AI Capsule Endoscopy Reading Platform Market
The AI Capsule Endoscopy Reading Platform Market is segmented by Platform type (Cloud, On-premise, Hybrid), Capsule type (Small bowel, Colon, Pan-enteric, Stomach), Core function (Lesion detection, Workflow triage, Report drafting, Cleansing scoring), End user (Hospitals, GI clinics, Ambulatory centers, Academic centers), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
AI Capsule Endoscopy Reading Platform Market Size, Market Forecast and Outlook By FMI
The AI capsule endoscopy reading platform market garnered a value of USD 71.9 million in 2025 and is estimated to reach at USD 82.0 million in 2026. Sector is projected to advance at a CAGR of 14.10% from 2026 to 2036, taking total valuation to USD 306.7 million by the end of the forecast period. Wider adoption across clinical gastroenterology is tied to one clear need i.e., reducing the time burden of manual capsule video review. Automated bounding-box protocols are becoming more relevant as departments try to manage higher case volumes without adding the same level of physician review time.
Summary of AI Capsule Endoscopy Reading Platform Market
- AI Capsule Endoscopy Reading Platform Market Snapshot
- The AI capsule endoscopy reading platform market is estimated at USD 71.9 million in 2025 and is projected to reach USD 306.7 million by 2036.
- The sector is forecast to expand at a 14.1% CAGR from 2026 to 2036, creating an incremental opportunity of USD 224.7 million over the period.
- This market sits within the capsule endoscopy software layer and centers on frame prioritization, lesion detection, faster study review, and report support rather than capsule hardware.
- The industry outlook remains shaped by compliance requirements and physician oversight, as FDA-cleared AI tools still need clinician review of the full study where required.
- AI Capsule Endoscopy Reading Platform Market Demand and Growth Drivers
- AI tools are gaining traction in routine workflow as they solve time restraints for the stakeholders. Published multicentre evidence supports their value in cutting review time while improving detection of bleeding-related findings.
- Small-bowel bleeding work remains the strongest adoption point because ESGE guidance already places small-bowel capsule endoscopy in a first-line diagnostic role.
- Cloud-connected reading setups are moving ahead because they support remote access, multi-site interpretation, and easier software updates than fixed desktop-based systems.
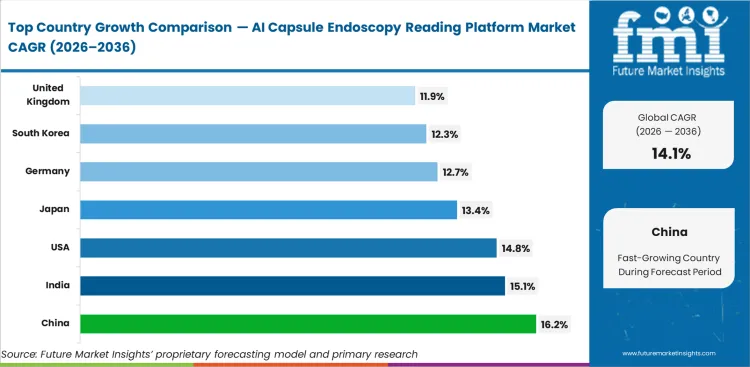
- China is set to lead key national markets at 16.2% CAGR by 2036, followed by India at 15.1%, the United States at 14.8%, Japan at 13.4%, Germany at 12.7%, South Korea at 12.3%, and the United Kingdom at 11.9%.
- The pace of rollout still depends on reimbursement clarity for AI software, legal caution around missed findings, and local validation requirements before hospitals shift away from established reading practice.
- AI Capsule Endoscopy Reading Platform Market Product and Segment View
- The market includes cloud, on-premise, and hybrid reading platforms used across small-bowel, colon, stomach, and pan-enteric capsule studies.
- Cloud platforms are expected to account for 42% share in 2026. Their lead comes from better fit with remote reading, cross-site case review, and simpler software maintenance.
- Small bowel is projected to secure 51% share in 2026, supported by the strongest clinical evidence base and the first cleared AI use case. This keeps commercial focus concentrated on that study type.
- Hospitals tend to adopt AI reading tools first when the purchase case rests on diagnostic confidence and quicker review. In 2026, lesion detection is expected to contribute 48% of total market share within the core function segment.
- Higher study throughput and access to GI specialists support buying decisions in larger care settings. The hospitals segment is likely to represent 44% of the market in 2026, which keeps this user group at the center of demand analysis.
- The market is still operating under physician-led review models, which limits the room for fully autonomous interpretation. Reader-assist is forecast to represent 57% share in 2026 within the deployment model segment, and that keeps commercial adoption tied to assisted rather than independent reading.
- The scope covers AI-assisted capsule reading software, cloud readers, workflow triage, lesion-detection engines, and reporting support. It does not include capsule hardware revenue, patency capsules, therapeutic enteroscopy, or non-capsule GI AI platforms.
- AI Capsule Endoscopy Reading Platform Market Geography and Competitive Outlook
- China, India, and the United States are the fastest-rising national markets, while Japan and Germany remain important high-value bases because their GI care systems are already well established.
- The competitive environment is defined by regulatory clearance, cloud-based workflow compatibility, dataset quality, and how well a platform connects with the installed base of capsule endoscopy systems.
