The global pharma moisture barrier film coating market is projected to reach USD 1,309.5 million by 2035, recording an absolute increase of USD 416.3 million over the forecast period. The pharma moisture barrier film coating market is valued at USD 893.2 million in 2025 and is set to rise at a CAGR of 3.9% during the assessment period. The pharma moisture barrier film coating market size is expected to grow by nearly 1.5 times during the same period, supported by increasing demand for pharmaceutical stability enhancement solutions worldwide, driving demand for efficient moisture protection systems and increasing investments in drug formulation quality improvement projects globally.
The pharmaceutical industry's focus on extending shelf life and maintaining drug efficacy through environmental protection creates sustained demand for advanced barrier coating technologies. Growing adoption of modified-release dosage forms and rising pharmaceutical manufacturing activities in emerging markets further support market expansion. Stringent regulatory requirements and raw material price fluctuations may pose challenges to market expansion. The pharma moisture barrier film coating market benefits from pharmaceutical companies' commitment to quality assurance and patient safety standards that necessitate reliable moisture barrier solutions across diverse therapeutic applications and dosage forms.
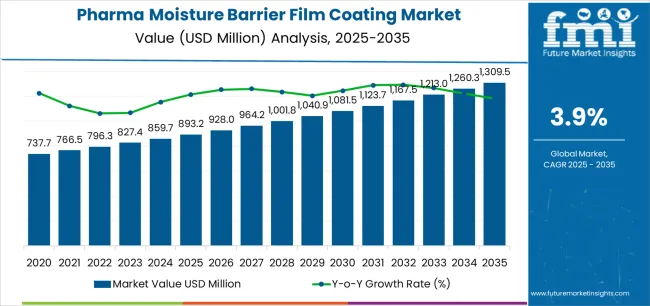
Between 2025 and 2030, the pharma moisture barrier film coating market is projected to expand from USD 893.2 million to USD 1,081.5 million, resulting in a value increase of USD 188.3 million, which represents 45.2% of the total forecast growth for the decade. This phase of development will be shaped by rising demand for pharmaceutical stability solutions and shelf life extension technologies, product innovation in polymer formulations and coating application systems, as well as expanding integration with pharmaceutical quality management and regulatory compliance programs. Companies are establishing competitive positions through investment in advanced coating technologies, pharmaceutical-grade material sourcing, and strategic market expansion across solid dosage, tablet coating, and specialty pharmaceutical applications.
From 2030 to 2035, the pharma moisture barrier film coating market is forecast to grow from USD 1,081.5 million to USD 1,309.5 million, adding another USD 228.0 million, which constitutes 54.8% of the ten-year expansion. This period is expected to be characterized by the expansion of specialized coating systems, including advanced enteric formulations and targeted gastric dissolving solutions tailored for specific drug delivery requirements, strategic collaborations between coating material manufacturers and pharmaceutical companies, and an enhanced focus on moisture protection efficiency and formulation stability. The growing emphasis on pharmaceutical quality enhancement and patient compliance improvement will drive demand for advanced, high-performance barrier coating solutions across diverse therapeutic applications.
| Metric | Value |
|---|---|
| Market Value (2025) | USD 893.2 million |
| Market Forecast Value (2035) | USD 1,309.5 million |
| Forecast CAGR (2025-2035) | 3.9% |
The pharma moisture barrier film coating market grows by enabling pharmaceutical manufacturers to achieve superior drug stability and extended shelf life while meeting regulatory standards for product quality. Pharmaceutical producers face mounting pressure to ensure medication efficacy throughout distribution chains, with moisture barrier coatings typically providing 40-60% improvement in hygroscopic drug protection over uncoated formulations, making barrier technologies essential for pharmaceutical quality assurance. The pharmaceutical industry's need for reliable environmental protection creates demand for advanced coating solutions that can prevent moisture ingress, maintain active ingredient stability, and ensure consistent drug performance across diverse storage conditions.
