The global isoprene rubber latex market is forecasted to reach USD 1,079.4 million by 2035, recording an absolute increase of USD 80.5 million over the forecast period. The market is valued at USD 996.7 million in 2025 and is set to rise at a CAGR of 0.8% during the assessment period. The overall market size is expected to grow by nearly 1.08 times during the same period, supported by increasing demand for latex-free medical products worldwide, driving demand for biocompatible polyisoprene systems and increasing investments in healthcare infrastructure and surgical procedure volumes globally. Raw material price volatility and complex manufacturing requirements for high-purity formulations may pose challenges to market expansion.
Between 2025 and 2030, the market is projected to expand from USD 996.7 million to USD 1,037.1 million, resulting in a value increase of USD 40.4 million, which represents 50.2% of the total forecast growth for the decade. This phase of development will be shaped by rising demand for latex allergy-free medical products and healthcare safety protocols, product innovation in high-purity polyisoprene formulations and biocompatible medical devices, as well as expanding integration with surgical procedure growth and infection prevention initiatives. Companies are establishing competitive positions through investment in automated manufacturing systems, medical-grade quality certifications, and strategic market expansion across hospital procurement channels, medical device OEMs, and regulatory compliance frameworks.
From 2030 to 2035, the market is forecast to grow from USD 1,037.1 million to USD 1,079.4 million, adding another USD 40.1 million, which constitutes 49.8% of the overall ten-year expansion. This period is expected to be characterized by the expansion of specialized applications, including advanced surgical glove formulations and integrated medical device solutions tailored for specific clinical requirements, strategic collaborations between latex manufacturers and healthcare providers, and an enhanced focus on sustainability and environmental compliance. The growing focus on patient safety optimization and biocompatibility standards will drive demand for advanced, high-performance isoprene rubber latex solutions across diverse medical and healthcare applications.
| Metric | Value |
|---|---|
| Market Value (2025) | USD 996.7 million |
| Market Forecast Value (2035) | USD 1,079.4 million |
| Forecast CAGR (2025-2035) | 0.8% |
The market grows by enabling healthcare providers to achieve superior biocompatibility and latex allergy prevention in critical medical applications, ranging from surgical procedures to medical device manufacturing. Healthcare manufacturers face mounting pressure to eliminate latex allergy risks and improve patient safety outcomes, with synthetic polyisoprene solutions typically providing equivalent performance to natural rubber latex while eliminating allergenic proteins that affect up to 8-17% of healthcare workers. The medical industry's need for maximum safety and performance creates demand for advanced polyisoprene solutions that can deliver surgical-grade tactile sensitivity, maintain sterility standards, and ensure consistent quality across diverse clinical environments. Government healthcare policies promoting infection prevention and patient safety standards drive adoption in hospital systems, surgical centers, and medical device manufacturing, where latex allergy prevention has a direct impact on clinical outcomes and healthcare worker safety. The higher production costs compared to natural rubber latex and the complexity of achieving consistent quality formulations may limit adoption rates among cost-sensitive healthcare facilities and developing regions with limited procurement budgets.
The market is segmented by application, end use, region, and form. By application, the market is divided into medical gloves, condoms, catheters, medical balloons, and adhesives. Based on end use, the market is categorized into medical, consumer goods, and industrial. By region, the market is segmented into Asia Pacific, North America, Europe, Latin America, and Middle East & Africa. Based on form, the market is classified into powder-free surgical gloves, examination gloves, catheter components, and medical adhesives.
