Tablet-activated Face Masks Market
Tablet-activated Face Masks Market Analysis - Size, Share, and Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Tablet-Activated Face Masks Market Forecast and Outlook 2026 to 2036
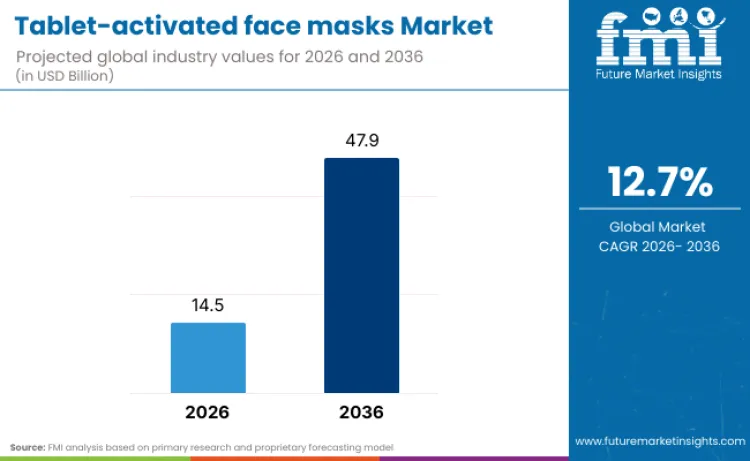
The global tablet-activated face masks market is projected to reach USD 14.5 billion in 2026 and expand to USD 47.9 billion by 2036, registering a CAGR of 12.7%. This growth signals the convergence of two high-impact shifts: consumers increasingly rejecting preservative-laden, pre-soaked sheet masks, and brands responding with dry-to-wet delivery systems that activate only at the point of use. These tablet masks, typically composed of compressed plant-based fibers or biocellulose, eliminate the need for synthetic preservatives, bulky packaging, and excess water content-delivering cleaner, more compact, and travel-friendly skincare rituals.
Product development is also evolving. Brands are infusing tablets with serum-adherent matrices that allow for even absorption of active concentrates such as peptides, probiotics, niacinamide, and ceramides. Paired with refillable glass or mono-material packaging, the format is winning traction among dermatology-led, waterless, and eco-forward beauty startups. Notably, skincare players are integrating tablet systems with ampoule droppers or concentrated serums to let consumers customise hydration levels and layer actives based on skin needs.
"The majority of the water in liquid shampoo, conditioner or face wash is just creating the illusion of value by filling a big bottle... Replacing water-which tends to actually dehydrate skin-with botanical butters and oils will increase the efficacy of the product."
- Brianne West, Founder and CEO of Ethique
The rise of waterless formats like tablet masks aligns with this philosophy. With growing pressure from packaging regulations and tightening restrictions on volatile preservatives, tablet-activated face masks are no longer niche innovations. They are emerging as a new delivery architecture in both clinical and prestige skincare, optimised for efficacy, sustainability, and shelf-stable personalization.
Quick Stats for Tablet-activated Face Masks Market
- Tablet-activated Face Masks Industry Value (2026): USD 14.5 Billion
- Tablet-activated Face Masks Industry Forecast Value (2036): USD 47.9 Billion
- Tablet-activated Face Masks Industry Forecast CAGR: 12.7%
- Tablet-activated Face Masks Industry Leading Product: Compressed Mask Tablets (42.6%)
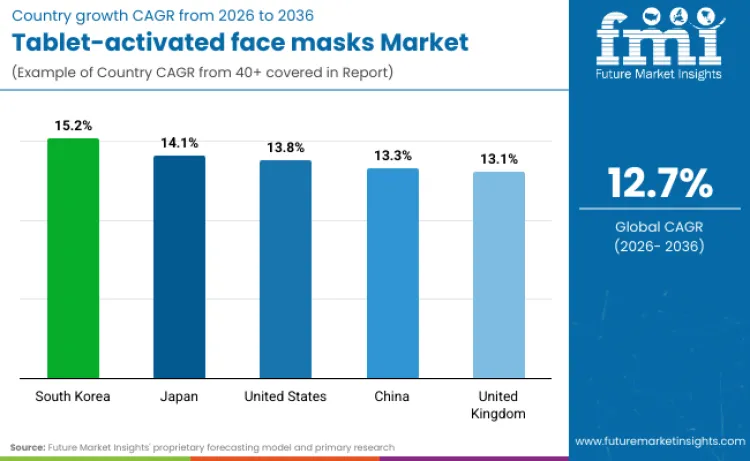
- Tablet-activated Face Masks Industry Key Growth Regions: South Korea, Japan, United States, China
- Tablet-activated Face Masks Industry Key Players: L'Oréal, Estée Lauder, Unilever, Shiseido, P&G
Tablet-activated Face Masks Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 14.5 billion |
| Market Forecast Value (2036) | USD 47.9 billion |
| Forecast CAGR (2026-2036) | 12.7% |
Source: Future Market Insights - analysis driven by proprietary forecasting models and primary research
Key Shift in the Tablet-Activated Face Masks Market During 2026 to 2036
Prior to 2026, tablet-activated masks, specifically dry compressed coins, were largely relegated to the DIY and travel sectors, often dismissed for their lack of inherent efficacy. Consumer confidence was stifled by the manual effort required to hydrate the substrate and the perceived inferiority of add-your-own liquid systems compared to high-performance serum-saturated sheet masks.
By 2026, the segment enters what innovators describe as the Precision Rehydration Era. Advances in freeze-dried flash release technology and stimuli responsive polymers allow these tablets to transform from passive carriers into active delivery systems. This shift enables dry formats to compete on, and occasionally exceed, liquid masks in bioavailability, active ingredient stability, and targeted transdermal penetration.
