Hemp Oil-Based Formulas Market
Hemp Oil-Based Formulas Market Forecast and Outlook 2026 to 2036
Hemp Oil-Based Formulas Market Forecast and Outlook 2026 to 2036
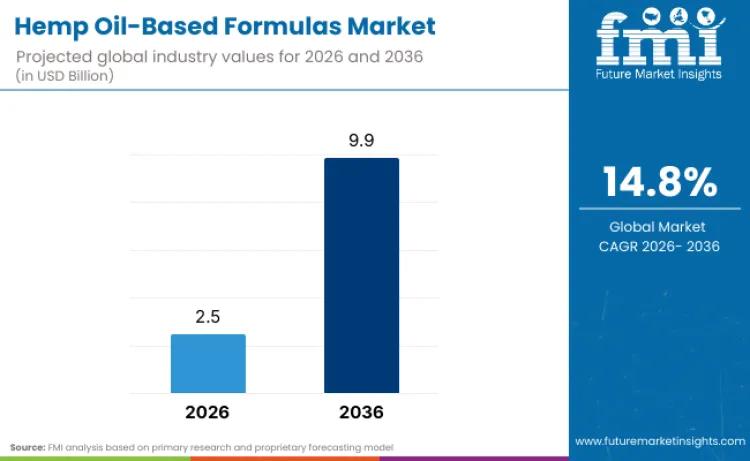
Hemp oil based formulas are valued at USD 2.5 billion in 2026, with expansion shaped by rising consumer acceptance of cannabinoid led wellness and clearer commercialization pathways created by evolving hemp regulations. The category is projected to reach USD 9.9 billion by 2036 at a 14.8% CAGR, supported by higher expectations around product consistency, documented composition, and repeatable user outcomes across beauty, personal care, and wellness applications. Companies that lead in this space treat extraction, purification, and cannabinoid standardization as front line competitive capabilities rather than back end processing steps, because retailers and regulators increasingly expect traceability, batch level testing, and specification driven supply.
Government regulation is a direct influence on how this category scales. In the United States, hemp derivatives are governed by the 2018 Farm Bill, while the FDA regulates how CBD and related ingredients can be positioned in cosmetics, foods, and wellness products, with strict limits on therapeutic claims. In the European Union, hemp extracts fall under cosmetic regulation and Novel Food authorization pathways, requiring safety dossiers and ingredient documentation. Similar compliance frameworks are emerging in parts of Asia Pacific and Latin America, where authorities are formalizing THC limits, labeling rules, and import controls. These policies are reducing room for unverified products and favoring suppliers that can meet testing and documentation standards.
As formulation teams align cannabinoid profiles with defined use cases and brands move from curiosity led products to protocol led routines, demand is shifting toward standardized extracts that make downstream scaling easier while maintaining regulatory readiness and consumer trust.
Quick Stats for Hemp Oil-Based Formulas Market
- Hemp Oil-Based Formulas Industry Value (2026): USD 2.5 Billion
- Hemp Oil-Based Formulas Industry Forecast Value (2036): USD 9.9 Billion
- Hemp Oil-Based Formulas Industry Forecast CAGR: 14.8%
- Hemp Oil-Based Formulas Industry Leading Product Type: Hemp CBD Oil (34.7%)
- Hemp Oil-Based Formulas Industry Key Growth Regions: North America, Europe, Asia-Pacific
- Hemp Oil-Based Formulas Industry Key Players: Cannavest, Pharmahemp, ENDOCA, BAFA GmbH, Protect Pharma Rakitovica
Hemp Oil-Based Formulas Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 2.5 Billion |
| Industry Value (2036) | USD 9.9 Billion |
| CAGR (2026-2036) | 14.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
What Drivers Are Accelerating Hemp CBD Oil Adoption?
Hemp CBD oil adoption is being accelerated by a shift from “wellness curiosity” to outcomes-led, formulation-driven demand. Buyers are increasingly selecting products based on predictable performance, clarity on what is inside the bottle, and confidence that the product meets non-psychoactive expectations. This is pushing the category away from loose botanical positioning and toward cannabinoid profiles that are engineered, repeatable, and easier to scale across mass and premium channels without brand risk.
On the product side, advances in isolation and purification are strengthening consistency and removing variability that previously limited repeat purchase and retailer confidence. That technical progress is widening use cases because brands can now design broad-spectrum offerings across multiple delivery formats while keeping composition aligned to labeling and compliance requirements. The result is faster portfolio expansion, especially where consumers want natural alternatives but still expect proof, transparency, and a controlled experience.
How Is the Hemp Oil-Based Formulas Market Segmented Across Key Categories?
Global marketplace is comprehensively segmented to address varied requirements of the hemp-derived wellness industry, categorizing sector by product type, application, distribution channel, source, and end-user. This structural organization enables stakeholders to identify specific therapeutic opportunities, such as increasing demand for organic hemp extracts in premium cosmetic formulations or preference for cold-pressed seed oils in functional food applications. Through analyzing these segments, manufacturers can customize extraction processes to meet distinct requirements of pharmaceutical-grade applications versus consumer wellness products. Segmentation also reveals transition from basic hemp seed oils to sophisticated full-spectrum cannabinoid formulations. As regulatory frameworks advance, detailed segmentation becomes essential for understanding therapeutic pathways and compliance requirements, ensuring product development aligns with evolving cannabis industry standards.
Why Do Hemp CBD Oil Products Maintain Leadership?