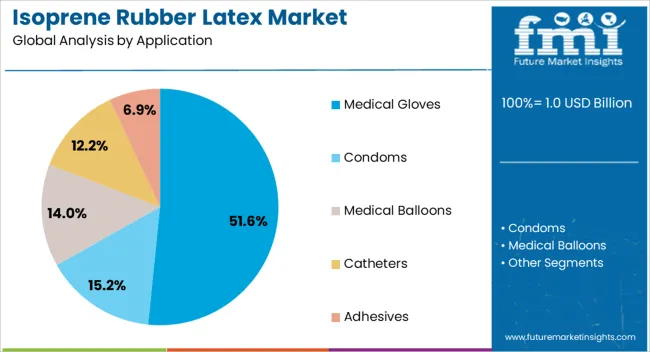
The medical gloves segment represents the dominant force in the isoprene rubber latex market, capturing approximately 51.6% of total market share in 2025. This advanced application category encompasses surgical and examination gloves featuring sophisticated biocompatibility profiles, including latex protein-free formulations and optimized tactile sensitivity that enable superior clinical performance and healthcare worker safety characteristics. The medical gloves segment's market leadership stems from its exceptional safety performance capabilities, with gloves capable of eliminating latex allergy risks while maintaining equivalent dexterity and barrier protection compared to natural rubber latex alternatives.
The segment benefits from increasing surgical procedure volumes globally and expanding healthcare infrastructure investments. Within medical gloves, surgical powder-free variants represent 62.0% of the segment, driven by operating room requirements and sterile procedure protocols, while examination gloves account for 38.0%, supported by outpatient care and diagnostic procedure growth.
Key technological advantages driving the medical gloves segment include:
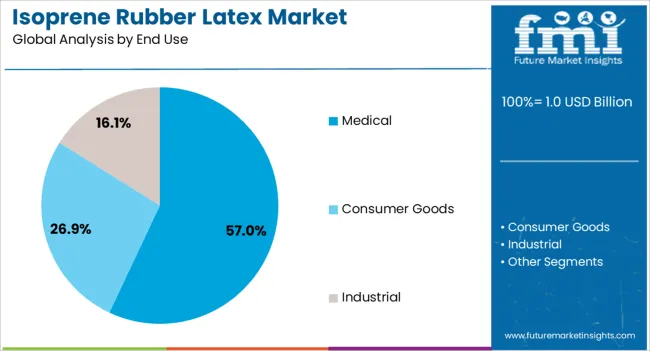
Medical applications dominate the isoprene rubber latex market with approximately 57.0% market share in 2025, reflecting the critical role of synthetic polyisoprene in healthcare safety protocols and patient protection initiatives supporting global medical device manufacturing growth. The medical segment's market leadership is reinforced by accelerating healthcare expenditure, government safety regulations, and rising demand for latex allergy prevention that directly correlates with clinical outcomes and healthcare worker protection requirements.
Consumer goods represent the second-largest end use category, capturing 28.0% market share through specialized requirements for condoms and personal care products requiring biocompatible materials. The industrial segment accounts for 15.0% market share, serving adhesives manufacturers and specialty coating applications.
Within the medical segment, surgical supplies constitute 55.0% of medical end use through gloves, catheters, and surgical device components. Patient care disposables represent 25.0% including examination gloves and medical balloon applications, while diagnostics and specialty applications capture 20.0% covering catheter systems and medical adhesive formulations.
Key market dynamics supporting medical segment growth include:
Powder-free surgical gloves are estimated to hold premium market positioning due to enhanced safety characteristics, reduced contamination risks, and surgical-grade performance specifications that eliminate powder-related complications in sterile clinical environments. These specialty products offer regulatory compliance benefits for both healthcare facilities and medical device manufacturers while maintaining superior tactile sensitivity compared to powdered alternatives that can cause tissue irritation and surgical site complications.
The segment provides advanced clinical capabilities for specialized surgical procedures requiring maximum dexterity and contamination control. Powder-free formulations meet strict regulatory requirements including FDA medical device regulations and European MDR compliance standards for surgical applications.
Manufacturing advantages supporting powder-free surgical gloves include automated dipping technologies enabling consistent quality control and specialized curing processes that eliminate residual chemicals while maintaining optimal physical properties for surgical use.
