The Capsule & Tablet Counter market is estimated at USD 1 billion in 2026 and is projected to reach USD 2 billion by 2036, representing a CAGR of 6.5%. Automatic counters dominate the product segment with a 45% share, primarily serving pharmaceutical manufacturing applications at 55%. End users are largely direct OEMs at 45%, complemented by distributors and CMOs/CDMOs. Leading companies in this space include IMA Group, Syntegon, ACG, Romaco, and Marchesini.
Market growth is driven by increasing demand for high-speed, accurate counting solutions in pharma and nutraceutical production. Automation adoption is expanding to improve throughput and reduce human error. Semi-automatic and manual counters remain relevant for smaller-scale operations and after-market service requirements. Technological integration, such as vision systems and connectivity, enhances operational efficiency. Growth is supported by regulatory requirements for precise dosage packaging and rising production capacities globally.
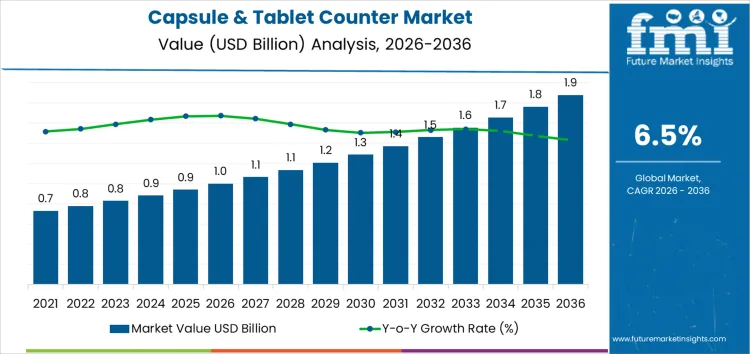
From 2026 to 2031, the capsule and tablet counter market grows from USD 1.0 billion to approximately USD 1.3 billion, representing the early adoption phase. Annual value additions increase from USD 0.08 billion in 2026 to USD 0.10 billion in 2031. Growth is driven by adoption in pharmaceutical manufacturing, contract packaging facilities, and hospital pharmacies, where high-speed counting, accuracy, and reduced manual handling improve operational efficiency. North America and Europe account for roughly 60% of early adoption, with uptake concentrated in high-volume production facilities, validating equipment performance, throughput, and compliance with regulatory counting standards.
Between 2031 and 2036, the market expands from roughly USD 1.3 billion to USD 1.9 billion, illustrating the late adoption phase characterized by portfolio-wide deployment and broad commercialization. Annual increments rise steadily from USD 0.10 billion to USD 0.14 billion, supported by multi-site installations, repeat procurement, and expansion into emerging pharmaceutical markets in Asia Pacific and Latin America. Growth is reinforced by regulatory compliance requirements, demand for high-precision counting, and structural integration of automated counters across global production and packaging facilities. Early growth relied on selective high-value plants, while later expansion reflects normalized adoption and global market penetration.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.0 billion |
| Forecast Value (2036) | USD 1.9 billion |
| Forecast CAGR (2026 to 2036) | 6.5% |
Demand for capsule and tablet counters arose from practical inefficiencies in manual counting and hand packaging rather than from broad automation initiatives alone. Pharmacies, compounding facilities, and small-scale manufacturers traditionally relied on staff to count pills by hand or with simple trays. These methods were slow, error-prone, and increasingly strained as product varieties and prescription volumes grew. Accuracy issues created risks for patient safety, inventory discrepancies, and regulatory non-compliance. Early adoption of dedicated capsule and tablet counters focused on improving throughput and accuracy where manual processes had become untenable rather than across all dispensaries or production lines. Technicians and operators selected equipment that fit existing workflows, with adoption levels tied directly to error rates and workload.
