The immunosuppressants kits market is forecasted to reach USD 1,340.4 million in 2026 and expand to USD 2,920.7 million by 2036, at a CAGR of 8.1%. Value behavior reflects the structural requirement for routine therapeutic drug monitoring in transplant medicine and chronic autoimmune care, where dosing precision directly affects clinical risk. Spending remains anchored to repeat testing volumes rather than episodic diagnostics, with laboratories prioritizing assay reliability, lot consistency, and uninterrupted reagent supply. Capital allocation favors platforms supporting standardized workflows across inpatient and outpatient monitoring, reinforcing stable procurement cycles within hospital networks and reference laboratories.
Adoption patterns are shaped by regulatory oversight, assay validation demands, and integration with automated laboratory systems. Utilization intensity increases where transplant programs scale longitudinal follow-up and where combination immunosuppressive regimens require frequent exposure assessment. Cost sensitivity persists in smaller centers due to reagent expense and validation overhead, limiting indiscriminate expansion. Market structure remains constrained by requirements for analytical specificity, cross-platform comparability, and continuous quality control. These constraints reinforce predictable growth tied to protocolized care pathways, long-term patient management, and institutional monitoring obligations rather than expansion of therapeutic indications.
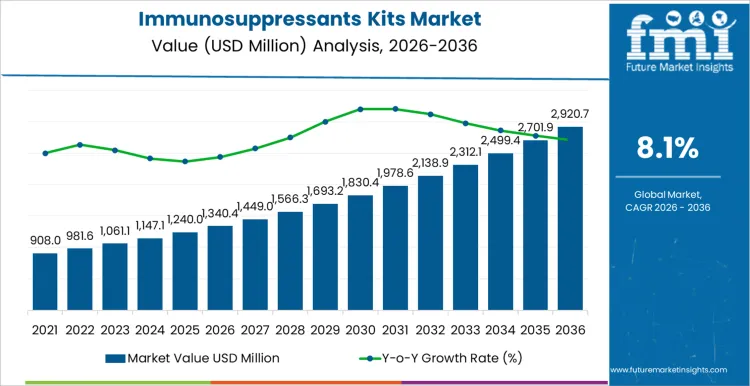
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1,340.4 million |
| Market Forecast Value (2036) | USD 2,920.7 million |
| Forecast CAGR 2026 to 2036 | 8.1% |
Demand for immunosuppressants kits is expanding as healthcare providers and transplant centers require standardized panels to support therapeutic drug monitoring and dose optimization in patients undergoing organ transplantation and treatment for autoimmune disorders. These kits include reagents, controls, and calibrators designed to measure blood concentrations of immunosuppressive agents such as tacrolimus, cyclosporine, and sirolimus, enabling clinicians to maintain drug levels within narrow therapeutic ranges that prevent rejection while minimizing toxicity. Clinical pharmacology laboratories value kits that deliver reproducible results with minimal assay variability because precise measurement influences dosing decisions that affect graft survival and patient outcomes. Procurement teams assess kit stability, lot-to-lot consistency, and compatibility with various analytical platforms to ensure integration with existing instrumentation and laboratory workflows under routine and high-volume testing conditions.
Growth in solid organ transplant procedures, rising prevalence of autoimmune conditions requiring long-term immunosuppression, and expanded use of personalized medicine strategies reinforce adoption of comprehensive monitoring solutions that reduce complications and readmissions. Providers are implementing structured immunosuppressant testing algorithms that support early detection of subtherapeutic or supratherapeutic exposure, which reduces incidence of opportunistic infections and drug-related organ toxicity. Research into drug interactions, pharmacogenomics, and immune response variability encourages laboratories to adopt multi-analyte kits that support broader clinical interpretation. Training for clinical laboratory scientists on assay performance and interpretation enhances confidence in therapeutic monitoring. These clinical performance, workflow integration, and patient management considerations are driving sustained demand growth in the immunosuppressants kits market.
Demand for immunoassay kits is shaped by therapeutic drug monitoring requirements, narrow therapeutic windows, and post-transplant outcome optimization. Laboratories prioritize analytical accuracy, turnaround time, and method standardization to support dose adjustment decisions. Adoption reflects growth in transplant procedures, broader use of combination regimens, and expansion of LC-MS/MS workflows. Selection decisions emphasize assay specificity, cross-reactivity control, and integration with laboratory systems. Segmentation clarifies how kit focus, monitoring objective, and testing environment influence utilization patterns across transplant medicine and immunotherapy management.