Key market characteristics include:
The market is driven by three concrete demand factors tied to healthcare safety optimization outcomes. First, healthcare industry growth and medical safety protocol expansion create increasing demand for latex allergy prevention solutions, with global healthcare expenditure projected to grow by 5-7% annually in major markets worldwide, requiring specialized biocompatible materials for maximum patient and worker safety. Second, surgical procedure advancement and medical device innovation drive the adoption of superior polyisoprene formulations, with healthcare facilities seeking complete latex allergy elimination and enhanced clinical performance optimization. Third, technological advancements in polymer chemistry and manufacturing processes enable more effective and consistent polyisoprene solutions that reduce contamination risks while improving long-term clinical outcomes and cost-effectiveness.
Market restraints include raw material price volatility that can impact production costs and profitability margins, particularly during periods of supply chain disruption or petroleum price fluctuations affecting synthetic polymer production. Manufacturing complexity in achieving consistent biocompatibility poses another significant challenge, as producing medical-grade polyisoprene requires specialized expertise and quality control systems, potentially causing production delays and increased manufacturing costs. Regulatory compliance requirements and validation processes create additional complexity for manufacturers, demanding ongoing investment in clinical testing and certification programs to meet varying regional medical device standards.
The growing development of high-purity polyisoprene formulations is enabling enhanced patient safety and clinical performance that meet USP Class VI biocompatibility standards and FDA medical device requirements. Advanced polymer processing with latex protein elimination maintains surgical performance throughout sterilization cycles while delivering complete allergy prevention compared to natural rubber latex alternatives that contain allergenic proteins affecting healthcare workers. Medical-grade formulations help reduce adverse reactions while maintaining tactile sensitivity, making them valuable for surgical procedures and medical device applications where patient safety and clinical reliability are essential.
Manufacturing capabilities also support development of specialized applications incorporating catheter balloon systems and medical adhesive formulations for cardiovascular procedures seeking biocompatible material solutions with proven clinical performance.
| Country | CAGR (2025-2035) |
|---|---|
| India | 12.0% |
| China | 8.9% |
| Malaysia | 7.4% |
| Spain | 6.1% |
| United States | 4.5% |
| Germany | 3.9% |
| United Kingdom | 3.4% |
The global market is expected to experience notable growth from 2025 to 2035, driven by rising demand for medical gloves, adhesives, and personal care products. India leads with a robust CAGR of 12.0%, supported by strong growth in healthcare manufacturing and expanding glove production capacity. China follows at 8.9%, driven by industrial expansion and rising use of latex in medical and hygiene products. Malaysia records a 7.4% CAGR, benefiting from its strong base in natural rubber processing and export-oriented glove manufacturing. Spain grows at 6.1%, supported by expanding healthcare infrastructure and industrial applications. The United States shows a 4.5% CAGR, reflecting steady medical and industrial demand. Germany and the United Kingdom record modest growth of 3.9% and 3.4% respectively, indicating mature markets with stable consumption in healthcare and industrial protective applications.
The report covers an in-depth analysis of 40+ countries, top-performing countries are highlighted below.
India demonstrates the strongest growth potential in the market with a CAGR of 12.0% through 2035. The country's leadership position stems from massive healthcare infrastructure projects, government-backed medical device manufacturing programs, and aggressive hospital expansion targets, driving the adoption of latex allergy-free medical systems. Growth is concentrated in major healthcare regions, including Maharashtra, Karnataka, Tamil Nadu, and Gujarat, where hospital networks and medical device manufacturers are implementing advanced polyisoprene solutions for enhanced patient safety and international competitiveness. Distribution channels through established medical suppliers and direct hospital relationships expand deployment across surgical facilities and medical device production lines. The country's Ayushman Bharat healthcare initiative provides policy support for advanced medical materials development, including high-performance biocompatible systems.