Future demand for capsule and tablet counters will be shaped by operational standardisation, regulatory expectations for traceability, and integration with inventory and dispensing systems rather than by incremental speed improvements alone. Healthcare facilities and pharmaceutical manufacturers are moving toward digital record keeping and automated audit trails to support compliance with drug safety reporting and supply chain transparency. Counters that integrate with software systems to log batches, reconcile stock, and interface with dispensing records will be specified as part of broader quality frameworks. Demand growth will follow defined performance and compliance criteria rather than isolated throughput gains, reinforcing the role of counting technology as an integral control point in medication safety and inventory management.
The capsule and tablet counter market is driven by rising solid dosage production, regulatory pressure on counting accuracy, and the need to improve packaging line efficiency. Demand is concentrated in automated solutions used in regulated manufacturing environments where throughput, traceability, and error reduction are critical. Market leadership reflects operational scale, compliance requirements, and labor optimization rather than basic counting functionality. Adoption patterns are shaped by production volumes and outsourcing trends across pharmaceutical and nutraceutical supply chains.
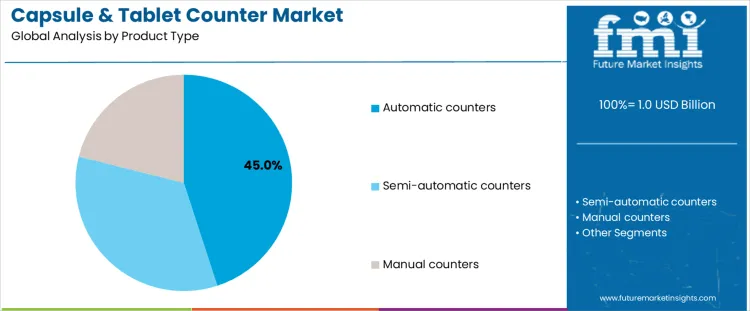
Automatic counters account for about 45% of demand because they align with high speed, high accuracy requirements in modern packaging lines. These systems integrate vision inspection, reject mechanisms, and electronic validation to ensure count accuracy under regulatory scrutiny. Pharmaceutical manufacturers favor automatic counters to reduce manual handling, limit contamination risk, and maintain consistent output across large batch sizes. Their ability to integrate with blister and bottle filling lines further strengthens adoption. Semi-automatic counters serve mid-scale operations where moderate throughput is required but full automation is not justified. They balance cost control with improved accuracy over manual methods. Manual counters remain in use for small batch runs, pilot production, or low volume specialty products but face limitations in speed and repeatability. Product type leadership is therefore driven by compliance intensity, labor efficiency, and scalability requirements rather than basic affordability or mechanical simplicity.
Automatic counters also benefit from longer service life and predictable maintenance cycles, making them suitable for capital intensive environments. Semi-automatic and manual systems persist in niche roles but do not influence overall market direction. As production volumes increase and regulatory oversight tightens, demand continues to shift toward fully automated counting technologies.
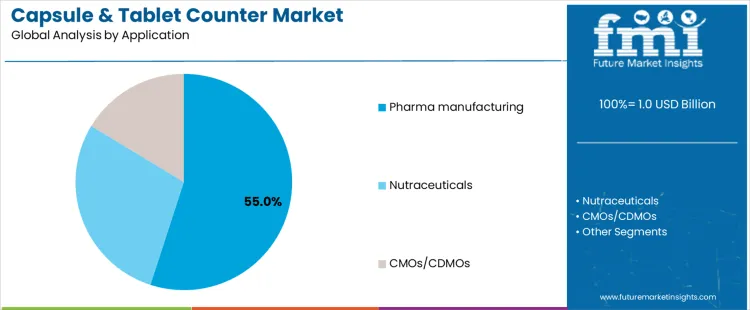
Pharmaceutical manufacturing represents about 55% of demand because solid oral dosage forms remain the dominant drug delivery format. These facilities require precise counting to meet labeling accuracy, serialization, and quality control standards. Automatic counters support continuous production while minimizing deviations and rework. Regulatory audits further reinforce the need for validated counting systems. Nutraceutical manufacturers also contribute to demand but operate under more flexible compliance frameworks, allowing wider use of semi-automatic equipment. Contract manufacturing organizations and CDMOs adopt counters based on client requirements, often investing in automation to meet diverse regulatory expectations across multiple customers.
