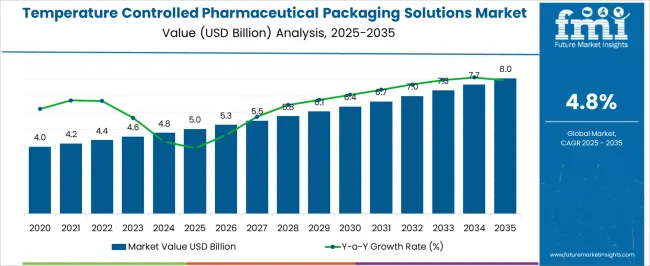
The temperature controlled pharmaceutical packaging solutions market was valued at USD 4.99 billion in 2025, projected to reach USD 5.24 billion in 2026, and is forecast to expand to USD 8.37 billion by 2036 at a 4.8% CAGR. Pharmaceutical supply chain operators face simultaneous regulatory pressure to demonstrate validated thermal performance documentation and commercial pressure from parcel carrier dimensional weight pricing models that impose surcharges on bulky expanded polystyrene cold chain shipments. This dual constraint is accelerating qualification investment in compact, high-performance vacuum insulation panel and rigid polyurethane container systems that maintain temperature compliance across extended lane durations while occupying significantly reduced cubic volume in air freight and courier consignments.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 5.24 Billion |
| Industry Value (2036) | USD 8.37 Billion |
| CAGR (2026-2036) | 4.8% |
| Source | Future Market Insights, 2026 |
Source: Future Market Insights, 2026
The incremental opportunity of USD 3.13 billion over the forecast decade reflects the structural shift in pharmaceutical distribution toward direct-to-patient specialty drug shipments, clinical trial biological sample returns, and cell therapy product deliveries that each require individually qualified packaging solutions rather than bulk freight cold chain systems. E-pharmacy channel growth is driving pharmaceutical retailers to invest in consumer-facing cold chain packaging validated for last-mile delivery exposures, where temperature fluctuation risks during residential delivery attempts differ fundamentally from controlled pharmaceutical warehouse-to-warehouse transfer profiles. Packaging solution providers that offer pre-validated lane studies and regulatory submission documentation packages are reducing customer qualification lead times from months to weeks, creating a commercial differentiation that commands premium pricing in specialty packaging procurement.
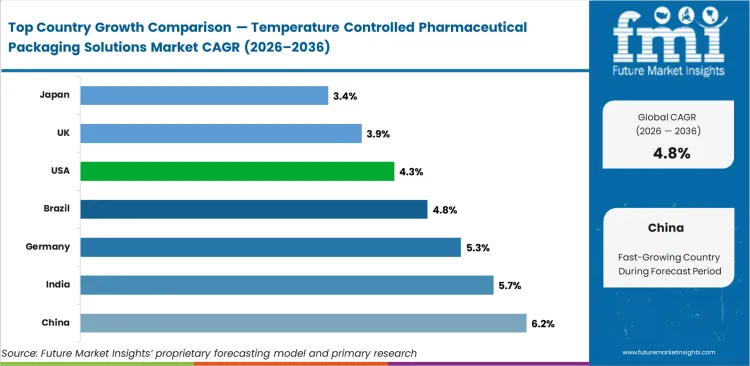
China leads at a 6.17% CAGR through 2036, driven by NMPA distribution quality standards requiring validated thermal packaging for all biologics and vaccine distribution across the domestic pharmaceutical supply chain. India advances at 5.71%, supported by CDSCO Schedule M amendments and Universal Immunisation Programme cold chain modernisation investment. Germany registers 5.26% growth anchored by EMA GDP guideline enforcement and biosimilar export packaging qualification requirements from domestic manufacturers. Brazil at 4.80% reflects ANVISA cold chain compliance investment, while the USA at 4.34%, UK at 3.89%, and Japan at 3.43% each represent structured replacement-cycle markets driven by regulatory audit schedules and specialty pharmaceutical pipeline growth.