Key market factors driving Indian growth include:
In Beijing, Shanghai, Guangzhou, and Shenzhen, the adoption of isoprene rubber latex systems is accelerating across medical device manufacturing facilities and healthcare networks, driven by healthcare modernization targets and domestic manufacturing competitiveness initiatives. The market demonstrates strong growth momentum with a CAGR of 8.9% through 2035, linked to comprehensive medical device capacity expansion and increasing focus on healthcare safety solutions. Chinese manufacturers are implementing advanced polyisoprene systems and quality control platforms to enhance product performance while meeting growing demand for medical gloves and healthcare products in domestic and export markets. The country's healthcare reform programs create constant demand for high-performance medical materials, while increasing focus on patient safety drives adoption of advanced biocompatible technologies.
Leading manufacturing expansion factors include:
Malaysia's established rubber processing industry demonstrates sophisticated implementation of isoprene rubber latex systems, with documented case studies showing enhanced safety performance in medical glove applications through optimized biocompatible formulations. The country's manufacturing infrastructure in major industrial centers, including Selangor, Penang, Johor, and Perak, showcases integration of advanced polyisoprene technologies with existing production systems, leveraging expertise in latex processing and precision manufacturing. Malaysian manufacturers prioritize quality standards and export competitiveness, creating demand for high-performance polyisoprene solutions that support international market requirements and regulatory compliance. The market maintains steady growth through focus on technology innovation and manufacturing excellence, with a CAGR of 7.4% through 2035.
Key development areas supporting Malaysian market growth:
Spain's market expansion is driven by diverse healthcare modernization demand, including hospital upgrades in Madrid and Barcelona, medical procurement improvements in Valencia and Seville, and comprehensive healthcare system enhancement across multiple regions. The country demonstrates promising growth potential with a CAGR of 6.1% through 2035, supported by EU healthcare development programs and regional medical facility modernization initiatives. Spanish healthcare facilities face implementation challenges related to budget constraints and procurement complexity, requiring cost-effective approaches and supplier partnership support. Growing patient safety requirements and healthcare quality standards create compelling business cases for polyisoprene adoption, particularly in surgical departments where latex allergy prevention has a direct impact on clinical outcomes.
Market characteristics supporting Spanish growth:
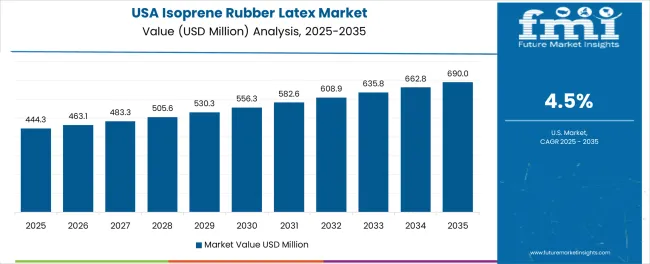
The USA market leads in advanced polyisoprene innovation based on integration with next-generation medical technologies and sophisticated healthcare applications for enhanced patient safety characteristics. The country shows solid potential with a CAGR of 4.5% through 2035, driven by healthcare quality improvement programs and advanced medical device manufacturing initiatives across major industrial regions, including California, Texas, North Carolina, and Massachusetts. American manufacturers are adopting advanced polyisoprene systems for clinical performance optimization and regulatory compliance, particularly in states with stringent healthcare standards and advanced medical facilities requiring superior product differentiation. Technology deployment channels through established medical distributors and direct healthcare relationships expand coverage across hospital systems and medical device production facilities.
Leading market segments driving USA growth:
In England, Scotland, Wales, and Northern Ireland, healthcare facilities are implementing isoprene rubber latex solutions to enhance patient safety and improve clinical outcomes, with documented case studies showing significant reduction in latex allergy incidents through optimized polyisoprene applications. The market shows moderate growth potential with a CAGR of 3.4% through 2035, linked to NHS modernization programs, healthcare safety initiatives, and emerging medical technology adoption projects. British healthcare providers are adopting advanced polyisoprene systems to enhance patient care while maintaining safety standards demanded by UK healthcare regulations and international clinical guidelines. The country's established healthcare system creates steady demand for technology upgrade solutions that integrate with existing medical protocols.
