Demand for Anti-counterfeit Packaging in Japan
Demand for Anti-counterfeit Packaging in Japan Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Demand for Anti-counterfeit Packaging in Japan 2026 to 2036
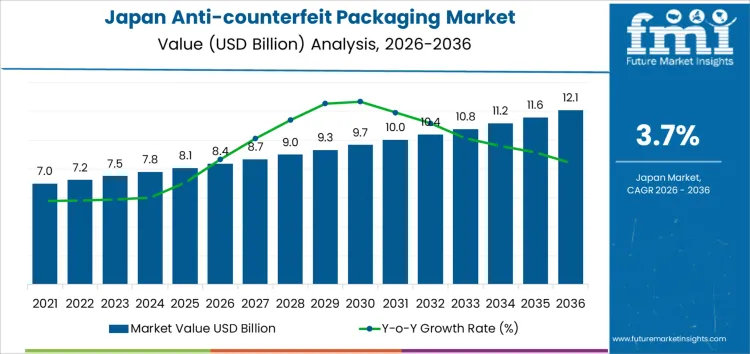
Demand for anti-counterfeit packaging in Japan is valued at USD 8.4 billion in 2026 and is projected to reach USD 12.1 billion by 2036, reflecting a CAGR of 3.7%. Demand grows as manufacturers address rising risks of counterfeit medicines, product diversion, and tampering within regulated supply chains. Strong regulatory oversight, patient safety priorities, and serialization requirements support continued adoption of authentication solutions.
Quick Stats for Demand for Anti-counterfeit Packaging in Japan
- Japan Anti-counterfeit Packaging Sales Value (2026): USD 8.4 billion
- Japan Anti-counterfeit Packaging Forecast Value (2036): USD 12.1 billion
- Japan Anti-counterfeit Packaging Forecast CAGR (2026 to 2036): 3.7%
- Leading Packaging Format in Japan: Bottles and Jars
- Leading End-Use Industry in Japan: Healthcare
- Key Growth Regions in Japan: Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku
- Top Players in Japan: SICPA, Avery Dennison, CCL Industries, DuPont, Authentix
Bottles and jars lead packaging-format usage because these formats dominate pharmaceuticals, nutraceuticals, and liquid healthcare products exposed to high counterfeiting risk. Integration of tamper-evident closures, serialized labels, covert inks, and track-and-trace identifiers strengthens protection without disrupting filling and packaging operations. Compatibility with digital verification systems further enhances implementation across production lines. Healthcare leads end-use adoption due to strict compliance requirements, high brand liability exposure, and sensitivity to patient safety outcomes. Pharmaceutical manufacturers prioritize packaging solutions that ensure traceability, authenticity verification, and regulatory alignment across domestic and export channels.
Kyushu & Okinawa, Kanto, Kansai, Chubu, and Tohoku represent key growth regions supported by pharmaceutical manufacturing clusters and distribution infrastructure. SICPA, Avery Dennison, CCL Industries, DuPont, and Authentix anchor competitive activity through secure materials, authentication technologies, and integrated traceability platforms aligned with Japan’s regulatory and quality assurance standards.
Japan Anti-counterfeit Packaging Key Takeaways
| Metric | Value |
|---|---|
| Japan Anti-counterfeit Packaging Sales Value (2026) | USD 8.4 billion |
| Japan Anti-counterfeit Packaging Forecast Value (2036) | USD 12.1 billion |
| Japan Anti-counterfeit Packaging Forecast CAGR (2026-2036) | 3.7% |
Why is the Demand for Anti-counterfeit Packaging in Japan Growing?
Demand for anti-counterfeit packaging in Japan grows due to heightened protection requirements across pharmaceuticals, consumer electronics, cosmetics, and premium food categories. Brand owners prioritize safeguards that preserve product integrity within complex domestic distribution networks. Pharmaceutical regulation emphasizes traceability and patient safety, which drives adoption of tamper evident seals, serialization, and authentication features. Growth in e commerce increases exposure to illicit replication risks, prompting manufacturers to integrate verification elements that support consumer and retailer validation. Luxury goods and cosmetics rely on packaging security to protect brand value and ensure authenticity in resale channels.