Pharma manufacturing maintains leadership due to consistent demand for large volume, standardized packaging operations. Nutraceutical applications grow with consumer health product demand but remain more cost sensitive. CMOs and CDMOs expand adoption as outsourcing increases, though their purchasing behavior varies by contract mix. Application leadership therefore reflects regulatory intensity, production scale, and risk management priorities rather than the total number of production sites.
Use appears in manufacturing facilities, compounding pharmacies, and quality control laboratories where accurate counting of solid dosage forms is critical. Automated or semi-automated counters handle capsules and tablets for packaging, quality assurance, and inventory management, ensuring batch consistency and regulatory compliance. Pharmaceutical producers integrate counters into production lines for blister packs, bottles, or sachets. Nutraceutical and supplement manufacturers rely on them to maintain dosage accuracy across high-volume operations. These applications reflect operational and compliance priorities rather than decorative or marketing objectives, with adoption driven by speed, precision, and reliability.
Selection aligns with production lines handling varied shapes, sizes, and coating types. Machines are calibrated to prevent damage to delicate or coated tablets while ensuring counting accuracy. Integration with packaging lines and data capture systems allows traceability and regulatory documentation. Operators and maintenance teams monitor sensors, vibration, and feed rates to maintain consistent throughput. These conditions emerge from operational efficiency, dosage accuracy, and quality control priorities in structured pharmaceutical and nutraceutical workflows.
High initial investment and maintenance requirements can limit adoption in smaller facilities. Counters may require recalibration for different tablet or capsule dimensions. Fragile or irregularly shaped products may be damaged during counting, affecting yield. Compliance with GMP and data integrity regulations adds operational oversight. These factors lead to selective deployment where counting precision, throughput efficiency, and regulatory compliance justify incremental cost and process management.
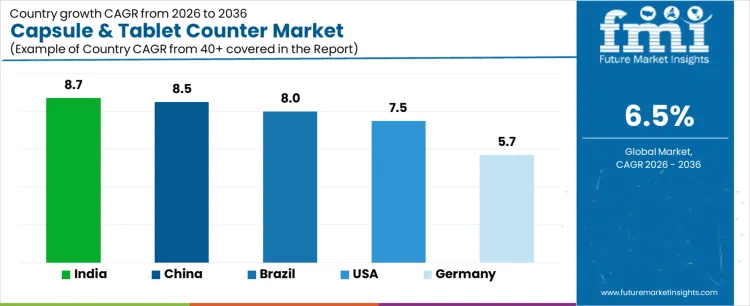
| Country | CAGR (%) |
|---|---|
| USA | 7.5% |
| Brazil | 8.0% |
| China | 8.5% |
| India | 8.7% |
| Germany | 5.7% |
The demand for capsule and tablet counters varies across countries, driven by pharmaceutical manufacturing growth, quality control requirements, and increasing production of oral solid dosage forms. India leads with an 8.7% CAGR, supported by expanding pharmaceutical manufacturing, growing generics production, and rising exports. China follows at 8.5%, driven by large scale pharmaceutical facilities and regulatory compliance for accurate dosing. Brazil records 8.0%, shaped by domestic pharmaceutical production and increasing healthcare demand. The USA grows at 7.5%, reflecting established pharmaceutical infrastructure and steady adoption of automated counting systems. Germany posts 5.7%, supported by mature pharmaceutical markets and regulated manufacturing standards.
In the United States, revenue from the Capsule & Tablet Counter Market is expanding at a CAGR of 7.5% through 2036, driven by adoption of automated counting systems in pharmaceutical manufacturing and packaging facilities. Manufacturers are integrating high-speed counters to improve accuracy, reduce manual errors, and increase production efficiency in tablet and capsule filling operations. Demand is concentrated in pharmaceutical plants, contract manufacturing organizations, and nutraceutical production facilities. Domestic suppliers provide high-performance, processing-compatible counting systems suitable for integration with packaging lines. Regulatory compliance, recurring production, and quality assurance requirements are sustaining predictable adoption nationwide.
