High Barrier Pharmaceutical Packaging Films for Blister Market
This report covers the high barrier pharmaceutical packaging films for blister market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, industry trends, supply chain developments, and strategic growth opportunities.
High Barrier Pharmaceutical Packaging Films for Blister Market Size, Market Forecast and Outlook By FMI
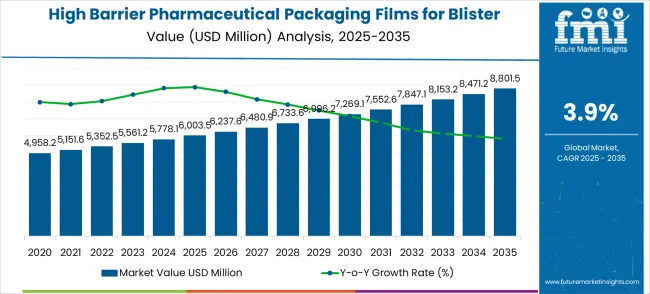
In 2025, the high barrier pharmaceutical packaging films for blister market was valued at USD 6.00 billion. Based on Future Market Insights' analysis, demand is estimated to grow to USD 6.24 billion in 2026 and USD 9.14 billion by 2036. FMI projects a CAGR of 3.90% during the forecast period.
The market is set to add approximately USD 2.91 billion in absolute terms between 2026 and 2036. Pharmaceutical stability requirements for moisture-sensitive and photosensitive drug formulations are the primary demand driver, with regulatory agencies (FDA, EMA, PMDA) mandating ICH Q1A stability data demonstrating dosage form integrity through expiration dating that only high-barrier blister films can support. Cold-form aluminum blister adoption for biologics-adjacent oral formulations and tropical climate distribution is expanding the premium segment.
Summary of High Barrier Pharmaceutical Packaging Films for Blister Market
- High Barrier Pharmaceutical Packaging Films for Blister Market Definition
- High barrier pharmaceutical packaging films for blister applications are multi-layer polymer and aluminum laminate films engineered to provide moisture vapor transmission rate (MVTR) and oxygen transmission rate (OTR) barriers protecting hygroscopic and oxidation-sensitive tablet and capsule dosage forms in thermoformed blister cavity and push-through lidding configurations.
- Demand Drivers in the Market
- ICH Q1A stability testing requirements mandate pharmaceutical manufacturers to demonstrate drug product stability through labeled shelf life under Zone IVb climatic conditions (30C/75% RH), driving procurement of PVDC-coated and cold-form aluminum films that deliver MVTR below 0.25 g/m2/day.
- Generic pharmaceutical production growth in India and China, with over 60% of global generic solid oral dosage form volume, sustains high-volume blister film procurement as thermoformed PVC/PVDC remains the default primary packaging format for cost-sensitive generic tablet products.
- Cold-form aluminum blister adoption for humidity-sensitive biologics-adjacent oral formulations (enzyme replacement therapies, targeted oral peptides) is creating a premium packaging segment requiring OTR below 0.001 cc/m2/day.
- Key Segments Analyzed in the FMI Report
- PVC Film material type: 48.0% share in 2026, reflecting its structural position as the primary category across established procurement channels.
- Tablets application: 58.0% share in 2026, reflecting its structural position as the primary category across established procurement channels.
- China: 5.30% compound growth through 2036, indicating concentrated demand acceleration.
- Analyst Opinion at FMI
- Ismail Sutaria Principal Consultant for Healthcare observe that The high barrier pharmaceutical blister film market is a specification-driven packaging category where moisture and oxygen barrier performance determines drug product shelf life compliance. FMI analysts observe that PVC film commands 48.0% material share because standard PVC/PVDC thermoformed blisters provide the cost-performance balance required for the vast majority of solid oral dosage forms where moderate barrier protection is sufficient. Tablet application at 58.0% share dominates because tablets represent over 70% of global solid oral dosage form production volume. China leads at 5.3% CAGR because domestic pharmaceutical packaging film manufacturers are scaling PVDC coating and cold-form aluminum lamination capacity to serve the country's expanding generic drug production base.
- Strategic Implications / Executive Takeaways
- Blister film converters must maintain PVDC coating weight consistency within pharmaceutical customer specifications (typically 40 to 90 gsm) to ensure barrier performance meets ICH stability requirements across production lot variability.
- Pharmaceutical packaging engineers should evaluate mono-PP high-barrier blister films as PVC-free alternatives to address EU PPWR recyclability requirements while maintaining the MVTR performance needed for Zone IVb stability compliance.
- Cold-form aluminum blister film suppliers should target the growing oral biologics and peptide therapeutic segment where ultra-high barrier performance justifies the 3x to 5x cost premium over standard thermoformed PVC/PVDC packaging.