Food producers apply anti-counterfeit measures to address safety concerns and maintain trust in origin labeling. Technological capability within Japan supports deployment of QR codes, RFID, holography, and covert markers compatible with high precision packaging lines. Export oriented manufacturers adopt standardized security features to meet partner expectations across regulated destinations. Retailers and logistics providers prefer packaging that enables tracking and inspection efficiency. Government focus on intellectual property protection reinforces compliance across sectors. Investment in smart packaging platforms supports scalable implementation without disrupting existing production throughput, sustaining consistent demand across applications.
How Are the Segments Classified in the Demand for Anti-counterfeit Packaging in Japan?
Demand for anti-counterfeit packaging in Japan is shaped by strict regulatory enforcement, patient safety priorities, and high expectations for product authenticity. Buyers assess traceability features, tamper evidence, serialization compatibility, and integration with automated packaging lines. Adoption patterns reflect strong uptake in regulated industries, where packaging functions as a control mechanism supporting supply-chain integrity, brand protection, and compliance with Japanese quality and safety standards.
Which Packaging Format Accounts for the Largest Share in Japan?

Bottles and jars hold 28.4%, making them the leading packaging-format segment in Japan. These formats are widely used in pharmaceuticals, nutraceuticals, and personal care products requiring tamper evidence and authentication features. Blisters hold 23.5%, supporting unit-dose integrity and traceability in solid-dose pharmaceuticals. Vials and ampoules account for 22.6%, where protection against counterfeiting is critical for injectable medicines. Pouches and sachets hold 13.0%, while trays account for 12.5%, serving food and specialty applications. Packaging-format distribution reflects concentration of anti-counterfeit requirements in container-based healthcare packaging aligned with Japan’s dispensing and regulatory practices.
Key Points:
- Bottles and jars dominate due to pharmaceutical and personal care usage.
- Blisters support unit-dose control and traceability needs.
- Vials and ampoules require strong protection for injectables.
Which End-Use Industry Represents the Largest Share in Japan?
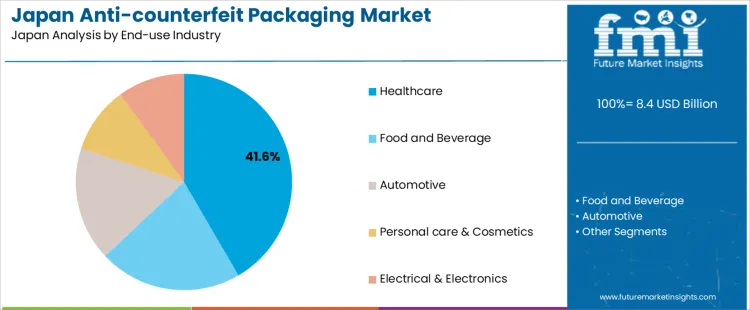
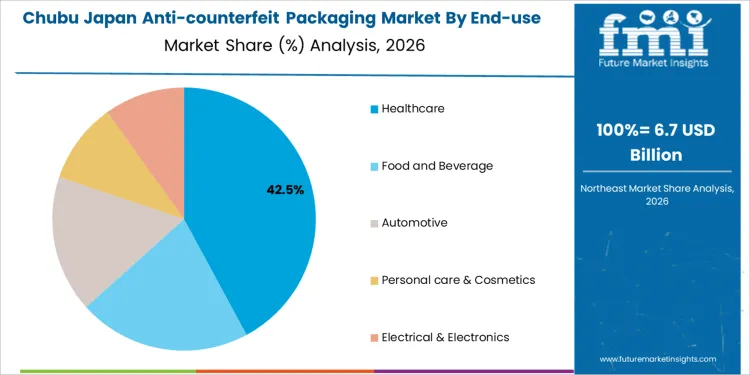
Healthcare holds 41.6%, making it the leading end-use industry segment in Japan. Pharmaceutical manufacturers apply anti-counterfeit packaging to meet serialization, traceability, and patient-safety requirements. Food and beverage accounts for 21.4%, addressing brand protection and supply-chain trust concerns. Automotive holds 17.0%, focusing on authentication of replacement parts and components. Personal care and cosmetics hold 10.0%, protecting premium brands and formulations. Electrical and electronics also hold 10.0%, targeting component authenticity and warranty protection. End-use distribution reflects strongest regulatory pressure and safety risk concentration within healthcare supply chains in Japan.