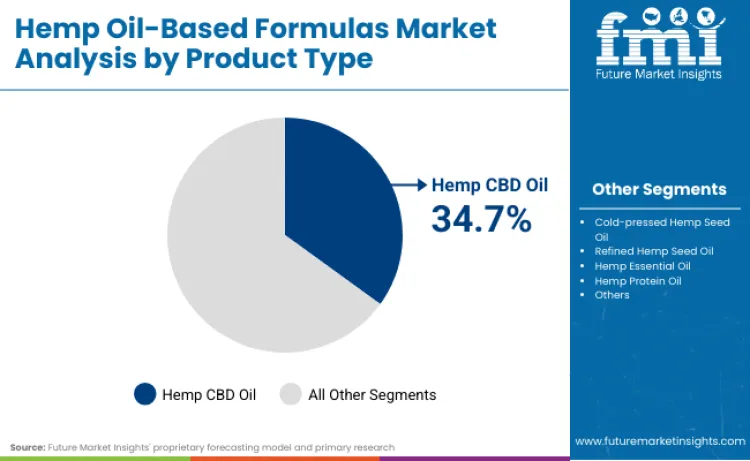
Hemp CBD oil products secure 34.7% of product type share in 2026, driven by extensive therapeutic research and proven efficacy across multiple wellness applications. Market leadership stems from established extraction technologies and comprehensive safety profiles that wellness brands can leverage effectively. Segment benefits from continuous purification improvements, including chromatographic separation techniques and molecular distillation removing impurities. Research focused on CBD pharmacokinetics is revitalizing interest among pharmaceutical manufacturers. Critical role in pain management and anxiety reduction protocols further strengthens market position, as these formulations provide targeted therapeutic relief. As wellness guidelines emphasize CBD therapeutic potential, demand for high-concentration oil formulations is expected to remain robust.
Why Do Cosmetics & Personal Care Applications Lead Market Adoption?
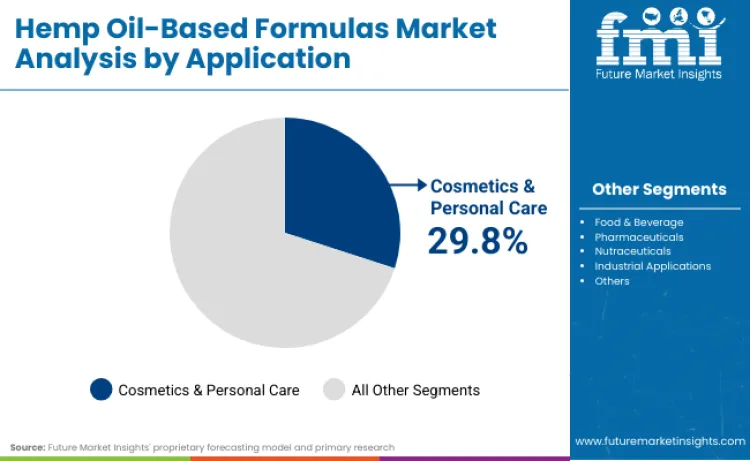
Cosmetics and personal care applications command 29.8% of application share in 2026, reflecting growing consumer interest in cannabinoid-infused beauty products and anti-aging formulations. Leadership position is connected to hemp oil's proven anti-inflammatory properties and compatibility with sensitive skin types. Beauty manufacturers depend on hemp-derived ingredients to achieve natural wellness positioning while maintaining product efficacy standards. These applications are vital for delivering antioxidant protection and skin barrier restoration, establishing priority status among premium skincare brands. Continuous pursuit of organic certification ensures formulations designed for daily use remain the preferred choice. Strong consumer acceptance drives substantial ingredient demand, supporting sustained revenue growth for hemp extract suppliers.
Why Do Online Retail Channels Dominate Distribution Strategies?
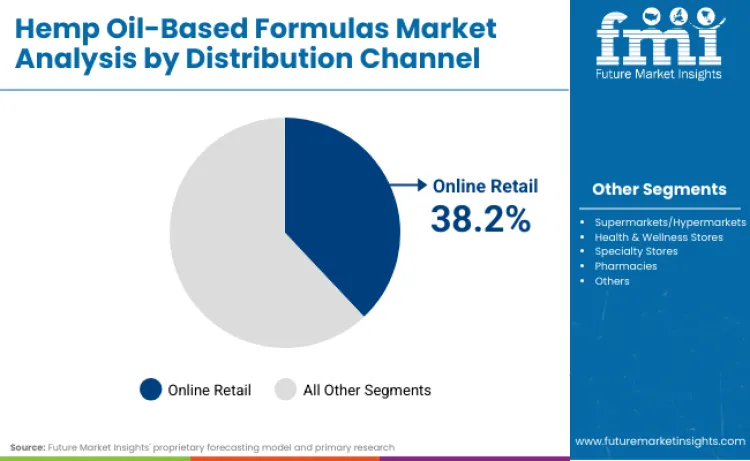
Online retail distribution captures 38.2% of channel share in 2026, owing to direct-to-consumer marketing advantages and comprehensive product education capabilities. Dominance originates from regulatory complexity requiring specialized knowledge that e-commerce platforms can provide through detailed product information. Consumers appreciate these channels for their convenience and access to third-party testing results and certification documentation. Implementation in digital platforms drives continuous adoption growth among tech-savvy wellness consumers. Compatibility with subscription models allows for consistent product delivery across different dosing requirements. Hemp brands focusing on direct online sales for specialized cannabinoid formulations secure substantial customer loyalty among educated wellness enthusiasts.