At the same time, the industrial base is pivoting toward sustainable high throughput manufacturing. Traditional non-biodegradable materials are being replaced by polylactic acid (PLA) and bamboo-fiber matrices that offer 100 percent natural degradation without sacrificing mechanical strength. Global leaders are now localizing production in Asia and North America to reduce carbon intensity and meet stringent environmental regulations, shifting the focus from disposable to compostable high-performance skincare.
What are the Various Segments of the Tablet-activated Face Masks Market?
The tablet-activated face masks market is segmented by product type into compressed mask tablets, powder mask tablets, and multi-actives/enzyme tablets; by function/benefit into hydrating masks, brightening/tone-evening, detox/purifying masks, anti-aging/firming, and acne/oil control; by skin type into combination skin, normal skin, dry skin, oily skin, and sensitive skin; by distribution channel into online retail/e-commerce, beauty specialty stores, mass retailers/supermarkets, and pharmacies/drug stores; by end-user into women, men, and unisex; and by region into Asia Pacific (China, Japan, South Korea, India, Australia & New Zealand, ASEAN, rest of Asia Pacific), Europe (Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, rest of Europe), North America (U.S., Canada, Mexico), Latin America (Brazil, Chile, rest of Latin America), and Middle East & Africa (Kingdom of Saudi Arabia, other GCC countries, Turkey, South Africa, other African Union, rest of Middle East & Africa).
Which Product Type Holds the Largest Share in Tablet-Activated Face Masks?
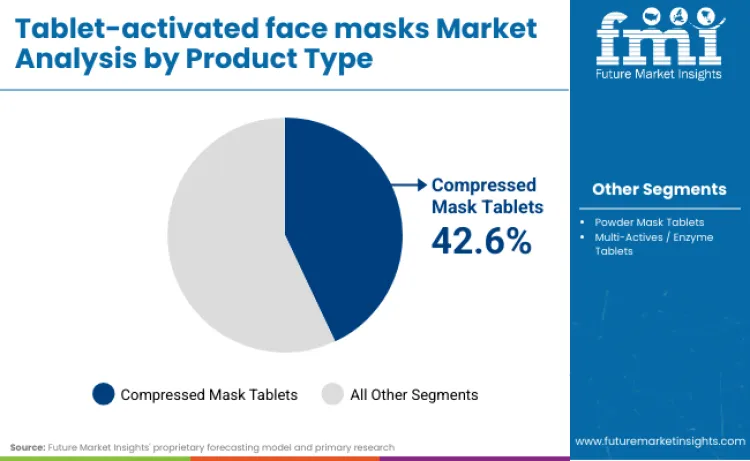
Compressed tablet masks hold the largest share of demand, accounting for 42.6 percent of total market consumption. Their dominance stems from their structural versatility and ability to support a wide range of concentrated actives across dermaceutical and premium skincare. These dry formats are favoured for their compactness, preservative-free delivery, and compatibility with custom hydration systems, especially in regions with high climate variability or retail constraints on liquid formats.
Real brand behavior reflects this strategic concentration. Estée Lauder captures the prestige share of tablet-style demand by utilizing PowerFoil architecture, a compressed, dual-matrix format that acts as a one-way valve to prevent evaporation and force 2X concentrated Hyaluronic Acid into the skin. L’Oréal dominates the tablet-activated sector by leveraging its Fresh Mix patent, which separates high-potency dry masks from liquid activators to prevent the premature degradation of unstable actives like Hyaluronic Acid and Vitamin C.
How Do Skin Type Applications Influence Tablet-Activated Face Mask Preferences?
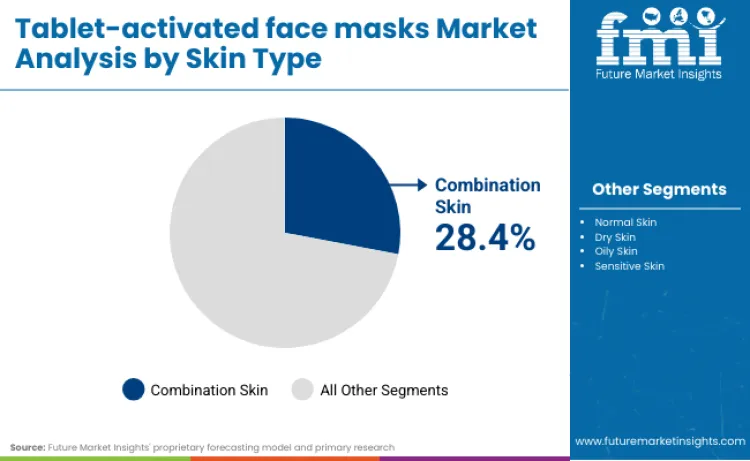
Combination skin represents the largest application segment, contributing 28.4 percent of total demand. The broad appeal comes from its dual-need profile, where oily zones require sebum regulation while dry areas demand hydration. This complexity makes it ideal for customized tablet-activated routines that allow users to vary serum volume, activation time, and ingredient layering.
Real market behavior reflects this formulation logic. Kao Corporation is refining tablet-activated protocols for oily and combination skin by using Sebum RNA (SSL-RNA) monitoring to identify gene expression profiles that dictate barrier function, allowing for Skin Gene Mode determination to guide precise activation levels. Beiersdorf targets oily and acne-prone segments by incorporating pH-independent gelling agents and Sebum Regulating Technology (Decanediol) into mask matrices to ensure that activation does not trigger rebound oiliness or comedogenic responses. Johnson & Johnson optimizes tablet-mask preference for dry skin by utilizing patented separation mechanisms (slits and tabs) in the substrate, which increase skin-to-mask contact (occlusion) to maximize the penetration of moisture-retaining actives like C-xyloside.
