence-based medical device adoption requirements
- Strong medical technology distribution networks capturing substantial market share through clinical validation and healthcare professional support
- Healthcare system sophistication driving demand for advanced monitoring technologies and detailed clinical integration capabilities
France Demonstrates Strong Growth with Digital Health Integration
The glucose monitoring devices industry in France is expanding at a steady CAGR of 7.1%, substantially supported by increasing French healthcare system focus on digital health solutions and comprehensive diabetes care programs, growing adoption of connected medical technologies, and sophisticated healthcare infrastructure reflecting French medical excellence traditions.
France's well-established healthcare system and premium positioning strategies across medical sectors are systematically driving demand for high-quality glucose monitoring devices across diverse healthcare and patient care segments.
Specialized medical device suppliers including French healthcare technology companies, traditional medical distributors, major hospital systems (Assistance Publique-Hôpitaux de Paris, regional health networks), and professional diabetes care centers are establishing comprehensive product ranges featuring advanced imported and French-distributed glucose monitoring devices emphasizing clinical validation and digital connectivity. The French market particularly benefits from strong healthcare system emphasis on clinical quality traditions, digital health integration techniques, and evidence-based care methods that align perfectly with glucose monitoring devices positioning as essential clinical components for superior diabetes management results.
- Digital health development programs creating opportunities for professional glucose monitoring device knowledge advancement and healthcare integration
- Professional healthcare specialist training and clinical expert recommendations enhancing glucose monitoring device expertise and market penetration
- Healthcare system emphasis on clinical excellence supporting higher-value glucose monitoring device positioning strategies
Italy Maintains Strong Growth with Healthcare System Modernization
The glucose monitoring devices industry in Italy is growing at a consistent CAGR of 7.2%, fundamentally driven by increasing healthcare system modernization and diabetes care improvement initiatives, growing recognition of advanced monitoring technology benefits, and strong Italian healthcare system commitment to patient care enhancement and clinical outcome optimization.
Italy's established healthcare infrastructure is systematically incorporating advanced glucose monitoring devices to enhance patient care protocols, improve clinical outcomes, and modernize diabetes management approaches while maintaining comprehensive healthcare coverage characteristics.
Professional healthcare systems, specialized medical device distributors including Italian suppliers, leading healthcare networks (regional health authorities), and traditional medical device suppliers are strategically investing in technology adoption programs and clinical training addressing growing Italian healthcare interest in advanced glucose monitoring solutions.
The Italian market particularly benefits from strong healthcare system appreciation for clinical effectiveness, traditional patient-centered care approaches reflecting established Italian healthcare values, and growing healthcare technology capacity particularly in Northern Italian regions supporting advanced medical device adoption.
- Healthcare system innovation programs facilitating glucose monitoring device integration in traditional Italian diabetes care protocols
- Professional medical training programs enhancing technical knowledge among healthcare providers and diabetes care specialists
- Regional healthcare development incorporating advanced medical technologies and evidence-based Italian healthcare methods
Spain Focuses on Innovation and Healthcare System Enhancement
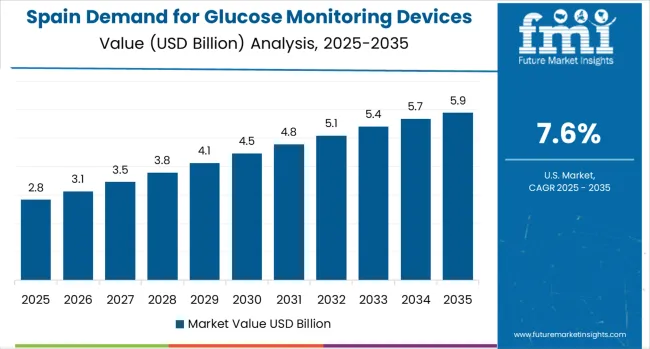
Demand for Glucose Monitoring Devices in EU in Spain is projected to grow at a strong CAGR of 7.6%, substantially supported by rapidly expanding healthcare modernization initiatives that actively promote advanced diabetes care technologies, increasing Spanish healthcare system focus on chronic disease management, and growing healthcare infrastructure particularly in major metropolitan areas including Madrid, Barcelona, Valencia, and Seville.
Spanish healthcare system is experiencing significant technological advancement with expansion of diabetes care centers, advanced medical device adoption, and major healthcare networks systematically increasing glucose monitoring device investments and introducing premium clinical capabilities.