How Will Regulatory Evolution Impact Product Development?
Regulatory evolution is becoming the primary “rules-of-scale” factor for hemp oil-based formulas. As standards mature, the commercial advantage shifts toward operators that can demonstrate repeatable quality through standardized testing, documentation, and auditable manufacturing controls. This directly influences product development: formulations increasingly need to be built around defined cannabinoid profiles and consistent terpene composition, supported by methods that can reliably reproduce those specifications at volume. For CEOs, the implication is clear, regulation is not only a compliance requirement, it is a gating mechanism for distribution access, partner credibility, and long-term brand defensibility.
A tightening compliance environment also raises the cost of inconsistency. Companies that cannot align extraction precision, quality assurance, and labeling discipline risk channel restrictions and reputational exposure. Conversely, firms that invest early in compliance-aligned processes create faster pathways into regulated and reputation-sensitive categories, where buyers scrutinize traceability, testing credibility, and manufacturing controls before committing shelf space or recommending usage.
How Will Scientific Research Transform Therapeutic Applications?
Scientific progress is pushing the category toward more targeted, evidence-oriented use cases and away from broad, ambiguous wellness claims. As clinical work and product testing improve the understanding of how different cannabinoid profiles perform, brands can move toward more specific formulation strategies, built to deliver a consistent experience and supported by clearer product rationale. That transformation benefits suppliers capable of delivering standardized extracts that align with defined therapeutic intentions and can be integrated into controlled product development pipelines.
From a market standpoint, research momentum raises the bar for differentiation. Competitive advantage increasingly comes from translating research into repeatable formulations, defensible product positioning, and more disciplined protocols for measuring consistency and customer outcomes, particularly as healthcare-adjacent stakeholders become more selective about what they are willing to recommend or distribute.
How Will Supply Chain Innovation Affect Market Growth?
Supply chain innovation is increasingly tied to product credibility and margin stability. Cultivation practices, cannabinoid expression consistency, processing capability, and documentation integrity all influence whether finished formulations can meet the specifications demanded by premium brands and stricter channels. The strongest operators are building tighter control from cultivation through processing to finished extract delivery, reducing variability and improving reliability for downstream manufacturing schedules and product consistency expectations.
Sustainability and certification expectations are also shaping procurement and branding strategies. As organic and traceability-driven sourcing gains importance, producers that can validate farming practices and maintain integrity across the chain are better positioned to support premium product lines and long-term supply agreements. For executives, the supply chain is no longer just an input cost lever, it is a strategic asset that determines scale readiness, channel eligibility, and the ability to defend quality claims in a more scrutinized competitive environment.
How Will the Hemp Oil-Based Formulas Industry Evolve in Major Nations?
Global landscape for hemp-derived wellness products is characterized by diverse regulatory environments, influenced by cannabis legislation and therapeutic goods administration policies. Established markets prioritize pharmaceutical-grade extraction systems and comprehensive clinical validation, whereas developing economies focus on agricultural hemp cultivation and basic processing capabilities. North America is emerging as regulatory leader due to progressive cannabis legislation and advanced research infrastructure. Conversely, European markets emphasize organic certification and traditional herbal medicine integration models. As per FMI's projection, government initiatives in developed nations supporting cannabis research ensure sustained demand for hemp-derived therapeutic products across all wellness categories.
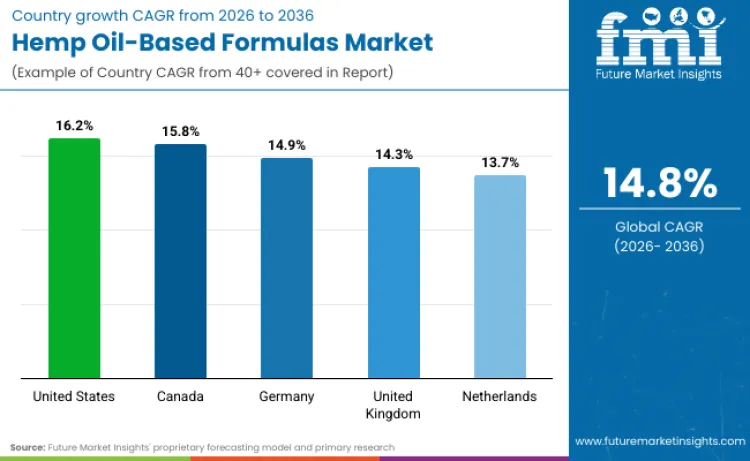
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 16.2% |
| Canada | 15.8% |
| Germany | 14.9% |
| United Kingdom | 14.3% |
| Netherlands | 13.7% |
Source: Future Market Insights - analysis driven by proprietary forecasting models and primary research
Why is the Hemp Oil Based Formulas Industry in United States Growing?
Development of hemp oil based formulas in the United States is positioned to grow at 16.2% CAGR through 2036 because federal hemp legalization created a large upstream cultivation and ingredient supply base, while downstream pathways are being shaped by federal product classification rules. The Agriculture Improvement Act of 2018 carved out hemp from the Controlled Substances Act definition of marijuana, and USDA continues to administer the domestic hemp production framework, which supports scalable sourcing for hemp derived inputs. At the same time, FDA has stated that existing foods and dietary supplement regulatory frameworks are not appropriate for cannabidiol, which has pushed many brands toward channels where claims and product positioning can be kept compliant, such as cosmetics and other non ingestible formats, or toward drug development routes where formal approval pathways exist.
