The global gelatin filters market is valued at USD 99.8 million in 2025. It is slated to reach USD 219.5 million by 2035, recording an absolute increase of USD 119.7 million over the forecast period. The global gelatin filters market expansion by nearly 2.2X is being shaped by the growing use of biocompatible filtration media in pharmaceutical production, microbial testing, food safety, and biotechnology research. The adoption of gelatin-based membrane technologies is accelerating as manufacturers seek filtration systems that preserve biological activity, ensure sterility assurance, and minimize protein adsorption. Increasing deployment of continuous bioprocessing and integrated manufacturing systems is creating a strong preference for gelatin filters that offer gentle separation, rapid microbial recovery, and consistent performance under regulatory validation protocols.
The microfiltration membrane segment holds a 52.7% market share, serving as the core filtration grade across sterile processing, clarification, and quality control operations. The microbial detection segment, representing 34.5% of global demand, continues to dominate as pharmaceutical laboratories and food testing facilities adopt gelatin filters for membrane transfer techniques, microbial enumeration, and aseptic validation workflows. Growth between 2025 and 2030 will be supported by increasing biopharmaceutical output, disposable filtration modules, and protein-compatible membranes, while the 2030–2035 phase will be defined by cell and gene therapy manufacturing, vaccine purification, and regenerative medicine applications requiring gentle biomolecule retention. China (11.1% CAGR) and India (10.3% CAGR) lead global adoption due to expanding biopharmaceutical infrastructure and biosimilar manufacturing programs, followed by Germany (9.4%), supported by pharmaceutical precision manufacturing and biotechnology leadership. SKC, Sartorius, ADOX Fotowerke GmbH, DRM Chemical Process Services, and Dorstener Drahtwerke dominate competition through single-use filtration integration, composite membrane innovation, and validation-ready gelatin filter formats designed for GMP environments.
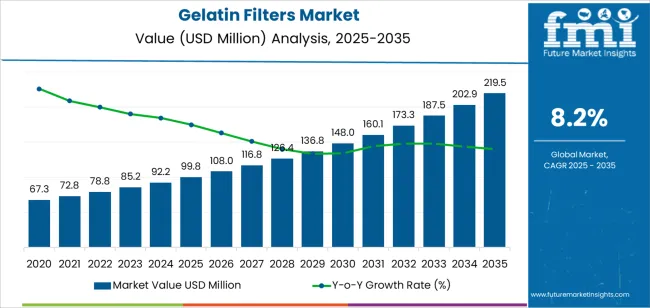
From 2030 to 2035, the market is forecast to grow from USD 148.0 million to USD 219.5 million, adding another USD 71.5 million, which constitutes 59.7% of the overall ten-year expansion. This period is expected to be characterized by the expansion of cell therapy manufacturing and regenerative medicine applications, the development of advanced composite gelatin membranes with enhanced selectivity, and the growth of specialized implementations for vaccine production and monoclonal antibody purification. The growing adoption of continuous manufacturing processes and integrated bioprocessing systems will drive demand for gelatin filters with enhanced flow rates and comprehensive quality validation.
Between 2020 and 2025, the market experienced steady growth, driven by increasing pharmaceutical quality standards and growing recognition of gelatin filters as essential filtration media for maintaining biological compatibility and protecting sensitive biomolecules in diverse pharmaceutical processing and laboratory filtration applications. The market developed as process engineers and quality control specialists recognized the potential for gelatin filter technology to provide protein-friendly filtration, minimize product losses, and support comprehensive bioburden reduction while meeting regulatory validation requirements. Technological advancement in membrane casting techniques and pore size control began focusing the critical importance of maintaining consistent filtration performance and batch-to-batch reproducibility in regulated manufacturing environments.
| Metric | Value |
|---|---|
| Estimated Value in (2025E) | USD 99.8 million |
| Forecast Value in (2035F) | USD 219.5 million |
| Forecast CAGR (2025 to 2035) | 8.2% |
Market expansion is being supported by the increasing global biopharmaceutical production volumes driven by biologics market growth and personalized medicine development, alongside the corresponding need for biocompatible filtration technologies that can provide gentle biomolecule handling, prevent protein adsorption, and maintain filtration efficiency across various sterile filtration, microbial removal, and clarification applications. Modern pharmaceutical processors and laboratory managers are increasingly focused on implementing gelatin filter solutions that can deliver low protein binding characteristics, support regulatory compliance, and provide reliable contamination control in sensitive biological processes.