High Barrier Pharmaceutical Packaging Films for Blister Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 6.24 Billion |
| Industry Value (2036) | USD 9.14 Billion |
| CAGR (2026 to 2036) | 3.90% |
Source: Future Market Insights, 2026
As per FMI, country-level growth rates through 2036 are projected as follows: China at 5.30%, India at 4.90%, Germany at 4.50%, Brazil at 4.10%, USA at 3.70%, UK at 3.30%, Japan at 2.90%. China records the fastest expansion driven by concentrated industry investment.
High Barrier Pharmaceutical Packaging Films for Blister Market Definition
The high barrier pharmaceutical packaging films for blister market encompasses multi-layer barrier film structures engineered for thermoformed and cold-formed blister packaging of solid oral dosage forms. Material categories span PVC base films, PVDC-coated barrier films, polypropylene films, and aluminum cold-form laminates, each delivering different moisture and oxygen barrier performance levels matched to drug product stability requirements. As per FMI, the market is driven by pharmaceutical stability compliance mandates under ICH guidelines, with generic drug production expansion in Asia Pacific generating the largest volume demand while cold-form aluminum blisters capture premium pricing in biologics-adjacent oral formulation packaging.
High Barrier Pharmaceutical Packaging Films for Blister Market Inclusions
Market scope covers PVC, PVDC-coated, polypropylene, and aluminum cold-form blister films used in thermoformed and cold-formed pharmaceutical blister packaging for tablet, capsule, and unit-dose solid oral dosage form applications.
High Barrier Pharmaceutical Packaging Films for Blister Market Exclusions
Standard PVC films without barrier coating or lamination, strip packaging films, bottle and vial packaging, and parenteral (injectable) packaging materials are excluded.
High Barrier Pharmaceutical Packaging Films for Blister Market Research Methodology
- Primary Research: Analysts conducted structured interviews with procurement managers, product engineers, and distribution channel operators to map purchase decision triggers and specification requirements across key verticals.
- Desk Research: Data collection aggregated regulatory filings, industry standards documentation, patent registries, trade body publications, and company annual reports across all target geographies.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of product shipment volumes and average selling prices, validated against publicly disclosed revenue figures from leading suppliers.
- Data Validation and Update Cycle: Projections are cross-referenced against quarterly earnings data, import-export trade statistics, and industry association production reports to maintain forecast integrity.
Why is the High Barrier Pharmaceutical Packaging Films for Blister Market Growing?
Market expansion is being supported by the rapid advancement of pharmaceutical manufacturing across developing economies and the corresponding need for protective packaging systems for drug stability and shelf life extension in diverse environmental conditions. Modern pharmaceutical operations require superior moisture and oxygen barrier properties to ensure optimal drug efficacy and regulatory compliance. The exceptional barrier performance and chemical resistance characteristics of high barrier pharmaceutical packaging films make them essential components in demanding pharmaceutical environments where product integrity is critical.
The growing emphasis on drug safety and regulatory compliance is driving demand for advanced packaging film technologies from certified manufacturers with proven track records of quality and regulatory approval. Pharmaceutical operators are increasingly investing in high-performance barrier systems that offer enhanced protection and extended shelf life over traditional packaging solutions. Regulatory standards and quality requirements are establishing performance benchmarks that favor precision-engineered high barrier packaging film solutions with advanced protective capabilities.
Segmental Analysis
The market is segmented by material type, application, and region. By material type, the market is divided into PVC film, PVDC coated film, PP film, aluminum film, and other configurations. Based on application, the market is categorized into tablets, capsules, and other applications. Regionally, the market is divided into North America, Europe, East Asia, South Asia & Pacific, Latin America, and Middle East & Africa.
By Material Type, PVC Film Segment Accounts for 47% Market Share
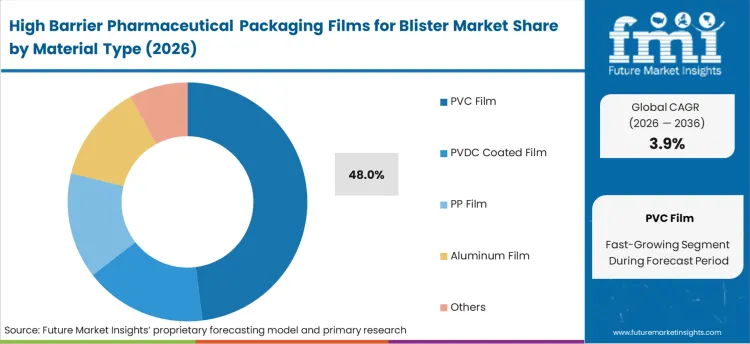
PVC film configurations are projected to account for 48% of the high barrier pharmaceutical packaging films for blister market in 2026. This leading share is supported by the increasing demand for cost-effective barrier solutions in pharmaceutical packaging applications and growing requirements for versatile processing characteristics. PVC films provide excellent thermoforming properties and chemical resistance, making them the preferred choice for tablet packaging, generic drug applications, and standard pharmaceutical blister packs. The segment benefits from technological advancements that have improved the barrier properties and processing efficiency of PVC film formulations while maintaining cost-effectiveness and regulatory compliance.