What Structural Drivers Make Tablet-Activated Face Masks Win Share?
Tablet-activated face masks gain traction when the adoption barrier shifts from novelty to logistical necessity. The strongest structural driver is transportation and storage density. The waterless tablet format removes the weight and volume of pre-soaked serums, reducing shipping emissions and lowering the carbon footprint across global e-commerce supply chains. This advantage aligns with ISO 14040/14044-compliant Life Cycle Assessments.
A second driver is packaging economics shaped by regulatory fees. Under the UK’s Plastic Extended Producer Responsibility (pEPR), traditional sheet masks in multilayer sachets face Red-rated fees up to £846/tonne. In contrast, tablet masks in bulk recyclable jars qualify for Green-rated discounts and avoid 50% “empty space ratio” penalties under the EU Packaging and Packaging Waste Regulation (PPWR 2025/40).
A third driver is institutional demand from hospitality. The Global Tourism Plastics Initiative mandates the removal of problematic single-use plastics by 2025. Tablet formats enable hotels to eliminate plastic sachets in favour of compact, paper-wrapped alternatives.
These shifts reflect deep supply chain logic. Brands are responding not to trends but to clear structural incentives driven by compliance costs, bulk logistics, and institutional procurement mandates.
Which Constraints Slow Adoption Even When Interest Exists?
Constraints emerge when compliance obligations or product format introduce user or regulatory risk. In the US, the Modernization of Cosmetics Regulation Act (MoCRA) mandates safety substantiation and adverse event reporting. Tablet masks activated by consumers may violate these expectations if mixed with unregulated or unsanitary liquids.
A second constraint is the variability in mask substrate performance. While synthetic options are consistent, biodegradable alternatives like PLA or cellulose require specialised coatings to avoid hygiene issues during activation. A 2025 study shows 70% biodegradation within 45 days, but highlights the need for jade or antimicrobial coatings to maintain skin safety and structural integrity.
Adoption friction arises when brands must redesign formulations, packaging, and safety protocols. Unlike conventional pre-soaked masks, tablet formats involve more than format swapping, they affect ingredient exposure, microbial risk, and legal liability, especially in cross-border launches.
What Recent Shifts Explain Why Momentum Has Increased Lately?
Momentum increased sharply after regulatory timelines converted into active enforcement. As of February 11, 2025, the EU PPWR 2025/40 transitioned from a directive to a binding regulation. This forces multinational brands to adopt harmonised packaging formats that meet recyclability and minimisation standards across all member states.
Simultaneously, material science breakthroughs in nanocellulose and compostable antimicrobials created higher-performance biodegradable substrates. These options now act as functional serum carriers while avoiding plastic stigma, satisfying compostability and barrier requirements in one design.
This is no longer about innovation hype. With pEPR fees rising, the 2030 EU HORECA packaging ban approaching, and high street retailers demanding lower-packaging SKUs, the tablet format benefits from an entire regulatory and logistics system aligning around its core characteristics. It becomes a market default-not a niche innovation.
How Is the Tablet-activated Face Masks Market Evolving Globally?
Global demand for tablet-activated face masks is rising as the category shifts from niche experimentation to routine adoption, supported by formulation advances that make compressed, water-activated masks competitive on ingredient delivery, treatment intensity, active stability, and result consistency. Growth is increasingly anchored in performance-led conversion rather than novelty positioning, with brands scaling compression technology, controlled-release systems, and stabilization platforms to deliver consistent results across skin types. Expansion is also being reinforced by retail mainstreaming and format normalization, as consumers encounter tablet masks in mass beauty doors and structured planograms, reducing discovery friction and increasing repeat usage.
| Country | CAGR (2026-2036) |
|---|---|
| South Korea | 15.2% |
| Japan | 14.1% |
| United States | 13.8% |
| China | 13.3% |
| United Kingdom | 13.1% |
Source: Future Market Insights proprietary forecasting model and primary research
United Kingdom Tablet Activated Face Masks Market Gaining Ground
The United Kingdom tablet activated face masks market is projected to grow at a CAGR of 13.1 percent from 2026 to 2036. Growth is driven by regulatory mechanisms such as the Plastic Packaging Tax and the 2025 to 2026 rollout of Extended Producer Responsibility which penalise pre soaked single use sachets. Brands like L’Oréal and Unilever benefit by switching to compressed tablet masks which reduce liquid volume and packaging weight, key cost centres under UK law.
L’Oréal’s Revitalift Fresh Mix Mask separates serum and tissue to preserve ingredient potency until activation. Boots and Superdrug support this transition by retailing dry mask accessories with refillable essences, creating a walk in retail logic. As compressed formats align with policy led savings and merchandising visibility, the format gains ground beyond eco conscious niche into regulatory compliance infrastructure.
South Korea Tablet Activated Face Masks Market Anchored in Customisation Rituals
The South Korean tablet activated face masks market is expected to grow at a CAGR of 15.2 percent between 2026 and 2036. Growth is rooted in ritualised skincare behaviour and active layering preferences. Consumers use compressed tablets with high performance essences, creating DIY mask packs aligned with Skinification, a strategic priority for players like Shiseido.
Amorepacific brands such as Innisfree integrate compressed mask sets into global retail pipelines, with North American sales up 80 percent in early 2023. The appeal lies in control. Users mix fresh actives on demand and bypass preservatives and format rigidity. Recyclable cotton and dry formats align with South Korea’s high recycling standards. Beauty hubs and e commerce tutorials reinforce the format’s routine role. Mask tablets are no longer accessories. They are format infrastructure inside professionalised skincare routines.