The growing focus on product quality and regulatory compliance is driving demand for gelatin filters that can offer validated filtration performance, enable documentation of microbial retention, and ensure comprehensive process consistency across diverse biopharmaceutical applications. Pharmaceutical manufacturer preference for filtration media that combine biological compatibility with proven sterility assurance and operational reliability is creating opportunities for innovative gelatin filter implementations. The rising influence of cell and gene therapy manufacturing and biosimilar production is also contributing to increased adoption of gelatin filters that can provide superior biomolecule preservation without compromising filtration throughput or process economics.
The market is segmented by filtration grade, application, and region. By filtration grade, the market is divided into microfiltration membrane and ultrafiltration membrane. Based on application, the market is categorized into microbial detection, food, pharmaceutical industry, medical research, and others. Regionally, the market is divided into East Asia, Europe, North America, South Asia, Latin America, Middle East & Africa, and Eastern Europe.
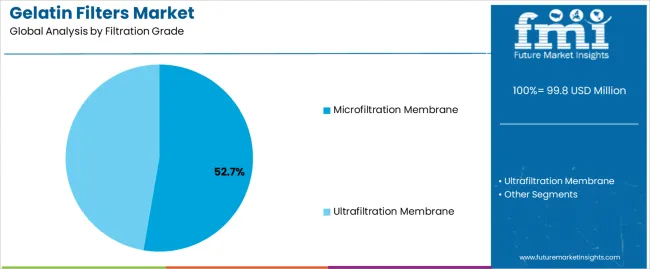
The microfiltration membrane segment is projected to maintain its leading position in the market in 2025 with a 52.7% market share, reaffirming its role as the preferred filtration grade for sterile filtration and particulate removal in pharmaceutical and food processing applications. Pharmaceutical manufacturers and laboratory technicians increasingly utilize gelatin microfiltration membranes for their effective microbial retention capabilities, gentle handling of sensitive biologics, and proven effectiveness in removing bacteria and particles while maintaining protein integrity. Microfiltration membrane technology's proven effectiveness and application versatility directly address the industry requirements for reliable sterile filtration and comprehensive bioburden reduction across diverse pharmaceutical manufacturing and quality control operations.
This filtration grade segment forms the foundation of biopharmaceutical processing, as it represents the pore size range with the greatest contribution to sterility assurance and established validation record across multiple pharmaceutical applications and regulatory frameworks. Pharmaceutical industry investments in sterile manufacturing technologies continue to strengthen adoption among drug manufacturers and contract development organizations. With regulatory requirements demanding robust sterile filtration and validated microbial removal, gelatin microfiltration membranes align with both quality assurance objectives and biocompatibility requirements, making them the central component of comprehensive pharmaceutical filtration strategies.
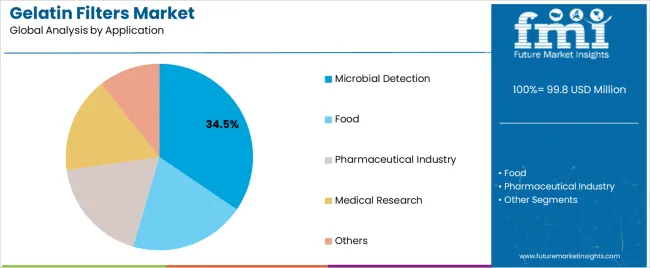
The microbial detection application segment is projected to represent the largest share of gelatin filters demand in 2025 with a 34.5% market share, underscoring its critical role as the primary driver for filter adoption across pharmaceutical quality control laboratories, food safety testing facilities, and environmental monitoring programs. Quality control specialists prefer gelatin filters for microbial detection due to their low background growth characteristics, excellent recovery of microorganisms, and ability to support membrane transfer techniques while enabling reliable enumeration and identification. Positioned as essential materials for modern microbiological analysis, gelatin filters offer both detection sensitivity advantages and handling convenience.