Modern PVC pharmaceutical films incorporate advanced polymer formulations, barrier coatings, and surface treatments that enhance moisture protection and extend drug shelf life. These innovations have significantly improved pharmaceutical packaging performance while reducing total cost of ownership through enhanced processability and elimination of packaging failures. The generic pharmaceuticals and over-the-counter drug sectors particularly drive demand for PVC film solutions, as these industries require cost-effective protection to maintain drug quality and comply with regulatory standards.
Additionally, the emerging market pharmaceutical industry increasingly adopts PVC films to balance protection requirements with cost considerations and meet international packaging standards. Market accessibility requirements and regulatory compliance initiatives further accelerate market adoption, as PVC systems provide reliable protection while maintaining competitive pricing and processing efficiency.
By Application, Tablets Segment Accounts for 58% Market Share
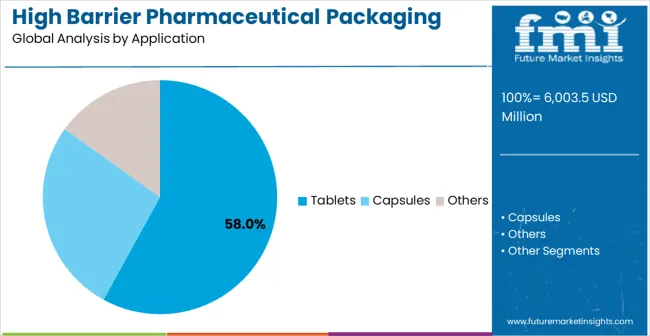
Tablets applications are expected to represent 58% of high barrier pharmaceutical packaging films for blister demand in 2026. This dominant share reflects the critical role of blister packaging in tablet protection and the need for reliable barrier solutions capable of maintaining drug stability in diverse storage and distribution conditions. Tablet manufacturers require effective and durable packaging films for moisture protection, oxygen barrier, and tamper evidence. The segment benefits from ongoing expansion of oral solid dosage form production in developing countries and increasing quality requirements demanding enhanced barrier systems in pharmaceutical manufacturing facilities.
Tablet packaging applications demand exceptional barrier performance to ensure drug efficacy and shelf life stability. These applications require films capable of handling various environmental conditions, storage requirements, and distribution challenges while maintaining drug integrity throughout the product lifecycle. The growing emphasis on pharmaceutical quality standards, particularly in generic drug manufacturing and international market distribution, drives consistent demand for high-performance barrier film solutions. Emerging pharmaceutical markets in Asia-Pacific, Latin America, and European regions contribute significantly to market growth as drug manufacturers invest in modern packaging technologies to improve product quality and market competitiveness.
Additionally, the trend toward patient-friendly packaging and unit-dose applications creates opportunities for specialized barrier film systems equipped with enhanced protective capabilities and user convenience features. The segment also benefits from increasing chronic disease medication production and aging population demographics driven by growing demand for long-term medication storage and patient compliance solutions.
What are the Drivers, Restraints, and Key Trends of the High Barrier Pharmaceutical Packaging Films for Blister Market?
The high barrier pharmaceutical packaging films for blister market is advancing steadily due to increasing pharmaceutical production and growing recognition of drug protection technology importance. However, the market faces challenges including regulatory compliance complexity, need for specialized processing expertise, and varying barrier requirements across different pharmaceutical applications. Standardization efforts and certification programs continue to influence film quality and market development patterns.
Integration of Smart Packaging and Track-and-Trace Technologies
The growing deployment of intelligent packaging systems and digital authentication capabilities is enabling real-time drug monitoring and anti-counterfeiting protection capabilities in pharmaceutical blister packaging. Smart sensors and tamper-evident technologies provide continuous product verification while enhancing supply chain security and extending drug traceability. These technologies are particularly valuable for high-value pharmaceuticals that require comprehensive protection against counterfeiting and supply chain integrity monitoring.
Development of Sustainable and High-Performance Solutions
Modern pharmaceutical film manufacturers are incorporating recyclable materials and bio-based barrier layers that improve environmental sustainability while maintaining protective performance through advanced material formulations and coating technologies. Integration of renewable raw materials and reduced environmental impact enables superior sustainability credentials and significant environmental benefits compared to traditional petroleum-based packaging films. Advanced manufacturing processes and quality control systems also support development of more consistent and reliable pharmaceutical packaging films for demanding regulatory environments.