Japan Tablet Activated Face Masks Market Built on Efficacy and Freshness
Japan’s tablet activated face masks market is forecast to grow at a CAGR of 14.1 percent from 2026 to 2036. The format has transitioned from novelty to a precision driven application method within Japanese skincare systems. Shiseido’s 2030 strategy formalises Skinification and treats every step as therapeutic, not cosmetic.
Compressed cotton masks allow control over actives and timing. Consumers combine tablets with toners like Hada Labo Gokujyun. This supports skin barrier targeting with balanced actives. Domestic brands and drugstores distribute multipacks and essence pairings, framing tablets as professional delivery systems. Japan’s high ingredient transparency and storage conscious routines match the dry format’s advantages. Sheet masks in liquid packaging are increasingly seen as inefficient. The compressed alternative wins on format logic, therapeutic trust, and sensory customisation.
United States Tablet Activated Face Masks Market Shaped by Transparency
The United States tablet activated face masks market is projected to expand at a CAGR of 13.8 percent between 2026 and 2036. Ingredient safety and label clarity are the central drivers. The EWG Verified program requires full transparency and exclusion of nearly 2000 substances, including fragrance disclosures.
Retailers such as Sephora, Target, and Ulta list dry masks under their Clean merchandising hubs, creating front of shelf visibility for compressed formats. In response, brands reformulate tablet activated options to exclude silicones, parabens, PFAS, and dyes. Some publish in home usage testing data to validate hydration and irritation claims. Dry tablets provide preservative free delivery and lower environmental load, appealing to clean label shoppers. The US market rewards ingredient assurance and retail trust, turning compressed tablets into the preferred clean format delivery vehicle.
China Tablet Activated Face Masks Market Driven by Skinification and Upgrade
China’s tablet activated face masks market is projected to grow at a CAGR of 13.3 percent from 2026 to 2036. This growth stems from the skinification shift where masks are now evidence backed vehicles for actives rather than pampering accessories. L’Oréal notes that Chinese consumers are adopting more advanced routines with openness to new formats.
Shiseido frames compressed tablets as delivery mechanisms for functional treatments within its global innovation messaging. These dry formats are biodegradable, low preservative, and easy to integrate into e commerce routines. Short form content and skincare influencers demonstrate soak and press rituals, driving both adoption and correct usage. Packaging light benefits align with China’s material reduction targets. The compressed tablet format gains share by uniting ritual, regulation, retail, and online beauty education into a single routine framework.
Which Brands Are Gaining Share in 2026 Beauty Tech and Advanced Skin Systems?
In 2026, the competitive dynamic in beauty tech and advanced skin systems favors players that directly own product development narratives and integrate digital diagnostics, wearable delivery, or biotech-adjacent materials into their skin solutions. Leadership is not purely ingredient-led, it reflects integration between formulation science, delivery innovation, and consumer habit formation.
L’Oréal demonstrates clear leadership in this convergence. Its 2026 flagship launch is a flexible silicone LED Face Mask, developed with I-Smart Developments and positioned under its “Beauty Tech” banner. This launch is not an isolated device, it is marketed as an anti-aging system calibrated to skincare routines and supported by the group’s AI-driven diagnostics and personalized skincare ecosystem. This ownership of both the hardware and the surrounding software loop (e.g., Skin Genius, ModiFace) allows L’Oréal to frame the product as a core infrastructure of high-performance aging care.
Shiseido holds strategic strength in Asia and selective premium markets by investing in “second skin” polymer technologies and skin microbiome science. Through its acquisition of Gallinée, the group reinforces its “Skin Beauty” positioning with prebiotic and probiotic actives. Its "breathable polymer" skin barrier systems show strong appeal in dermatology-adjacent segments.
Unilever, while less active in beauty devices, is innovating in skin delivery systems through generative AI-driven formulation. Its 2026 focus lies in “Serum Shower” formats and microbiome-friendly lines, which deliver sensorial and functional upgrades inside mainstream body care habits.
Each player stakes leadership via distinct innovation levers, hardware delivery (L’Oréal), microbiome-biopolymer fusion (Shiseido), and AI-enabled format agility (Unilever).
Key Players in the Tablet-activated Face Masks Market
- L'Oréal
- Estée Lauder
- Unilever
- Shiseido
- P&G
- Beiersdorf
- Amorepacific
- Johnson & Johnson
- Kao Corporation
- Natura & Co
- Coty Inc.
- Revlon
- Caudalie
- Tatcha
- SK-II
- The Face Shop
- Innisfree
- Laneige
- Sulwhasoo
- Hada Labo
Market Definition
The tablet-activated face masks market refers to revenues generated from solid skincare products formulated in compressed or powdered tablet form that convert into facial mask treatments when activated with water or other liquids. These products are engineered to deliver targeted facial treatment functions including hydration, brightening, detoxification, anti-aging support, and acne control through controlled-release active systems. The market is measured in USD billion and evaluated across the 2026 to 2036 period. Tablet-activated face masks are positioned as primary facial treatment solutions rather than supplemental skincare steps. Classification is based on activation and delivery mechanism rather than ingredient origin. The analysis treats tablet-activated face masks as a standalone skincare category, distinct from sheet masks, clay masks, gel masks, and peel-off treatments. Revenue attribution is limited strictly to products that are explicitly marketed, sold, and used as tablet-activated facial mask treatments within consumer, professional, or clinical skincare settings.