The segment is supported by continuous tightening of pharmaceutical contamination control standards and the growing implementation of rapid microbiological methods that require reliable membrane filtration with enhanced organism recovery and reduced testing time. The pharmaceutical manufacturers are investing in comprehensive microbial monitoring programs to support increasingly stringent sterility requirements and operational demands for real-time bioburden assessment throughout aseptic processing areas and product release testing. As pharmaceutical quality standards advance and food safety awareness increases, the microbial detection application will continue to dominate the market while supporting advanced testing methodologies and quality assurance optimization.
The gelatin filters market is advancing steadily due to increasing biopharmaceutical production volumes driven by biologics market expansion and growing implementation of sterile manufacturing requirements that necessitate filtration technologies providing biocompatible membrane surfaces and reliable microbial retention capabilities across diverse pharmaceutical processing, laboratory testing, and food safety applications. The market faces challenges, including limited chemical resistance compared to synthetic membranes, moisture sensitivity requiring proper storage conditions, and competition from alternative biocompatible filtration materials including polyethersulfone and cellulose derivatives. Innovation in membrane stabilization techniques and composite material development continues to influence product development and market expansion patterns.
The growing biopharmaceutical industry is driving demand for specialized filtration solutions that address sensitive biomolecule handling requirements including minimal protein adsorption, gentle cell separation, and preservation of biological activity during filtration processes. Biopharmaceutical manufacturing applications require gelatin filters that deliver consistent performance across production batches while supporting validation documentation and regulatory compliance objectives. Process development engineers are increasingly recognizing the filtration advantages of gelatin membranes for protecting high-value biologics and maintaining product quality, creating opportunities for pharmaceutical-grade gelatin filters specifically designed for monoclonal antibody production, vaccine manufacturing, and cell therapy processing applications.
Modern gelatin filter manufacturers are incorporating single-use filtration formats and disposable capsule configurations to enhance process flexibility, eliminate cross-contamination risks, and support comprehensive biopharmaceutical manufacturing through pre-sterilized filtration assemblies and ready-to-use filter formats. Leading companies are developing gelatin filters with validated gamma irradiation compatibility, implementing integrity testing capabilities, and advancing capsule designs that provide simplified installation and disposal procedures. These technologies improve manufacturing efficiency while enabling new operational models, including rapid product changeovers, reduced cleaning validation requirements, and flexible production campaigns. Advanced single-use integration also allows manufacturers to support comprehensive contamination prevention objectives and operational simplicity beyond traditional reusable filtration systems.
The expansion of demanding filtration applications and increasing requirements for improved chemical stability is driving development of composite gelatin membranes and reinforced filter structures with enhanced mechanical strength and expanded compatibility ranges. These advanced applications require specialized membrane engineering and support material integration that enable operation under challenging process conditions, creating premium market segments with differentiated value propositions. Manufacturers are investing in membrane reinforcement technologies and stabilization treatments to serve applications requiring extended filtration times and elevated pressure differentials while supporting innovation in continuous manufacturing, high-throughput screening, and automated filtration platforms.