Analysis of High Barrier Pharmaceutical Packaging Films for Blister Market by Key Country
.webp)
| Country | CAGR |
|---|---|
| China | 5.3% |
| India | 4.9% |
| Germany | 4.5% |
| Brazil | 4.1% |
| United States | 3.7% |
| United Kingdom | 3.3% |
| Japan | 2.9% |
Source: FMI analysis based on primary research and proprietary forecasting model
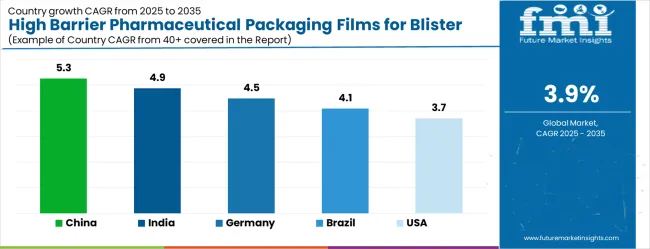
The high barrier pharmaceutical packaging films for blister market is growing steadily, with China leading at a 5.3% CAGR through 2036, driven by massive pharmaceutical manufacturing expansion, generic drug production growth, and domestic pharmaceutical market development. India follows at 4.9%, supported by rising pharmaceutical investments and increasing adoption of advanced packaging technologies in drug manufacturing and export operations. Germany records strong growth at 4.5%, emphasizing precision engineering, pharmaceutical excellence, and advanced packaging technology capabilities. Brazil grows steadily at 4.1%, integrating high barrier packaging films into expanding pharmaceutical production and healthcare infrastructure facilities. The United States shows moderate growth at 3.7%, focusing on pharmaceutical innovation and packaging technology advancement improvements. The United Kingdom maintains steady expansion at 3.3%, supported by pharmaceutical manufacturing programs. Japan demonstrates stable growth at 2.9%, emphasizing technological innovation and pharmaceutical packaging excellence.
The report covers an in-depth analysis of 40+ countries, the top-performing countries are highlighted below.
Sales Forecast for High Barrier Pharmaceutical Packaging Films for Blister Market in China
The high barrier pharmaceutical packaging films for blister market in China is projected to exhibit the highest growth rate with a CAGR of 5.3% through 2036, driven by rapid pharmaceutical manufacturing expansion and massive generic drug production development programs across pharmaceutical sectors. The country's growing API manufacturing and expanding pharmaceutical export industries are creating significant demand for high-performance packaging film systems. Major pharmaceutical companies are establishing comprehensive packaging installations to support large-scale drug production operations and meet international quality standards.
China's pharmaceutical sector continues expanding rapidly, with major investments in generic drug manufacturing, API production, and pharmaceutical export operations. The country's position as a global pharmaceutical manufacturing hub drives substantial demand for reliable packaging film solutions that ensure drug quality and regulatory compliance across diverse pharmaceutical applications.
- Pharmaceutical manufacturing modernization programs are supporting widespread adoption of advanced packaging film technologies across production facilities, driving demand for reliable and high-performance barrier protection solutions.
- Generic drug production expansion and pharmaceutical export initiatives are creating substantial opportunities for specialized packaging applications in demanding pharmaceutical environments requiring international quality standards.
Revenue Prospects for High Barrier Pharmaceutical Packaging Films for Blister Market in India
The high barrier pharmaceutical packaging films for blister market in India is expanding at a CAGR of 4.9%, supported by increasing pharmaceutical investments across manufacturing sectors and growing adoption of advanced packaging technologies in drug manufacturing and export operations. The country's expanding generic drug industry is driving demand for specialized packaging systems capable of handling diverse regulatory and quality requirements. Pharmaceutical facilities are investing in advanced barrier technologies to improve product quality and comply with international standards.
India's pharmaceutical development initiatives and export expansion create significant opportunities for high barrier packaging film applications. The country's growing pharmaceutical market drives demand for generic drugs and API production, requiring reliable packaging solutions to maintain drug quality and market competitiveness.
- Pharmaceutical sector growth and generic drug expansion are creating opportunities for specialized packaging film applications across diverse manufacturing facilities requiring reliable drug protection solutions.
- Government pharmaceutical programs and export promotion initiatives are driving investments in advanced packaging systems for drug manufacturing throughout major pharmaceutical production regions.