- The sector remains fragmented, with market positions still taking shape as hospitals and specialist centers evaluate software performance, clinical reliability, and integration fit before standardizing adoption.
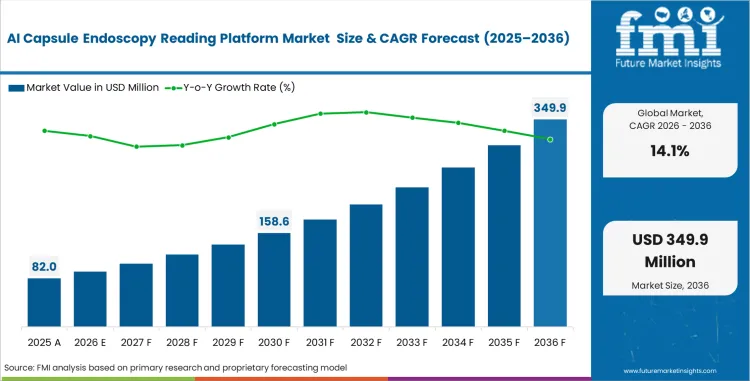
AI Capsule Endoscopy Reading Platform Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 82.0 million |
| Industry Value (2036) | USD 306.7 million |
| CAGR (2026–2036) | 14.10% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Manual assessment of a single small bowel study can take close to 45 minutes, which places real pressure on routine screening economics. Reimbursement pressure across major healthcare systems makes that model harder to sustain, especially where departments are already dealing with reporting delays. Reading software improves workflow by removing large volumes of normal mucosa frames before physician review begins, which can cut interpretation time to less than 10 minutes. Clinical teams that postpone adoption are more likely to face constrained throughput and a heavier backlog across scheduled studies.
Integration quality also shapes the industry outlook more than it first appears. Standalone tools add friction because video files still need to be uploaded manually before review can begin. Native integration with endoscopy visualization systems or PACS environments gives physicians direct access within existing clinical workflow, which makes implementation more practical at department level.
Adoption tends to accelerate once validation work supports a strong negative predictive value for active bleeding. High-volume clinics start changing their workflow at that point, especially when algorithm triage is considered reliable enough to clear straightforward negative studies without the same level of secondary review. That shift changes the role of the software materially. What begins as a support tool for faster reading becomes a more central diagnostic layer in routine capsule endoscopy practice.
Growth in the AI capsule endoscopy reading platform market reflects clear regional priorities. China is projected to expand at a 16.2% CAGR through 2036 as high‑volume urban hospitals rely on automated triage to manage screening demand. India is projected to rise at 15.1% CAGR by 2036, supported by private diagnostic chains scaling centralized remote reading hubs. In the United States, 14.8% CAGR expansion is owing to value‑based care models that penalize missed early‑stage lesions, while Japan’s 13.4% expansion is set to be driven by structured gastric cancer screening mandates. Germany (12.7%), South Korea (12.3%), and the United Kingdom (11.9%) show steadier growth, suggesting that adoption will be grounded in clinical accuracy and centralized interpretation models.
Segmental Analysis
AI Capsule Endoscopy Reading Platform Market Analysis by Platform type
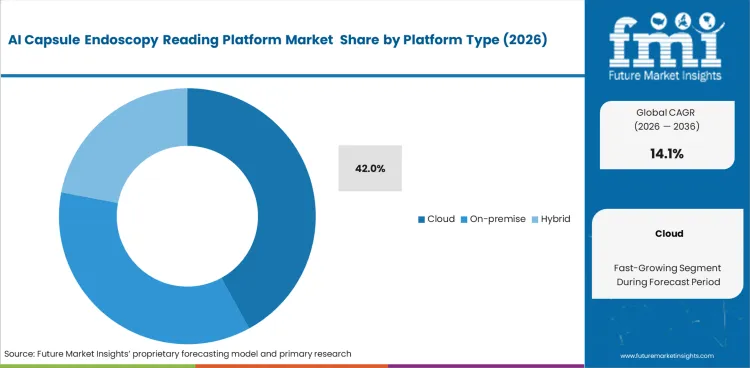
Deployment models favor remote hosting as neural networks require heavy computational power for rapid video processing. The cloud segment is expected to account for 42.0% share in 2026. IT directors at mid-size practices prefer paying per-study fees rather than carrying capital expenditure for onsite GPUs. Remote processing enables continuous algorithm improvements without requiring hospital staff to install local updates. One hidden operational reality involves data privacy regulations. Sending unencrypted patient anatomy videos across external networks violates compliance protocols, forcing vendors to anonymize files before transmission. Medical facilities delaying this transition face deteriorating processing speeds as new, more complex mobile endoscopic workstations models outgrow aging local hardware. FMI also notes that clinical ai model governance rules heavily favor centralized models for version control. These centralization policies ensure that all affiliated clinics operate on the identical diagnostic standard.
- Early adopters: High-volume screening centers implemented remote processing first to manage video backlogs. IT directors gained immediate processing capacity without requesting additional server room space.
- Secondary wave: Mid-sized gastroenterology practices follow suit to access enterprise-grade algorithms previously unaffordable. Clinical managers convert variable labor costs into predictable software subscription expenses.
- Late converts: Rural clinics eventually transition once reliable high-speed internet reaches remote areas. Rural administrators finally achieve diagnostic parity with major urban academic centers.