Regulatory requirements enforcing pharmaceutical stability standards drive adoption in solid dosage manufacturing, enteric applications, and specialty formulations, where moisture control has a direct impact on drug efficacy and patient safety. The global expansion of pharmaceutical manufacturing in emerging markets and increasing focus on generic drug quality accelerates barrier coating demand as manufacturers seek solutions to maintain product integrity during extended distribution. The rising prevalence of hygroscopic active pharmaceutical ingredients requiring environmental protection and growing pharmaceutical export activities necessitating tropical climate stability further support market growth. Complex regulatory approval processes for coating materials and variability in pharmaceutical manufacturing standards across regions may limit adoption rates among smaller pharmaceutical companies and contract manufacturing organizations with limited technical capabilities.
The pharma moisture barrier film coating market is segmented by polymer type, application, and region. By polymer type, the pharma moisture barrier film coating market is divided into PVA type, HPMC type, and other polymer formulations. Based on application, the pharma moisture barrier film coating market is categorized into gastric dissolving solid dosage and enteric dissolving solid dosage. Regionally, the pharma moisture barrier film coating market is divided into Asia Pacific, Europe, North America, Latin America, and the Middle East & Africa.
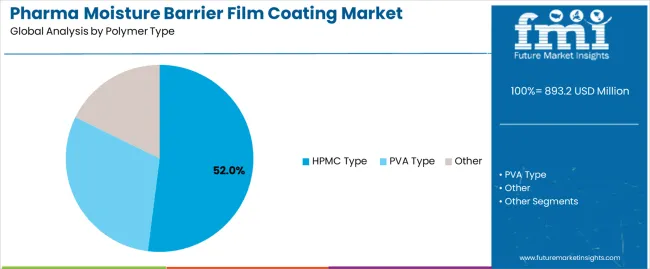
The HPMC type segment represents the dominant force in the pharma moisture barrier film coating market, capturing approximately 52.0% of total market share in 2025. This advanced category encompasses formulations featuring hydroxypropyl methylcellulose polymer systems that deliver comprehensive moisture protection with enhanced pharmaceutical compatibility and regulatory acceptance. The HPMC segment's market leadership stems from its exceptional film-forming properties, broad regulatory approval across major pharmaceutical markets, and compatibility with diverse active pharmaceutical ingredients requiring environmental protection.
The PVA type segment maintains a substantial 31.0% market share, serving pharmaceutical manufacturers who require specialized coating solutions through polyvinyl alcohol-based formulations. The other polymer category accounts for 17.0% market share, featuring alternative polymer systems including specialized cellulose derivatives and novel coating materials.
Key advantages driving the HPMC type segment include:
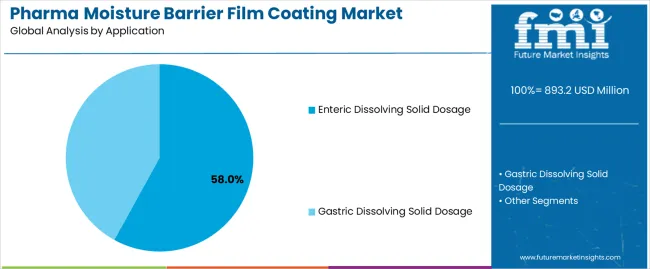
Enteric dissolving solid dosage applications dominate the pharma moisture barrier film coating market with approximately 58.0% market share in 2025, reflecting the critical importance of targeted drug delivery systems requiring gastric acid resistance combined with moisture protection. The enteric segment's market leadership is reinforced by widespread adoption in pharmaceutical formulations designed for intestinal absorption, where moisture barrier properties ensure drug stability while maintaining precise pH-dependent release characteristics.
The gastric dissolving solid dosage segment represents 42.0% market share through immediate-release formulations requiring moisture protection without enteric functionality. This segment benefits from broad application across standard pharmaceutical tablets where environmental stability enhancement is prioritized.