In Brazil, the market is growing at a CAGR of 8% through 2036, supported by expanding pharmaceutical production and stricter quality control regulations. Manufacturers are deploying automated capsule and tablet counters to ensure precision in counting, reduce waste, and maintain packaging compliance. Demand is concentrated in pharmaceutical companies, contract manufacturers, and nutraceutical producers. Local suppliers provide high-quality, processing-compatible counting systems suitable for high-speed production lines. Growth in domestic pharmaceutical output, regulatory compliance, and recurring production cycles are sustaining adoption nationwide.
China is witnessing growth at a CAGR of 8.5% through 2036, driven by rapid expansion of pharmaceutical manufacturing and nutraceutical production. Manufacturers are integrating automated capsule and tablet counters to improve speed, accuracy, and reliability in high-volume packaging operations. Demand is concentrated in industrial pharmaceutical plants, contract manufacturing organizations, and functional food producers. Domestic suppliers are scaling high-performance counting systems compatible with various packaging lines. Expanding production infrastructure, quality standards, and recurring packaging cycles are sustaining rapid adoption nationwide.
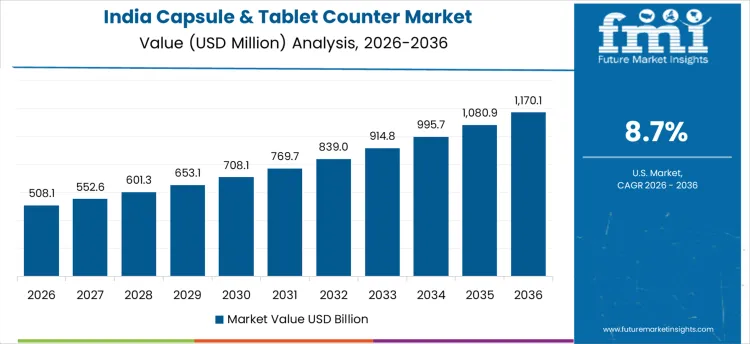
In India, revenue from the Capsule & Tablet Counter Market is expanding at a CAGR of 8.7% through 2036, supported by increasing pharmaceutical output and nutraceutical production. Manufacturers are adopting automated counting systems to ensure precise tablet and capsule packaging, reduce human error, and enhance operational efficiency. Demand is concentrated in pharmaceutical manufacturing units, contract production facilities, and supplement manufacturers. Domestic suppliers provide high-performance, processing-compatible counting systems suitable for integration with production and packaging lines. Growth in industrial production, regulatory compliance, and recurring packaging cycles are sustaining robust adoption nationwide.
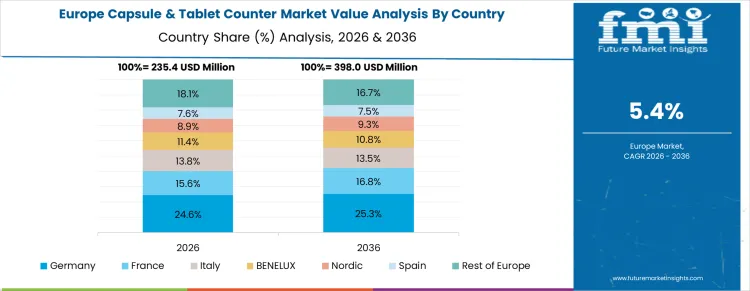
Germany is witnessing growth at a CAGR of 5.7% through 2036, driven by adoption of automated capsule and tablet counting systems in pharmaceutical and nutraceutical manufacturing. Manufacturers are integrating high-precision counters to improve production accuracy, compliance, and packaging efficiency. Demand is concentrated in pharmaceutical plants, contract manufacturers, and functional food producers. Domestic suppliers provide high-quality, processing-compatible counting systems suitable for integration with automated production lines. Regulatory compliance, recurring production, and quality assurance standards are sustaining consistent procurement nationwide.