The Spanish market is increasingly characterized by healthcare modernization trends reflecting broader European patterns, growing interest in glucose monitoring device applications addressing specific clinical requirements, and increasing healthcare system acceptance of advanced technology investments for devices delivering visible clinical benefits. Spain's substantial healthcare development focus, strong healthcare system commitment to clinical innovation reflecting Mediterranean healthcare values, and expanding diabetes care infrastructure create favorable conditions for glucose monitoring device market expansion.
- Healthcare system investments prioritizing advanced diabetes care technologies for clinical outcome improvement and patient care innovation
- Professional medical certification programs ensuring clinical expertise among healthcare providers and diabetes care specialists
- Healthcare development initiatives leveraging Spain's position in Mediterranean and Latin American healthcare relationships
Netherlands Emphasizes Innovation and Digital Health Leadership
Demand for Glucose Monitoring Devices in EU in the Netherlands is expanding at a robust CAGR of 7.4%, fundamentally driven by exceptionally strong Dutch commitment to digital health innovation, premium medical technology solutions, and healthcare consciousness that positions Netherlands among European digital health leaders.
Dutch healthcare providers and diabetes care specialists are increasingly selecting glucose monitoring devices based on comprehensive digital integration credentials, verified clinical outcomes through European standards, and complete documentation demonstrating patient benefits and clinical effectiveness throughout healthcare delivery processes.
The Netherlands market significantly benefits from exceptionally well-developed digital health research infrastructure including major healthcare innovation companies, specialized medical device suppliers, and professional healthcare technology distributors, combined with demonstrated healthcare system willingness to invest substantial resources in glucose monitoring devices with verified clinical certifications and digital health integration standards.
Dutch regulatory environment actively supports medical device innovation, digital health development, transparent clinical outcome requirements, and evidence-based validation initiatives that enhance healthcare provider confidence and market development.
- Digital health leadership driving healthcare demand for technologically validated premium glucose monitoring device products
- Healthcare innovation infrastructure supporting comprehensive clinical product development and professional healthcare access
- Clinical excellence emphasis encouraging development of evidence-based validation and healthcare advancement practices
Competitive Landscape
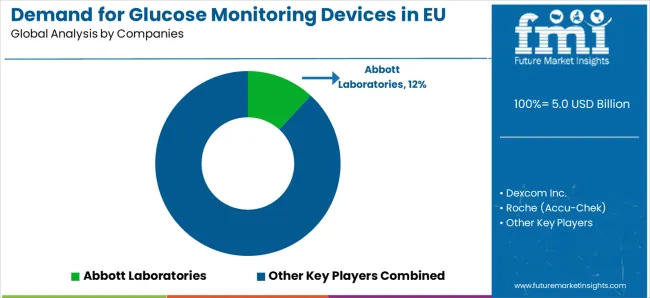
The Demand for Glucose Monitoring Devices in EU is defined by intense competition among multinational medical device corporations, regional European manufacturers, specialized diabetes care technology producers, and digital health companies from major healthcare technology suppliers.
Companies are investing in European manufacturing capacity expansion, advanced sensor technologies, digital health platform development through European innovation centers, clinical validation solutions aligned with EU medical device directives, and direct-to-patient distribution platforms serving diverse European diabetes patient populations. Strategic acquisitions, product portfolio expansion, geographic market penetration across Eastern European growth markets, healthcare professional partnership programs, and clinical certifications are central to strengthening market position and capturing share in this dynamic European category.
Major market participants include Abbott Laboratories with significant European market presence through FreeStyle Libre leadership and comprehensive continuous glucose monitoring solutions distributed through European healthcare channels. Dexcom Inc. maintains substantial European market leadership through CGM integration and AI-focused applications across advanced diabetes care sectors including professional and patient markets.
Roche (Accu-Chek) emphasizes SMBG and digital solution technologies with technical support services for professional healthcare providers across European markets. Medtronic represents significant European diabetes technology integration with specialized CGM plus insulin pump systems and comprehensive diabetes management technologies.
Regional European producers and specialized digital health brands are establishing significant market presence through premium technology positioning, CE marking certifications, specialty glucose monitoring device formulations including enhanced connectivity alternatives and patient-focused options, and direct-to-patient business models.