Market Inclusion
Included in the tablet-activated face masks market are finished, ready-to-use tablet mask products sold through online retail, physical beauty stores, professional spa networks, dermatology clinics, and direct-to-consumer platforms. The scope covers hydrating, brightening, detoxifying, anti-aging, and acne-control tablet masks intended for repeated use by consumers or professionals. Products formulated with compressed actives, effervescent activation systems, controlled-release carriers, botanical extracts, peptides, and performance-oriented blends are included when their primary function is facial treatment after liquid activation. Revenues from tablet masks sold within bundled skincare kits, professional spa treatment sets, or subscription programs are included where the tablet mask component is separately identifiable and priced. Geographic coverage spans North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa, with country-level analysis applied where retail penetration, consumer education, or clinical adoption materially influences demand.
Market Exclusion
Excluded from the market are traditional sheet masks, cream masks, clay masks, gel masks, and peel-off masks, regardless of packaging or concentration claims. Tablet cleansers, exfoliating tablets, ingestible supplements, and multipurpose beauty tablets are excluded unless explicitly positioned and functionally used as facial mask treatments. DIY formulations, bulk professional treatments not sold in consumer tablet form, and experimental or prototype products without commercial distribution are outside the scope. Oral beauty products such as collagen tablets, effervescent drinks, or ingestible powders are excluded unless marketed for topical facial mask activation. Revenues from activation accessories, mixing tools, applicators, or packaging innovations are not counted. Spot treatments, eye masks, lip masks, and body treatment tablets intended for non-facial use are excluded from the defined market boundary.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Product Type | Compressed Mask Tablets; Powder Mask Tablets; Multi-Actives/Enzyme Tablets |
| Function/Benefit | Hydrating Masks; Brightening/Tone-Evening; Detox/Purifying Masks; Anti-Aging/Firming; Acne/Oil Control |
| Skin Type Application | Combination Skin; Normal Skin; Dry Skin; Oily Skin; Sensitive Skin |
| Distribution Channel | Online Retail/E-Commerce; Beauty Specialty Stores; Mass Retailers/Supermarkets; Pharmacies/Drug Stores |
| End-User | Women; Men; Unisex |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | South Korea, Japan, United States, China, United Kingdom, France, Germany, Australia, and 40+ countries |
| Key Companies Profiled | L'Oréal; Estée Lauder; Unilever; Shiseido; P&G; Beiersdorf; Amorepacific; Others |
Immersion Cooling Fluid Test Equipment Market by Segment
Test Function:
- Viscosity & Rheology
- Thermal Properties
- Dielectric Strength & Resistivity
- Contamination & Aging Analysis
Fluid Type:
- Synthetic Hydrocarbons
- Esters
- Water Blends
- Fluorinated Fluids (Legacy)
Buyer Type:
- Cooling Fluid Formulators
- Cooling System OEMs
- Data Center Operators
- Independent Test Labs
Application:
- Server Immersion
- Power Electronics Cooling
- Energy Storage
- R&D or Pilot Rigs
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- Personal Care Products Council (PCPC). (2024). Safety and regulatory framework for cosmetic products in the United States. PCPC.
- UK Government, Office for Product Safety and Standards (OPSS). (2024). Guidance on cosmetic product regulation and safety. GOV.UK.
- International Organization for Standardization (ISO). (2023). ISO 22716: Cosmetics - Good Manufacturing Practices (GMP). ISO.
- International Fragrance Association (IFRA). (2024). IFRA standards for safe use of fragrance ingredients in cosmetics. IFRA.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Y-o-Y Growth Trend Analysis 2021 to 2025
- Absolute $ Opportunity Analysis 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Product Type, 2026 to 2036
- Compressed Mask Tablets
- Powder Mask Tablets
- Multi-Actives/Enzyme Tablets
- Y-o-Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Function/Benefit
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Function/Benefit, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Function/Benefit, 2026 to 2036
- Hydrating Masks
- Brightening/Tone-Evening
- Detox/Purifying Masks
- Anti-Aging/Firming
- Acne/Oil Control
- Y-o-Y Growth Trend Analysis By Function/Benefit, 2021 to 2025
- Absolute $ Opportunity Analysis By Function/Benefit, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Skin Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Skin Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Skin Type, 2026 to 2036
- Combination Skin
- Normal Skin
- Dry Skin
- Oily Skin
- Sensitive Skin
- Y-o-Y Growth Trend Analysis By Skin Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Skin Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Online Retail/E-Commerce
- Beauty Specialty Stores
- Mass Retailers/Supermarkets
- Pharmacies/Drug Stores
- Y-o-Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Function/Benefit
- By Skin Type
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- L'Oréal
- Estée Lauder
- Unilever
- Shiseido
- P&G
- Beiersdorf
- Amorepacific
- Johnson & Johnson
- Kao Corporation
- Natura & Co
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Tablet-Activated Face Masks Market Key Takeaways
- Global Tablet-Activated Face Masks Market Size (USD Billion), 2021-2036
- Global Tablet-Activated Face Masks Market Y-o-Y Growth (%), 2021-2036
- Global Tablet-Activated Face Masks Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Global Tablet-Activated Face Masks Market Pricing Analysis (USD/Unit), 2021-2036
- Global Tablet-Activated Face Masks Market Size (USD Billion) By Product Type, 2021-2036
- Global Tablet-Activated Face Masks Market Size (USD Billion) By Function/Benefit, 2021-2036
- Global Tablet-Activated Face Masks Market Size (USD Billion) By Skin Type, 2021-2036
- Global Tablet-Activated Face Masks Market Size (USD Billion) By Distribution Channel, 2021-2036
- Global Tablet-Activated Face Masks Market Size (USD Billion) By Region, 2021-2036
- North America Tablet-Activated Face Masks Market Size (USD Billion) By Country, 2021-2036
- North America Tablet-Activated Face Masks Market Size (USD Billion) By Product Type, 2021-2036
- North America Tablet-Activated Face Masks Market Size (USD Billion) By Function/Benefit, 2021-2036
- North America Tablet-Activated Face Masks Market Size (USD Billion) By Skin Type, 2021-2036
- North America Tablet-Activated Face Masks Market Size (USD Billion) By Distribution Channel, 2021-2036
- North America Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- North America Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- North America Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- North America Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- North America Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- Latin America Tablet-Activated Face Masks Market Size (USD Billion) By Country, 2021-2036
- Latin America Tablet-Activated Face Masks Market Size (USD Billion) By Product Type, 2021-2036
- Latin America Tablet-Activated Face Masks Market Size (USD Billion) By Function/Benefit, 2021-2036
- Latin America Tablet-Activated Face Masks Market Size (USD Billion) By Skin Type, 2021-2036
- Latin America Tablet-Activated Face Masks Market Size (USD Billion) By Distribution Channel, 2021-2036
- Latin America Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- Latin America Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- Latin America Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- Latin America Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- Latin America Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- Western Europe Tablet-Activated Face Masks Market Size (USD Billion) By Country, 2021-2036
- Western Europe Tablet-Activated Face Masks Market Size (USD Billion) By Product Type, 2021-2036
