The global reduced-crosslinking gelatin capsule market is projected to reach USD 239.1 million by 2035, recording an absolute increase of USD 99.1 million over the forecast period. This market is valued at USD 140 million in 2025 and is set to rise at a CAGR of 5.5% during the assessment period. Expansion is supported by rising demand for stable, patient-friendly oral dosage forms across pharmaceutical, nutraceutical, and functional food sectors, alongside increasing adoption of gelatin capsules for temperature-sensitive and moisture-sensitive formulations.
Development reflects evolving pharmaceutical manufacturing processes, where advanced gelatin formulation techniques enable reduced crosslinking while maintaining capsule integrity, dissolution uniformity, and enhanced bioavailability. Pharmaceutical manufacturers and supplement producers face increasing pressure to improve formulation stability, shelf-life, and patient acceptability. Precision-controlled encapsulation, automated drying, and inline quality inspection systems are being deployed to ensure consistent capsule performance, reduce batch variability, and meet global pharmacopoeia standards.
Government regulatory frameworks and industry standards accelerate development. Compliance with FDA, USP, and European Pharmacopeia guidelines ensures consistent product quality and safety. Manufacturers are investing in workforce training, equipment modernization, and process optimization to meet evolving regulatory and consumer expectations. Consumer trends indicate growing preference for convenient, reliable oral dosage forms, supporting sustained demand for reduced-crosslinking gelatin capsules in both developed and emerging markets.
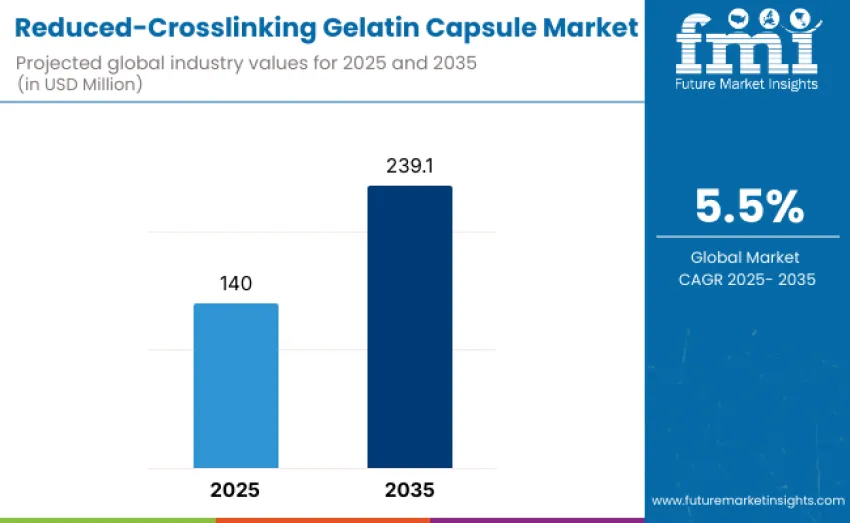
Between 2025 and 2030, the reduced-crosslinking gelatin capsule market is projected to expand from USD 140 million to USD 182.9 million, resulting in a value increase of USD 42.9 million, which represents 43.3% of total forecast growth for the decade. The period will be driven by increasing demand for targeted pharmaceutical applications requiring controlled-release formulations and improved capsule stability. Innovation in capsule manufacturing processes, including optimization of gelatin crosslinking techniques and incorporation of functional excipients, will shape competitive dynamics.
From 2030 to 2035, growth is forecast from USD 182.9 million to USD 239.1 million, adding USD 56.1 million, constituting 56.7% of overall ten-year expansion. This period is expected to witness maturation of advanced reduced-crosslinking gelatin formulations, including tailored release profiles and enhanced compatibility with novel drug delivery technologies. Strategic collaborations between capsule manufacturers and pharmaceutical innovators will accelerate adoption in high-value therapeutic areas. Sustainable manufacturing practices and cost-efficient production methods will become increasingly important as demand scales.