Key market dynamics supporting application preferences include:
The pharma moisture barrier film coating market is driven by three concrete demand factors tied to pharmaceutical quality and regulatory compliance. First, pharmaceutical stability requirements create increasing demand for moisture barrier solutions, with global pharmaceutical production growing 5-7% annually in major markets worldwide, requiring reliable coating systems for shelf life assurance and regulatory compliance maintenance. Second, hygroscopic drug formulations and climate challenges drive manufacturers toward protective coating technologies, with barrier films reducing moisture-related degradation by 50-70% annually while ensuring product efficacy throughout distribution chains spanning tropical and humid environments. Third, regulatory enforcement of pharmaceutical quality standards through good manufacturing practices, stability testing requirements, and pharmacopeial compliance accelerates adoption across North America, Europe, and Asia-Pacific regions where pharmaceutical production receives rigorous oversight.
Market restraints include raw material sourcing complexities affecting polymer availability and coating material consistency, particularly for pharmaceutical-grade HPMC where quality specifications vary significantly by supplier and manufacturing process. Regulatory approval timelines of 12-24 months for new coating formulations pose adoption barriers for pharmaceutical companies seeking rapid product launches and contract manufacturers serving multiple clients with diverse formulation requirements. Technical expertise gaps regarding coating optimization create additional challenges, as pharmaceutical manufacturers require specialized knowledge in coating thickness control, plasticizer selection, and process parameter optimization to achieve target moisture barrier performance.
Key trends indicate accelerated adoption in Asia-Pacific pharmaceutical manufacturing hubs, particularly China and India, where expanding pharmaceutical production and export-oriented manufacturing incentivize quality enhancement investments through coating technology deployment. Technology advancement trends toward multilayer coating systems with enhanced barrier properties, modified-release formulations combining moisture protection with controlled drug delivery, and sustainable coating materials addressing environmental concerns are enabling next-generation product development. The pharma moisture barrier film coating market thesis could face disruption if pharmaceutical packaging innovations or desiccant technologies produce cost-effective environmental protection alternatives that reduce reliance on moisture barrier coatings while maintaining comparable drug stability outcomes.
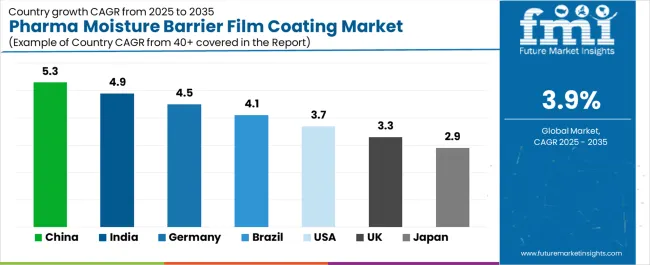
| Country | CAGR (2025-2035) |
|---|---|
| China | 5.3% |
| India | 4.9% |
| Germany | 4.5% |
| Brazil | 4.1% |
| USA | 3.7% |
| UK | 3.3% |
| Japan | 2.9% |
The pharma moisture barrier film coating market is gaining momentum worldwide, with China taking the lead thanks to aggressive pharmaceutical manufacturing expansion and generic drug production intensification programs. Close behind, India benefits from pharmaceutical export growth and contract manufacturing development, positioning itself as a strategic growth hub in the Asia-Pacific region. Germany shows strong advancement, where established pharmaceutical manufacturing infrastructure and quality standards strengthen its role in European pharmaceutical supply chains. Brazil demonstrates robust growth through expanding domestic pharmaceutical production and regulatory modernization programs, signaling continued investment in pharmaceutical quality infrastructure. Meanwhile, the USA maintains steady development through pharmaceutical innovation and regulatory compliance emphasis, while the UK and Japan continue to record consistent progress driven by pharmaceutical manufacturing excellence and quality assurance standards. Together, China and India anchor the global expansion story, while established markets build stability and consistency into the pharma moisture barrier film coating market's growth path.
The report covers an in-depth analysis of 40 countries, with top-performing countries highlighted below.