Growth Outlook for High Barrier Pharmaceutical Packaging Films for Blister Market in Germany
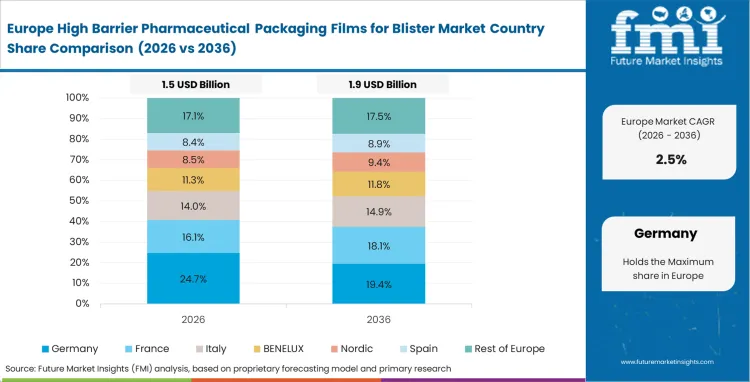
The high barrier pharmaceutical packaging films for blister market in Germany is projected to grow at a CAGR of 4.5%, supported by the country's emphasis on pharmaceutical excellence and advanced packaging technology capabilities. German pharmaceutical facilities are implementing high-performance packaging systems that meet stringent quality standards and regulatory reliability requirements. The market is characterized by focus on technological innovation, advanced materials, and compliance with comprehensive pharmaceutical regulations.
Germany's advanced pharmaceutical sector and packaging excellence capabilities drive demand for high-quality barrier film solutions. The country's emphasis on pharmaceutical innovation, technological advancement, and quality precision creates opportunities for advanced packaging technologies that meet demanding pharmaceutical specifications.
- Pharmaceutical industry investments are prioritizing advanced packaging film technologies that demonstrate superior performance and reliability while meeting German pharmaceutical quality and regulatory standards.
- Packaging excellence programs and pharmaceutical innovation initiatives are driving adoption of high-performance barrier systems that support optimal drug protection conditions and enhanced regulatory compliance.
Market Assessment for High Barrier Pharmaceutical Packaging Films for Blister Market in Brazil
The high barrier pharmaceutical packaging films for blister market in Brazil is growing at a CAGR of 4.1%, driven by expanding pharmaceutical production and increasing healthcare infrastructure development across pharmaceutical sectors. The country's growing generic drug manufacturing is investing in advanced packaging systems to improve drug quality and performance in competitive market conditions. Pharmaceutical facilities are adopting modern barrier technologies to support growing production requirements and quality standards.
Brazil's expanding pharmaceutical infrastructure and healthcare development create substantial opportunities for packaging film applications. The country's growing pharmaceutical market and domestic production expansion drive investments in drug protection solutions that enhance product competitiveness and operational efficiency.
- Pharmaceutical infrastructure expansion and production development are facilitating adoption of reliable packaging film systems capable of consistent performance in diverse pharmaceutical manufacturing environments.
- Healthcare development programs and pharmaceutical enhancement initiatives are increasing demand for advanced barrier solutions that meet evolving pharmaceutical standards and quality requirements.
Demand Assessment for High Barrier Pharmaceutical Packaging Films for Blister Market in United States
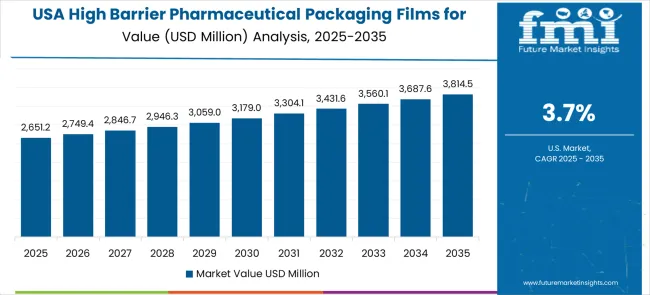
The high barrier pharmaceutical packaging films for blister market in the United States is expanding at a CAGR of 3.7%, driven by ongoing pharmaceutical innovation and increasing emphasis on packaging technology advancement solutions. Pharmaceutical facilities are upgrading existing packaging systems with advanced technologies that provide improved performance and enhanced drug protection capabilities. The market benefits from replacement demand and facility modernization programs across multiple pharmaceutical sectors.
The United States market emphasizes pharmaceutical innovation, technology advancement, and packaging improvement in barrier film applications. Advanced research and development activities drive the adoption of next-generation packaging technologies that offer superior performance and regulatory compliance capabilities.
- Pharmaceutical facility upgrades and innovation programs are driving demand for advanced packaging film systems that offer superior drug protection and enhanced pharmaceutical capabilities.
- Technology development initiatives and pharmaceutical advancement mandates are supporting adoption of innovative barrier technologies that demonstrate compliance with pharmaceutical standards and regulatory requirements.
