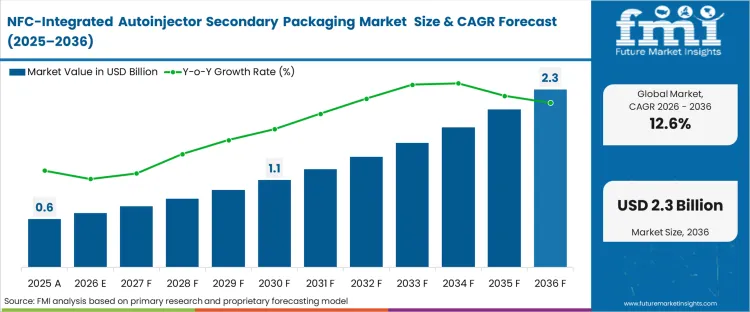
The NFC‑integrated autoinjector secondary packaging market was valued at USD 0.6 billion in 2025 and is projected to reach USD 0.7 billion in 2026, growing at a CAGR of 12.63%. Continued investment is expected to drive the market to USD 2.3 billion by 2036, as the need for verifiable temperature‑chain integrity makes NFC a standard requirement for biologic therapy packaging rather than a premium feature.
Biopharma procurement directors are currently forced into a decision between lower-cost passive packaging and the rising demand for real-time authentication in clinical trials. The stakes of delay are not just related to smart label adoption, but the potential loss of high-value biologic batches due to patient mishandling. While the obvious narrative centers on patient engagement, the structural shift is actually occurring in automated pharmacy dispensing where NFC eliminates manual entry errors. One non-obvious observation is that the integration of NFC into secondary cardboard structures is often more about preventing counterfeiting in emerging trade lanes than it is about providing dosage reminders to patients in developed regions.
The growth becomes self-reinforcing once the cost of NFC inlay assembly drops below the cost of manual serialized labeling for high-volume autoinjectors. Tier-1 packaging converters trigger this gate by embedding packaging NFC tags directly into the paperboard converting process rather than applying them as secondary labels. Once this threshold is crossed, the incremental cost of "smartness" becomes negligible compared to the insurance value it provides for a USD 5,000 autoinjector.
India is expected to advance at a CAGR of 15.2%, followed by China at 14.5% and Germany at 11.5%. The USA sector is poised to register 10.2%, while the UK tracks at 9.8%, Brazil at 9.2%, and Japan at 8.4%. The structural divergence across these regions stems from the local manufacturing capacity for high-density NFC inlays versus the reliance on imported pre-converted smart cartons.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.7 billion |
| Industry Value (2036) | USD 2.3 billion |
| CAGR (2026-2036) | 12.24% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The market encompasses the design and manufacturing of secondary protective structures for autoinjectors, primarily cartons and sleeves, that feature embedded Near Field Communication (NFC) technology. This technology enables touchless data exchange between the package and an NFC-enabled device to verify authenticity, monitor temperature logs, and provide digital instructions for use. It is distinct from QR-code systems as it requires no line-of-sight and provides a unique hardware-level cryptographic signature.
Scope includes paperboard cartons with integrated antennas, plastic protective cases with NFC-enabled closures, and adhesive pharmaceutical smart labels designed for application on existing secondary boxes. It also covers NFC-integrated tamper-evident seals and multi-pack secondary containers used for chronic disease management kits. Service components such as pre-conversion programming and secure cloud-key management for unique ID tracking are included when bundled with the physical package.
The market excludes primary packaging components such as the autoinjector device itself, pre-filled syringes, or needle shields. External scanning hardware, generic RFID tags used exclusively for pallet-level logistics, and secondary packaging that relies solely on passive visual indicators (like thermochromic inks) are outside the scope. Additionally, mobile applications and back-end health data platforms are excluded unless they are provided as a direct integrated feature of the physical packaging sale.