Reduced-Crosslinking Gelatin Capsule Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 140 million |
| Market Forecast Value (2035) | USD 239.1 million |
| Forecast CAGR (2025 to 2035) | 5.5% |
The reduced-crosslinking gelatin capsule market grows by enabling pharmaceutical manufacturers to achieve superior capsule performance and bioavailability while maintaining cost-effective production processes for oral dosage forms. Pharmaceutical companies face mounting pressure to deliver high-quality formulations within compressed development timelines, with reduced-crosslinking capsules providing significant advantages in dissolution consistency and drug release control. Rising demand for specialty pharmaceuticals and nutraceuticals creates opportunities for capsule manufacturers who can deliver reliable performance across diverse therapeutic applications.
Government initiatives promoting pharmaceutical quality and regulatory compliance drive adoption in drug manufacturing facilities, contract development organizations, and research institutions, where capsule performance directly impacts product approval and commercialization success. The global shift toward personalized medicine and specialty drug formulations accelerates demand for capsules that can accommodate sensitive compounds while maintaining stability. Premium pricing ranging from specialized manufacturing requirements and stringent quality standards may limit adoption among smaller pharmaceutical companies and generic manufacturers.
The reduced-crosslinking gelatin capsule market is segmented by product, application, and region. By product, division includes standard RCC capsules, pharma-grade low-crosslink capsules, nutraceutical grade capsules, custom sizes and colors, and others. Based on application, categorization covers prescription drug encapsulation, OTC and nutraceuticals, clinical trial supply, and research and specialty use. Regionally, segmentation spans Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
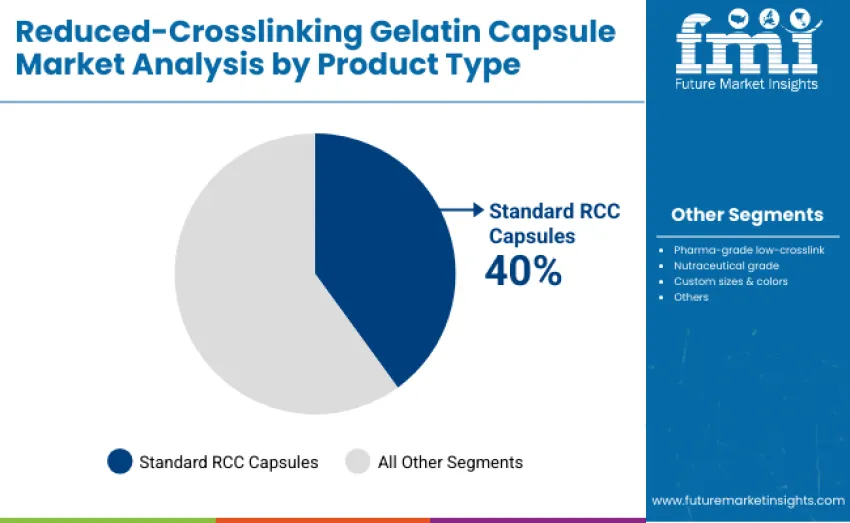
The standard RCC capsules segment represents the dominant force, capturing 40% of total revenue in 2025. This segment includes conventional gelatin capsules widely adopted across prescription and OTC pharmaceutical applications due to their cost-effectiveness, reliable dissolution performance, and compatibility with standard encapsulation processes. Leadership is driven by broad acceptance among pharmaceutical manufacturers for controlled drug delivery and general oral dosage applications.
The pharma-grade low-crosslink segment maintains a significant 25% share, serving specialized pharmaceutical formulations that require enhanced dissolution, improved bioavailability, and regulatory compliance. Nutraceutical grade capsules account for 15%, supporting the growing dietary supplement and functional health products sector. Custom sizes and colors contribute 12% of revenue, catering to personalized and specialty formulations, while other capsule types represent 8%, addressing niche or experimental applications.