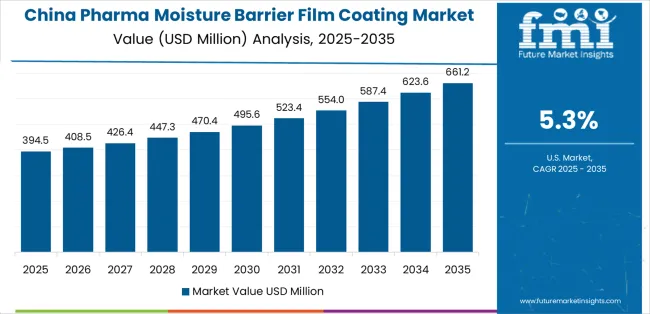
China demonstrates the strongest growth potential in the Pharma Moisture Barrier Film Coating Market with a CAGR of 5.3% through 2035. The country's leadership position stems from comprehensive pharmaceutical manufacturing capacity expansion, intensive generic drug production programs, and aggressive pharmaceutical quality improvement targets driving adoption of advanced coating technologies. Growth is concentrated in major pharmaceutical manufacturing regions, including Jiangsu, Zhejiang, Shandong, and Guangdong, where tablet manufacturers and pharmaceutical companies are implementing moisture barrier coating programs for domestic supply and international export competitiveness. Distribution channels through pharmaceutical ingredient suppliers, coating material distributors, and direct manufacturer relationships expand deployment across pharmaceutical production clusters and contract manufacturing facilities. The country's pharmaceutical quality initiatives provide policy support for coating technology adoption, including incentives for pharmaceutical manufacturing upgrades and quality system improvements.
Key market factors:
In pharmaceutical manufacturing hubs across Maharashtra, Gujarat, Andhra Pradesh, and Telangana, the adoption of moisture barrier coating systems is accelerating across generic drug manufacturers, contract pharmaceutical organizations, and specialty formulation companies, driven by international export requirements and domestic pharmaceutical quality initiatives. The pharma moisture barrier film coating market demonstrates strong growth momentum with a CAGR of 4.9% through 2035, linked to comprehensive pharmaceutical manufacturing expansion and increasing focus on pharmaceutical stability enhancement solutions. Indian manufacturers are implementing advanced coating programs and quality assurance platforms to improve drug stability while meeting stringent regulatory standards in key export markets including USA, Europe, and emerging pharmaceutical markets. The country's pharmaceutical export growth creates sustained demand for coating technologies, while increasing emphasis on pharmaceutical quality assurance drives adoption of barrier solutions that enhance shelf life stability.
Advanced pharmaceutical sector in Germany demonstrates sophisticated implementation of moisture barrier coating systems, with documented case studies showing 30-40% improvement in pharmaceutical stability through optimized coating technologies. The country's pharmaceutical infrastructure in major manufacturing regions, including North Rhine-Westphalia, Bavaria, Baden-Württemberg, and Hesse, showcases integration of advanced coating technologies with existing pharmaceutical manufacturing systems, leveraging expertise in pharmaceutical engineering and quality assurance protocols. German manufacturers emphasize regulatory compliance and pharmaceutical excellence, creating demand for reliable coating solutions that support medication stability commitments and international market approval requirements. The pharma moisture barrier film coating market maintains strong growth through focus on pharmaceutical innovation and regulatory adherence, with a CAGR of 4.5% through 2035.
Key development areas:
The Brazilian pharmaceutical market leads in coating technology implementation based on integration with pharmaceutical manufacturing expansion and domestic drug production growth programs. The country shows solid potential with a CAGR of 4.1% through 2035, driven by pharmaceutical manufacturing capacity expansion and increasing pharmaceutical quality standards across major production regions, including São Paulo, Rio de Janeiro, Minas Gerais, and Paraná. Brazilian manufacturers are adopting moisture barrier coatings for compliance with pharmaceutical stability requirements, particularly in generic drug manufacturing requiring shelf life assurance and in tropical climate markets demanding enhanced environmental protection. Technology deployment channels through pharmaceutical ingredient distributors, coating equipment suppliers, and pharmaceutical manufacturing service relationships expand coverage across diverse drug manufacturing operations.