- Western Europe Tablet-Activated Face Masks Market Size (USD Billion) By Function/Benefit, 2021-2036
- Western Europe Tablet-Activated Face Masks Market Size (USD Billion) By Skin Type, 2021-2036
- Western Europe Tablet-Activated Face Masks Market Size (USD Billion) By Distribution Channel, 2021-2036
- Western Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- Western Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- Western Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- Western Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- Western Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- Eastern Europe Tablet-Activated Face Masks Market Size (USD Billion) By Country, 2021-2036
- Eastern Europe Tablet-Activated Face Masks Market Size (USD Billion) By Product Type, 2021-2036
- Eastern Europe Tablet-Activated Face Masks Market Size (USD Billion) By Function/Benefit, 2021-2036
- Eastern Europe Tablet-Activated Face Masks Market Size (USD Billion) By Skin Type, 2021-2036
- Eastern Europe Tablet-Activated Face Masks Market Size (USD Billion) By Distribution Channel, 2021-2036
- Eastern Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- Eastern Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- Eastern Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- Eastern Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- Eastern Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- East Asia Tablet-Activated Face Masks Market Size (USD Billion) By Country, 2021-2036
- East Asia Tablet-Activated Face Masks Market Size (USD Billion) By Product Type, 2021-2036
- East Asia Tablet-Activated Face Masks Market Size (USD Billion) By Function/Benefit, 2021-2036
- East Asia Tablet-Activated Face Masks Market Size (USD Billion) By Skin Type, 2021-2036
- East Asia Tablet-Activated Face Masks Market Size (USD Billion) By Distribution Channel, 2021-2036
- East Asia Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- East Asia Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- East Asia Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- East Asia Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- East Asia Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- South Asia and Pacific Tablet-Activated Face Masks Market Size (USD Billion) By Country, 2021-2036
- South Asia and Pacific Tablet-Activated Face Masks Market Size (USD Billion) By Product Type, 2021-2036
- South Asia and Pacific Tablet-Activated Face Masks Market Size (USD Billion) By Function/Benefit, 2021-2036
- South Asia and Pacific Tablet-Activated Face Masks Market Size (USD Billion) By Skin Type, 2021-2036
- South Asia and Pacific Tablet-Activated Face Masks Market Size (USD Billion) By Distribution Channel, 2021-2036
- South Asia and Pacific Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- South Asia and Pacific Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- South Asia and Pacific Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- South Asia and Pacific Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- South Asia and Pacific Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Tablet-Activated Face Masks Market Size (USD Billion) By Country, 2021-2036
- Middle East & Africa Tablet-Activated Face Masks Market Size (USD Billion) By Product Type, 2021-2036
- Middle East & Africa Tablet-Activated Face Masks Market Size (USD Billion) By Function/Benefit, 2021-2036
- Middle East & Africa Tablet-Activated Face Masks Market Size (USD Billion) By Skin Type, 2021-2036
- Middle East & Africa Tablet-Activated Face Masks Market Size (USD Billion) By Distribution Channel, 2021-2036
- Middle East & Africa Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- Middle East & Africa Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- Middle East & Africa Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- Middle East & Africa Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- Middle East & Africa Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- USA Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- USA Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- USA Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- USA Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- USA Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Canada Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Canada Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Canada Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Canada Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Canada Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Mexico Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Mexico Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Mexico Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Mexico Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Mexico Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Brazil Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Brazil Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Brazil Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Brazil Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Brazil Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Chile Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Chile Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Chile Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Chile Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Chile Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Germany Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Germany Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Germany Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Germany Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Germany Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- UK Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- UK Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- UK Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- UK Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- UK Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Italy Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Italy Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Italy Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Italy Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Italy Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Spain Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Spain Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Spain Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Spain Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Spain Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- France Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- France Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- France Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- France Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- France Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- India Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- India Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- India Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- India Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- India Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- ASEAN Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- ASEAN Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- ASEAN Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- ASEAN Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- ASEAN Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Australia & New Zealand Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Australia & New Zealand Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Australia & New Zealand Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Australia & New Zealand Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- China Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- China Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- China Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- China Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- China Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Japan Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Japan Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Japan Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Japan Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Japan Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- South