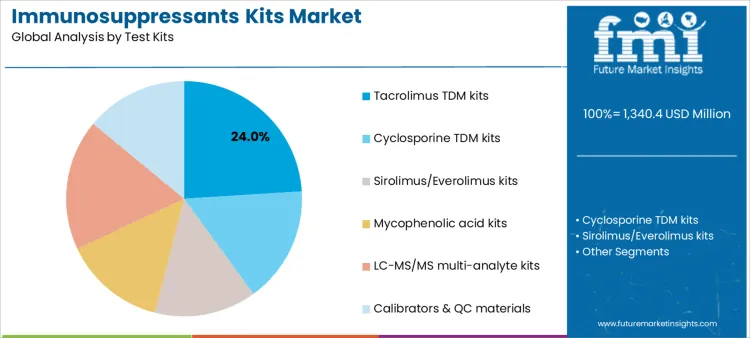
Tacrolimus TDM kits account for 24.0%, reflecting widespread use as a first-line calcineurin inhibitor in solid organ transplantation. These kits support routine trough level monitoring and frequent dose adjustments. LC-MS/MS multi-analyte kits at 18.0% enable simultaneous quantification of multiple immunosuppressants, improving efficiency and comparability. Cyclosporine TDM kits at 16.0% remain relevant in specific transplant protocols. Sirolimus or everolimus kits and mycophenolic acid kits each at 14.0% support mTOR inhibitor and antiproliferative monitoring. Calibrators and QC materials at 14.0% underpin assay reliability. Kit segmentation reflects emphasis on high-volume drugs supported by multiplexing and quality control.
Key Points:
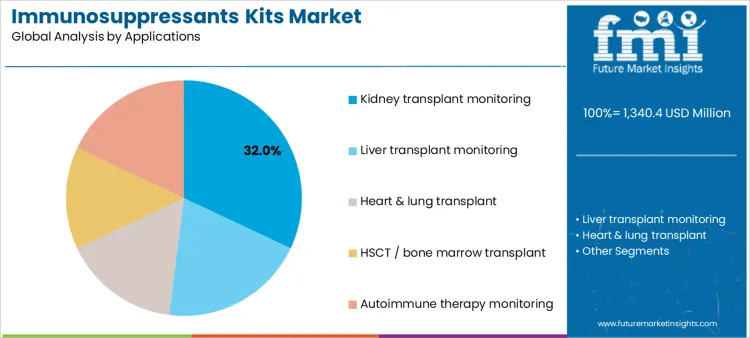
Kidney transplant monitoring represents 32.0%, driven by high transplant volumes and long-term immunosuppression management. Liver transplant monitoring at 20.0% requires close exposure control due to variable metabolism. Autoimmune therapy monitoring at 18.0% reflects expanding off-transplant use of immunosuppressants. Heart and lung transplant applications at 16.0% involve complex regimens and critical dosing precision. HSCT or bone marrow transplant at 14.0% applies monitoring during conditioning and graft maintenance. Application-based segmentation highlights concentration where dosing precision directly affects graft survival, toxicity risk, and therapeutic outcomes.
Key Points:
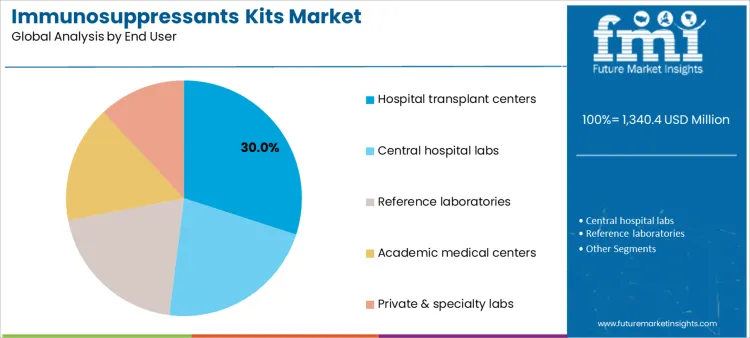
Hospital transplant centers account for 30.0%, reflecting proximity to patients and need for rapid therapeutic decisions. Central hospital laboratories at 22.0% provide high-throughput testing supporting multiple clinical units. Reference laboratories at 20.0% offer standardized methods and LC-MS/MS expertise for external clients. Academic medical centers at 16.0% support complex cases and protocol-driven care. Private and specialty laboratories at 12.0% address outpatient monitoring needs. End-user segmentation indicates concentration where clinical urgency, testing volume, and analytical capability guide kit adoption.
Key Points:
Demand for immunosuppressants kits reflects clinical monitoring requirements for drugs with narrow therapeutic windows used in transplantation and autoimmune disease management. Adoption concentrates in hospital laboratories, transplant centers, and specialized diagnostic facilities. Global scope aligns with long-term graft survival protocols and chronic immunosuppression management. Usage centers on assay kits designed to quantify immunosuppressive drug levels in blood to support dose optimization and toxicity avoidance.