Leading market segments:
The USA pharmaceutical market demonstrates sophisticated implementation focused on regulatory compliance and pharmaceutical quality assurance, with documented integration of coating programs achieving FDA stability requirements in commercial drug manufacturing. The country maintains steady growth momentum with a CAGR of 3.7% through 2035, driven by pharmaceutical regulatory standards and manufacturers' emphasis on drug quality assurance across major pharmaceutical production regions, including New Jersey, North Carolina, Indiana, and Puerto Rico. Major pharmaceutical companies showcase advanced deployment of coating systems that integrate seamlessly with existing pharmaceutical manufacturing infrastructure and quality management programs.
Key market characteristics:
In pharmaceutical manufacturing regions across Southeast England, Northwest England, and Scotland, pharmaceutical companies are implementing moisture barrier coating programs to enhance drug stability and meet regulatory requirements, with documented case studies showing sustained stability improvements in commercial pharmaceutical products. The pharma moisture barrier film coating market shows solid growth potential with a CAGR of 3.3% through 2035, linked to pharmaceutical manufacturing standards, regulatory compliance requirements, and increasing emphasis on pharmaceutical quality assurance programs. British manufacturers are adopting advanced coating formulations and stability testing protocols to maintain pharmaceutical standards while complying with MHRA requirements and international pharmaceutical regulations.
Market development factors:
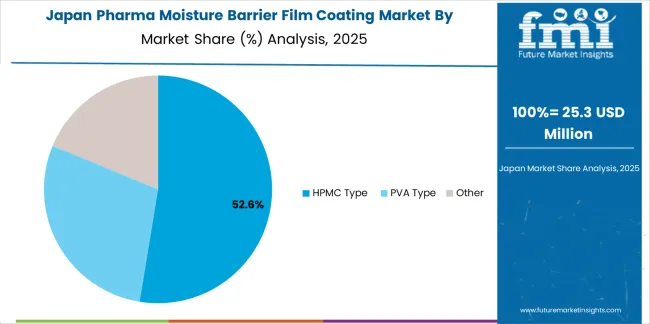
Pharma moisture barrier film coating market in Japan demonstrates sophisticated implementation focused on pharmaceutical manufacturing excellence and quality assurance protocols, with documented integration of coating programs achieving PMDA regulatory standards in commercial pharmaceutical production. The country maintains steady growth momentum with a CAGR of 2.9% through 2035, driven by pharmaceutical quality emphasis and manufacturers' commitment to medication stability across major pharmaceutical production regions, including Tokyo, Osaka, and Shizuoka. Japanese pharmaceutical companies showcase advanced coating technology deployment that integrates seamlessly with existing pharmaceutical manufacturing systems and comprehensive quality control programs.
Key market characteristics:
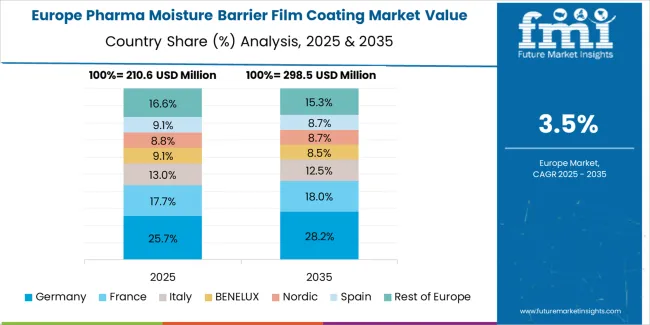
The pharma moisture barrier film coating market in Europe is projected to grow from USD 267.5 million in 2025 to USD 398.3 million by 2035, registering a CAGR of 4.1% over the forecast period. Germany is expected to maintain its leadership position with a 28.5% market share in 2025, declining slightly to 27.8% by 2035, supported by its advanced pharmaceutical manufacturing infrastructure and major production centers, including North Rhine-Westphalia, Bavaria, and Baden-Württemberg regions.