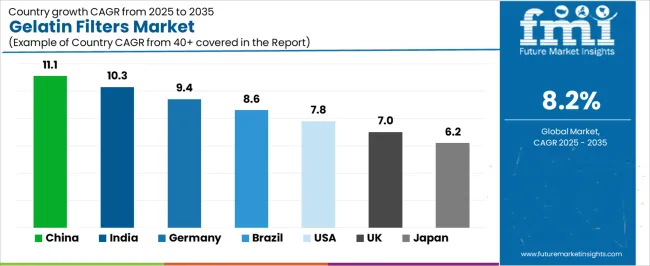
| Country | CAGR (2025-2035) |
|---|---|
| China | 11.1% |
| India | 10.3% |
| Germany | 9.4% |
| Brazil | 8.6% |
| United States | 7.8% |
| United Kingdom | 7.0% |
| Japan | 6.2% |
The market is experiencing solid growth globally, with China leading at a 11.1% CAGR through 2035, driven by expanding biopharmaceutical manufacturing capacity, growing pharmaceutical quality control infrastructure, and increasing adoption of sterile filtration technologies. India follows at 10.3%, supported by pharmaceutical industry expansion, growing biosimilar production, and increasing focus on quality control testing. Germany shows growth at 9.4%, focusing pharmaceutical manufacturing excellence, biotechnology industry leadership, and comprehensive quality assurance standards. Brazil demonstrates 8.6% growth, supported by pharmaceutical market development, food safety regulations, and growing laboratory testing capabilities. The United States records 7.8%, focusing on established biopharmaceutical industry, comprehensive FDA regulations, and advanced pharmaceutical manufacturing. The United Kingdom exhibits 7.0% growth, focusing pharmaceutical research excellence, biotechnology innovation, and comprehensive quality control standards. Japan shows 6.2% growth, supported by pharmaceutical industry maturity, biotechnology advancement, and precision manufacturing capabilities.
The report covers an in-depth analysis of 40+ countries, top-performing countries are highlighted below.
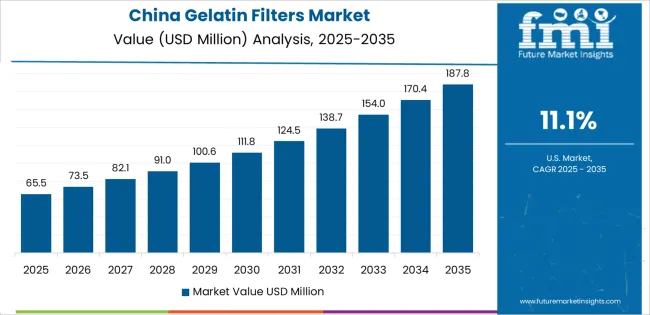
China is projected to exhibit exceptional growth with a CAGR of 11.1% through 2035, driven by massive biopharmaceutical manufacturing expansion and rapidly growing pharmaceutical quality control infrastructure supported by government pharmaceutical industry development policies and healthcare quality improvement initiatives. The country's extensive pharmaceutical production sector and increasing focus on manufacturing quality are creating substantial demand for gelatin filter solutions. Major filtration media manufacturers and international companies are establishing comprehensive production capabilities to serve both domestic pharmaceutical manufacturers and export markets.
India is expanding at a CAGR of 10.3%, supported by the country's pharmaceutical industry expansion, growing biosimilar production capabilities, and increasing adoption of advanced quality control practices among pharmaceutical manufacturers. The country's comprehensive pharmaceutical manufacturing infrastructure and rising quality focus are driving demand for gelatin filter solutions throughout diverse pharmaceutical sectors. Leading filtration media suppliers and pharmaceutical companies are establishing distribution networks and technical support capabilities to address growing demand.
Germany is expanding at a CAGR of 9.4%, supported by the country's pharmaceutical manufacturing excellence, biotechnology industry leadership, and comprehensive quality assurance standards requiring advanced filtration solutions. The nation's pharmaceutical expertise and regulatory framework are driving sophisticated gelatin filter implementations throughout pharmaceutical sectors. Leading pharmaceutical manufacturers and filtration media companies are investing extensively in advanced filtration technology development and quality validation.
Brazil is expanding at a CAGR of 8.6%, supported by the country's pharmaceutical market development, food safety regulations, and growing laboratory testing capabilities. Brazil's healthcare sector expansion and regulatory advancement are driving demand for gelatin filter technologies. Pharmaceutical suppliers and filtration media distributors are investing in market development to serve both pharmaceutical and food testing sectors.