Key Points:
- Healthcare leads due to strict regulation and patient safety focus.
- Food and beverage adopt measures for brand integrity.
- Automotive and electronics address counterfeit component risks.
What are the Key Dynamics in the Anti-counterfeit Packaging Industry in Japan?
Demand increases as Japanese manufacturers and brand owners strengthen protection against counterfeiting, diversion, and parallel imports across regulated and premium product categories. Anti-counterfeit packaging supports product authenticity, consumer safety, and supply-chain integrity. Adoption aligns with strict quality expectations, traceability culture, and export-oriented manufacturing. Usage concentrates in pharmaceuticals, electronics, automotive components, cosmetics, and premium food products distributed through complex domestic and cross-border channels.
How do quality assurance standards and export-oriented manufacturing drive demand in Japan?
Japanese pharmaceutical companies implement serialization, tamper-evident features, and verification codes to ensure patient safety and distribution control. Electronics and automotive component manufacturers apply covert markers and traceability features to prevent counterfeit parts entering OEM supply chains. Cosmetic and personal care brands use authentication labels and QR-based verification to protect brand value in domestic and Asian export markets. Food producers adopt tamper-evident packaging to maintain trust and manage recall risk. Strong emphasis on manufacturing precision and zero-defect culture supports adoption of high-reliability packaging solutions integrated with enterprise systems and logistics tracking.
How do cost sensitivity, system integration complexity, and operational discipline influence demand stability?
Implementation requires investment in specialized printing, inspection, and data management infrastructure. Small and mid-sized manufacturers assess return on investment carefully due to tight cost controls. Integration with existing packaging lines and IT systems demands precise planning to avoid operational disruption. Data security and governance expectations increase system complexity. Consumer interaction with overt authentication features remains limited, affecting perceived value in some segments. Counterfeit methods evolve, requiring continuous technology updates. Demand remains stable in sectors where authenticity risk is high, while broader adoption depends on cost efficiency, seamless integration, and alignment with Japan’s disciplined manufacturing and quality management practices.
How Is Demand for Anti-counterfeit Packaging in Japan Evolving by Region?
Demand for anti-counterfeit packaging in Japan is increasing due to brand protection priorities, pharmaceutical traceability requirements, and rising scrutiny across food, beverages, and consumer goods. Kyushu and Okinawa lead with a 4.6% CAGR, supported by export-oriented manufacturing and port-linked distribution. Kanto follows at 4.2%, driven by pharmaceutical concentration, advanced packaging technologies, and compliance requirements. Kansai records a 3.7% CAGR, shaped by packaged food, cosmetics, and regional brand protection needs. Chubu posts 3.3%, reflecting manufacturing-linked adoption. Tohoku shows 2.9%, supported by regulated food and healthcare packaging. Rest of Japan records 2.7%, reflecting stable replacement-driven implementation. Regional variation reflects regulatory exposure, brand density, and supply chain complexity across Japan.
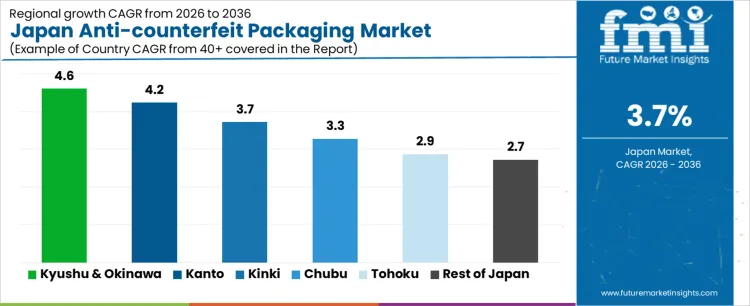
| Region | CAGR (2026 to 2036) |
|---|---|
| Kyushu & Okinawa | 4.6% |
| Kanto | 4.2% |
| Kansai | 3.7% |
| Chubu | 3.3% |
| Tohoku | 2.9% |
| Rest of Japan | 2.7% |
How is Kyushu and Okinawa driving demand for Anti-counterfeit Packaging in Japan?