Immunosuppressive therapies require precise dosing to balance rejection prevention against infection and organ toxicity risk. Demand increases where interpatient variability affects drug metabolism and exposure. Kits enable standardized measurement of drug concentrations to guide individualized dosing decisions. Transplant programs rely on routine monitoring to maintain stable immunosuppression during early post-transplant and maintenance phases. Autoimmune disease management uses monitoring to adjust long-term therapy and reduce adverse effects. Assay reliability, turnaround time, and compatibility with existing analyzers influence laboratory selection. Integration into routine clinical workflows supports frequent testing without excessive burden. Adoption reflects clinical dependence on objective drug level data to manage high-risk therapies safely.
Immunosuppressant assays must distinguish parent drugs from metabolites to avoid dosing error. Demand sensitivity rises where cross-reactivity or analytical variability affects clinical confidence. Regulatory oversight requires validation, quality control, and proficiency testing, increasing operational burden for laboratories. Reagent cost and instrument compatibility influence adoption in smaller centers. Method standardization varies across platforms, complicating result comparison between institutions. Supply continuity remains critical due to uninterrupted monitoring needs. Staff training and assay calibration add overhead. Scalability remains constrained by regulatory compliance demands, assay performance expectations, and economic considerations tied to sustained testing volume.
Demand for immunosuppressants kits is expanding globally as transplant programs, autoimmune disease management, and therapeutic drug monitoring gain clinical importance. Routine monitoring of drug levels supports dose optimization, toxicity prevention, and long-term graft survival. Laboratory standardization and automation improve testing consistency across hospital networks. Reimbursement frameworks and guideline inclusion influence testing frequency. Growth rates in India at 10.6%, Brazil at 10.1%, China at 9.6%, the USA at 6.6%, and Germany at 6.5% indicate sustained expansion driven by transplant volume growth, protocolized monitoring, and laboratory capacity scaling rather than increases in disease incidence.
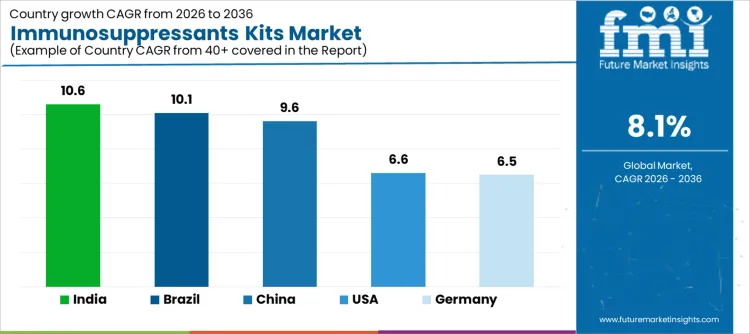
| Country | CAGR (%) |
|---|---|
| India | 10.6% |
| Brazil | 10.1% |
| China | 9.6% |
| USA | 6.6% |
| Germany | 6.5% |
Demand for immunosuppressants kits in India is growing at a CAGR of 10.6%, supported by rising organ transplant activity and post-transplant care standardization. Tertiary hospitals expand therapeutic drug monitoring to manage calcineurin inhibitors and mTOR inhibitors effectively. Growth of private transplant centers increases routine testing volumes. Improved access to automated immunoassay platforms reduces turnaround time. Gradual insurance coverage expansion supports sustained monitoring. Growth reflects transplant program maturation and protocol adherence rather than episodic testing linked to acute complications.
Immunosuppressants kit demand in Brazil is expanding at a CAGR of 10.1%, driven by public transplant programs and long-term graft management. Public hospitals require regular drug level monitoring to support universal transplant coverage. Standardized follow-up protocols increase testing consistency. Regional laboratories expand access beyond major metropolitan centers. Government procurement supports stable kit supply. Growth reflects institutional commitment to transplant outcomes and monitoring discipline rather than expansion of transplant indications.
Demand for immunosuppressants kits in China is growing at a CAGR of 9.6%, aligned with expanding transplant infrastructure and hospital capacity. Large transplant centers integrate drug monitoring within routine inpatient and outpatient care. Reimbursement inclusion for therapeutic monitoring improves access. Domestic kit manufacturers supply cost-competitive assays at scale. Centralized hospital laboratories support high-throughput testing. Growth reflects systematized transplant care delivery and laboratory scaling rather than decentralized testing expansion.
Immunosuppressants kit demand in the United States is expanding at a CAGR of 6.6%, supported by established transplant programs and guideline-driven care. Therapeutic drug monitoring remains integral to post-transplant management across organ types. Broad payer coverage ensures routine testing continuity. Advanced laboratory automation supports assay precision and throughput. Replacement demand and assay upgrades sustain volumes. Growth reflects protocol adherence and quality assurance rather than growth in transplant rates.