The United States is expanding at a CAGR of 7.8%, supported by the country's established biopharmaceutical industry leadership, comprehensive FDA regulatory framework, and advanced pharmaceutical manufacturing capabilities. The nation's mature pharmaceutical sector and innovation focus are driving demand for sophisticated gelatin filter technologies. Pharmaceutical manufacturers and filtration media suppliers are investing in product development and regulatory compliance to serve demanding pharmaceutical applications.
The United Kingdom is expanding at a CAGR of 7.0%, supported by the country's pharmaceutical research excellence, biotechnology innovation capabilities, and comprehensive quality control standards. The UK's pharmaceutical sector strength and research focus are driving demand for gelatin filter solutions. Pharmaceutical companies and research institutions are establishing advanced filtration programs and quality assurance initiatives.
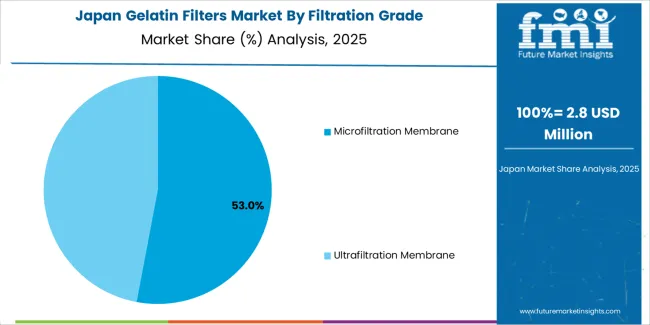
Japan is expanding at a CAGR of 6.2%, supported by the country's pharmaceutical industry maturity, biotechnology advancement, and precision manufacturing capabilities. Japan's quality standards and pharmaceutical expertise are driving demand for high-specification gelatin filter products. Pharmaceutical manufacturers and filtration media suppliers are investing in product development for Japanese pharmaceutical requirements and quality expectations.
What is the market split by country in Europe?
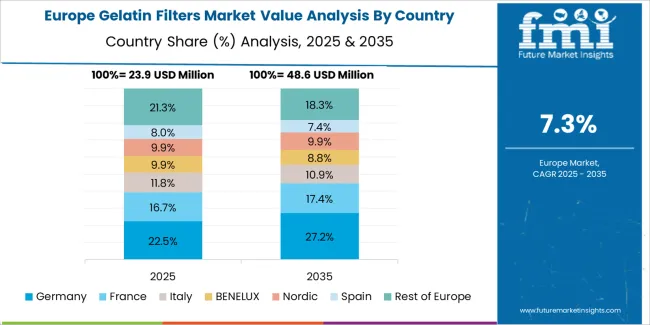
The gelatin filters market in Europe is projected to grow from USD 36.4 million in 2025 to USD 80.2 million by 2035, registering a CAGR of 8.2% over the forecast period. Germany is expected to maintain leadership with a 29.8% market share in 2025, moderating to 29.5% by 2035, supported by pharmaceutical manufacturing excellence, biotechnology industry leadership, and comprehensive quality assurance frameworks.
France follows with 19.0% in 2025, projected at 19.2% by 2035, driven by pharmaceutical industry capabilities, biotechnology research, and food safety testing requirements. The United Kingdom holds 17.3% in 2025, expected to reach 17.0% by 2035 due to pharmaceutical research excellence and biotechnology innovation. Italy commands 13.0% in 2025, rising slightly to 13.2% by 2035, while Spain accounts for 9.8% in 2025, reaching 10.0% by 2035 aided by pharmaceutical manufacturing growth and laboratory testing expansion. Switzerland maintains 4.5% in 2025, up to 4.6% by 2035 due to pharmaceutical industry concentration and biotechnology research excellence. The Rest of Europe region, including Nordics, Central & Eastern Europe, and other markets, is anticipated to hold 6.6% in 2025 and 6.5% by 2035, reflecting steady development in pharmaceutical manufacturing, quality control infrastructure, and biotechnology advancement.