AI Capsule Endoscopy Reading Platform Market Analysis by Capsule type
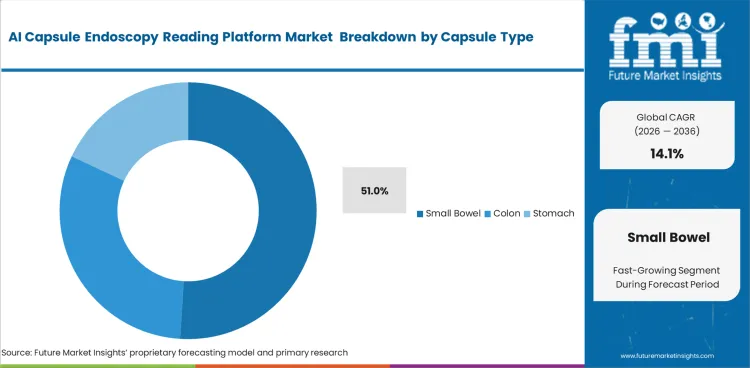
Gastroenterologists deciding on software focus entirely on anatomical regions where manual review fails most frequently. Small bowel algorithms are projected to secure 51.0% share in 2026 because human concentration degrades over eight-hour video files showing identical villi formations. Locating obscure angioectasia hidden within millions of similar-looking frames represents an ideal task for computer vision. Algorithms highlight single bleeding pixels that fatigued human eyes routinely miss. What share figures obscure is algorithm specificity drops sharply when bowel prep quality is poor. Software flags food debris as potential tumors. Physicians choosing platforms lacking advanced debris-filtering capabilities spend more time dismissing false positives than they previously spent conducting manual small bowel enteroscopes reviews. Clinics failing to assess these filtering capabilities end up with bloated review times.
- Optimal conditions: Algorithms demonstrate exceptional sensitivity on perfectly cleansed mucosa. Gastroenterologists detect subtle flat lesions with near-perfect accuracy under ideal preparation protocols.
- Edge limitations: Bile stains and opaque fluids trigger false positive cascades. Clinical readers must manually override algorithms constantly when patients fail strict fasting requirements.
- Acceptability thresholds: Hospitals require software to maintain positive predictive values above ninety percent regardless of preparation quality. Purchasing committees reject platforms generating excessive alerts that slow down final report generation.
AI Capsule Endoscopy Reading Platform Market Analysis by Core function
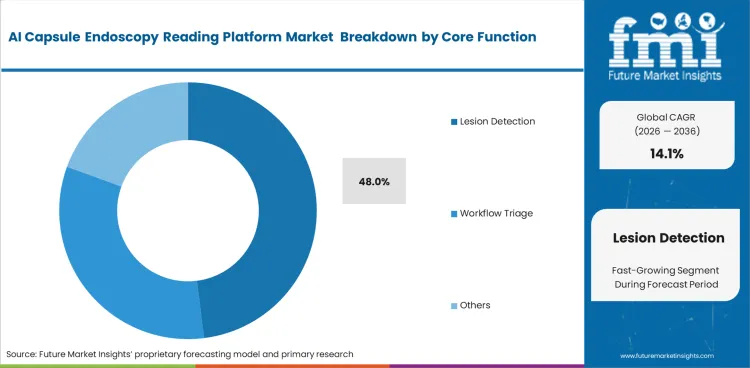
Regulatory clearance pathways dictate software functionality priorities across most major health systems. The lesion detection category is likely to represent 48.0% of the market in 2026 by directly improving diagnostic yield metrics required for value-based care bonuses. Clinical directors justify flexible endoscopes software purchases strictly on its ability to find pathology that human readers miss. Automated bounding boxes draw attention to suspicious areas immediately upon video upload. A practitioner reality remains that highly sensitive detection software generates significant anxiety for physicians who must legally document why they disagreed with an algorithmic warning. Clinics ignoring detection upgrades risk malpractice exposure when subsequent traditional procedures discover advanced cancers missed during manual pill reviews. Legal departments now view this software as essential risk mitigation.
- Purchasing savings: Software subscriptions cost less than hiring additional specialized gastroenterologists to handle rising video volumes. Financial officers reduce diagnostic labor spend while simultaneously increasing total screening capacity.
- Hidden operation expenses: IT departments incur data storage fees keeping annotated videos for legal compliance. System administrators must upgrade network capacity to handle rapid transmission of marked frames.
- Lifecycle comparison: Initial software licensing appears expensive compared to manual reading. Long-term analysis proves automated detection prevents costly missed-diagnosis lawsuits and reduces repeat procedures.