Market development factors supporting UK growth:
Germany's market demonstrates sophisticated implementation focused on precision engineering and quality excellence optimization, with documented integration of advanced polyisoprene systems achieving proven biocompatibility performance across medical device and healthcare applications. The country maintains steady growth momentum with a CAGR of 3.9% through 2035, driven by manufacturers' focus on quality standards and continuous improvement methodologies that align with medical device manufacturing principles applied to biocompatible materials processing operations. Major industrial regions, including North Rhine-Westphalia, Bavaria, Baden-Württemberg, and Lower Saxony, showcase advanced deployment of precision polyisoprene platforms where systems integrate seamlessly with existing quality control systems and comprehensive medical device management programs.
Key market characteristics supporting German development:
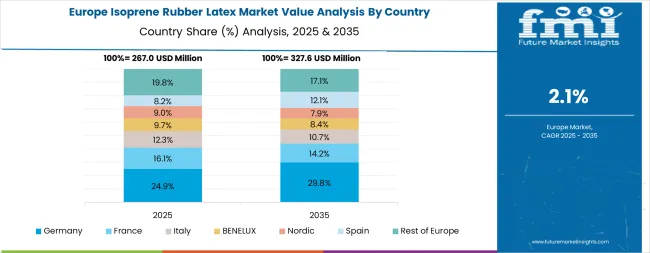
The isoprene rubber latex market in Europe is projected to grow from USD 239.2 million in 2025 to USD 258.6 million by 2035, registering a CAGR of 0.8% over the forecast period. Germany is expected to maintain its leadership position with approximately 20% market share in 2025, supported by its extensive chemical manufacturing infrastructure and major medical device production facilities, including Munich, Frankfurt, and Hamburg industrial centers.
The United Kingdom follows with a 16% share in 2025, driven by comprehensive NHS procurement programs and advanced healthcare initiatives implementing polyisoprene technologies. France holds a 14% share through ongoing medical facility upgrades and healthcare technology development. Italy commands a 12% share, while Spain accounts for 10% in 2025. The Rest of Europe region is anticipated to maintain steady momentum, with Nordic countries and emerging Eastern European healthcare facilities implementing advanced biocompatible materials programs.
In Japan, the market is largely driven by the medical applications segment, which accounts for advanced biocompatible material requirements in the domestic healthcare system and the precision manufacturing focus that characterizes Japanese medical device production. The healthcare focus in clinical safety protocols and the comprehensive quality management systems are key contributing factors supporting polyisoprene adoption. Industrial applications follow with specialized requirements in automotive and electronics sectors that integrate advanced material systems. Consumer applications contribute through medical device and healthcare product manufacturing.
Key market characteristics supporting Japanese medical segment dominance:
In South Korea, the market is expected to remain dominated by medical device applications, which hold a significant share in 2025. These applications are typically integrated within advanced healthcare systems where biocompatibility requirements demand specialized polyisoprene solutions. Healthcare facility procurement and medical device manufacturing each maintain substantial market presence, with growing adoption in specialized clinical applications. Consumer applications account for the remaining share, but are gradually gaining traction in healthcare product manufacturing due to safety and performance factors.