France follows with a 19.2% share in 2025, projected to reach 19.5% by 2035, driven by comprehensive pharmaceutical quality programs and coating technology adoption in pharmaceutical manufacturing facilities. The United Kingdom holds a 16.8% share in 2025, expected to decrease to 16.2% by 2035 reflecting pharmaceutical manufacturing consolidation. Italy commands a 13.5% share in both 2025 and 2035, backed by established pharmaceutical production capabilities. Spain accounts for 9.7% in 2025, rising to 10.1% by 2035 on pharmaceutical manufacturing expansion. Switzerland maintains 6.3% in 2025, reaching 6.5% by 2035 on pharmaceutical innovation focus. The Rest of Europe region is anticipated to hold 6.0% in 2025, expanding to 6.4% by 2035, attributed to increasing coating technology adoption in Nordic countries and emerging Central European pharmaceutical manufacturing programs.
The Japanese pharma moisture barrier film coating market demonstrates a mature and quality-focused landscape, characterized by sophisticated integration of HPMC type and pharmaceutical-grade coating systems with existing pharmaceutical manufacturing infrastructure across generic drug production, specialty pharmaceutical operations, and traditional pharmaceutical companies. Japan's emphasis on pharmaceutical quality and regulatory compliance drives demand for certified coating materials that support medication stability commitments and patient safety expectations in domestic pharmaceutical supply chains. The pharma moisture barrier film coating market benefits from strong partnerships between international coating material providers and domestic pharmaceutical manufacturers, creating comprehensive service ecosystems that prioritize material consistency and technical support programs. Pharmaceutical manufacturing centers in Tokyo, Osaka, Shizuoka, and other major production areas showcase advanced pharmaceutical implementations where coating programs achieve pharmaceutical stability requirements through validated manufacturing processes and quality assurance systems.
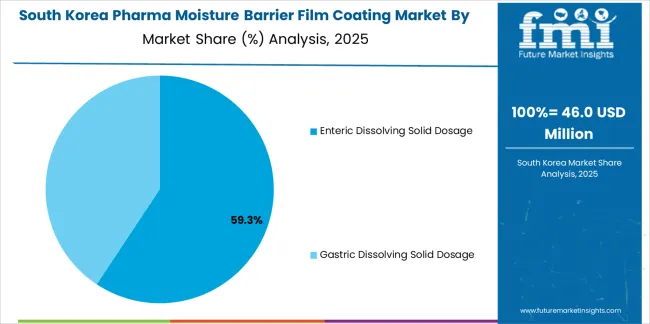
The South Korean pharma moisture barrier film coating market is characterized by growing international technology provider presence, with companies maintaining significant positions through comprehensive pharmaceutical support and technical services capabilities for pharmaceutical manufacturing and drug formulation applications. The pharma moisture barrier film coating market is seeing increased emphasis on pharmaceutical quality standards and international pharmaceutical compliance, as Korean manufacturers increasingly demand certified coating materials that integrate with domestic pharmaceutical infrastructure and sophisticated quality control systems deployed across major pharmaceutical complexes. Regional coating material manufacturers are gaining market share through strategic partnerships with international suppliers, offering specialized services including Korean pharmaceutical regulatory support and formulation-specific coating programs for pharmaceutical operations. The competitive landscape shows increasing collaboration between multinational coating material companies and Korean pharmaceutical technology specialists, creating hybrid service models that combine international material sourcing expertise with local pharmaceutical knowledge and regulatory compliance systems.
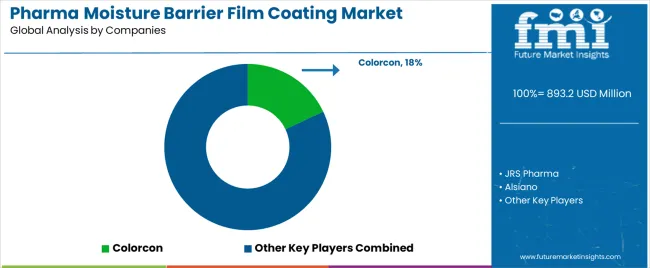
The pharma moisture barrier film coating market features approximately 20-25 meaningful players with moderate concentration, where the top three companies control roughly 35-40% of global market share through established pharmaceutical relationships and comprehensive coating material portfolios. Competition centers on regulatory compliance capability, material quality consistency, and pharmaceutical manufacturing support rather than price competition alone. Colorcon leads with approximately 18.0% market share through its comprehensive pharmaceutical coating solutions portfolio and global pharmaceutical industry presence.