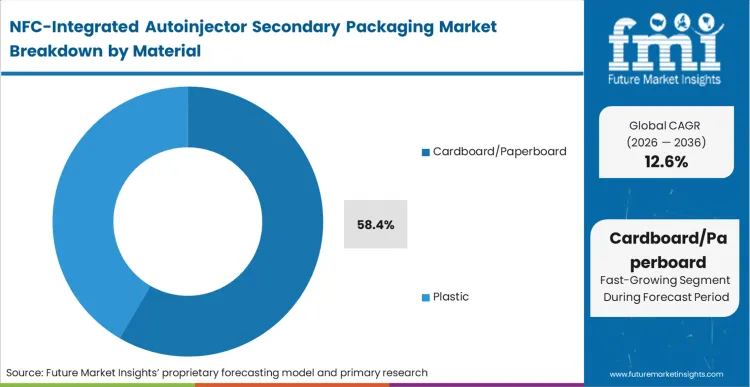
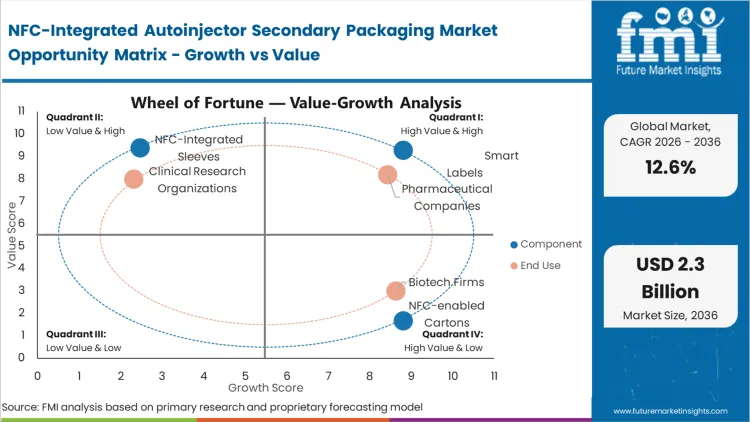
The displacement of traditional plastic-heavy secondary containers is accelerating as pharmaceutical brands seek to align their cold-chain logistics with corporate sustainability targets. This shift is not merely environmental; it is a response to the improved rigidity of engineered paperboard cartons that can now house delicate NFC antennas without the risk of circuit fracture. According to FMI's estimates, cardboard/paperboard holds 58.4% share in 2026 because it allows for high-speed integration of electronics during the printing stage, unlike plastic injection molding which requires secondary assembly steps. Packaging managers at biotech firms are increasingly qualifying fiber-based structures that offer the same structural integrity as polymers while reducing the carbon footprint of the protective packaging. Buyers who delay this transition face rising plastic taxes in European markets, which will eventually erode the margin of legacy secondary packs.
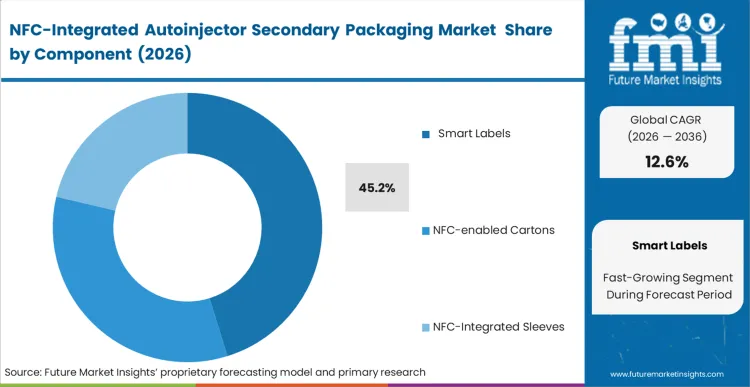
The reason smart labels hold 45.2% of this market comes down to a single operational reality: pharmaceutical companies cannot easily discard millions of dollars in existing carton packaging inventory to switch to integrated boxes. Smart labels offer a bridge, allowing for the addition of NFC functionality to legacy secondary packs without a complete line redesign. FMI analysts opine that this component segment leads because it offers the lowest barrier to entry for clinical trials where packaging formats are already fixed. The smart label serves as a digital skin, providing a secure link to electronic instructions for use (eIFU) and anti-tamper evidence. As the market moves toward 2036, the preference will shift toward fully integrated solutions, but for the current decade, adhesive digital twins remain the dominant choice for rapid deployment.
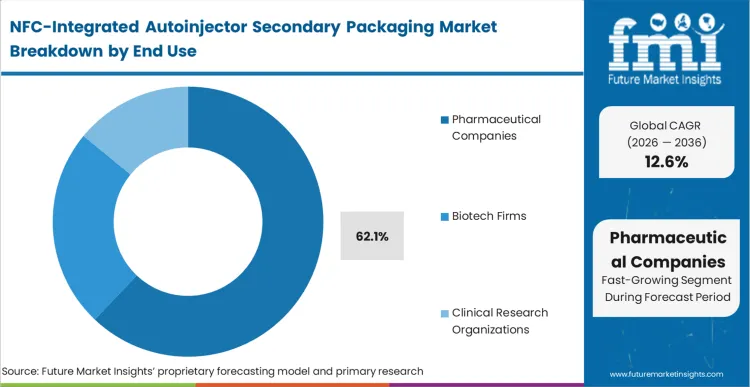
Pharmaceutical companies are currently facing the decision to either modernize their secondary packaging or face increasing pressure from payers to prove patient adherence. The stakes for delay are highest here, as reimbursement for specialty biologics is increasingly tied to "value-based" outcomes which require digital proof of injection. Based on FMI's assessment, these organizations dominate the market with a share of 62.1% because they control the global supply of the chronic disease medications that most require integrated packaging. One non-obvious dynamic is that the demand is being driven by the commercial teams rather than the R&D departments, as NFC becomes a key differentiator in crowded markets like GLP-1 agonists and TNF-alpha inhibitors. The outcome of choosing NFC-integrated packs is a direct increase in the quality of the real-world data collected during the drug's post-market surveillance phase.