Key market factors driving South Korean medical device focus:
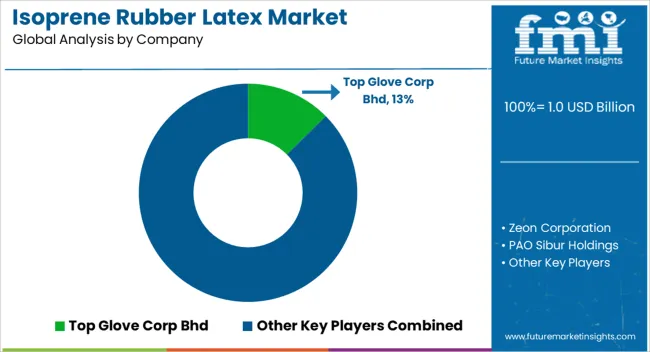
The market features approximately 10-15 meaningful players with moderate concentration, where the top three companies control roughly 35-40% of global market share through established manufacturing platforms and extensive medical industry relationships. Competition centers on biocompatibility performance, manufacturing consistency, and regulatory compliance expertise rather than price competition alone.
Market leaders include Top Glove Corp Bhd, Hartalega Holdings Bhd, and Kossan Rubber Industries Bhd, which maintain competitive advantages through comprehensive glove manufacturing portfolios, global production networks, and deep expertise in the medical protective equipment sectors, creating high switching costs for healthcare customers. These companies leverage research and development capabilities and ongoing technical support relationships to defend market positions while expanding into adjacent medical device and healthcare applications.
Challengers encompass Supermax Corp Bhd and Ansell Limited, which compete through specialized medical applications and strong regional presence in key healthcare markets. Technology specialists, including Zeon Corporation, JSR Corporation, and Kraton Corporation, focus on specific polymer formulations or vertical applications, offering differentiated capabilities in chemical synthesis, customization services, and specialized biocompatible characteristics.
Regional players and emerging chemical providers create competitive pressure through cost-effective solutions and rapid customization capabilities, particularly in high-growth markets including India and China, where local presence provides advantages in customer service and regulatory compliance. Market dynamics favor companies that combine advanced polymer technologies with comprehensive technical support offerings that address the complete product lifecycle from formulation development through ongoing clinical application optimization.
Isoprene rubber latex represents specialized biocompatible formulations that enable healthcare providers to achieve latex allergy elimination and enhanced patient safety in critical medical applications, delivering superior clinical performance with complete protein elimination compared to natural rubber latex that affects 8-17% of healthcare workers. With the market projected to grow from USD 996.7 million in 2025 to USD 1,079.4 million by 2035 at a 0.8% CAGR, these synthetic polyisoprene systems offer compelling advantages - complete allergen elimination, USP Class VI biocompatibility, and clinical-grade performance - making them essential for medical glove applications (51.6% market share), healthcare device manufacturing (57.0% medical end use), and surgical facilities seeking alternatives to natural rubber latex that compromise patient safety through allergenic protein exposure. Scaling market adoption and technological advancement requires coordinated action across healthcare policy frameworks, medical device regulations, specialty chemical manufacturers, healthcare providers, and biocompatible materials investment capital.
How Governments Could Spur Local Production and Adoption?
Healthcare Safety Integration: Include latex allergy prevention requirements in hospital safety standards, provide tax incentives for healthcare facilities using hypoallergenic polyisoprene products, and establish domestic manufacturing capabilities for medical-grade synthetic latex that reduce import dependencies in critical healthcare infrastructure. Government healthcare programs like Medicare and Medicaid could prioritize reimbursement for facilities demonstrating latex allergy prevention protocols through certified polyisoprene adoption.
Medical Device Regulatory Support: Fund research initiatives on USP Class VI compliant formulations, biocompatibility testing protocols, and next-generation medical device materials that meet FDA medical device regulations. Invest in university-industry partnerships that advance polymer chemistry and precision manufacturing techniques for healthcare applications requiring stringent biocompatibility standards.
Manufacturing Development Incentives: Provide grants for establishing polyisoprene production facilities near medical device manufacturing centers, offer technical assistance for medical-grade quality control systems, and support supply chain development that ensures reliable chemical availability for domestic healthcare manufacturers. Create specialized zones for medical device manufacturing with streamlined regulatory processes for biocompatible materials.