Key Advantages Driving the Standard RCC Capsules Segment Include:
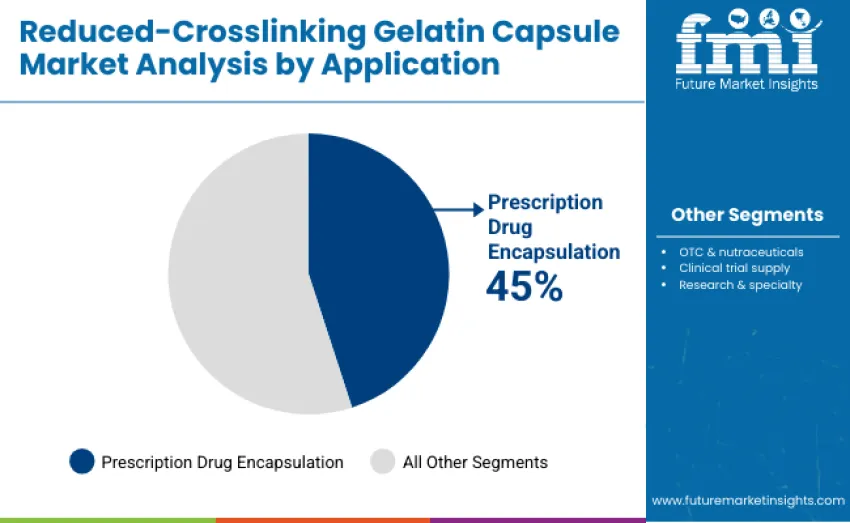
Prescription drug encapsulation dominates, capturing 45% of total revenue in 2025. This leadership is attributed to widespread adoption of gelatin capsules in controlled oral dosage forms, where consistent dissolution and bioavailability are critical for therapeutic efficacy. Manufacturers increasingly prefer reduced-crosslinking formulations to overcome crosslinking-related challenges that can compromise drug release, ensuring compliance with regulatory standards and maintaining product quality.
The OTC and nutraceuticals segment accounts for 30% of revenue, driven by growing consumer awareness of health, wellness, and functional nutrition. Rising demand for dietary supplements and nutraceutical products has led to increased adoption of gelatin capsules that deliver reliable performance, convenient dosage, and enhanced stability for active ingredients. Clinical trial supply represents 15%, supporting investigational studies and early-stage drug development.
Key Market Dynamics Supporting Application Preferences Include:
What Are the Major Drivers, Restraints, and Emerging Trends Influencing the Reduced-Crosslinking Gelatin Capsule Market?
The reduced-crosslinking gelatin capsule market experiences steady growth, driven by increasing demand for improved oral dosage forms in pharmaceutical and nutraceutical applications. Expansion is supported by technological advancements in capsule manufacturing, rising adoption of low-crosslink formulations for enhanced dissolution performance, and expanding global healthcare infrastructure. Manufacturers increasingly invest in process innovations and product differentiation strategies to meet regulatory requirements and ensure consistent bioavailability, contributing to overall growth.
What Are the Major Restraints and Challenges Affecting the Growth of the Reduced-Crosslinking Gelatin Capsule Market?
Growth faces restraints from high manufacturing costs associated with specialized low-crosslink formulations and strict compliance requirements with regulatory standards. Challenges include formulation complexity for active pharmaceutical ingredients sensitive to gelatin crosslinking, requiring technical expertise and process optimization. Competition from alternative delivery systems, such as soft gels, tablets, and capsule substitutes, can limit adoption in certain segments.
What Emerging Trends Are Shaping the Reduced-Crosslinking Gelatin Capsule Market?
Emerging trends indicate strong focus on product differentiation and customization. Capsule manufacturers increasingly offer nutraceutical-grade, pharma-grade, and custom-size options to meet evolving client and consumer requirements. Efforts integrate advanced control systems and real-time quality monitoring during production to ensure consistent capsule performance and regulatory compliance. Sustainability and eco-friendly manufacturing initiatives gain traction, with efforts to reduce energy consumption, waste generation, and environmental impact during gelatin production.