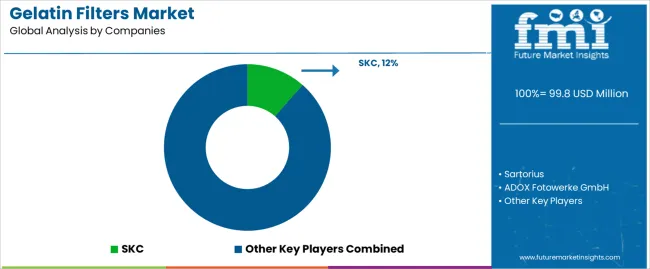
The market is characterized by competition among specialized filtration media manufacturers, pharmaceutical supply companies, and biotechnology material suppliers. Companies are investing in membrane quality optimization, biocompatibility validation, manufacturing scale-up capabilities, and regulatory documentation support to deliver reliable, compliant, and high-performance gelatin filter solutions. Innovation in membrane stabilization, composite material development, and single-use format integration is central to strengthening market position and competitive advantage.
SKC leads the market with comprehensive filtration media solutions focusing on pharmaceutical applications, quality control testing, and validated membrane products across diverse laboratory and manufacturing environments. Sartorius provides advanced bioprocess solutions with focus on single-use technologies and pharmaceutical manufacturing applications. ADOX Fotowerke GmbH delivers specialized gelatin products with focus on European markets and photographic applications. DRM Chemical Process Services offers chemical processing expertise with comprehensive filtration solutions. Dorstener Drahtwerke specializes in technical textiles and filter media manufacturing.
Gelatin filters represent a specialized filtration media segment within pharmaceutical manufacturing and microbiological testing applications, projected to grow from USD 99.8 million in 2025 to USD 219.5 million by 2035 at a 8.2% CAGR. These biocompatible filtration membranes, manufactured from natural gelatin materials for gentle biomolecule handling, serve as critical filtration media in pharmaceutical processing, laboratory testing, and food safety applications where protein compatibility, microbial retention, and biological activity preservation are paramount. Market expansion is driven by increasing biopharmaceutical production, growing pharmaceutical quality control requirements, expanding cell therapy manufacturing, and rising demand for biocompatible filtration solutions across diverse pharmaceutical manufacturers, quality control laboratories, and research institutions.
How Pharmaceutical Regulators Could Strengthen Quality Standards and Validation Requirements?
How Industry Associations Could Advance Best Practices and Technical Standards?
How Gelatin Filter Manufacturers Could Drive Innovation and Market Leadership?
How Pharmaceutical Manufacturers and Laboratories Could Optimize Filtration Processes?
How Research Institutions Could Enable Technology Advancement?
How Investors and Financial Enablers Could Support Market Growth and Innovation?
| Items | Values |
|---|---|
| Quantitative Units (2025) | USD 99.8 million |
| Filtration Grade | Microfiltration Membrane, Ultrafiltration Membrane |
| Application | Microbial Detection, Food, Pharmaceutical Industry, Medical Research, Others |
| Regions Covered | East Asia, Europe, North America, South Asia, Latin America, Middle East & Africa, Eastern Europe |
| Countries Covered | China, India, Germany, Brazil, United States, United Kingdom, Japan, and 40+ countries |
| Key Companies Profiled | SKC, Sartorius, ADOX Fotowerke GmbH, DRM Chemical Process Services, Dorstener Drahtwerke |
| Additional Attributes | Dollar sales by filtration grade and application category, regional demand trends, competitive landscape, technological advancements in membrane stabilization, biocompatibility enhancement, single-use format development, and pharmaceutical validation support |
How big is the gelatin filters market in 2025?
The global gelatin filters market is estimated to be valued at USD 99.8 million in 2025.
What will be the size of gelatin filters market in 2035?
The market size for the gelatin filters market is projected to reach USD 219.5 million by 2035.
How much will be the gelatin filters market growth between 2025 and 2035?
The gelatin filters market is expected to grow at a 8.2% CAGR between 2025 and 2035.
What are the key product types in the gelatin filters market?
The key product types in gelatin filters market are microfiltration membrane and ultrafiltration membrane.
Which application segment to contribute significant share in the gelatin filters market in 2025?
In terms of application, microbial detection segment to command 34.5% share in the gelatin filters market in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.