The temperature controlled pharmaceutical packaging solutions market encompasses thermal insulation systems, phase-change material components, and integrated packaging formats engineered to maintain defined temperature ranges during pharmaceutical product distribution, including insulated shippers, protective shipper systems, expanded polystyrene containers, polyurethane rigid containers, vacuum insulation panel assemblies, and fibreboard insulated packaging. The market covers both single-use disposable and reusable return-system configurations across frozen, chilled, and ambient temperature performance profiles serving pharmaceutical manufacturers, specialty distributors, clinical research logistics, and direct-to-patient delivery channels.
The scope covers global and regional revenue from insulated pharmaceutical shipper hardware, phase-change material components sold as integrated packaging elements, reusable container system rental and purchase contracts, and packaging qualification and validation study services. Forecast includes all product type variants from panels and envelopes to full rigid insulated container systems, all temperature application categories, and all distribution channel configurations serving the pharmaceutical industry through 2036.
The scope excludes refrigerated pharmaceutical transport vehicle infrastructure, cold storage warehouse equipment, and temperature monitoring data logger devices sold independently of packaging systems. General-purpose consumer cold chain packaging for food and beverage distribution, cosmetics temperature-sensitive packaging without pharmaceutical-grade validation, and active mechanical refrigeration container systems are excluded from the thermal packaging solution valuation.
Strict Temperature Control Regulations to Enhance Market Growth
The growing demand for biologics, vaccines, and specialized pharmaceuticals need stringent temperature control to ensure efficacy and safety, driving the need for innovative packaging solutions. Temperature controlled pharmaceutical packaging solutions are becoming more popular as pharmaceutical distribution networks expand into new areas with harsh climatic conditions.
Investments in dependable packaging solutions are prompted by the strict regulatory requirements for temperature sensitive medications, which demand adherence to temperature control standards. Pharmaceutical supply chains are becoming more global, which emphasizes how crucial temperature controlled pharmaceutical packaging solutions is to maintaining product integrity while in transit.
Challenges in the Cold Chain Management to Impede the Market Growth
Maintaining temperature control throughout the supply chain for temperature sensitive pharmaceuticals involves logistical problems that need specialized infrastructure and knowledge, thereby limiting market accessibility and scalability for some enterprises.
Adoption of current temperature controlled packaging technology is hindered, especially by smaller pharmaceutical businesses, by the high initial cost involved. Challenges in supply chain logistics, such as the requirement for specialized infrastructure and cold chain management, impede industry entrance and growth.
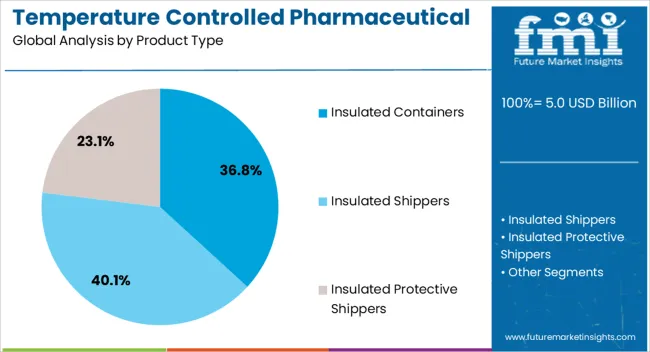
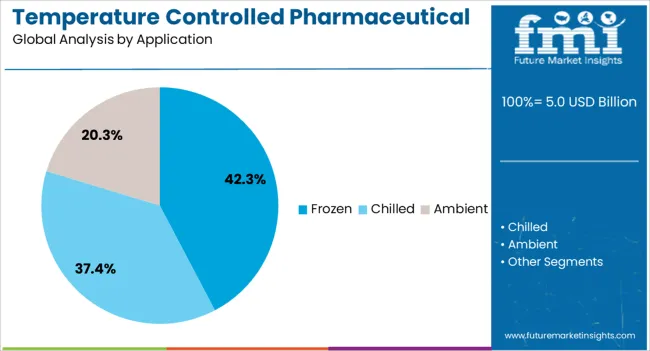
This section focuses on providing detailed analysis of two particular market segments for temperature controlled pharmaceutical packaging solutions, the dominant product type and the significant application. The two main segments discussed below are the insulated chillers and chilled segments.