AI Capsule Endoscopy Reading Platform Market Analysis by End user
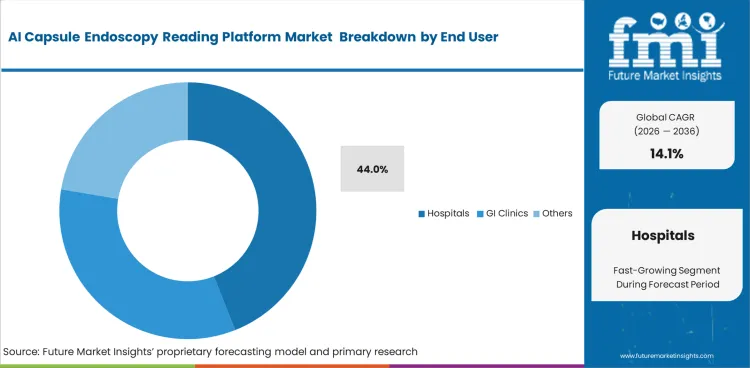
Standalone reading stations are rapidly disappearing from modern clinical environments. Hospitals are expected to contribute 44.0% of total market share in 2026 by integrating diagnostic AI directly into central electronic medical records. Independent GI specialists struggle to afford enterprise-level endoscopy video systems licenses, driving consolidation. Large academic centers deploy these tools across multiple campuses to standardize diagnostic quality regardless of which attending physician reads a study. Pure efficiency gains vanish inside large institutions because rigid bureaucratic workflows require junior doctors to review algorithm output before senior attendings provide final sign-off. Independent practices clinging to manual review lose referral networks as referring physicians demand standardized, AI-annotated reports. Larger health systems absorb these smaller practices at an accelerating pace.
- Integration failure: Platforms unable to connect seamlessly with existing PACS software create isolated data silos. IT directors ban standalone applications that force doctors to use separate login credentials.
- Residual liability: Algorithms missing obscure pathology transfer legal responsibility entirely to reviewing physicians. Risk managers must maintain extensive malpractice coverage despite deploying highly accurate diagnostic software.
- Capture requirement: Institutions must implement continuous physician training on software updates to capture full efficiency benefits. Department heads mandating new tools without workflow redesigns experience zero reduction in study turnaround times.
AI Capsule Endoscopy Reading Platform Market Analysis by Deployment model
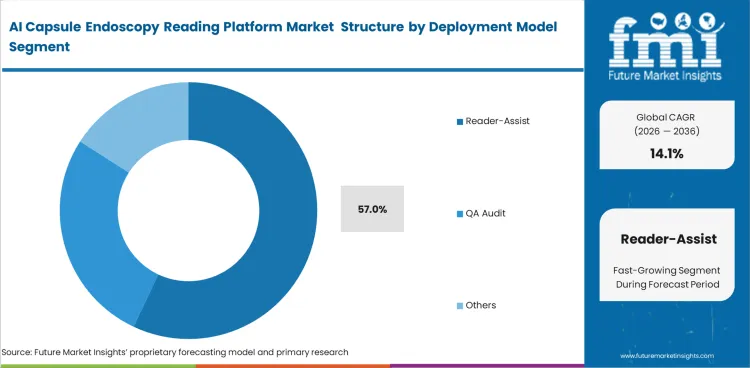
As current FDA guidelines require a human physician to make all final clinical determinations. Software acts merely as a highly advanced filter, pulling suspicious frames into a condensed timeline for rapid human validation. Near-term adoption is likely to remain centered on physician-supervised reader-assist models rather than fully autonomous interpretation. Reader-assist frameworks are anticipated to emerge with 57.0% market share in 2026. This setup protects visualization system components manufacturers from direct medical liability. Generalists assume AI will soon replace doctors entirely, but actual clinical implementation shows software is being used to upskill junior readers to expert levels rather than eliminate reading jobs. Facilities attempting fully autonomous workflows face immediate billing rejections from insurance providers demanding physician signatures. Revenue cycles depend entirely on these manual sign-offs.
- Initial trigger: Backlogged screening queues force clinical directors to seek efficiency tools. Reading delays stretching beyond two weeks typically initiate urgent software evaluations.
- Validation criteria: Purchasing committees demand independent peer-reviewed studies proving algorithms match expert human sensitivity. Hospital administrators require hard data showing software performs consistently across diverse patient demographics.
- Expansion driver: Successful implementation in small bowel departments usually prompts rapid expansion into colon capsule applications. Chief medical officers renew licenses easily once initial deployment proves measurable reductions in physician overtime hours.
AI Capsule Endoscopy Reading Platform Market Drivers, Restraints, and Opportunities
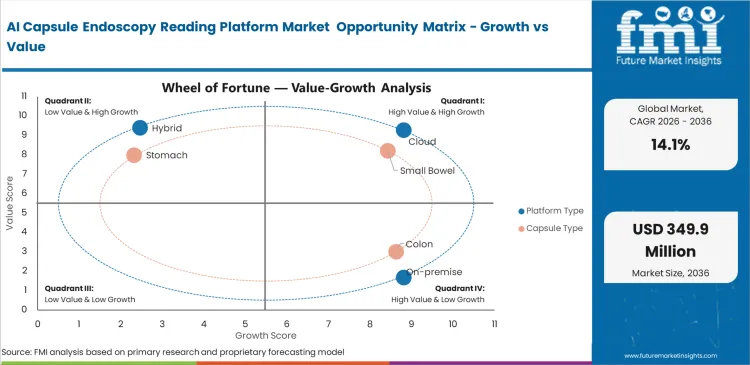
Severe gastroenterologist shortages force large medical networks to rethink diagnostic capacity. Clinical directors cannot physically process rising capsule video volumes using manual frame-by-frame techniques without delaying critical diagnoses. Reader-assist algorithms solve this immediate bottleneck by condensing an eight-hour video into a five-minute highlight reel. Buyer interest rises when AI triage can shorten review time enough to ease backlog pressure without adding specialist headcount. Delaying this transition leaves clinics completely unable to handle demographic-driven surges in gastric cancer screening requirements.