Market Analysis for High Barrier Pharmaceutical Packaging Films for Blister Market in United Kingdom
The high barrier pharmaceutical packaging films for blister market in the United Kingdom is projected to grow at a CAGR of 3.3%, supported by ongoing pharmaceutical manufacturing programs and packaging technology facility upgrades. Pharmaceutical operators are investing in reliable barrier film systems that provide consistent performance and meet regulatory compliance requirements. The market is characterized by focus on system reliability, operational efficiency, and pharmaceutical compliance across diverse drug manufacturing applications.
The United Kingdom's focus on pharmaceutical manufacturing and packaging excellence drives demand for high-performance barrier film solutions. The country's emphasis on drug quality, manufacturing efficiency, and pharmaceutical compliance creates opportunities for advanced packaging technologies.
- Pharmaceutical manufacturing initiatives and packaging enhancement programs are supporting adoption of advanced barrier film systems that meet contemporary pharmaceutical and regulatory standards.
- Drug production programs and efficiency improvement initiatives are creating demand for specialized packaging applications that provide reliable performance and pharmaceutical excellence.
Opportunity Analysis for High Barrier Pharmaceutical Packaging Films for Blister Market in Japan
The high barrier pharmaceutical packaging films for blister market in Japan is expanding at a CAGR of 2.9%, driven by the country's emphasis on technological innovation and pharmaceutical packaging excellence. Japanese manufacturers are developing advanced packaging film technologies that incorporate precision engineering and efficiency-optimized design principles. The market benefits from focus on quality, reliability, and continuous improvement in pharmaceutical packaging technology performance.
Japan's technological leadership and pharmaceutical expertise drive the development of advanced barrier film solutions. The country's emphasis on innovation, quality control, and packaging optimization creates opportunities for cutting-edge packaging technologies that set pharmaceutical industry standards.
- Pharmaceutical excellence programs and technological innovation initiatives are driving development of advanced packaging film systems that demonstrate superior performance and operational reliability.
- Packaging technology and precision manufacturing programs are supporting adoption of precision-engineered barrier solutions that optimize drug protection conditions and enhance pharmaceutical quality.
Competitive Landscape of High Barrier Pharmaceutical Packaging Films for Blister Market
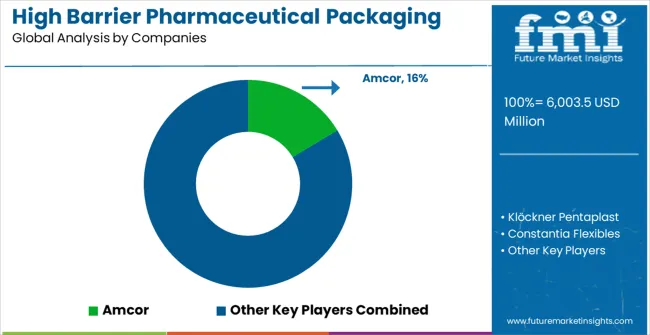
The high barrier pharmaceutical packaging films for blister market is defined by competition among established packaging materials manufacturers, specialized pharmaceutical packaging companies, and emerging sustainable packaging solution providers. Companies are investing in advanced barrier technologies, regulatory compliance capabilities, standardized quality systems, and technical support services to deliver reliable, compliant, and cost-effective pharmaceutical packaging solutions. Strategic partnerships, technological advancement, and geographic expansion are central to strengthening product portfolios and market presence.
Amcor, operating globally, offers comprehensive pharmaceutical packaging solutions with focus on barrier excellence, regulatory compliance, and technical support services. Klöckner Pentaplast, multinational manufacturer, provides advanced packaging film systems with emphasis on pharmaceutical applications and regulatory integration capabilities. Constantia Flexibles, specialized provider, delivers innovative barrier packaging solutions for pharmaceutical applications with focus on quality and performance. Perlen Packaging (CPH) offers comprehensive packaging technologies with standardized procedures and pharmaceutical service support.
Tekni-plex provides advanced pharmaceutical packaging systems with emphasis on specialty applications and technical expertise. Honeywell, Liveo Research GmbH deliver specialized barrier solutions with focus on high-performance pharmaceutical applications. Sumitomo Bakelite, HySum, Aluberg offer comprehensive pharmaceutical packaging technologies for demanding regulatory environments with established manufacturing and service capabilities.
Bilcare, SÜDPACK, FlexiPack, Etimex Primary Packaging, Uniworth, Sichuan Huili Industry, Jiangxi Chunguang New Materials, Hangzhou Plastics Industry, Jiangsu Fuxin, Huakang Packaging, LIAONING TOTEM PACKAGING provide advanced packaging film systems with regional manufacturing capabilities, pharmaceutical industry expertise, and specialized knowledge across packaging technology and pharmaceutical compliance sectors.