Kyushu and Okinawa drive demand through export-focused manufacturing, port-based logistics, and brand-sensitive product flows. Region’s CAGR of 4.6% reflects rising use of anti-counterfeit packaging for pharmaceuticals, specialty foods, and consumer goods shipped through international channels. Manufacturers adopt tamper-evident seals, security labels, and serialized codes to protect products during transit. Port proximity increases exposure to diversion risks, reinforcing adoption of authentication features. Packaging converters supply scalable solutions compatible with high-throughput operations. Demand favors physical security features combined with basic digital verification. Growth remains risk-mitigation driven, aligned with export compliance and brand integrity protection rather than premium packaging upgrades.
- Export-oriented pharmaceutical and food packaging
- Tamper-evident and security label adoption
- Port-linked logistics risk mitigation
- Scalable solutions for high-volume operations
How is Kanto driving demand for Anti-counterfeit Packaging in Japan?
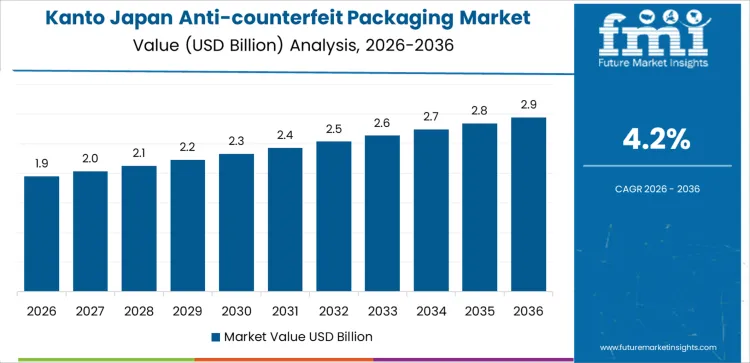
Kanto demand is shaped by pharmaceutical concentration, regulatory enforcement, and advanced packaging infrastructure. Region’s CAGR of 4.2% reflects extensive deployment of serialization, authentication codes, and track-and-trace systems. Pharmaceutical manufacturers prioritize compliance, auditability, and data integrity across packaging lines. Consumer electronics, cosmetics, and premium goods brands adopt layered security features to protect intellectual property. Packaging solutions emphasize integration with enterprise systems and supply chain visibility platforms. Demand remains compliance-led and technology-intensive, supported by strong collaboration between brand owners, packaging converters, and solution providers.
- Pharmaceutical serialization and traceability systems
- Advanced authentication and data integration
- High regulatory and audit requirements
- Strong technology and packaging ecosystem
How is Kansai driving demand for Anti-counterfeit Packaging in Japan?
Kansai demand reflects packaged food production, cosmetics manufacturing, and regional brand protection needs. Region’s CAGR of 3.7% is supported by use of tamper-evident closures, security printing, and holographic labels. Manufacturers prioritize cost efficiency, reliability, and compatibility with existing packaging equipment. Food and personal care brands adopt visible security features to maintain consumer trust. Demand remains steady, linked to routine packaging upgrades and customer-driven requirements rather than regulatory mandates. Growth is incremental and replacement-driven.
- Packaged food and cosmetics brand protection
- Tamper-evident and security printing usage
- Cost-controlled and equipment-compatible solutions
- Replacement-driven implementation patterns
How is Chubu driving demand for Anti-counterfeit Packaging in Japan?
Chubu demand is anchored in manufacturing-linked consumer goods, automotive components, and industrial products. Region’s CAGR of 3.3% reflects adoption of basic anti-counterfeit measures such as batch coding, security labels, and tamper-resistant packaging. Manufacturers emphasize operational simplicity and supply continuity. Anti-counterfeit features are often customer-mandated rather than internally driven. Demand growth remains moderate, supported by stable manufacturing output and gradual packaging modernization.
- Manufacturing-linked consumer and industrial goods
- Batch coding and basic security features
- Customer-driven adoption requirements
- Gradual packaging system upgrades
How is Tohoku driving demand for Anti-counterfeit Packaging in Japan?
Tohoku demand is influenced by regulated food production, healthcare supplies, and regional brand protection. Region’s CAGR of 2.9% reflects controlled adoption of anti-counterfeit packaging focused on compliance and safety assurance. Manufacturers favor simple, cost-effective solutions such as tamper seals and security inks. Smaller production scales limit adoption of advanced digital systems. Demand growth remains cautious and necessity-driven, aligned with regulatory expectations rather than competitive differentiation.