The move toward home-based healthcare for chronic conditions forces pharmaceutical companies to replace traditional paper inserts with interactive digital support. This structural pressure is most acute in the biologics market, where the complexity of autoinjector operation leads to high rates of patient error. Commercial leaders name the reduction in "usage-related waste" as the primary economic driver, as every misfired autoinjector represents a significant loss to both the payer and the manufacturer. According to FMI, this forcing condition ensures that NFC adoption is not a marketing experiment but a fundamental restructuring of the drug delivery interface.
The primary restraint in the market is the organizational friction caused by the fragmentation of data ownership between the packaging converter, the pharma company, and the digital health provider. This is not a technical barrier but a structural one, as departments must agree on who manages the cryptographic keys and who owns the patient interaction data. This friction slows adoption even when the hardware cost is justified, as internal legal and IT qualification cycles can take longer than the packaging design itself. FMI notes that a partial solution is emerging through standardized packaging platforms, but these are currently limited by a lack of cross-industry data protocols.
Based on the regional analysis, the NFC-Integrated Autoinjector Secondary Packaging market is segmented into North America, Europe, Asia Pacific, and others across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 15.2% |
| China | 14.5% |
| Germany | 11.5% |
| USA | 10.2% |
| United Kingdom | 9.8% |
| Brazil | 9.2% |
| Japan | 8.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research

Europe's adoption pattern is defined by the early implementation of the Falsified Medicines Directive (FMD) and the subsequent adoption of GS1 Digital Link standards, which enable NFC and 2D DataMatrix to coexist in a unified data architecture. The structural condition here is policy-led, as European healthcare systems push for greater transparency in the biologic supply chain to combat the rising trade in counterfeit medical packaging. FMI views the region as a testing ground for high-density NFC integration in cardboard, with German and Swiss converters leading the development of antenna-embedding techniques. According to FMI's assessment, procurement directors in European hospital networks are increasingly favoring vendors who can provide a digital birth certificate for every autoinjector, making NFC a competitive necessity.
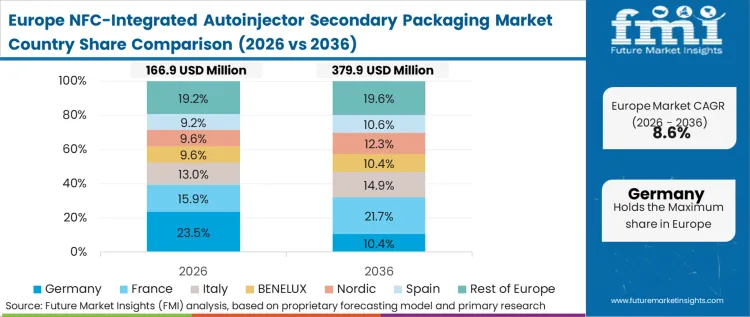
FMI's report includes coverage of additional markets such as France and Italy. These regions show a structural pattern where the harmonization of digital traceability standards supports cross-border trade and stabilizes the biologic supply chain against volatility.
North American market dynamics are shaped by the massive concentration of biotech innovation in hubs like Boston and San Francisco, where digital health is an expected component of every new therapy. The structural lens here is infrastructure-led, as the region already possesses the high-speed 5G networks and high smartphone penetration required to make NFC-enabled secondary packaging effective at the consumer level. Based on FMI's view, growth in the USA is driven by the patient experience competition between top-tier biopharma brands, each trying to offer the most frictionless home-injection support.