Why is the Hemp Oil Based Formulas Sector in Canada Expanding?
Demand in Canada is projected to advance at 15.8% CAGR because the regulatory environment draws a clear line between hemp seed derived products and cannabinoid concentrated products, which shapes what can scale through mainstream consumer channels. Health Canada explains that hemp seed oil can be exempt from the Cannabis Act only when phytocannabinoids such as THC and CBD are not added or concentrated by processing, allowing compliant hemp seed oil products to be sold across food, cosmetics, and natural health product channels under the applicable rules. Canada also maintains Industrial Hemp Regulations under federal law, reinforcing a defined compliance structure for cultivation and handling. On the category expansion side, Health Canada has been consulting on a potential pathway for health products containing cannabidiol, which signals active policy work that could broaden compliant product routes if implemented.
Why is the Hemp Oil Based Formulas Industry in Germany Showing Potential?
Germany is set to expand at 14.9% CAGR because the highest compliance ready demand concentrates around medically governed cannabinoid products, while broader consumer formats remain constrained by EU level food authorization rules. Germany’s Federal Institute for Drugs and Medical Devices states that medical cannabis and medical scientific cannabis are regulated under the MedCanG framework, and BfArM is the competent authority for application of the MedCanG, which supports a structured, pharma grade channel for oils and extracts used as medicines. In parallel, the European Commission maintains the Novel Food status catalogue and consultation process that member states use to determine whether substances fall under Novel Food rules, which affects how cannabinoid extract products can be commercialized as ingestible items across the EU.
Why is the Hemp Oil Based Formulas Industry in United Kingdom Growing?
Sales in the United Kingdom are set to grow at 14.3% CAGR because the CBD segment is being forced into a more formal compliance track that favors manufacturers able to support safety dossiers and stable quality systems. The Food Standards Agency positions CBD extracts and isolates for ingestible use within the novel foods authorization route and has published CBD guidance that expects products not linked to the novel food process to be voluntarily withdrawn. The FSA also maintains a public register of CBD products linked to novel food applications, which effectively drives market consolidation toward documented, traceable products that fit the regulator’s expectations for continued sale.
Why is the Hemp Oil Based Formulas Sector in Netherlands Expanding?
Revenue in the Netherlands is positioned to grow at 13.7% CAGR because the country has a formal government controlled architecture for medicinal cannabis that supports regulated production and cross border trade in medical grade products, which influences the ingredient and extract supply ecosystem. The Dutch Office of Medicinal Cannabis is the government agency responsible for overseeing production for medicinal and scientific purposes and holds the monopoly on supplying medicinal cannabis to pharmacies, as well as import and export of cannabis and extracts under the Single Convention framework. For ingestible cannabinoid products outside the medicines channel, EU novel food determination processes remain the gating mechanism for commercialization, which tends to favor compliant operators with strong regulatory documentation.
What is the Competitive Outlook for the Hemp Oil-Based Formulas Market?
Competition is shifting from “who sells CBD” to who can prove consistent chemistry, compliant claims, and repeatable manufacturing at scale. One clear signal is that legacy players are buying capability, not just adding SKUs: Cannavest (now operating as CV Sciences) has moved to widen control over formulation and production by signing a definitive agreement to acquire Extract Labs, a GMP-certified, FDA-registered manufacturer with gummies, topicals, and tinctures plus flexible production runs, an upgrade aimed at faster product development and better margin control. On the European bulk side, suppliers like PharmaHemp compete on B2B reliability through documented quality systems, highlighting ISO certifications and GMP-related certifications for cannabis-based raw materials and cosmetics, which matters for brands that need traceability and batch consistency.
Regulatory friction is a defining competitive filter, especially for ingestibles: EU authorities have repeatedly noted that CBD novel foods are not authorized at the EU level, which pushes many firms toward cosmetics/topicals, “wellness” positioning, or B2B ingredient supply until approvals mature. Innovation is concentrating in delivery science, nanoemulsions and other nano-systems are being explored to improve bioavailability and performance consistency, creating room for IP-led differentiation beyond commodity extracts.