Immunosuppressants kit demand in Germany is growing at a CAGR of 6.5%, shaped by evidence-based transplant care and reimbursement governance. Certified transplant centers apply standardized monitoring schedules. Statutory insurance covers approved assays for long-term therapy management. Central laboratory services ensure analytical consistency and quality control. Emphasis on clinical utility moderates testing intensity. Growth remains steady, reflecting structured adoption and health system oversight rather than rapid expansion of testing volumes.
Demand for immunosuppressants kits is driven by organ transplantation volumes, therapeutic drug monitoring requirements, and long-term graft survival management. Clinical laboratories evaluate assay accuracy at low trough levels, cross-reactivity control, turnaround time, and compatibility with immunoassay or chromatography platforms. Buyer assessment includes calibration stability, reagent lot consistency, sample matrix flexibility, and compliance with clinical guidelines. Procurement behavior reflects hospital standardization strategies, long-term reagent contracts, and preference for kits integrated with existing analyzers. Trend in the immunosuppressants kits market shows sustained monitoring intensity supported by protocol-driven dosing and increasing transplant program complexity.
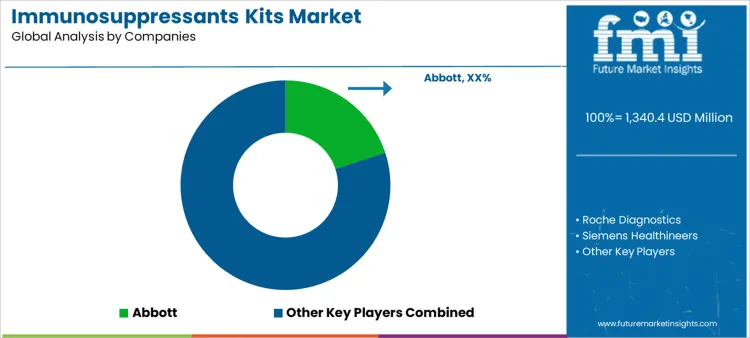
Abbott leads competitive positioning through immunosuppressant monitoring kits embedded within high-throughput clinical chemistry and immunoassay systems. Roche Diagnostics competes with standardized kits aligned with automated laboratory workflows and centralized testing environments. Siemens Healthineers supports demand by offering assays integrated with scalable laboratory automation and data management systems. Thermo Fisher Scientific maintains relevance through chromatography-based kits supporting high specificity monitoring in reference laboratories. Waters participates by aligning immunosuppressant kits with LC-MS workflows used for precise therapeutic drug monitoring. Competitive differentiation centers on analytical accuracy, workflow integration, regulatory compliance, and reliability of long-term supply.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Test Kits | Tacrolimus TDM kits; Cyclosporine TDM kits; Sirolimus/Everolimus kits; Mycophenolic acid kits; LC-MS/MS multi-analyte kits; Calibrators & QC materials |
| Applications | Kidney transplant monitoring; Liver transplant monitoring; Heart & lung transplant; HSCT / bone marrow transplant; Autoimmune therapy monitoring |
| End User | Hospital transplant centers; Central hospital labs; Reference laboratories; Academic medical centers; Private & specialty labs |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Brazil, China, USA, Germany, and 40+ countries |
| Key Companies Profiled | Abbott; Roche Diagnostics; Siemens Healthineers; Thermo Fisher Scientific; Waters; Others |
| Additional Attributes | Dollar sales by test kit type, application, and end user; analytical accuracy and inter-assay precision for immunosuppressant therapeutic drug monitoring across immunoassay and LC-MS/MS platforms; turnaround time and workflow integration in transplant laboratories; role of calibrators and QC materials in maintaining longitudinal result consistency; clinical decision support for dose optimization and toxicity avoidance; procurement dynamics shaped by transplant volumes, centralized lab testing, and regulatory quality requirements. |
How big is the immunosuppressants kits market in 2026?
The global immunosuppressants kits market is estimated to be valued at USD 1,340.4 million in 2026.
What will be the size of immunosuppressants kits market in 2036?
The market size for the immunosuppressants kits market is projected to reach USD 2,920.7 million by 2036.
How much will be the immunosuppressants kits market growth between 2026 and 2036?
The immunosuppressants kits market is expected to grow at a 8.1% CAGR between 2026 and 2036.
What are the key product types in the immunosuppressants kits market?
The key product types in immunosuppressants kits market are tacrolimus tdm kits, cyclosporine tdm kits, sirolimus/everolimus kits, mycophenolic acid kits, lc-ms/ms multi-analyte kits and calibrators & qc materials.
Which applications segment to contribute significant share in the immunosuppressants kits market in 2026?
In terms of applications, kidney transplant monitoring segment to command 32.0% share in the immunosuppressants kits market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.