Market leaders include Colorcon, JRS Pharma, and Alsiano, which maintain competitive advantages through global pharmaceutical distribution infrastructure, regulatory expertise across multiple jurisdictions, and deep pharmaceutical manufacturing knowledge, creating trust and reliability advantages with pharmaceutical operations worldwide. These companies leverage research capabilities in coating formulation optimization and ongoing technical support relationships to defend market positions while expanding into specialty pharmaceutical and modified-release applications.
Challengers encompass Ideal Cures and Huzhou Zhanwang Pharmaceutical, which compete through specialized coating formulations and strong regional presence in key pharmaceutical manufacturing markets. Product specialists, including Jiangsu Xinyu Pharmaceutical, Zhejiang Oulun Coating Technology, and Tianjing Aileyi, focus on specific polymer systems or pharmaceutical applications, offering differentiated capabilities in coating technologies, customization services, and specialized pharmaceutical-grade materials.
Pharma moisture barrier film coatings represent advanced pharmaceutical materials that enable drug manufacturers to achieve 40-60% improvement in hygroscopic drug protection compared to uncoated formulations, delivering superior stability and extended shelf life with enhanced environmental resistance in demanding pharmaceutical applications. With the pharma moisture barrier film coating market projected to grow from USD 893.2 million in 2025 to USD 1,309.5 million by 2035 at a 3.9% CAGR, these coating systems offer compelling advantages - drug stability enhancement, shelf life extension, and regulatory compliance support - making them essential for enteric dissolving solid dosage (58.0% market share), gastric dissolving solid dosage (42.0% share), and pharmaceutical operations seeking alternatives to unprotected formulations that compromise drug efficacy through moisture exposure. Scaling market adoption and pharmaceutical integration requires coordinated action across pharmaceutical regulation, coating material standards, pharmaceutical manufacturers, coating technology providers, and pharmaceutical quality investment capital.
| Item | Value |
|---|---|
| Quantitative Units | USD 893.2 million |
| Polymer Type | PVA Type, HPMC Type, Other |
| Application | Gastric Dissolving Solid Dosage, Enteric Dissolving Solid Dosage |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Country Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40 countries |
| Key Companies Profiled | Colorcon, JRS Pharma, Alsiano, Ideal Cures, Huzhou Zhanwang Pharmaceutical, Jiangsu Xinyu Pharmaceutical, Zhejiang Oulun Coating Technology, Tianjing Aileyi |
| Additional Attributes | Dollar sales by polymer type and application categories, regional adoption trends across Asia Pacific, Europe, and North America, competitive landscape with coating material manufacturers and pharmaceutical distribution networks, pharmaceutical manufacturing integration requirements and specifications, integration with pharmaceutical quality programs and regulatory compliance initiatives, innovations in coating technology and pharmaceutical formulation systems, and development of specialized coating materials with enhanced moisture barrier capabilities and pharmaceutical compatibility. |
How big is the pharma moisture barrier film coating market in 2025?
The global pharma moisture barrier film coating market is estimated to be valued at USD 893.2 million in 2025.
What will be the size of pharma moisture barrier film coating market in 2035?
The market size for the pharma moisture barrier film coating market is projected to reach USD 1,309.5 million by 2035.
How much will be the pharma moisture barrier film coating market growth between 2025 and 2035?
The pharma moisture barrier film coating market is expected to grow at a 3.9% CAGR between 2025 and 2035.
What are the key product types in the pharma moisture barrier film coating market?
The key product types in pharma moisture barrier film coating market are hpmc type , pva type and other.
Which application segment to contribute significant share in the pharma moisture barrier film coating market in 2025?
In terms of application, enteric dissolving solid dosage segment to command 58.0% share in the pharma moisture barrier film coating market in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.