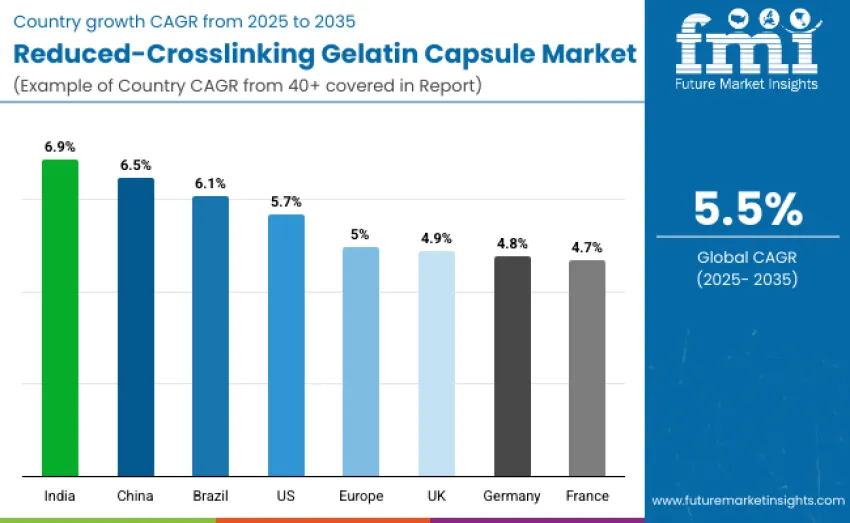
| Country | CAGR (2025-2035) |
|---|---|
| United States | 5.7% |
| Brazil | 6.1% |
| China | 6.5% |
| India | 6.9% |
| Europe | 5.0% |
| Germany | 4.8% |
| France | 4.7% |
| United Kingdom | 4.9% |
The reduced-crosslinking gelatin capsule market is seeing significant growth, with India leading at a 6.9% CAGR due to expanding pharmaceutical manufacturing and increased demand for high-quality oral drug delivery systems. China follows with a 6.5% CAGR, driven by government initiatives and investments in healthcare. Germany shows a 4.8% CAGR, supported by strong research and quality standards. Brazil records a 6.1% CAGR, driven by growing nutraceutical production and healthcare awareness. The USA stands at 5.7%, with a mature pharmaceutical industry, while the UK grows steadily at 4.9%, supported by product innovation and regulation.
The report covers an in-depth analysis of 40+ countries top-performing countries are highlighted below.
China demonstrates the strongest growth potential with a CAGR of 6.5% through 2035. Leadership is supported by large-scale pharmaceutical manufacturing expansion, increased production of nutraceutical products, and government initiatives to modernize healthcare infrastructure. Growth concentrates in major industrial clusters, including Beijing, Shanghai, Shenzhen, and Hangzhou, where capsule manufacturing facilities are equipped with advanced production technologies designed to minimize crosslinking and improve bioavailability.
Key market factors:
In India, growth is projected at a CAGR of 6.9% through 2035. Expansion is driven by increasing local pharmaceutical manufacturing capacity, improving healthcare accessibility, and rising demand for oral drug delivery systems and nutraceutical products. Major metropolitan hubs, including Bangalore, Hyderabad, Mumbai, and Pune, have emerged as production centers, implementing modern capsule technologies that reduce crosslinking and ensure compliance with international quality standards.
Germany's growth is expected at a CAGR of 4.8% through 2035, supported by stringent European regulatory standards and advanced manufacturing practices. Production clusters in Berlin, Munich, Frankfurt, and Hamburg have implemented automated capsule lines that minimize crosslinking, improve product consistency, and enhance bioavailability. Focus on precision engineering, sustainability, and environmental compliance has strengthened reputation for high-quality capsules.
Key development areas:
Brazil demonstrates strong potential with a CAGR of 6.1% through 2035. Expansion is driven by increasing healthcare access, growing local pharmaceutical production, and rising consumer awareness of preventive healthcare. Urban centers including São Paulo, Rio de Janeiro, Brasília, and Porto Alegre have become hubs for capsule production, with modern facilities designed to reduce crosslinking and improve stability.
Leading market segments:
The United States reduced-crosslinking gelatin capsule market is projected to grow at a CAGR of 5.7% through 2035. Growth is facilitated by adoption of advanced production technologies, automation, and significant research investment. Capsule manufacturing facilities in California, New Jersey, Texas, and New York have integrated automated lines that reduce crosslinking and improve bioavailability.