Standards & Quality Infrastructure: Establish national testing facilities for biocompatibility validation and USP compliance testing, develop certification programs for medical device manufacturing specialists, and create international standards harmonization that facilitates export of polyisoprene medical products to global markets. Support development of comprehensive testing laboratories meeting FDA and European MDR requirements.
Environmental & Sustainability Programs: Promote eco-friendly polyisoprene formulations through green chemistry initiatives, support recycling programs for medical device waste, and establish lifecycle assessment frameworks that validate the environmental benefits of synthetic latex alternatives over natural rubber harvesting. Create incentive programs for eco-friendly medical device manufacturing practices.
How Industry Bodies Could Support Market Development?
Performance Standards & Testing: Define standardized measurement protocols for biocompatibility performance, clinical safety validation, and regulatory compliance across surgical (powder-free) and examination glove systems, enabling reliable performance comparison and medical application-specific selection criteria. Develop comprehensive testing protocols that align with FDA medical device regulations and European MDR requirements for medical latex products.
Application Best Practices: Develop comprehensive guidelines for polyisoprene application in medical glove manufacturing (51.6% application dominance), surgical device production, and healthcare facility procurement, ensuring optimal clinical performance and patient safety consistency across different healthcare environments. Create standardized protocols for latex allergy prevention and clinical safety management.
Technology Integration Standards: Create compatibility frameworks for medical device manufacturing equipment, biocompatibility testing systems, and quality control platforms that facilitate seamless integration across different healthcare facilities and medical device production technologies. Establish interoperability standards for medical grade polyisoprene across diverse clinical applications.
Skills Development & Certification: Establish training programs for medical device technicians, quality control specialists, and clinical application engineers on advanced polyisoprene technologies, precision manufacturing techniques, and regulatory compliance methods that ensure healthcare excellence and patient safety. Create certification programs for latex allergy prevention specialists and biocompatible materials experts.
How OEMs and Technology Players Could Strengthen the Ecosystem?
Advanced Polyisoprene Formulations: Develop next-generation USP Class VI compliant systems with enhanced biocompatibility, improved clinical durability, and multi-functional properties including antimicrobial characteristics and enhanced tactile sensitivity that provide additional clinical value beyond basic latex allergy prevention. Focus on specialized formulations for surgical applications requiring maximum dexterity and barrier protection.
Precision Manufacturing Technologies: Provide automated dipping equipment with real-time quality monitoring, biocompatibility control systems, and regulatory compliance platforms that ensure consistent medical-grade production across high-volume healthcare manufacturing operations while minimizing contamination and quality defects. Develop advanced curing and processing technologies that optimize clinical performance characteristics.
Customization & Engineering Services: Offer application-specific polyisoprene development, including clinical-optimized formulations, healthcare facility-specific solutions, and performance-tailored systems that address unique requirements in surgical, examination, and medical device applications. Provide specialized consulting for regulatory compliance and biocompatibility validation.
Technical Support & Integration: Build comprehensive customer support, including clinical performance optimization, regulatory compliance assistance, and quality monitoring that help healthcare manufacturers achieve maximum patient safety benefits while maintaining consistent production quality and FDA compliance. Develop comprehensive technical documentation supporting regulatory submissions and clinical validation.
How Suppliers Could Navigate the Shift?
Application-Focused Product Development: Develop specialized polyisoprene lines for medical gloves (51.6% market dominance), healthcare devices (57.0% medical end use), and clinical applications, with formulations optimized for each sector's specific biocompatibility requirements and regulatory compliance conditions. Focus on USP Class VI compliance and FDA medical device regulations.
Geographic Market Strategy: Establish production and technical support capabilities in high-growth markets like India (12.0% CAGR) and China (8.9% CAGR), while maintaining R&D centers in established markets like Germany (3.9% CAGR) for advanced formulation development and regulatory compliance expertise. Build regional expertise in local healthcare regulations and compliance requirements.