| Product Type | Insulated Chillers |
|---|---|
| CAGR from 2026 to 2036 | 62.30% |
In 2026, the insulated chillers are anticipated to gain a 62.30% market share. Pharmaceutical supply chains are becoming more and more globalized, making dependable temperature control solutions necessary to preserve product integrity while shipping across several climatic zones.
The necessity for accurate temperature control is highlighted by the rising need for drugs that are sensitive to changes in temperature, such as biologics and vaccinations. Pharmaceutical firms must invest in insulated chillers to maintain compliance and product safety across the supply chain due to strict regulatory standards and quality assurance requirements and costs.
| Application | Chilled |
|---|---|
| Market Share in 2026 | 42.90% |
In 2026, the chilled application segment is anticipated to acquire almost 42.90% market share. A growing number of drugs, such as vaccinations and biologics, must be stored and transported at certain cold temperatures in order to be effective.
The use of chilled solutions is becoming increasingly practical due to advancements in refrigeration and cold chain logistics. Pharmaceutical firms are compelled by strict regulations and elevated quality standards to invest in dependable chilled packaging solutions in order to guarantee product safety and stability.
This section will go into detail on the temperature controlled pharmaceutical packaging solutions markets in a few key countries, including the United States, the United Kingdom, China, Japan and South Korea.
This part will focus on the key factors that are driving up demand in these countries for temperature controlled pharmaceutical packaging solutions.
.webp)
| Countries | Forecast CAGRs from 2026 to 2036 |
|---|---|
| The United States | 36.6% |
| The United Kingdom | 37% |
| France | 36.9% |
| China | 37.6% |
| Japan | 38.1% |
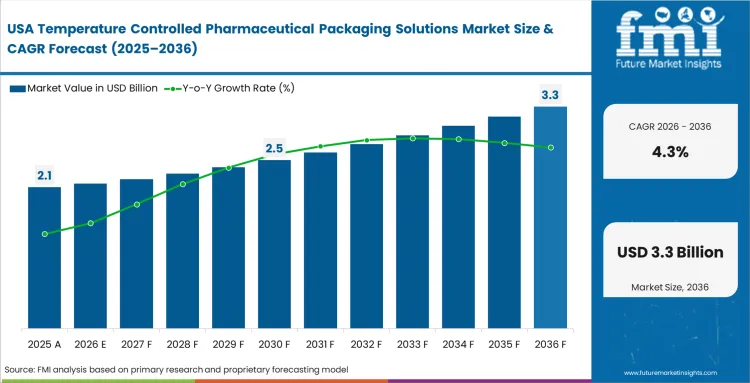
The United States temperature controlled pharmaceutical packaging solutions ecosystem is anticipated to gain a CAGR of 2% through 2035. Factors that are bolstering the growth are:
The temperature controlled pharmaceutical packaging solutions market in the United Kingdom is expected to expand with a 3.80% CAGR through 2035. The factors pushing the growth are:
The temperature controlled pharmaceutical packaging solutions ecosystem in China is anticipated to develop with a 5.10% CAGR from 2026 to 2036. The drivers behind this growth are:
The temperature controlled pharmaceutical packaging solutions industry in Japan is anticipated to reach a 2.60% CAGR from 2026 to 2036. The drivers propelling growth forward are:
The temperature controlled pharmaceutical packaging solutions ecosystem in South Korea is likely to evolve with a 3.40% CAGR during the forecast period. The factors bolstering the growth are:
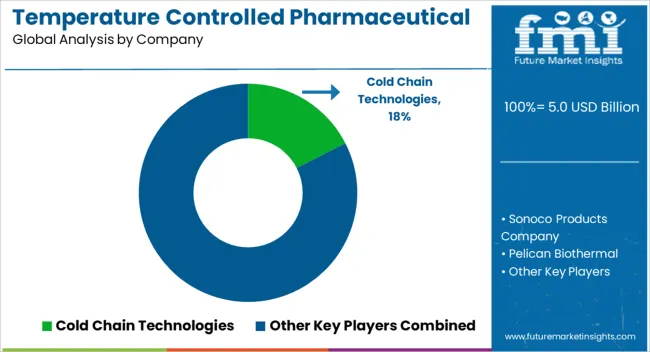
The key players in the market for temperature controlled pharmaceutical packaging solutions are concentrating on a number of tactics to satisfy the strict needs for the transportation and storage of drugs that are sensitive to temperature. Investing in research and development to improve temperature monitoring systems and insulating materials is part of this.