Data interoperability blocks seamless deployment across fragmented hospital networks. Software platforms often struggle to communicate with legacy PACS setups, forcing doctors to review videos on separate, non-integrated monitors. This disconnected workflow destroys efficiency gains because physicians must manually re-enter findings into official electronic medical records. API standardization remains poor across competing health systems. Until algorithm vendors build universal artificial intelligence healthcare integration tools, IT departments will continue rejecting advanced diagnostic software that disrupts established charting procedures.
Opportunities in the AI Capsule Endoscopy Reading Platform Market
- Bleeding detection specialization: Algorithms trained exclusively to identify obscure gastrointestinal bleeding secure expedited regulatory clearance. Emergency department directors gain rapid triage tools for critical patients.
- Cleansing score automation: Software automatically grading bowel preparation quality eliminates subjective human assessments. Clinical auditors standardize endoscope reprocessing devices protocol evaluations across entire hospital networks.
- Video summarization: Platforms generating written clinical narratives from visual data save doctors immense reporting time. Hospital administrators reduce administrative burden on over-worked specialists.
Regional Analysis
.webp)
Based on regional analysis, AI Capsule Endoscopy Reading Platform Market is segmented into East Asia, South Asia, North America, and Europe across 40 plus countries.
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 16.2% |
| India | 15.1% |
| United States | 14.8% |
| Japan | 13.4% |
| Germany | 12.7% |
| South Korea | 12.3% |
| United Kingdom | 11.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific AI Capsule Endoscopy Reading Platform Market Analysis
Asia Pacific remains central to the industry outlook because large screening populations and uneven specialist availability make manual capsule review difficult to scale. Health systems across the region are under pressure to process high study volumes without slowing diagnostic turnaround. That is pushing hospitals and diagnostic networks toward cloud-based reading platforms that can route raw capsule data from smaller centers to larger interpretation hubs. Speed, workflow stability, and the ability to handle daily case loads matter more here than highly specialized feature depth.
- China: Rural screening programs supported by the public health system generate a very large volume of capsule video, which keeps software throughput and case triage at the center of adoption decisions. Urban academic hospitals are using algorithm-assisted review to separate routine studies from cases needing closer clinical attention. The China market is set to advance at a CAGR of 16.2% through 2036. Suppliers that secure large hospital networks are likely to benefit from long contract cycles and a deeply embedded installed base.
- India: The industry outlook in India points to 15.1% CAGR through 2036. Private diagnostic chains are expanding remote reading models to connect smaller clinics with centralized interpretation capacity. Standardization matters in this setting because operators need consistent output across dispersed locations with uneven specialist coverage. The market is likely to stay favorable for platforms that can support scale without adding heavy operating complexity.
- Japan: National screening discipline and an aging patient base keep diagnostic workloads high across the country’s GI care network. Hospitals are adopting capsule reading tools to improve case handling efficiency and reduce pressure on specialist teams reviewing large image volumes. Japan is expected to record 13.4% CAGR in the market during the forecast period. Adoption is likely to remain concentrated in settings where workflow speed and diagnostic consistency carry equal weight.
- South Korea: Adoption in South Korea is anticipated to move ahead at a CAGR of 12.3% during the assessment period. Domestic electronics capability gives the country an advantage in aligning capsule hardware with reading software and broader digital clinical systems. Local clinics and hospital groups are upgrading diagnostic workflows with platforms that fit established technology systems and service support. Local technical alignment should continue to support platform preference across major care networks.
The FMI’s report includes detailed tracking of adoption patterns across China, India, Japan, and South Korea. Differences in screening volume, specialist availability, and digital health capacity continue to influence how software platforms expand across Asia Pacific diagnostic networks.
North America AI Capsule Endoscopy Reading Platform Market Analysis
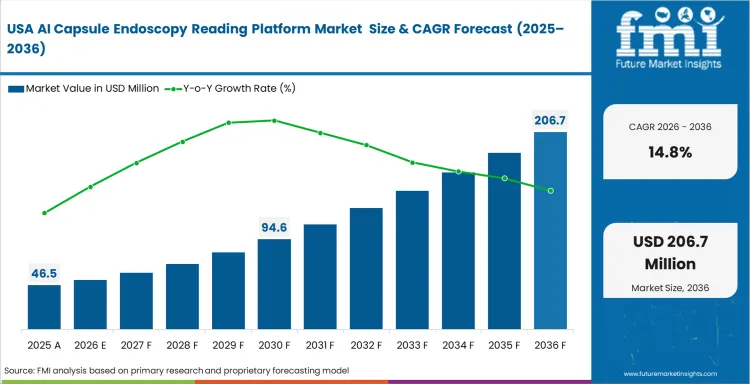
North America remains a commercially important region because clinical adoption is shaped as much by legal exposure and reimbursement logic as by workflow efficiency. Hospitals and specialist groups are under pressure to reduce missed findings, document review quality, and defend diagnostic decisions more clearly. That keeps interest high in AI-assisted reading platforms that can support secondary review and faster case handling. FDA clearance remains a critical commercial filter, since vendors need credible clinical evidence before hospitals treat these systems as deployable tools rather than pilot technologies.