Global High Barrier Pharmaceutical Packaging Films for Blister Market -- Stakeholder Contribution Framework
The high barrier pharmaceutical packaging films for blister market underpins pharmaceutical quality excellence, drug safety advancement, regulatory compliance optimization, and patient health protection. With drug safety mandates, stricter regulatory requirements, and demand for protective packaging systems, the sector must balance cost competitiveness, barrier performance, and regulatory compliance. Coordinated contributions from governments, industry bodies, OEMs/technology integrators, suppliers, and investors will accelerate the transition toward performance-optimized, regulatory-compliant, and technologically advanced pharmaceutical packaging systems.
How Governments Could Spur Market Growth and Adoption
- Pharmaceutical Quality Incentives: Provide subsidies or regulatory support for manufacturers upgrading to high-performance or sustainable pharmaceutical packaging solutions.
- Drug Safety Standards: Enforce stringent pharmaceutical quality, patient safety, and drug stability standards, boosting demand in generic drug, branded pharmaceutical, and specialty drug sectors.
- Healthcare Infrastructure Programs: Embed advanced packaging technologies into pharmaceutical manufacturing initiatives, drug safety projects, and healthcare quality enhancement programs.
- Trade Facilitation: Lower tariffs for importing pharmaceutical-grade materials and packaging components while supporting local exports of compliant packaging systems.
- Innovation Mandates: Recognize advanced and regulatory-compliant packaging solutions within national pharmaceutical competitiveness and healthcare quality frameworks.
How Industry Bodies Could Support Market Development
- Standardization & Certification: Develop harmonized guidelines for barrier performance, regulatory compliance benchmarks, and pharmaceutical quality testing procedures.
- Training & Workforce Development: Create curricula for packaging technology, regulatory compliance, and advanced pharmaceutical packaging systems to reduce skill shortages.
- Global Recognition: Position regional manufacturers as leaders in high-performance and regulatory-compliant pharmaceutical packaging solutions.
- Policy Advocacy: Engage with pharmaceutical regulators to shape favorable procurement policies and inclusion of barrier films in drug safety standards.
How OEMs and Technology Integrators Could Strengthen the Ecosystem
- Technology Integration: Embed smart packaging systems, track-and-trace capabilities, and quality monitoring into pharmaceutical packaging films.
- Partnerships: Collaborate with pharmaceutical companies, drug manufacturers, and regulatory bodies to design application-specific packaging configurations.
- Product Innovation: Advance barrier materials, sustainable packaging, and enhanced protection technologies.
- Comprehensive Solutions: Bundle packaging films with complete pharmaceutical packaging systems, offering end-to-end drug protection effectiveness.
How Suppliers Could Navigate the Market Evolution
- Advanced Materials: Provide pharmaceutical-grade polymers, barrier coatings, and compliance-enhancing additives to extend packaging performance and regulatory approval.
- Localized Manufacturing: Establish regional production facilities to reduce logistics costs and improve pharmaceutical market access.
- Portfolio Diversification: Expand into specialty pharmaceutical materials, regulatory-specific solutions, and sustainable packaging technologies.
- Customer Engagement: Build trust via regulatory compliance, performance guarantees, and comprehensive pharmaceutical service excellence.
How Investors and Financial Enablers Could Unlock Value
- Infrastructure Financing: Fund pharmaceutical packaging facilities, regional distribution centers, and production plants in high-growth pharmaceutical markets.
- Innovation Finance: Channel pharmaceutical-linked capital toward regulatory-compliant and high-performance packaging solutions.
- M&A and Consolidation: Back cross-border mergers to integrate fragmented regional players into scalable global pharmaceutical packaging suppliers.
- Technology Funding: Support startups developing advanced barrier technologies, smart packaging systems, or specialized pharmaceutical applications.
- Risk Management Tools: Provide insurance and hedging products against raw material cost volatility and regulatory compliance risks.