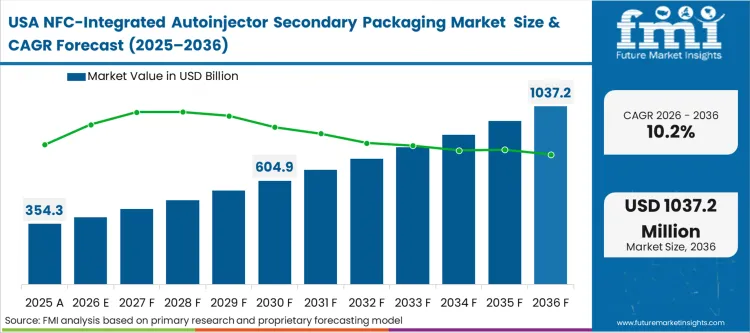
FMI's report includes additional coverage of the Canadian market. This region exhibits a trajectory where centralized healthcare procurement increasingly mandates digital verification to mitigate the economic impact of medication non-adherence.
South America’s adoption pattern is governed by the structural transition of its largest pharmaceutical markets from a reliance on branded generics toward complex, home-administered biologic therapies. The region's growth is primarily economics-led, as domestic manufacturers in Brazil and Argentina aggressively pivot toward biosimilar production to offset the high costs of imported specialty medicines. This shift creates an immediate requirement for secondary packaging that can ensure cold-chain integrity and patient safety in geographies with fragmented logistics infrastructure. FMI's assessment suggests that the integration of NFC technology is becoming a critical tool for local brands to differentiate their high-value injectables and secure their position within public health procurement frameworks.
FMI's report includes additional coverage of the Argentinian and Chilean markets, where the focus remains on the harmonization of digital traceability standards to support cross-border trade. The overarching regional pattern signals a transition toward a high-stakes rational market where packaging is no longer a commodity but a core component of therapeutic efficacy.
The structural condition making Asia's adoption pattern unique is the sheer volume of biosimilar production aimed at both domestic and export markets. This is an economics-led region where the priority is securing the export value of high-margin medicines against the high prevalence of local counterfeiting. FMI's report includes significant analysis of the packaging hubs in India and China that are now being forced to upgrade their flexible packaging and carton lines to meet Western digital standards.
FMI's report includes additional coverage of additional markets such as South Korea and Australia. These regions show a structural pattern where the professionalization of the local biopharma packaging industry is pushing the market toward high-fidelity digital solutions.
The structural reason for the market's moderate concentration level is the high capital requirement for cleanroom electronics assembly integrated with traditional printing. Leading companies like Schreiner MediPharm and Avery Dennison maintain their position not just through patent holdings, but through their ability to guarantee a 99.99% read-rate across millions of units, a benchmark that smaller converters struggle to meet. The primary variable buyers use to distinguish qualified vendors is the "antenna reliability score," which measures how many tags survive the high-pressure environment of an automated filling line.
Incumbent players like WestRock and Jones Healthcare Group hold a structural advantage because they own the relationship with both the biopharma brand and the machinery vendors. This allows them to design protective packaging that is perfectly tuned to the specific mechanical stresses of an autoinjector. A challenger must build a category of capability that includes both silicon expertise and fiber-science to be considered for tier-1 contracts. According to FMI, the competitive gap is widening between those who provide "commodity" boxes and those who provide "digital-ready" cartons.
Large buyers are resisting vendor lock-in by pushing for open-protocol NFC chips that can be read by any standardized mobile OS. This creates a structural tension between the dominant vendors' desire for proprietary ecosystems and the pharma industry's need for interoperability. Through 2036, the market will likely become more fragmented at the label level but more concentrated at the structural carton level, as only a few global firms can manage the complex supply chain required for global medicine launches.

| Metric | Value |
|---|---|
| Quantitative Units | USD 0.7 billion to USD 2.3 billion, at a CAGR of 12.24% |
| Market Definition | Functional segment of secondary packaging for drug delivery systems featuring embedded Near Field Communication technology for security, compliance, and patient support. |
| Material Segmentation | Cardboard/Paperboard, Plastic |
| Component Segmentation | Smart Labels, NFC-enabled Cartons, NFC-Integrated Sleeves |
| End Use Segmentation | Pharmaceutical Companies, Biotech Firms, Clinical Research Organizations |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East, Africa |
| Countries Covered | India, China, Germany, USA, UK, Brazil, Japan, and 40 plus countries |
| Key Companies Profiled | Schreiner MediPharm, Avery Dennison, Jones Healthcare Group, WestRock, Amcor, Constantia Flexibles, CCL Industries, Körber AG |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews with pharmaceutical supply chain leaders, baseline anchoring to global autoinjector volumes, and validation through NFC chip shipment data. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the NFC-Integrated Autoinjector Secondary Packaging Market in 2026?
The market is valued at USD 0.7 billion in 2026. This figure signals that the technology is moving out of the "niche" clinical trial stage into the early-majority stage of commercial biologic launches.
What will it be valued at by 2036?