Key Players in Hemp Oil-Based Formulas Market
- Cannavest
- Pharmahemp
- ENDOCA
- BAFA GmbH
- Protect Pharma Rakitovica
- Biobloom Hemp
- Deep Nature Project
- Harmony
- Dragonfly CBD
- MH medical hemp GmbH
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product Type Segments | Hemp CBD Oil; Cold-pressed Hemp Seed Oil; Refined Hemp Seed Oil; Hemp Essential Oil; Hemp Protein Oil; Others |
| Application Categories | Cosmetics & Personal Care; Food & Beverage; Pharmaceuticals; Nutraceuticals; Industrial Applications; Others |
| Distribution Channel Types | Online Retail; Supermarkets/Hypermarkets; Health & Wellness Stores; Specialty Stores; Pharmacies; Others |
| Source Categories | Organic Hemp; Conventional Hemp; Others |
| End-User Categories | Cosmetic Brands; Households; Food Manufacturers; Nutraceutical Companies; Medical Professionals; Others |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, Middle East & Africa |
| Key Countries | United States, Canada, Germany, United Kingdom, Netherlands |
| Key Companies Profiled | Cannavest; Pharmahemp; ENDOCA; BAFA GmbH; Protect Pharma Rakitovica; Biobloom Hemp; Deep Nature Project; Harmony; Dragonfly CBD; MH medical hemp GmbH |
| Additional Attributes | Dollar sales measured for hemp oil-based formulas used in therapeutic applications and wellness products, specified by extraction method (CO2, ethanol, cold-pressed), cannabinoid concentration levels, product certification status, application focus (topical, oral, inhalation), distribution model (direct-to-consumer vs. retail), and compliance alignment with evolving cannabis regulations and therapeutic goods standards. |
Hemp Oil-Based Formulas Market by Segments
Product Type
- Hemp CBD Oil
- Cold-pressed Hemp Seed Oil
- Refined Hemp Seed Oil
- Hemp Essential Oil
- Hemp Protein Oil
- Others
Application
- Cosmetics & Personal Care
- Food & Beverage
- Pharmaceuticals
- Nutraceuticals
- Industrial Applications
- Others
Distribution Channel
- Online Retail
- Supermarkets/Hypermarkets
- Health & Wellness Stores
- Specialty Stores
- Pharmacies
- Others
Source
- Organic Hemp
- Conventional Hemp
- Others
End-User
- Cosmetic Brands
- Households
- Food Manufacturers
- Nutraceutical Companies
- Medical Professionals
- Others
Bibliography
- USA Food and Drug Administration. (2023). Regulatory considerations for cannabis and cannabis‑derived compounds: Guidance for industry. USA Department of Health and Human Services, FDA.
- European Medicines Agency. (2024). Guideline on quality and safety of botanical and herbal medicinal products containing cannabinoids. European Medicines Agency.
- National Institutes of Health, National Center for Complementary and Integrative Health. (2024). Cannabinoids: What we know and what we don’t - A scientific review. NIH NCCIH.
- European Industrial Hemp Association. (2024). Sustainable cultivation practices and organic certification requirements for hemp production. European Industrial Hemp Association.
Frequently Asked Questions
How big is the global hemp oil-based formulas market?
The global hemp oil-based formulas market is valued at USD 2.5 billion in 2026.
What is the growth outlook for the hemp oil-based formulas market over the next 10 years?
The market is projected to grow at a 14.8% CAGR from 2026 to 2036, reaching USD 9.9 billion.
Which applications or end uses drive demand for hemp oil-based formulas?
Demand is primarily driven by cosmetics and personal care, nutraceuticals, pharmaceuticals, and functional wellness products that require standardized cannabinoid profiles.
What are the key regional differences in the hemp oil-based formulas market?
North America leads in commercial scale and regulatory clarity, Europe emphasizes organic certification and pharmaceutical alignment, while Asia-Pacific is emerging through formalization of hemp regulations and wellness adoption.
What are the main risks and constraints affecting this market?
Key constraints include regulatory uncertainty, strict limits on therapeutic claims, variability in cannabinoid composition, and high compliance and testing costs.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Y-o-Y Growth Trend Analysis 2021 to 2025
- Absolute $ Opportunity Analysis 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Product Type, 2026 to 2036
- Hemp CBD Oil
- Cold-pressed Hemp Seed Oil
- Refined Hemp Seed Oil
- Hemp Essential Oil
- Hemp Protein Oil
- Others
- Y-o-Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Application, 2026 to 2036
- Cosmetics & Personal Care
- Food & Beverage
- Pharmaceuticals
- Nutraceuticals
- Industrial Applications
- Others
- Y-o-Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Online Retail
- Supermarkets/Hypermarkets
- Health & Wellness Stores
- Specialty Stores
- Pharmacies
- Others
- Y-o-Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Source
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Source, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Source, 2026 to 2036
- Organic Hemp
- Conventional Hemp
- Others
- Y-o-Y Growth Trend Analysis By Source, 2021 to 2025
- Absolute $ Opportunity Analysis By Source, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-User
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By End-User, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By End-User, 2026 to 2036
- Cosmetic Brands
- Households
- Food Manufacturers
- Nutraceutical Companies
- Medical Professionals
- Others
- Y-o-Y Growth Trend Analysis By End-User, 2021 to 2025
- Absolute $ Opportunity Analysis By End-User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By Distribution Channel
- By Source
- By End-User
- Competition Analysis
- Competition Deep Dive
- Cannavest
- Pharmahemp
- ENDOCA
- BAFA GmbH
- Protect Pharma Rakitovica
- Biobloom Hemp
- Deep Nature Project
- Harmony
- Dragonfly CBD
- MH medical hemp GmbH
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Hemp Oil-Based Formulas Market Key Takeaways
- Global Hemp Oil-Based Formulas Market Size (USD Billion), 2021-2036
- Global Hemp Oil-Based Formulas Market Y-o-Y Growth (%), 2021-2036
- Global Hemp Oil-Based Formulas Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Global Hemp Oil-Based Formulas Market Pricing Analysis (USD/Unit), 2021-2036
- Global Hemp Oil-Based Formulas Market Size (USD Billion) By Product Type, 2021-2036
- Global Hemp Oil-Based Formulas Market Size (USD Billion) By Application, 2021-2036
- Global Hemp Oil-Based Formulas Market Size (USD Billion) By Distribution Channel, 2021-2036
- Global Hemp Oil-Based Formulas Market Size (USD Billion) By Source, 2021-2036
- Global Hemp Oil-Based Formulas Market Size (USD Billion) By End-User, 2021-2036
- Global Hemp Oil-Based Formulas Market Size (USD Billion) By Region, 2021-2036
- North America Hemp Oil-Based Formulas Market Size (USD Billion) By Country, 2021-2036
- North America Hemp Oil-Based Formulas Market Size (USD Billion) By Product Type, 2021-2036
- North America Hemp Oil-Based Formulas Market Size (USD Billion) By Application, 2021-2036
- North America Hemp Oil-Based Formulas Market Size (USD Billion) By Distribution Channel, 2021-2036
- North America Hemp Oil-Based Formulas Market Size (USD Billion) By Source, 2021-2036
- North America Hemp Oil-Based Formulas Market Size (USD Billion) By End-User, 2021-2036
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- Latin America Hemp Oil-Based Formulas Market Size (USD Billion) By Country, 2021-2036
- Latin America Hemp Oil-Based Formulas Market Size (USD Billion) By Product Type, 2021-2036
- Latin America Hemp Oil-Based Formulas Market Size (USD Billion) By Application, 2021-2036
- Latin America Hemp Oil-Based Formulas Market Size (USD Billion) By Distribution Channel, 2021-2036
- Latin America Hemp Oil-Based Formulas Market Size (USD Billion) By Source, 2021-2036
- Latin America Hemp Oil-Based Formulas Market Size (USD Billion) By End-User, 2021-2036