Korea Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- South Korea Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- South Korea Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- South Korea Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- South Korea Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Russia Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Russia Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Russia Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Russia Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Russia Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Poland Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Poland Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Poland Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Poland Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Poland Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Hungary Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Hungary Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Hungary Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Hungary Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Hungary Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Kingdom of Saudi Arabia Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Kingdom of Saudi Arabia Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Kingdom of Saudi Arabia Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Kingdom of Saudi Arabia Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Turkiye Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- Turkiye Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- Turkiye Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- Turkiye Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- Turkiye Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- South Africa Tablet-Activated Face Masks Pricing Analysis (USD/Unit), 2021-2036
- South Africa Tablet-Activated Face Masks Market Share Analysis (%) By Product Type, 2025
- South Africa Tablet-Activated Face Masks Market Share Analysis (%) By Function/Benefit, 2025
- South Africa Tablet-Activated Face Masks Market Share Analysis (%) By Skin Type, 2025
- South Africa Tablet-Activated Face Masks Market Share Analysis (%) By Distribution Channel, 2025
- Tablet-Activated Face Masks Market Competition Dashboard
- Tablet-Activated Face Masks Market Competition Benchmarking Analysis
- Tablet-Activated Face Masks Market Share Analysis of Top Players By Regional, 2025
- Tablet-Activated Face Masks Market Share Analysis of Top Players By Product Type, 2025
- Tablet-Activated Face Masks Market Share Analysis of Top Players By Function/Benefit, 2025
- Tablet-Activated Face Masks Market Share Analysis of Top Players By Skin Type, 2025
- Tablet-Activated Face Masks Market Share Analysis of Top Players By Distribution Channel, 2025
- L'Oréal - Company Profile
- Estée Lauder - Company Profile
- Unilever - Company Profile
- Shiseido - Company Profile
- P&G - Company Profile
- Beiersdorf - Company Profile
- Amorepacific - Company Profile
- Johnson & Johnson - Company Profile
- Kao Corporation - Company Profile
- Natura & Co - Company Profile
- Assumptions & Acronyms Used in Tablet-Activated Face Masks Market Report
List of Figures
- Tablet-Activated Face Masks Market Size (USD Billion), 2021-2036
- Tablet-Activated Face Masks Market Y-o-Y Growth (%), 2021-2036
- Tablet-Activated Face Masks Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Tablet-Activated Face Masks Market Value Chain Analysis
- Tablet-Activated Face Masks Market Supply Chain Analysis
- Tablet-Activated Face Masks Market Investment Feasibility Matrix
- Tablet-Activated Face Masks Market PESTLE Analysis
- Tablet-Activated Face Masks Market Porter's Five Forces Analysis
- Tablet-Activated Face Masks Market Product Life Cycle Analysis
- Tablet-Activated Face Masks Market Opportunity Map Analysis
- Tablet-Activated Face Masks Market Scenario Forecast Analysis
- Tablet-Activated Face Masks Market Production and Consumption Statistics
- Tablet-Activated Face Masks Market Import and Export Statistics
- Global Tablet-Activated Face Masks Market Share (%) By Product Type, 2025
- Global Tablet-Activated Face Masks Market Share (%) By Product Type, 2036
- Global Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2025
- Global Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2036
- Global Tablet-Activated Face Masks Market Share (%) By Skin Type, 2025
- Global Tablet-Activated Face Masks Market Share (%) By Skin Type, 2036
- Global Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2025
- Global Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2036
- Global Tablet-Activated Face Masks Market Share (%) By Region, 2025
- Global Tablet-Activated Face Masks Market Share (%) By Region, 2036
- Global Tablet-Activated Face Masks Market Attractiveness Analysis By Region
- North America Tablet-Activated Face Masks Market Size (USD Billion), 2021-2036
- North America Tablet-Activated Face Masks Market Share (%) By Country, 2025
- North America Tablet-Activated Face Masks Market Share (%) By Country, 2036
- North America Tablet-Activated Face Masks Market Share (%) By Product Type, 2025
- North America Tablet-Activated Face Masks Market Share (%) By Product Type, 2036
- North America Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2025
- North America Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2036
- North America Tablet-Activated Face Masks Market Share (%) By Skin Type, 2025
- North America Tablet-Activated Face Masks Market Share (%) By Skin Type, 2036
- North America Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2025
- North America Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2036
- North America Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- North America Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- North America Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- North America Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- North America Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- Latin America Tablet-Activated Face Masks Market Size (USD Billion), 2021-2036
- Latin America Tablet-Activated Face Masks Market Share (%) By Country, 2025
- Latin America Tablet-Activated Face Masks Market Share (%) By Country, 2036
- Latin America Tablet-Activated Face Masks Market Share (%) By Product Type, 2025
- Latin America Tablet-Activated Face Masks Market Share (%) By Product Type, 2036
- Latin America Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2025
- Latin America Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2036
- Latin America Tablet-Activated Face Masks Market Share (%) By Skin Type, 2025
- Latin America Tablet-Activated Face Masks Market Share (%) By Skin Type, 2036
- Latin America Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2025
- Latin America Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2036
- Latin America Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- Latin America Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- Latin America Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- Latin America Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- Latin America Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- Western Europe Tablet-Activated Face Masks Market Size (USD Billion), 2021-2036
- Western Europe Tablet-Activated Face Masks Market Share (%) By Country, 2025
- Western Europe Tablet-Activated Face Masks Market Share (%) By Country, 2036
- Western Europe Tablet-Activated Face Masks Market Share (%) By Product Type, 2025
- Western Europe Tablet-Activated Face Masks Market Share (%) By Product Type, 2036
- Western Europe Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2025
- Western Europe Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2036