Established pharmaceutical companies leverage economies of scale to produce both prescription and nutraceutical capsules efficiently, while FDA regulations ensure consistent quality, stability, and safety. The mature nutraceutical segment, coupled with rising preventive healthcare demand, has supported increased adoption. Distribution networks spanning hospitals, retail pharmacies, and online platforms enable comprehensive coverage. Collaborative research with universities and private institutions fosters innovation in formulations, capsule stability, and process optimization.
In the United Kingdom, growth is expected at a CAGR of 4.9% through 2035. Expansion is supported by advanced manufacturing practices, robust regulatory frameworks, and consistent pharmaceutical innovation. Production clusters in London, Manchester, Bristol, and Edinburgh emphasize GMP compliance, ensuring capsules with minimal crosslinking and high stability.
Investments in process optimization, workforce training, and quality control have enhanced productivity and product reliability. Collaborations between pharmaceutical companies and universities have accelerated adoption of advanced production technologies and facilitated workforce skill development. Penetration is supported through hospital networks, retail pharmacies, and nutraceutical distribution channels. Rising consumer preference for preventive healthcare and dietary supplements has contributed to increased capsule usage across diverse segments.
France's reduced-crosslinking gelatin capsule market is projected to grow at a 4.7% CAGR through 2035, supported by strong regulatory frameworks and continuous advancements in manufacturing processes. Production facilities in Paris, Lyon, and Toulouse have adopted cutting-edge encapsulation technologies that minimize crosslinking, improve dissolution rates, and enhance bioavailability. France’s adherence to stringent European standards and commitment to innovation in pharmaceutical manufacturing ensure high-quality capsule production.
Key development areas:
The reduced-crosslinking gelatin capsule market in Europe is projected to grow from USD 42 million in 2025 to USD 66.9 million by 2035, registering a CAGR of 5.0% over the forecast period. Germany is expected to maintain leadership with an 18.5% share in 2025, rising to 19.5% by 2035, supported by advanced pharmaceutical manufacturing infrastructure, strong research capabilities, and extensive capsule technology integration within formulation plants.
France follows with a 16% share in 2025, projected to decline slightly to 15.5% by 2035, driven by consistent demand from prescription drug and nutraceutical applications supported by well-established pharmaceutical clusters. Italy holds a 12% share in 2025, expected to decrease to 11.5% by 2035, reflecting steady capsule usage in contract manufacturing and dietary supplement production.
Spain accounts for a 9% share in 2025, declining to 8.5% by 2035, backed by modernization of encapsulation facilities and increasing adoption for temperature-sensitive formulations. The BENELUX region records moderate expansion from 6% in 2025 to 6.5% by 2035, driven by advanced capsule filling technologies and multinational excipient producers serving Western Europe.
The Japanese reduced-crosslinking gelatin capsule market is characterized by a focus on formulation precision, controlled crosslinking, and capsule uniformity for high-value drug applications. The pharmaceutical industry demands capsules with superior dissolution performance and consistent bioavailability, supported by strict regulatory compliance and advanced encapsulation technologies. Domestic producers and international suppliers collaborate with Japanese pharmaceutical firms to deliver capsules meeting stringent quality and stability standards. Japan’s robust R&D base, adherence to JP and global pharmacopeial requirements, and leadership in adopting reduced-crosslinking technologies reinforce its position in ensuring optimal drug release profiles.
The South Korean reduced-crosslinking gelatin capsule market is increasingly influenced by global suppliers offering advanced encapsulation materials, analytical support, and formulation services to domestic pharmaceutical and nutraceutical manufacturers. International producers provide comprehensive technical assistance to optimize gelatin capsule performance in temperature- and humidity-sensitive formulations. Rising collaboration between global gelatin producers and Korean pharmaceutical technology providers ensures compatibility with high-performance filling equipment and compliance with MFDS quality standards. Multinational suppliers expand their presence through partnerships with local distributors and contract manufacturers, meeting Korea’s growing demand for premium capsule materials in health supplements and innovative dosage forms.