They are also diversifying their product lines to provide a greater selection of packaging alternatives that are suited to various temperature needs. To guarantee the integrity of the supply chain from beginning to finish, cooperation with pharmaceutical firms and logistical partners is also given top priority.
Significant corporations are progressively implementing sustainable practices in order to lessen their impact on the environment and adhere to legal requirements. The key players in this market include:
Significant advancements in the temperature controlled pharmaceutical packaging solutions market are being made by key market participants, and these include:

| Metric | Value |
|---|---|
| Quantitative Units | USD 5.24 Billion to USD 8.37 Billion, at a CAGR of 4.8% |
| Market Definition | The temperature controlled pharmaceutical packaging solutions market encompasses thermal insulation systems, phase-change material components, and integrated packaging formats engineered to maintain defined temperature r... |
| Product Type Segmentation | Insulated Shippers, Insulated Protective Shippers, Insulated Containers |
| Application Segmentation | Frozen, Chilled, Ambient |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | India, China, USA, Germany, France, UK, Japan, Brazil, and 40 plus countries |
| Key Companies Profiled | Sonoco Products Company, Pelican Biothermal, Sofrigam SA Ltd., Cryopak, Cold Chain Technologies, Envirotainer Ltd., Cencora Inc., Inmark Packaging |
| Forecast Period | 2026 to 2036 |
| Approach | Bottom-up methodology applied to segment-level deployment data, cross-validated against publicly reported manufacturer and operator revenue disclosures |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
How large is the Temperature Controlled Pharmaceutical Packaging Solutions Market in 2026?
The temperature controlled pharmaceutical packaging solutions is estimated to be valued at USD 5.24 Billion in 2026.
What will the Temperature Controlled Pharmaceutical Packaging Solutions Market size be by 2036?
The market is projected to reach USD 8.37 Billion by 2036.
What is the expected CAGR for the Temperature Controlled Pharmaceutical Packaging Solutions Market between 2026 and 2036?
The market is expected to grow at a CAGR of 4.8% between 2026 and 2036.
Which product type category leads the Temperature Controlled Pharmaceutical Packaging Solutions Market?
Insulated Shippers leads due to its broad deployment across primary end-use applications and established procurement frameworks.
Which country records the fastest growth in the Temperature Controlled Pharmaceutical Packaging Solutions Market?
China leads with a 6.17% CAGR through 2036, driven by NMPA distribution quality management standards requiring validated thermal packaging for all biologics and vaccine shipments.
What is driving demand in India?
India is advancing at 5.71% CAGR, supported by CDSCO Schedule M compliance mandates and National Cold Chain Management System upgrades under the Universal Immunisation Programme.
Who are the key players in the Temperature Controlled Pharmaceutical Packaging Solutions Market?
Key companies include Sonoco Products Company, Pelican Biothermal, Sofrigam SA Ltd., Cryopak, Cold Chain Technologies, and others detailed in the competitive landscape section.
What incremental opportunity does the Temperature Controlled Pharmaceutical Packaging Solutions Market offer through 2036?
FMI analysts project an incremental market opportunity of USD 3.13 Billion over the 2026 to 2036 forecast period.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.