- United States: The United States is likely to post 14.8% CAGR in the sector by 2036. Reimbursement policies and medico-legal pressure both support adoption across large gastroenterology networks and hospital systems. Providers are looking for platforms that improve turnaround time while strengthening confidence in routine interpretation. Clinics that delay implementation may find it harder to match the reporting speed and workflow consistency of better-equipped competitors.
The FMI’s report includes detailed tracking of adoption patterns across the United States and Canada. Regulatory scrutiny, reimbursement discipline, and hospital IT integration requirements continue to shape how quickly AI capsule endoscopy reading platforms scale across North American diagnostic networks.
Europe AI Capsule Endoscopy Reading Platform Market Analysis
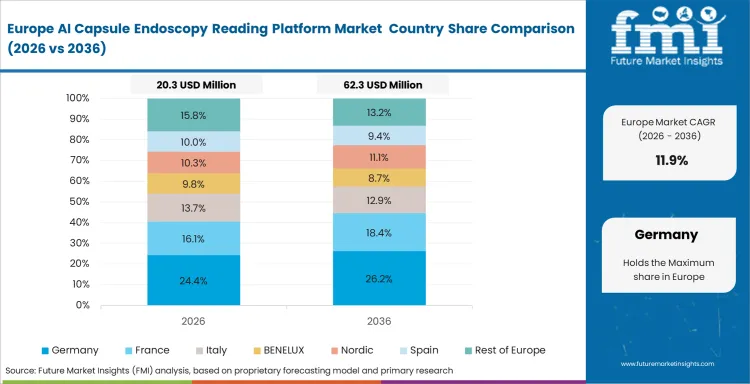
Europe’s market is shaped by data governance, public procurement discipline, and the operational demands of national health systems. Software deployment decisions depend heavily on how well vendors handle patient privacy, local hosting requirements, and integration with established clinical IT environments. Public buyers tend to prioritize data control, traceability, and platform reliability before they prioritize raw processing speed. This creates a market where regulatory fit and implementation discipline matter as much as product capability.
- Germany: Clinical digitization funding and a organized hospital environment support software adoption where approval and reimbursement routes are clear. Departments are more willing to invest when platforms fit existing documentation standards and can be introduced without creating friction in clinical workflow. Germany is forecast to register 12.7% CAGR in this market over the study period. Vendors that manage regulatory and hospital onboarding requirements well are positioned to expand faster in the country.
- United Kingdom: The market in the United Kingdom is projected to expand at a CAGR of 11.9% through 2036. Screening backlogs and pressure on diagnostic waiting times continue to shape platform adoption across the care system. Trust administrators are prioritizing tools that help triage higher-risk cases earlier and use specialist reading time more efficiently. Deployment is likely to remain strongest where workflow relief can be demonstrated clearly at the hospital level.
The FMI’s report includes detailed tracking of adoption patterns across France, Italy, and Spain. Data sovereignty requirements across Europe continue to limit how easily international vendors can scale across multiple national diagnostic networks.
Competitive Aligners for Market Players
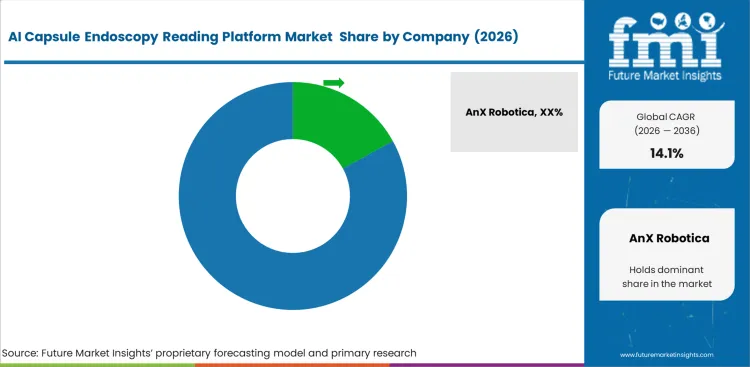
Diagnostic software competition centers entirely on clinical validation library size rather than pure algorithmic architecture. Companies like Medtronic and Olympus maintain dominance because they possess millions of annotated clinical videos gathered over decades of hardware sales. Startup developers can write elegant code, but without heavy proprietary datasets to train their neural networks, their platforms generate unacceptable false positive rates on complex mucosa. Major gastroesophageal reflux devices manufacturers actively bundle their reading algorithms with physical capsule sales, effectively locking out third-party software vendors from hospital purchasing evaluations.
Incumbents defend their positions through deep integration with hospital electronic medical records. AnX Robotica and CapsoVision invest heavily in HL7 and FHIR interoperability standards. Challengers must build identical API connections to even qualify for hospital software trials. Pure software firms like DigestAID face severe headwinds because IT directors reject adding standalone applications that require doctors to maintain separate login credentials. Vendors must ensure their diagnostic outputs flow directly into a physician's final report using healthcare ai computer vision standards.
Large academic hospital networks resist vendor lock-in by demanding software platforms capable of reading videos from any capsule manufacturer. This interoperability requirement forces hardware makers to open their proprietary video formats to third-party analysis. Algorithms capable of drafting a complete, legally compliant clinical narrative win major ai enabled medical devices enterprise contracts over simple bounding-box applications. Forward-thinking developers prioritize generating readable text reports directly from pixel data.