- Western Europe Tablet-Activated Face Masks Market Share (%) By Skin Type, 2025
- Western Europe Tablet-Activated Face Masks Market Share (%) By Skin Type, 2036
- Western Europe Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2025
- Western Europe Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2036
- Western Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- Western Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- Western Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- Western Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- Western Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- Eastern Europe Tablet-Activated Face Masks Market Size (USD Billion), 2021-2036
- Eastern Europe Tablet-Activated Face Masks Market Share (%) By Country, 2025
- Eastern Europe Tablet-Activated Face Masks Market Share (%) By Country, 2036
- Eastern Europe Tablet-Activated Face Masks Market Share (%) By Product Type, 2025
- Eastern Europe Tablet-Activated Face Masks Market Share (%) By Product Type, 2036
- Eastern Europe Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2025
- Eastern Europe Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2036
- Eastern Europe Tablet-Activated Face Masks Market Share (%) By Skin Type, 2025
- Eastern Europe Tablet-Activated Face Masks Market Share (%) By Skin Type, 2036
- Eastern Europe Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2025
- Eastern Europe Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2036
- Eastern Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- Eastern Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- Eastern Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- Eastern Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- Eastern Europe Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- East Asia Tablet-Activated Face Masks Market Size (USD Billion), 2021-2036
- East Asia Tablet-Activated Face Masks Market Share (%) By Country, 2025
- East Asia Tablet-Activated Face Masks Market Share (%) By Country, 2036
- East Asia Tablet-Activated Face Masks Market Share (%) By Product Type, 2025
- East Asia Tablet-Activated Face Masks Market Share (%) By Product Type, 2036
- East Asia Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2025
- East Asia Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2036
- East Asia Tablet-Activated Face Masks Market Share (%) By Skin Type, 2025
- East Asia Tablet-Activated Face Masks Market Share (%) By Skin Type, 2036
- East Asia Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2025
- East Asia Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2036
- East Asia Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- East Asia Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- East Asia Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- East Asia Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- East Asia Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- South Asia and Pacific Tablet-Activated Face Masks Market Size (USD Billion), 2021-2036
- South Asia and Pacific Tablet-Activated Face Masks Market Share (%) By Country, 2025
- South Asia and Pacific Tablet-Activated Face Masks Market Share (%) By Country, 2036
- South Asia and Pacific Tablet-Activated Face Masks Market Share (%) By Product Type, 2025
- South Asia and Pacific Tablet-Activated Face Masks Market Share (%) By Product Type, 2036
- South Asia and Pacific Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2025
- South Asia and Pacific Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2036
- South Asia and Pacific Tablet-Activated Face Masks Market Share (%) By Skin Type, 2025
- South Asia and Pacific Tablet-Activated Face Masks Market Share (%) By Skin Type, 2036
- South Asia and Pacific Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2025
- South Asia and Pacific Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2036
- South Asia and Pacific Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- South Asia and Pacific Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- South Asia and Pacific Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- South Asia and Pacific Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- South Asia and Pacific Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Tablet-Activated Face Masks Market Size (USD Billion), 2021-2036
- Middle East & Africa Tablet-Activated Face Masks Market Share (%) By Country, 2025
- Middle East & Africa Tablet-Activated Face Masks Market Share (%) By Country, 2036
- Middle East & Africa Tablet-Activated Face Masks Market Share (%) By Product Type, 2025
- Middle East & Africa Tablet-Activated Face Masks Market Share (%) By Product Type, 2036
- Middle East & Africa Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2025
- Middle East & Africa Tablet-Activated Face Masks Market Share (%) By Function/Benefit, 2036
- Middle East & Africa Tablet-Activated Face Masks Market Share (%) By Skin Type, 2025
- Middle East & Africa Tablet-Activated Face Masks Market Share (%) By Skin Type, 2036
- Middle East & Africa Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2025
- Middle East & Africa Tablet-Activated Face Masks Market Share (%) By Distribution Channel, 2036
- Middle East & Africa Tablet-Activated Face Masks Market Attractiveness Analysis By Country
- Middle East & Africa Tablet-Activated Face Masks Market Attractiveness Analysis By Product Type
- Middle East & Africa Tablet-Activated Face Masks Market Attractiveness Analysis By Function/Benefit
- Middle East & Africa Tablet-Activated Face Masks Market Attractiveness Analysis By Skin Type
- Middle East & Africa Tablet-Activated Face Masks Market Attractiveness Analysis By Distribution Channel
- USA Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Canada Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Mexico Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Brazil Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Chile Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Germany Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- UK Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Italy Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Spain Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- France Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- India Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- ASEAN Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- China Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Japan Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- South Korea Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Russia Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Poland Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Hungary Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Turkiye Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- South Africa Tablet-Activated Face Masks Pricing Trend Analysis (USD/Unit), 2021-2036
- Tablet-Activated Face Masks Market Competitive Landscape
- Tablet-Activated Face Masks Market Structure Analysis
- Tablet-Activated Face Masks Market Competition Dashboard
- Tablet-Activated Face Masks Market Competition Benchmarking Analysis
- Tablet-Activated Face Masks Market Share Analysis of Top Players By Regional
- Tablet-Activated Face Masks Market Share Analysis of Top Players By Product Type
- Tablet-Activated Face Masks Market Share Analysis of Top Players By Function/Benefit
- Tablet-Activated Face Masks Market Share Analysis of Top Players By Skin Type
- Tablet-Activated Face Masks Market Share Analysis of Top Players By Distribution Channel
- L'Oréal - Revenue Analysis (USD Billion), 2019-2025
- Estée Lauder - Revenue Analysis (USD Billion), 2019-2025
- Unilever - Revenue Analysis (USD Billion), 2019-2025
- Shiseido - Revenue Analysis (USD Billion), 2019-2025
- P&G - Revenue Analysis (USD Billion), 2019-2025
- L'Oréal - Market Position Analysis
- Estée Lauder - Market Position Analysis
- Unilever - Market Position Analysis
- Shiseido - Market Position Analysis
- P&G - Market Position Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