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- Western Europe Hemp Oil-Based Formulas Market Size (USD Billion) By Country, 2021-2036
- Western Europe Hemp Oil-Based Formulas Market Size (USD Billion) By Product Type, 2021-2036
- Western Europe Hemp Oil-Based Formulas Market Size (USD Billion) By Application, 2021-2036
- Western Europe Hemp Oil-Based Formulas Market Size (USD Billion) By Distribution Channel, 2021-2036
- Western Europe Hemp Oil-Based Formulas Market Size (USD Billion) By Source, 2021-2036
- Western Europe Hemp Oil-Based Formulas Market Size (USD Billion) By End-User, 2021-2036
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- Eastern Europe Hemp Oil-Based Formulas Market Size (USD Billion) By Country, 2021-2036
- Eastern Europe Hemp Oil-Based Formulas Market Size (USD Billion) By Product Type, 2021-2036
- Eastern Europe Hemp Oil-Based Formulas Market Size (USD Billion) By Application, 2021-2036
- Eastern Europe Hemp Oil-Based Formulas Market Size (USD Billion) By Distribution Channel, 2021-2036
- Eastern Europe Hemp Oil-Based Formulas Market Size (USD Billion) By Source, 2021-2036
- Eastern Europe Hemp Oil-Based Formulas Market Size (USD Billion) By End-User, 2021-2036
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- East Asia Hemp Oil-Based Formulas Market Size (USD Billion) By Country, 2021-2036
- East Asia Hemp Oil-Based Formulas Market Size (USD Billion) By Product Type, 2021-2036
- East Asia Hemp Oil-Based Formulas Market Size (USD Billion) By Application, 2021-2036
- East Asia Hemp Oil-Based Formulas Market Size (USD Billion) By Distribution Channel, 2021-2036
- East Asia Hemp Oil-Based Formulas Market Size (USD Billion) By Source, 2021-2036
- East Asia Hemp Oil-Based Formulas Market Size (USD Billion) By End-User, 2021-2036
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- South Asia and Pacific Hemp Oil-Based Formulas Market Size (USD Billion) By Country, 2021-2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Size (USD Billion) By Product Type, 2021-2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Size (USD Billion) By Application, 2021-2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Size (USD Billion) By Distribution Channel, 2021-2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Size (USD Billion) By Source, 2021-2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Size (USD Billion) By End-User, 2021-2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- Middle East & Africa Hemp Oil-Based Formulas Market Size (USD Billion) By Country, 2021-2036
- Middle East & Africa Hemp Oil-Based Formulas Market Size (USD Billion) By Product Type, 2021-2036
- Middle East & Africa Hemp Oil-Based Formulas Market Size (USD Billion) By Application, 2021-2036
- Middle East & Africa Hemp Oil-Based Formulas Market Size (USD Billion) By Distribution Channel, 2021-2036
- Middle East & Africa Hemp Oil-Based Formulas Market Size (USD Billion) By Source, 2021-2036
- Middle East & Africa Hemp Oil-Based Formulas Market Size (USD Billion) By End-User, 2021-2036
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- USA Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- USA Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- USA Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- USA Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- USA Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- USA Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Canada Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Canada Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Canada Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Canada Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Canada Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Canada Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Mexico Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Mexico Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Mexico Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Mexico Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Mexico Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Mexico Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Brazil Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Brazil Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Brazil Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Brazil Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Brazil Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Brazil Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Chile Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Chile Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Chile Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Chile Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Chile Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Chile Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Germany Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Germany Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Germany Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Germany Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Germany Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Germany Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- UK Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- UK Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- UK Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- UK Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- UK Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- UK Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Italy Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Italy Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Italy Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Italy Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Italy Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Italy Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Spain Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Spain Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Spain Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Spain Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Spain Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Spain Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- France Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- France Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- France Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- France Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- France Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- France Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- India Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- India Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- India Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- India Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- India Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- India Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- ASEAN Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- ASEAN Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- ASEAN Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- ASEAN Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- ASEAN Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- ASEAN Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Australia & New Zealand Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Australia & New Zealand Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Australia & New Zealand Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Australia & New Zealand Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Australia & New