Key Players in AI Capsule Endoscopy Reading Platform Market
- AnX Robotica
- Medtronic
- Olympus
- CapsoVision
- IntroMedic
- Jinshan Science & Technology
- DigestAID
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 82.0 million to USD 306.7 million, at a CAGR of 14.10% |
| Market Definition | Diagnostic software specifically engineered to analyze ingestible camera video feeds. These platforms utilize machine learning to highlight gastrointestinal lesions automatically. |
| Segmentation | Platform type, Capsule type, Core function, End user, Deployment model, and Region |
| Regions Covered | North America, Latin America, Europe, Asia Pacific, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Brazil, Mexico, GCC Countries, South Africa |
| Key Companies Profiled | AnX Robotica, Medtronic, Olympus, CapsoVision, IntroMedic, Jinshan Science & Technology, DigestAID |
| Forecast Period | 2026 to 2036 |
| Approach | Annual software licensing volume and per-study transaction fees within clinical gastroenterology networks. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
AI Capsule Endoscopy Reading Platform Market Analysis by Segments
Platform type
- Cloud
- On-premise
- Hybrid
Capsule type
- Small bowel
- Colon
- Pan-enteric
- Stomach
Core function
- Lesion detection
- Workflow triage
- Report drafting
- Cleansing scoring
End user
- Hospitals
- GI clinics
- Ambulatory centers
- Academic centers
Deployment model
- Reader-assist
- First-pass review
- Concurrent review
- QA audit
Region
- North America
- Latin America
- Europe
- Asia Pacific
- Middle East and Africa
Bibliography
- Andrade, P., Rosa, B., Toth, E., Koulaouzidis, A., Spada, C., Eliakim, R., Leenhardt, R., Mascarenhas-Saraiva, M., Afonso, J., Carvalho, P. B., & Sidhu, R. (2025, October 08). Artificial intelligence-assisted capsule endoscopy for detection of ulcers and erosions: A multicenter study. Clinical Gastroenterology and Hepatology. https://www.cghjournal.org/article/S1542-3565(25)00861-4/abstract
- Bin, Y., Xiao, T., Liu, Z., Shi, Z., Yao, J., Li, H., & Tong, Y. (2025, April 01). Artificial intelligence-assisted capsule endoscopy for gastrointestinal disorders: A comprehensive review. Frontiers in Artificial Intelligence, 8, 1531362. https://www.frontiersin.org/journals/artificial-intelligence/articles/10.3389/frai.2025.1531362/full
- Chen, J., Chen, Z., Li, C., Tang, Y., Li, P., Tang, C., He, Q., & Chen, H. (2024, November 06). Establishing an AI model and application for automated diagnosis of 12 small bowel diseases by capsule endoscopy. BMC Gastroenterology, 24, Article 348. https://link.springer.com/article/10.1186/s12876-024-03482-7
- Dhali, A., Kipkorir, V., Hann, A., Sanders, D. S., & Sidhu, R. (2025, March 13). Artificial intelligence-assisted capsule endoscopy versus conventional capsule endoscopy: A systematic review and meta-analysis. Journal of Gastroenterology and Hepatology. https://onlinelibrary.wiley.com/doi/10.1111/jgh.16931
- Giordano, A., Leenhardt, R., Rosa, B., Carvalho, P. B., & Afonso, J. (2025, June 09). A review of market-available tools for clinical practice: Integration of artificial intelligence-enhanced capsule endoscopy in small bowel diagnostics. Digestive Diseases and Sciences. https://link.springer.com/article/10.1007/s10620-025-09099-4
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Regional adoption trajectories for cloud-based diagnostic reading software across forty nations.
- Competitive positioning strategies utilizing extensive proprietary clinical validation libraries.
- Workflow bottlenecks preventing fully autonomous capsule review inside modern hospital networks.
- Malpractice liability shifts accompanying algorithmic diagnostic triage implementation.
- Reimbursement coding updates driving American hospital software timelines.
- False positive rates associated with poor bowel preparation quality.
- Hardware interoperability standards demanded by academic medical centers.
- Rural screening capacity expansion supported by remote interpretation hubs.
Frequently Asked Questions
What valuation does FMI project for AI capsule software in 2036?
Revenue expands to USD 306.7 million by 2036. This growth signals a complete shift from manual video review toward automated triage frameworks across major gastroenterology departments.
Why does cloud architecture secure major platform share?
The cloud segment is expected to account for 42.0% share in 2026 because it eliminates expensive local server requirements. IT directors prefer subscription models that allow continuous algorithm updates without heavy onsite hardware investments.
Which core function dominates clinical procurement?
The lesion detection category is likely to represent 48.0% of the market in 2026. Clinical directors justify software purchases strictly on its ability to find subtle pathology that fatigued human reviewers routinely miss during manual evaluations.
Why does China lead international adoption rates?
The market in China is projected to expand at a CAGR of 16.2% through 2036. State-funded rural screening programs generate millions of video hours annually, pushing urban academic centers to rely entirely on algorithms to filter out normal cases.
What specific friction slows enterprise deployment?
Data interoperability blocks seamless deployment. Software platforms often struggle to communicate with legacy PACS setups, forcing doctors to review videos on separate, non-integrated monitors.