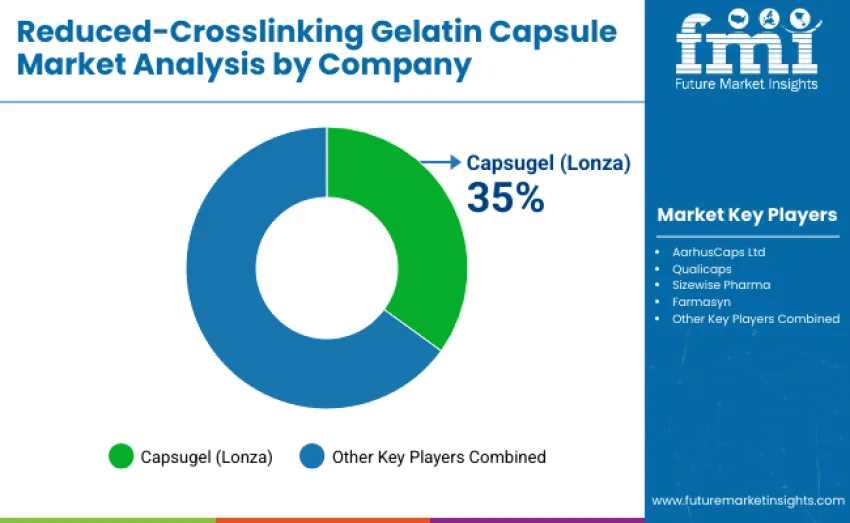
The global reduced-crosslinking gelatin capsule market is moderately concentrated, with 12 to 15 key players shaping industry dynamics through product quality, regulatory compliance, and formulation support. The top three companies control a significant share of global revenue, with competition driven by capsule material innovation, dissolution stability, and formulation compatibility, rather than price alone.
Capsugel (Lonza) leads with a 35% market share, bolstered by its extensive pharmaceutical-grade gelatin technology portfolio and global supply partnerships with leading formulation developers. Qualicaps maintains a strong presence in Japan, the USA, and Europe, offering consistent product performance, GMP-certified manufacturing, and advanced quality assurance systems. AarhusCaps Ltd stands out with its customized capsule solutions for high-potency drugs and nutraceuticals, focusing on low-crosslinking gelatin formulations to enhance bioavailability.
Emerging mid-tier players like Sizewise Pharma and Farmasyn target regional supply and formulation-specific capsule designs, competing on flexibility and cost-effectiveness for niche therapeutic applications and small-to-medium contract manufacturers. Regional and specialized producers contribute to a diverse market by offering tailored solutions for dietary supplements, herbal preparations, and temperature-sensitive drug products. Competitive dynamics increasingly favor companies combining proven gelatin processing technology with technical service expertise, ensuring product consistency across varying conditions.
| Items | Values |
|---|---|
| Quantitative Units | USD 140 million |
| Product | Standard RCC capsules, Pharma-grade low-crosslink, Nutraceutical grade, Custom sizes & colors, Others |
| Application | Prescription drug encapsulation, OTC & nutraceuticals, Clinical trial supply, Research & specialty |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Country Covered | USA, Brazil, China, India, Europe, Germany, France, UK |
| Key Companies Profiled | AarhusCaps Ltd, Capsugel ( Lonza ), Qualicaps, Sizewise Pharma, Farmasyn |
| Additiona Attributes | Dollar sales by product and application, regional trends across Asia Pacific, Europe, and North America, competitive landscape with pharmaceutical capsule manufacturers, technical specifications, integration with formulation workflows, innovations in low-crosslink gelatin processing, and enhanced stability and bioavailability in capsule configurations |
How big is the reduced-crosslinking gelatin capsule market in 2025?
The global reduced-crosslinking gelatin capsule market is valued at USD 140 million in 2025.
What will be the size of the reduced-crosslinking gelatin capsule market in 2035?
The market is projected to reach USD 239.1 million by 2035.
How fast will the reduced-crosslinking gelatin capsule market grow between 2025 and 2035?
The market will grow at a CAGR of 5.5% from 2025 to 2035.
Which product leads the reduced-crosslinking gelatin capsule market in 2025?
Standard RCC capsules lead the market with a 40% share in 2025.
Who are the major players in the reduced-crosslinking gelatin capsule market?
Key players include AarhusCaps Ltd, Capsugel (Lonza), Qualicaps, Sizewise Pharma, and Farmasyn.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.