Zealand Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- China Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- China Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- China Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- China Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- China Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- China Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Japan Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Japan Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Japan Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Japan Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Japan Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Japan Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- South Korea Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- South Korea Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- South Korea Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- South Korea Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- South Korea Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- South Korea Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Russia Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Russia Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Russia Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Russia Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Russia Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Russia Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Poland Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Poland Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Poland Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Poland Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Poland Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Poland Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Hungary Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Hungary Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Hungary Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Hungary Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Hungary Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Hungary Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Kingdom of Saudi Arabia Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Kingdom of Saudi Arabia Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Kingdom of Saudi Arabia Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Kingdom of Saudi Arabia Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Kingdom of Saudi Arabia Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Turkiye Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- Turkiye Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- Turkiye Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- Turkiye Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- Turkiye Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- Turkiye Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- South Africa Hemp Oil-Based Formulas Pricing Analysis (USD/Unit), 2021-2036
- South Africa Hemp Oil-Based Formulas Market Share Analysis (%) By Product Type, 2025
- South Africa Hemp Oil-Based Formulas Market Share Analysis (%) By Application, 2025
- South Africa Hemp Oil-Based Formulas Market Share Analysis (%) By Distribution Channel, 2025
- South Africa Hemp Oil-Based Formulas Market Share Analysis (%) By Source, 2025
- South Africa Hemp Oil-Based Formulas Market Share Analysis (%) By End-User, 2025
- Hemp Oil-Based Formulas Market Competition Dashboard
- Hemp Oil-Based Formulas Market Competition Benchmarking Analysis
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By Regional, 2025
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By Product Type, 2025
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By Application, 2025
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By Distribution Channel, 2025
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By Source, 2025
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By End-User, 2025
- Cannavest - Company Profile
- Pharmahemp - Company Profile
- ENDOCA - Company Profile
- BAFA GmbH - Company Profile
- Protect Pharma Rakitovica - Company Profile
- Biobloom Hemp - Company Profile
- Deep Nature Project - Company Profile
- Harmony - Company Profile
- Dragonfly CBD - Company Profile
- MH medical hemp GmbH - Company Profile
- Assumptions & Acronyms Used in Hemp Oil-Based Formulas Market Report
List of Figures
- Hemp Oil-Based Formulas Market Size (USD Billion), 2021-2036
- Hemp Oil-Based Formulas Market Y-o-Y Growth (%), 2021-2036
- Hemp Oil-Based Formulas Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Hemp Oil-Based Formulas Market Value Chain Analysis
- Hemp Oil-Based Formulas Market Supply Chain Analysis
- Hemp Oil-Based Formulas Market Investment Feasibility Matrix
- Hemp Oil-Based Formulas Market PESTLE Analysis
- Hemp Oil-Based Formulas Market Porter's Five Forces Analysis
- Hemp Oil-Based Formulas Market Product Life Cycle Analysis
- Hemp Oil-Based Formulas Market Opportunity Map Analysis
- Hemp Oil-Based Formulas Market Scenario Forecast Analysis
- Hemp Oil-Based Formulas Market Production and Consumption Statistics
- Hemp Oil-Based Formulas Market Import and Export Statistics
- Global Hemp Oil-Based Formulas Market Share (%) By Product Type, 2025
- Global Hemp Oil-Based Formulas Market Share (%) By Product Type, 2036
- Global Hemp Oil-Based Formulas Market Share (%) By Application, 2025
- Global Hemp Oil-Based Formulas Market Share (%) By Application, 2036
- Global Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2025
- Global Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2036
- Global Hemp Oil-Based Formulas Market Share (%) By Source, 2025
- Global Hemp Oil-Based Formulas Market Share (%) By Source, 2036
- Global Hemp Oil-Based Formulas Market Share (%) By End-User, 2025
- Global Hemp Oil-Based Formulas Market Share (%) By End-User, 2036
- Global Hemp Oil-Based Formulas Market Share (%) By Region, 2025
- Global Hemp Oil-Based Formulas Market Share (%) By Region, 2036
- Global Hemp Oil-Based Formulas Market Attractiveness Analysis By Region
- North America Hemp Oil-Based Formulas Market Size (USD Billion), 2021-2036
- North America Hemp Oil-Based Formulas Market Share (%) By Country, 2025
- North America Hemp Oil-Based Formulas Market Share (%) By Country, 2036
- North America Hemp Oil-Based Formulas Market Share (%) By Product Type, 2025
- North America Hemp Oil-Based Formulas Market Share (%) By Product Type, 2036
- North America Hemp Oil-Based Formulas Market Share (%) By Application, 2025
- North America Hemp Oil-Based Formulas Market Share (%) By Application, 2036
- North America Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2025
- North America Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2036
- North America Hemp Oil-Based Formulas Market Share (%) By Source, 2025
- North America Hemp Oil-Based Formulas Market Share (%) By Source, 2036
- North America Hemp Oil-Based Formulas Market Share (%) By End-User, 2025
- North America Hemp Oil-Based Formulas Market Share (%) By End-User, 2036
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- North America Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- Latin America Hemp Oil-Based Formulas Market Size (USD Billion), 2021-2036
- Latin America Hemp Oil-Based Formulas Market Share (%) By Country, 2025
- Latin America Hemp Oil-Based Formulas Market Share (%) By Country, 2036
- Latin America Hemp Oil-Based Formulas Market Share (%) By Product Type, 2025
- Latin America Hemp Oil-Based Formulas Market Share (%) By Product Type, 2036
- Latin America Hemp Oil-Based Formulas Market Share (%) By Application, 2025
- Latin America Hemp Oil-Based Formulas Market Share (%) By Application, 2036
- Latin America Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2025
- Latin America Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2036
- Latin America Hemp Oil-Based Formulas Market Share (%) By Source, 2025
- Latin America Hemp Oil-Based Formulas Market Share (%) By Source, 2036
- Latin America Hemp Oil-Based Formulas Market Share (%) By End-User, 2025
- Latin America Hemp Oil-Based Formulas Market Share (%) By End-User, 2036