How do incumbents maintain competitive dominance?
Incumbents defend their positions using extensive proprietary datasets. Startup developers struggle to train highly accurate neural networks without decades of annotated clinical videos gathered through hardware sales.
What anatomical segment requires algorithmic assistance most?
Small bowel algorithms are projected to secure 51.0% share in 2026. Human concentration degrades severely over long videos showing identical villi formations, making computer vision ideal for finding obscure bleeding pixels.
Why do standalone reading stations fail commercially?
Hospitals ban standalone applications that force doctors to use separate login credentials. Platforms unable to connect seamlessly with existing EMR software create isolated, inefficient data silos.
How does India approach algorithm deployment differently?
In India, remote reading models may gain traction where private diagnostic chains need to standardize interpretation across distributed clinic networks. Algorithms help standardize diagnostic quality across widely distributed clinic networks.
What capability defines future enterprise contracts?
Algorithms capable of drafting a complete, legally compliant clinical narrative win major contracts. Forward-thinking developers prioritize generating readable text reports directly from pixel data.
How do IT departments evaluate new platform acquisitions?
System administrators focus entirely on EHR integration capabilities. A highly accurate algorithm fails evaluation if its output cannot attach seamlessly to a physician's final legally binding report.
What false assumption do generalists make about this technology?
Generalists assume AI will replace doctors entirely. Actual clinical implementation shows software is being used to upskill junior readers and manage volume rather than eliminate professional reading jobs.
Why do independent specialists join larger networks?
Independent GI specialists struggle to afford enterprise-level software licenses. Referring physicians demand standardized, AI-annotated reports, forcing consolidation among smaller practices.
What limits rural clinic transition timelines?
Rural clinics require reliable high-speed internet to transmit heavy video files to cloud servers. Once data transmission improves, administrators quickly achieve diagnostic parity with major urban centers.
How do diagnostic service providers protect profit margins?
Service providers transition from manual reading to AI-assisted triage models. Algorithms convert variable, expensive specialist labor costs into predictable software subscription expenses.
What specialized detection modules gain regulatory clearance fastest?
Algorithms trained exclusively to identify obscure gastrointestinal bleeding secure expedited clearance. Emergency department directors prioritize rapid triage tools for critical patients.
Why do hardware manufacturers open proprietary formats?
Large academic hospital networks resist vendor lock-in by demanding software capable of reading videos from any capsule manufacturer. This interoperability requirement forces open API development.
What drives Japanese screening network investments?
Aggressive national cancer screening protocols mandate regular evaluations for older citizens. Hospitals utilize automated tools to manage heavy diagnostic workloads and accelerate early detection.
How do administrative officers calculate return on investment?
Administrators calculate ROI based on physician hours saved. Software subscriptions cost less than hiring additional specialized gastroenterologists to handle rising screening video volumes.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Platform Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Platform Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Platform Type , 2026 to 2036
- Cloud
- On-premise
- Hybrid
- Cloud
- Y to o to Y Growth Trend Analysis By Platform Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Platform Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Capsule Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Capsule Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Capsule Type, 2026 to 2036
- Small Bowel
- Colon
- Stomach
- Small Bowel
- Y to o to Y Growth Trend Analysis By Capsule Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Capsule Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Core Function
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Core Function, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Core Function, 2026 to 2036
- Lesion Detection
- Workflow Triage
- Others
- Lesion Detection
- Y to o to Y Growth Trend Analysis By Core Function, 2021 to 2025
- Absolute $ Opportunity Analysis By Core Function, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- GI Clinics
- Others
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment Model
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment Model, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment Model, 2026 to 2036
- Reader-Assist
- QA Audit
- Others
- Reader-Assist
- Y to o to Y Growth Trend Analysis By Deployment Model, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment Model, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Platform Type
- By Capsule Type
- By Core Function
- By End User
- By Deployment Model
- Competition Analysis
- Competition Deep Dive
- AnX Robotica
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Medtronic
- Olympus
- CapsoVision
- IntroMedic
- Jinshan Science & Technology
- AnX Robotica
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Capsule Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Capsule Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Capsule Type, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Capsule Type, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Capsule Type, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Capsule Type, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Capsule Type, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Capsule Type, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Platform Type
- Figure 6: Global Market Value Share and BPS Analysis by Capsule Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Capsule Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Capsule Type
- Figure 9: Global Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Core Function
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Deployment Model
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Platform Type
- Figure 32: North America Market Value Share and BPS Analysis by Capsule Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Capsule Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Capsule Type
- Figure 35: North America Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Core Function
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Deployment Model
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Platform Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Capsule Type, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Capsule Type, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Capsule Type
- Figure 51: Latin America Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Core Function
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Deployment Model
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Platform Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Capsule Type, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Capsule Type, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Capsule Type
- Figure 67: Western Europe Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Core Function
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Deployment Model
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Platform Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Capsule Type, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Capsule Type, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Capsule Type
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Core Function
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Deployment Model
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Platform Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Capsule Type, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Capsule Type, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Capsule Type
- Figure 99: East Asia Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Core Function
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Deployment Model
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Platform Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Capsule Type, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Capsule Type, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Capsule Type
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Core Function
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Deployment Model
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Platform Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Capsule Type, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Capsule Type, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Capsule Type
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Core Function
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Deployment Model
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