Technology Differentiation: Invest in proprietary biocompatible polyisoprene technologies, regulatory compliance systems, and advanced quality control capabilities that provide superior clinical performance and enable premium positioning in competitive healthcare markets. Develop comprehensive intellectual property portfolios around medical-grade formulations.
Customer Partnership Models: Develop long-term relationships with medical glove manufacturers, healthcare device producers, and hospital systems through collaborative product development, clinical performance guarantees, and regulatory consulting services that strengthen customer loyalty and market positioning. Create strategic partnerships with major healthcare procurement organizations.
How Investors and Financial Enablers Could Unlock Value?
Biocompatible Technology Investment: Finance established specialty chemical companies like Zeon Corporation, JSR Corporation, and Kraton Corporation for advanced R&D programs, USP Class VI production capacity expansion, and new medical-grade formulation development that serve growing demand in healthcare and medical device markets. Support development of next-generation biocompatible polymer technologies.
Manufacturing Infrastructure Development: Provide capital for establishing regional medical-grade polyisoprene production facilities, automated healthcare manufacturing equipment development, and quality control systems that reduce costs while ensuring consistent biocompatibility performance across global healthcare operations. Finance comprehensive regulatory compliance infrastructure and testing capabilities.
Innovation & Materials Science: Back specialty chemical start-ups developing breakthrough biocompatible technologies, smart medical materials, and eco-friendly formulations that enhance clinical performance while addressing healthcare safety concerns and regulatory requirements. Support research into advanced polymer chemistry and medical device applications.
Market Integration & Expansion: Support strategic partnerships between polyisoprene suppliers and healthcare providers, finance technology transfers initiatives from research institutions, and enable market consolidation that creates comprehensive medical materials solution providers serving multiple healthcare application segments. Fund development of integrated healthcare supply chain solutions with regulatory expertise.
| Item | Value |
|---|---|
| Quantitative Units | USD 996.7 million |
| Application | Medical Gloves, Condoms, Catheters, Medical Balloons, Adhesives |
| End Use | Medical, Consumer Goods, Industrial |
| Form | Powder-free Surgical, Examination, Catheter Components, Medical Adhesive |
| Grade | Medical-grade, Industrial-grade, Consumer-grade, Specialty Applications |
| Regions Covered | Asia Pacific, North America, Europe, Latin America, Middle East & Africa |
| Country Covered | India, China, Malaysia, Spain, United States, Germany, United Kingdom, and 40+ countries |
| Key Companies Profiled | Top Glove Corp Bhd, Hartalega Holdings Bhd, Kossan Rubber Industries Bhd, Supermax Corp Bhd, Ansell Limited, Zeon Corporation, JSR Corporation, Kraton Corporation, PAO Sibur Holdings, Kuraray Co., Ltd. |
| Additional Attributes | Dollar sales by application and end use categories, regional adoption trends across Asia Pacific, Europe, and North America, competitive landscape with glove manufacturers and chemical providers, healthcare facility requirements and specifications, integration with medical safety initiatives and regulatory compliance systems, innovations in polyisoprene technology and manufacturing processes, and development of specialized formulations with biocompatibility and clinical performance capabilities. |
How big is the isoprene rubber latex market in 2025?
The global isoprene rubber latex market is estimated to be valued at USD 996.7 million in 2025.
What will be the size of isoprene rubber latex market in 2035?
The market size for the isoprene rubber latex market is projected to reach USD 1,079.4 million by 2035.
How much will be the isoprene rubber latex market growth between 2025 and 2035?
The isoprene rubber latex market is expected to grow at a 0.8% CAGR between 2025 and 2035.
What are the key product types in the isoprene rubber latex market?
The key product types in isoprene rubber latex market are medical gloves, condoms, catheters, medical balloons and adhesives.
Which end use segment to contribute significant share in the isoprene rubber latex market in 2025?
In terms of end use, medical segment to command 57.0% share in the isoprene rubber latex market in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.