- Regulated food and healthcare packaging
- Preference for simple security measures
- Cost-sensitive procurement behavior
- Compliance-focused implementation
How is Rest of Japan driving demand for Anti-counterfeit Packaging in Japan?
Rest of Japan shows stable demand driven by routine packaging maintenance and replacement requirements. Region’s CAGR of 2.7% reflects continued use of basic anti-counterfeit features across local brands and small manufacturers. Buyers prioritize affordability, ease of implementation, and supplier reliability. Adoption remains limited to visible security measures rather than advanced digital authentication. Growth aligns with maintaining existing standards rather than expanding security sophistication.
- Local brand and small-scale manufacturing usage
- Replacement-driven security feature adoption
- Cost-efficient and simple packaging solutions
- Stable demand without expansion drivers
What is the competitive landscape of demand for anti-counterfeit packaging in Japan?
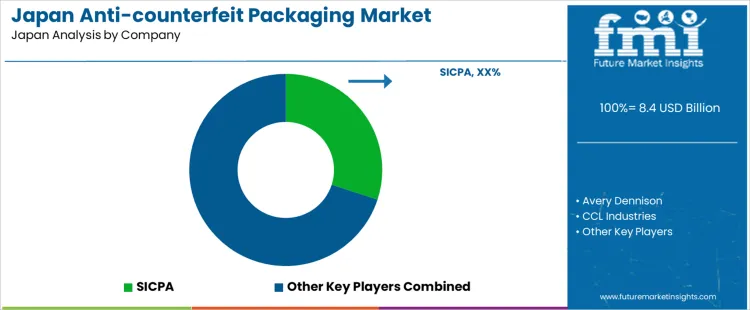
Demand for anti-counterfeit packaging in Japan is shaped by stringent brand protection requirements across pharmaceuticals, cosmetics, consumer electronics, and luxury goods. Growth in e-commerce and cross-border trade increases exposure to counterfeiting, prompting adoption of secure packaging and authentication technologies. Buyers evaluate serialization capabilities, tamper-evident features, covert markers, and integration with supply chain tracking systems. Procurement teams prioritize suppliers with Japan-based technical support, alignment with domestic regulatory expectations, and proven implementation track records. Trend in the Japan market reflects rising interest in digital track-and-trace systems, secure labeling solutions, and forensic verification tools supporting both product integrity and consumer confidence.
SICPA Japan maintains a strong position through secure ink technologies, covert tagging systems, and authentication solutions applied in pharmaceutical and regulated goods packaging across Japan. Avery Dennison operates locally with intelligent labeling and RFID-based anti-counterfeit platforms integrated into retail and industrial supply chains. CCL Industries supports Japanese brand owners with secure labels, holographic features, and tamper-evident components tailored to domestic packaging needs. DuPont Japan participates with material-based security and covert marker technologies used in high-value packaging applications. Authentix maintains visibility through digital authentication and forensic traceability solutions deployed by manufacturers addressing counterfeit risks. Competitive positioning in Japan reflects technology robustness, local service networks, regulatory compliance, and ability to integrate anti-counterfeit systems into existing packaging operations.
Key Players in Japan Anti-counterfeit Packaging Demand
- SICPA
- Avery Dennison
- CCL Industries
- DuPont
- Authentix
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Packaging Format | Bottles and Jars; Vials & Ampoules; Blisters; Trays; Pouches & Sachets |
| End-use Industry | Healthcare; Food and Beverage; Automotive; Personal Care & Cosmetics; Electrical & Electronics |
| Regions Covered | Kyushu & Okinawa; Kanto; Kansai; Chubu; Tohoku; Rest of Japan |
| Key Companies Profiled | SICPA; Avery Dennison; CCL Industries; DuPont; Authentix |
| Additional Attributes | Dollar sales by packaging format and end-use industry; regional CAGR trends led by Kanto and Kansai; rising adoption of track-and-trace, tamper-evident, and authentication technologies; demand driven by pharmaceuticals and premium consumer goods; regulatory pressure supporting serialization and brand protection solutions. |
Japan Anti-counterfeit Packaging Demand by Segments
Packaging Format:
- Bottles and Jars
- Vials & Ampoules
- Blisters
- Trays
- Pouches & Sachets
End-use Industry:
- Healthcare
- Food and Beverage
- Automotive
- Personal care & Cosmetics
- Electrical & Electronics
Region:
- Kyushu & Okinawa
- Kanto
- Kansai
- Chubu
- Tohoku
- Rest of Japan
Frequently Asked Questions
How big is the demand for anti-counterfeit packaging in Japan in 2026?