The valuation is projected to reach USD 2.3 billion by 2036. This growth represents a fundamental shift in how secondary packaging is perceived, from a passive commodity to a critical digital health asset.
What CAGR is projected for this market?
A CAGR of 12.24% is expected during the forecast period. This rate reflects the pace of capital equipment upgrades in the global packaging industry as printers transition to electronic-integration lines.
Which component segment leads the market?
Smart Labels currently lead with 45.2% share. This leadership is sustained by the mechanism of retrofitting, where pharma companies add digital intelligence to existing inventory without re-qualifying their entire carton supply.
Which end use segment leads?
Pharmaceutical Companies lead with 62.1% share. They lead because the massive scale of diabetes and immunology medication programs requires standardized, high-volume digital solutions that only major players can implement.
Which material segment leads?
Cardboard/Paperboard leads with 58.4% share. This is driven by the structural mechanism of "embedding," where antennas are printed directly onto the board, offering a more sustainable and high-speed alternative to plastic.
What drives rapid growth in this market?
The primary driver is the structural pressure to prove patient adherence for high-cost biologics. Payers are increasingly tying reimbursement to digital proof of injection, a non-obvious mechanism that forces pharma brands to adopt NFC.
What is the primary restraint on adoption?
The primary restraint is the structural friction of data ownership between the packaging vendor and the pharma company. This non-obvious gate often takes longer to resolve than the actual engineering of the package.
Which country grows fastest in this sector?
India is the fastest-growing market at 15.2% CAGR. Compared to China's 14.5%, India's growth is more focused on export-led compliance for biosimilars heading to Western markets, requiring immediate alignment with global digital standards.
Is NFC technology compliant with global serialization laws?
Yes, NFC provides a structural layer that exceeds current 2D barcode requirements by offering hardware-level encryption. This allows for a more secure "digital twin" that supports the Drug Supply Chain Security Act (DSCSA) in the USA.
How does NFC improve cold-chain integrity?
NFC chips can be integrated with temperature-sensitive sensors that log thermal excursions directly onto the package. This cause-effect chain allows a patient to tap their phone to the box and instantly see if the medicine was ever stored incorrectly.
Why is there a shift toward cardboard in smart packaging?
Cardboard is being favored because it allows for "in-line" electronics integration, which is faster than applying labels to plastic cases. This operational shift reduces the unit cost of smartness, making it viable for high-volume medicines.
What is the risk of using "dumb" packaging for autoinjectors?
Manufacturers who resist NFC face the structural risk of being excluded from the "value-based" reimbursement models emerging in the USA and Europe. Without digital usage data, they cannot prove the efficacy of their drug in a real-world setting.
How does NFC help decentralized clinical trials?
In decentralized trials, drugs are shipped to homes; NFC provides the structural verification that the correct patient received the kit. This reduces the risk of protocol violations and helps supply chain managers maintain 100% chain of custody.
Can NFC packaging be recycled?
Yes, modern NFC antennas are being printed with aluminum or conductive inks that are compatible with standard paper recycling processes. This comparative advantage over multi-material plastic boxes is a major driver for sustainability-focused brands.
What distinguishes NFC from QR codes in this market?
NFC requires no line-of-sight and cannot be easily copied or photographed like a QR code. This comparative security makes it the preferred choice for high-value autoinjectors where counterfeiting is a significant threat.
Does the FMI report include the cost of mobile apps?
No, the report focuses on the physical secondary packaging and the integrated NFC chip. The software development for mobile apps is excluded unless it is provided as a direct value-add by the packaging converter.
How do packaging converters guarantee 100% read rates?
Converters utilize high-speed "read-after-write" testing stations on their production lines to verify every chip. This mechanism ensures that no dead antennas reach the pharma filling facility, preventing line stoppages.
What is the "digital birth certificate" for medicine?
This refers to the unique, encrypted identity stored in the NFC chip at the moment of manufacture. It creates a structural audit trail that follows the package from the factory to the patient's phone, ensuring authenticity.
How does Japan's market differ from the USA?
Japan focuses more on structural accessibility for its elderly population, using NFC to trigger audio instructions. The USA market is more focused on the economics of insurance reimbursement and adherence tracking.
What is the role of GS1 in this market?
GS1 provides the standardized "Digital Link" protocol that allows NFC chips to resolve to a web URL. This structural standard ensures that a package made in India can be read by a health system in Germany without custom software.
Are there any exclusions to the report scope?
The report excludes the primary autoinjector device and the medicine inside. It also excludes generic RFID tags used for warehouse pallet tracking that do not provide consumer-level NFC interaction.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.