Key Players in the High Barrier Pharmaceutical Packaging Films for Blister Market
- Amcor
- Klöckner Pentaplast
- Constantia Flexibles
- Perlen Packaging (CPH)
- Tekni-plex
- Honeywell
- Liveo Research GmbH
- Sumitomo Bakelite
- HySum
- Aluberg
- Bilcare
- SÜDPACK
- FlexiPack
- Etimex Primary Packaging
- Uniworth
- Sichuan Huili Industry
- Jiangxi Chunguang New Materials
- Hangzhou Plastics Industry
- Jiangsu Fuxin
- Huakang Packaging
- LIAONING TOTEM PACKAGING
Scope of the Report
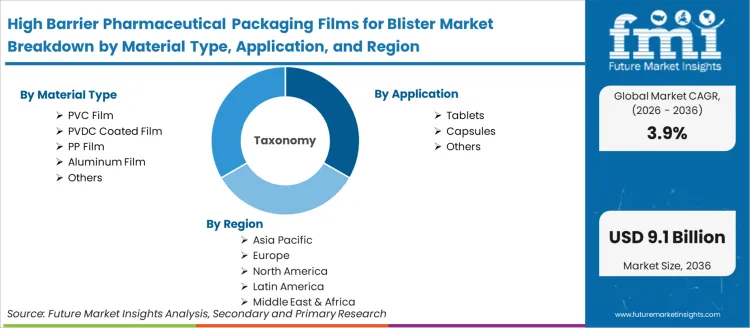
| Metric | Value |
|---|---|
| Quantitative Units | USD 6.24 Bn to USD 9.14 Bn, at a CAGR of 3.90% |
| Market Definition | High barrier pharmaceutical packaging films for blister applications are multi-layer polymer and aluminum laminate films engineered to provide moisture vapor transmission rate (MVTR) and oxygen transmission rate (OTR) barriers protecting hygroscopic and oxidation-sensitive tablet and capsule dosage forms in thermoformed blister cavity and push-through lidding configurations. |
| Material Type Segmentation | PVC Film, PVDC Coated Film, PP Film, Aluminum Film, Others |
| Application Segmentation | Tablets, Capsules, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40 plus countries |
| Key Companies Profiled | Amcor, Klockner Pentaplast, Constantia Flexibles, Perlen Packaging (CPH), Tekni-plex, Honeywell, Liveo Research GmbH, Sumitomo Bakelite, HySum, Aluberg, Bilcare, SUDPACK, FlexiPack, Etimex Primary Packaging, Uniworth, Sichuan Huili Industry |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down modeling validated against primary research inputs and publicly available industry data. |
High Barrier Pharmaceutical Packaging Films for Blister Market by Segments
Material Type:
- PVC Film
- PVDC Coated Film
- PP Film
- Aluminum Film
- Others
Application:
- Tablets
- Capsules
- Others
Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia & Pacific
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] International Council for Harmonisation. (2024). ICH Q1A(R2): Stability Testing of New Drug Substances and Products. ICH.
- [2] Amcor Plc. (2024). Annual Report 2024: Healthcare Packaging Division. Amcor.
- [3] European Commission. (2024). Packaging and Packaging Waste Regulation (PPWR): Pharmaceutical Packaging Recyclability Assessment. European Commission.
- [4] Klockner Pentaplast. (2024). Pharmaceutical Films Product Portfolio: Barrier Performance Specifications. Klockner Pentaplast.
- [5] US Food and Drug Administration. (2024). Guidance for Industry: Container Closure System Guidance for Packaging Human Drugs. FDA.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for High Barrier Pharmaceutical Packaging Films for Blister in the global market in 2026?
Demand for High Barrier Pharmaceutical Packaging Films for Blister in the global market is estimated to be valued at USD 6.24 billion in 2026.
What will be the market size of High Barrier Pharmaceutical Packaging Films for Blister by 2036?
Market size for High Barrier Pharmaceutical Packaging Films for Blister is projected to reach USD 9.14 billion by 2036.
What is the expected demand growth between 2026 and 2036?
Demand is expected to grow at a CAGR of 3.90% between 2026 and 2036.
Which Material Type is poised to lead by 2026?
PVC Film accounts for 48.0% in 2026.
How significant is Tablets in driving adoption?
Tablets represents 58.0% of segment share in 2026.
What country records the fastest growth?
China is projected to grow at a CAGR of 5.30% during 2026 to 2036.
What is the projected growth for India?
India is projected to expand at a CAGR of 4.90% during 2026 to 2036.
What is included in the scope of this report?
Market scope covers PVC, PVDC-coated, polypropylene, and aluminum cold-form blister films used in thermoformed and cold-formed pharmaceutical blister packaging for tablet, capsule, and unit-dose solid oral dosage form applications.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material Type , 2026 to 2036
- PVC Film
- PVDC Coated Film
- PP Film
- Aluminum Film
- Others
- PVC Film
- Y to o to Y Growth Trend Analysis By Material Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Material Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Tablets
- Capsules
- Others
- Tablets
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Material Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Amcor
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Klöckner Pentaplast
- Constantia Flexibles
- Perlen Packaging (CPH)
- Tekni-plex
- Honeywell
- Liveo Research GmbH
- Sumitomo Bakelite
- HySum
- Aluberg
- Bilcare
- SÜDPACK
- FlexiPack
- Etimex Primary Packaging
- Uniworth
- Sichuan Huili Industry
- Jiangxi Chunguang New Materials
- Hangzhou Plastics Industry
- Jiangsu Fuxin
- Huakang Packaging
- Amcor
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Material Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Material Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Material Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Material Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Material Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Material Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Material Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Material Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