Digital health programs from major European healthcare initiatives including established diabetes care programs provide technologically advanced alternatives supporting market development, capturing meaningful market share particularly in innovation-driven healthcare markets. European co-manufacturing specialists including specialized medical device processors operate comprehensive production facilities serving both branded manufacturers and healthcare customers, representing critical infrastructure enabling market expansion and product innovation across European healthcare technology markets.
Key Players in the Demand for Glucose Monitoring Devices in EU
- Abbott Laboratories
- Dexcom Inc.
- Roche (Accu-Chek)
- Medtronic
- LifeScan (OneTouch)
- Ascensia Diabetes Care
- Senseonics
- AgaMatrix
- Ypsomed
- European Regional Manufacturers
Scope of the Report
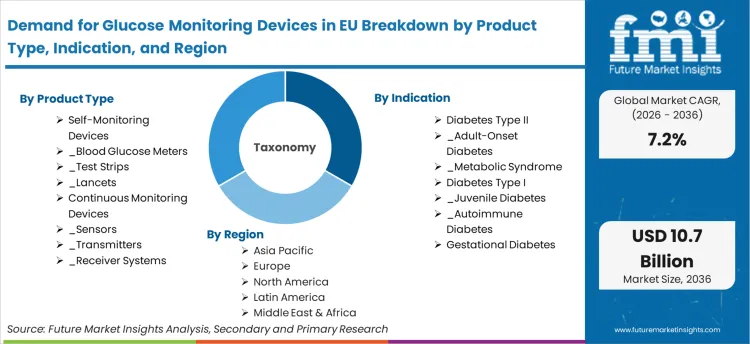
| Metric | Value |
|---|---|
| Quantitative Units | USD 5.36 billion to USD 10.74 billion, at a CAGR of 7.2% |
| Market Definition | Demand for Glucose Monitoring Devices in EU encompasses blood glucose measurement systems including self-monitoring blood glucose meters, test strips, lancets, and continuous glucose monitoring sensors, transmitters, and receiver systems used by diabetic patients and clinical professionals across European Union member states for glycemic management. |
| Segmentation | Product Type (Self-Monitoring Devices, Continuous Monitoring Devices); Indication (Diabetes Type II, Diabetes Type I, Gestational Diabetes); Distribution Channel (Hospitals and Clinics, Pharmacies, Online Retail) |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | Spain, Netherlands, Italy, France, Germany, and 40 plus countries |
| Key Companies Profiled | Abbott Laboratories, Dexcom Inc., Roche (Accu-Chek), Medtronic, LifeScan (OneTouch), Ascensia Diabetes Care, Senseonics, AgaMatrix, Ypsomed, European Regional Manufacturers |
| Forecast Period | 2026 to 2036 |
| Approach | Bottom-up revenue modeling anchored to verified shipment and pricing benchmarks, with scenario adjustments applied for regulatory, policy, and macroeconomic inputs |
Key Segments
Product Type:
- Self-Monitoring Devices
- Blood Glucose Meters
- Test Strips
- Lancets
- Continuous Monitoring Devices
- Sensors
- Transmitters
- Receiver Systems
Indication:
- Diabetes Type II
- Adult-Onset Diabetes
- Metabolic Syndrome
- Diabetes Type I
- Juvenile Diabetes
- Autoimmune Diabetes
- Gestational Diabetes
Distribution Channel:
- Hospitals & Clinics
- Healthcare Facilities
- Diabetes Centers
- Pharmacies
- Community Pharmacies
- Hospital Pharmacies
- Online Retail
- E-commerce Platforms
- Direct-to-Consumer Websites
Countries:
- Germany
- France
- Italy
- Spain
- Netherlands
- Rest of Europe
Bibliography
- European Centre for Disease Prevention and Control. (2024, March). Diabetes surveillance in the EU and EEA: Epidemiological data and monitoring framework. ECDC.
- European Medicines Agency. (2024, February). EU medical device regulation implementation: Guidance for glucose monitoring device manufacturers. EMA.
- German Federal Joint Committee. (2024, April). G-BA resolution: Continuous glucose monitoring reimbursement extension for type 2 diabetes patients. G-BA.
- French National Health Authority. (2024, June). HAS recommendation: Flash glucose monitoring reimbursement criteria update. HAS.
- International Diabetes Federation. (2024, November). IDF diabetes atlas 11th edition: European region data. IDF.
- European Commission, Directorate-General for Health and Food Safety. (2024, January). State of health in the EU: Companion report 2024 diabetes chapter. EC.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the Demand for Glucose Monitoring Devices in EU in 2026?