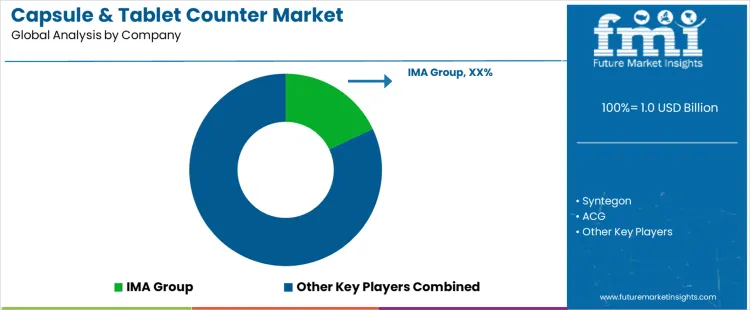
Competition in the capsule and tablet counter market is driven by how reliably machines count and reject units at high speed without disrupting downstream packaging. IMA Group and Marchesini influence this segment through integrated counting platforms designed to align with bottle filling, capping, and inspection stages in regulated pharmaceutical lines. Syntegon approaches the market with vision based counting systems positioned for flexibility across solid dose formats, where changeover time and validation effort matter. ACG participates by aligning counters with its broader solid dosage and packaging equipment ecosystem, emphasizing compatibility and service continuity. Purchasing decisions prioritize count accuracy under dust and vibration, audit readiness, and stable performance during long production runs rather than peak speed claims.
Process ownership and lifecycle support shape competitive outcomes beyond core hardware. Romaco competes by tailoring counting modules to compact line layouts and contract manufacturing environments where space and rapid reconfiguration are critical. Smaller and regional suppliers participate through niche counters or retrofit modules used to upgrade existing lines. Across the market, competitive positioning depends on validation support, documentation quality, and responsiveness during qualification and requalification cycles. End users favor suppliers that minimize downtime during format changes and inspections, reinforcing conservative adoption behavior. Market dynamics reflect regulatory scrutiny, installed base protection, and long capital planning horizons rather than rapid technology turnover or frequent supplier switching.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Product Type | Automatic counters, Semi-automatic counters, Manual counters |
| Application | Pharma manufacturing, Nutraceuticals, CMOs/CDMOs |
| End-Use Industry | Direct OEM sales, Distributors, Aftermarket/services |
| Region | Asia Pacific (China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific), Europe (Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe), North America (United States, Canada, Mexico), Latin America (Brazil, Chile, Rest of Latin America), Middle East & Africa (KSA, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa) |
| Key Companies Profiled | IMA Group, Syntegon, ACG, Romaco, Marchesini, Others |
| Additional Attributes | Dollar by sales by product type, Dollar by sales by application, Dollar by sales by end-user, Dollar by sales by region, Throughput per minute, Counting accuracy and reject rate, Integration with blister and bottle filling lines, Compliance with GMP and serialization, Vision system and sensor calibration, Maintenance and service cycles, Compatibility with capsule/tablet sizes and coatings, Batch tracking and digital audit trails, Adoption in high-volume pharmaceutical plants, Regulatory compliance monitoring, Data logging and software integration, Repeat procurement and replacement cycles, Operational efficiency and labor optimization, Expansion in emerging pharmaceutical and nutraceutical markets |
How big is the capsule & tablet counter market in 2026?
The global capsule & tablet counter market is estimated to be valued at USD 1.0 billion in 2026.
What will be the size of capsule & tablet counter market in 2036?
The market size for the capsule & tablet counter market is projected to reach USD 1.9 billion by 2036.
How much will be the capsule & tablet counter market growth between 2026 and 2036?
The capsule & tablet counter market is expected to grow at a 6.5% CAGR between 2026 and 2036.
What are the key product types in the capsule & tablet counter market?
The key product types in capsule & tablet counter market are automatic counters, semi-automatic counters and manual counters.
Which application segment to contribute significant share in the capsule & tablet counter market in 2026?
In terms of application, pharma manufacturing segment to command 55.0% share in the capsule & tablet counter market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.