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- Latin America Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- Western Europe Hemp Oil-Based Formulas Market Size (USD Billion), 2021-2036
- Western Europe Hemp Oil-Based Formulas Market Share (%) By Country, 2025
- Western Europe Hemp Oil-Based Formulas Market Share (%) By Country, 2036
- Western Europe Hemp Oil-Based Formulas Market Share (%) By Product Type, 2025
- Western Europe Hemp Oil-Based Formulas Market Share (%) By Product Type, 2036
- Western Europe Hemp Oil-Based Formulas Market Share (%) By Application, 2025
- Western Europe Hemp Oil-Based Formulas Market Share (%) By Application, 2036
- Western Europe Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2025
- Western Europe Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2036
- Western Europe Hemp Oil-Based Formulas Market Share (%) By Source, 2025
- Western Europe Hemp Oil-Based Formulas Market Share (%) By Source, 2036
- Western Europe Hemp Oil-Based Formulas Market Share (%) By End-User, 2025
- Western Europe Hemp Oil-Based Formulas Market Share (%) By End-User, 2036
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- Western Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- Eastern Europe Hemp Oil-Based Formulas Market Size (USD Billion), 2021-2036
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By Country, 2025
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By Country, 2036
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By Product Type, 2025
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By Product Type, 2036
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By Application, 2025
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By Application, 2036
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2025
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2036
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By Source, 2025
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By Source, 2036
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By End-User, 2025
- Eastern Europe Hemp Oil-Based Formulas Market Share (%) By End-User, 2036
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- Eastern Europe Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- East Asia Hemp Oil-Based Formulas Market Size (USD Billion), 2021-2036
- East Asia Hemp Oil-Based Formulas Market Share (%) By Country, 2025
- East Asia Hemp Oil-Based Formulas Market Share (%) By Country, 2036
- East Asia Hemp Oil-Based Formulas Market Share (%) By Product Type, 2025
- East Asia Hemp Oil-Based Formulas Market Share (%) By Product Type, 2036
- East Asia Hemp Oil-Based Formulas Market Share (%) By Application, 2025
- East Asia Hemp Oil-Based Formulas Market Share (%) By Application, 2036
- East Asia Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2025
- East Asia Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2036
- East Asia Hemp Oil-Based Formulas Market Share (%) By Source, 2025
- East Asia Hemp Oil-Based Formulas Market Share (%) By Source, 2036
- East Asia Hemp Oil-Based Formulas Market Share (%) By End-User, 2025
- East Asia Hemp Oil-Based Formulas Market Share (%) By End-User, 2036
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- East Asia Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- South Asia and Pacific Hemp Oil-Based Formulas Market Size (USD Billion), 2021-2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By Country, 2025
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By Country, 2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By Product Type, 2025
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By Product Type, 2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By Application, 2025
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By Application, 2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2025
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By Source, 2025
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By Source, 2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By End-User, 2025
- South Asia and Pacific Hemp Oil-Based Formulas Market Share (%) By End-User, 2036
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- South Asia and Pacific Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- Middle East & Africa Hemp Oil-Based Formulas Market Size (USD Billion), 2021-2036
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By Country, 2025
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By Country, 2036
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By Product Type, 2025
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By Product Type, 2036
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By Application, 2025
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By Application, 2036
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2025
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By Distribution Channel, 2036
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By Source, 2025
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By Source, 2036
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By End-User, 2025
- Middle East & Africa Hemp Oil-Based Formulas Market Share (%) By End-User, 2036
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By Country
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By Product Type
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By Application
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By Source
- Middle East & Africa Hemp Oil-Based Formulas Market Attractiveness Analysis By End-User
- USA Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Canada Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Mexico Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Brazil Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Chile Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Germany Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- UK Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Italy Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Spain Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- France Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- India Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- ASEAN Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- China Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Japan Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- South Korea Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Russia Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Poland Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Hungary Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Turkiye Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- South Africa Hemp Oil-Based Formulas Pricing Trend Analysis (USD/Unit), 2021-2036
- Hemp Oil-Based Formulas Market Competitive Landscape
- Hemp Oil-Based Formulas Market Structure Analysis
- Hemp Oil-Based Formulas Market Competition Dashboard
- Hemp Oil-Based Formulas Market Competition Benchmarking Analysis
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By Regional
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By Product Type
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By Application
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By Distribution Channel
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By Source
- Hemp Oil-Based Formulas Market Share Analysis of Top Players By End-User
- Cannavest - Revenue Analysis (USD Billion), 2019-2025
- Pharmahemp - Revenue Analysis (USD Billion), 2019-2025
- ENDOCA - Revenue Analysis (USD Billion), 2019-2025
- BAFA GmbH - Revenue Analysis (USD Billion), 2019-2025
- Protect Pharma Rakitovica - Revenue Analysis (USD Billion), 2019-2025
- Cannavest - Market Position Analysis
- Pharmahemp - Market Position Analysis
- ENDOCA - Market Position Analysis
- BAFA GmbH - Market Position Analysis
- Protect Pharma Rakitovica - Market Position Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