The demand for glucose monitoring devices in eu is estimated at USD 5.36 billion in 2026, based on FMI proprietary bottom-up revenue modeling and primary research validation.
What will the Demand for Glucose Monitoring Devices in EU size be by 2036?
FMI projects the demand for glucose monitoring devices in eu to reach USD 10.74 billion by 2036, supported by sustained demand across primary end-use segments and geographies.
What is the CAGR for the Demand for Glucose Monitoring Devices in EU between 2026 and 2036?
The demand for glucose monitoring devices in eu is projected to grow at a CAGR of 7.2% between 2026 and 2036.
Which product type leads in 2026?
Self-Monitoring Devices is estimated to hold 57.9% of the product type segment in 2026, driven by established procurement preference and performance consistency relative to alternative configurations.
Which country shows the fastest growth in the Demand for Glucose Monitoring Devices in EU?
Spain registers the highest CAGR at 7.6% through 2036, driven by policy-backed demand creation and expanding end-user industry capacity.
What is the leading application in the Demand for Glucose Monitoring Devices in EU?
Diabetes Type II is the leading application sub-segment in 2026, anchored by institutional procurement cycles and volume commitments that create predictable replacement demand.
What is included in the scope of this report?
The scope includes self-monitoring blood glucose meters, associated test strips and lancets, real-time and intermittently scanned continuous glucose monitoring systems, sensor applicators, transmitters, and compatible data management software sold through EU healthcare channels. Products are analyzed across all major segments and geographies defined in the FMI market framework.
What does FMI use to validate the Demand for Glucose Monitoring Devices in EU forecast?
FMI applies a bottom-up methodology anchored to verified unit shipment data and average selling price benchmarks, cross-referenced against top-down total addressable market checks and validated through primary interviews with market participants.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Desk Research Programme (Secondary Evidence)
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Self-Monitoring Devices
- Blood Glucose Meters
- Test Strips
- Lancets
- Continuous Monitoring Devices
- Sensors
- Transmitters
- Receiver Systems
- Self-Monitoring Devices
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Diabetes Type II
- Adult-Onset Diabetes
- Metabolic Syndrome
- Diabetes Type I
- Juvenile Diabetes
- Autoimmune Diabetes
- Gestational Diabetes
- Diabetes Type II
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel:
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel:, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel:, 2026 to 2036
- Hospitals & Clinics
- Healthcare Facilities
- Diabetes Centers
- Pharmacies
- Community Pharmacies
- Hospital Pharmacies
- Online Retail
- E-commerce Platforms
- Direct-to-Consumer Websites
- Hospitals & Clinics
- Y to o to Y Growth Trend Analysis By Distribution Channel:, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel:, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Indication
- By Distribution Channel:
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel:
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Indication
- By Distribution Channel:
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel:
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Indication
- By Distribution Channel:
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel:
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Indication
- By Distribution Channel:
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel:
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Indication
- By Distribution Channel:
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel:
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Indication
- By Distribution Channel:
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel:
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Indication
- By Distribution Channel:
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel:
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel:
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Indication
- By Distribution Channel:
- Competition Analysis
- Competition Deep Dive
- Abbott Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Dexcom Inc.
- Roche (Accu-Chek)
- Medtronic
- LifeScan (OneTouch)
- Ascensia Diabetes Care
- Senseonics
- AgaMatrix
- Ypsomed
- European Regional Manufacturers
- Abbott Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Distribution Channel:, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Distribution Channel:, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Distribution Channel:, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Distribution Channel:, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel:, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Distribution Channel:, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel:, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel:, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Indication
- Figure 9: Global Market Value Share and BPS Analysis by Distribution Channel:, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Distribution Channel:, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Distribution Channel:
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Indication
- Figure 29: North America Market Value Share and BPS Analysis by Distribution Channel:, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Distribution Channel:, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Distribution Channel:
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Indication
- Figure 39: Latin America Market Value Share and BPS Analysis by Distribution Channel:, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Distribution Channel:, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Distribution Channel:
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Indication
- Figure 49: Western Europe Market Value Share and BPS Analysis by Distribution Channel:, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel:, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Distribution Channel:
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel:, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel:, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Distribution Channel:
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Indication
- Figure 69: East Asia Market Value Share and BPS Analysis by Distribution Channel:, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Distribution Channel:, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Distribution Channel:
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel:, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel:, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel:
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel:, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel:, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Distribution Channel:
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