The demand for anti-counterfeit packaging in Japan is estimated to be valued at USD 8.4 billion in 2026.
What will be the size of anti-counterfeit packaging in Japan in 2036?
The market size for the anti-counterfeit packaging in Japan is projected to reach USD 12.1 billion by 2036.
How much will be the demand for anti-counterfeit packaging in Japan growth between 2026 and 2036?
The demand for anti-counterfeit packaging in Japan is expected to grow at a 3.7% CAGR between 2026 and 2036.
What are the key packaging format types in the anti-counterfeit packaging in Japan?
The key packaging format types in anti-counterfeit packaging in Japan are bottles and jars, vials & ampoules, blisters, trays and pouches & sachets.
Which end-use industry segment is expected to contribute significant share in the anti-counterfeit packaging in Japan in 2026?
In terms of end-use industry, healthcare segment is expected to command 41.6% share in the anti-counterfeit packaging in Japan in 2026.
Table of Content
- Executive Summary
- Japan Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Japan Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Japan Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Japan Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging Format , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging Format , 2026 to 2036
- Bottles and Jars
- Vials & Ampoules
- Blisters
- Trays
- Pouches & Sachets
- Bottles and Jars
- Y to o to Y Growth Trend Analysis By Packaging Format , 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging Format , 2026 to 2036
- Japan Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-use Industry
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-use Industry, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-use Industry, 2026 to 2036
- Healthcare
- Food and Beverage
- Automotive
- Personal care & Cosmetics
- Electrical & Electronics
- Healthcare
- Y to o to Y Growth Trend Analysis By End-use Industry, 2021 to 2025
- Absolute $ Opportunity Analysis By End-use Industry, 2026 to 2036
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Packaging Format
- By End-use Industry
- Competition Analysis
- Competition Deep Dive
- SICPA
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Avery Dennison
- CCL Industries
- DuPont
- Authentix
- SICPA
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Japan Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Japan Market Value (USD Million) Forecast by Packaging Format , 2021 to 2036
- Table 3: Japan Market Value (USD Million) Forecast by End-use Industry, 2021 to 2036
- Table 4: Japan Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: Japan Market Value (USD Million) Forecast by Packaging Format , 2021 to 2036
- Table 6: Japan Market Value (USD Million) Forecast by End-use Industry, 2021 to 2036
List of Figures
- Figure 1: Japan Market Pricing Analysis
- Figure 2: Japan Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Japan Market Value Share and BPS Analysis by Packaging Format , 2026 to 2036
- Figure 4: Japan Market Y-o-Y Growth Comparison by Packaging Format , 2026 to 2036
- Figure 5: Japan Market Attractiveness Analysis by Packaging Format
- Figure 6: Japan Market Value Share and BPS Analysis by End-use Industry, 2026 to 2036
- Figure 7: Japan Market Y-o-Y Growth Comparison by End-use Industry, 2026 to 2036
- Figure 8: Japan Market Attractiveness Analysis by End-use Industry
- Figure 9: Japan Market Value (USD Million) Share and BPS Analysis by Region, 2026 to 2036
- Figure 10: Japan Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Japan Market Attractiveness Analysis by Region
- Figure 12: Japan Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Japan Market Value Share and BPS Analysis by Country, 2026 to 2036
- Figure 14: Japan Market Value Share and BPS Analysis by Packaging Format , 2026 to 2036
- Figure 15: Japan Market Y-o-Y Growth Comparison by Packaging Format , 2026 to 2036
- Figure 16: Japan Market Attractiveness Analysis by Packaging Format
- Figure 17: Japan Market Value Share and BPS Analysis by End-use Industry, 2026 to 2036
- Figure 18: Japan Market Y-o-Y Growth Comparison by End-use Industry, 2026 to 2036
- Figure 19: Japan Market Attractiveness Analysis by End-use Industry
- Figure 20: Japan Market - Tier Structure Analysis
- Figure 21: Japan Market - Company Share Analysis

