The toxicology screening systems market is expected to reach USD 6,366.0 million in 2026 and expand to USD 13,617.0 million by 2036, at a CAGR of 7.9%. Value expansion reflects sustained reliance on analytical toxicology to support emergency medicine, compliance programs, and forensic determinations across regulated environments. Utilization intensity remains linked to testing frequency, legal defensibility requirements, and need for rapid decision support rather than episodic enforcement activity. Capital allocation favors platforms combining high throughput screening with confirmatory capability, workflow automation, and stable reagent supply. This structure concentrates spending within centralized laboratories and hospital networks operating under accreditation and audit pressure.
Screening programs expand where occupational testing mandates, pain management monitoring, and emergency overdose protocols require consistent turnaround and traceable results. Method complexity, accreditation obligations, and confirmatory testing costs moderate indiscriminate deployment outside defined clinical or legal indications. Automation adoption improves scalability, yet skilled staffing, consumable expense, and quality control oversight remain persistent constraints. Market behavior remains anchored to compliance driven demand, repeat testing cycles, and infrastructure investment supporting defensible toxicology outcomes. These factors sustain predictable growth without reliance on discretionary testing expansion globally.
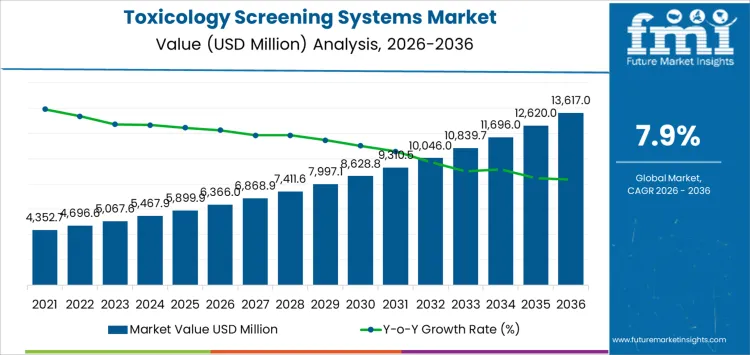
| Metric | Value |
|---|---|
| Market Value (2026) | USD 6,366.0 million |
| Market Forecast Value (2036) | USD 13,617.0 million |
| Forecast CAGR 2026 to 2036 | 7.9% |
Demand for toxicology screening systems is rising as clinical laboratories, forensic units, and occupational health services require comprehensive, rapid identification of chemicals and drugs in biological samples to support patient care and safety decisions. These systems detect a wide spectrum of substances including prescription drugs, illicit compounds, and environmental toxicants that can affect metabolic function, behavior, or organ systems. Clinicians specify screening platforms with high sensitivity, broad analyte libraries, and streamlined workflows that reduce time to results, because accurate toxicology data directly influences diagnosis, treatment planning, and risk mitigation. Procurement teams evaluate analyzer throughput, maintenance requirements, and software interoperability to ensure selected systems integrate with existing laboratory information management systems and support consistent performance under varied test volumes and sample types. Standardization initiatives often bundle instruments, consumables, and software under broader drug testing systems refresh cycles across multi-site networks.
Growth in regulatory requirements for workplace drug testing and expanded use of toxicology data in emergency departments and public health surveillance reinforces adoption of advanced screening solutions. Hospitals and reference laboratories are investing in instruments that accommodate confirmatory testing methods such as mass spectrometry, which enhance specificity and support legal defensibility in forensic contexts. Rising awareness of polysubstance use, novel psychoactive compounds, and therapeutic drug monitoring needs drives laboratories to broaden their testing capabilities. Manufacturers of toxicology instruments are enhancing automation, reporting features, and user support services to help facilities maintain quality standards and improve operational efficiency. These application-focused clinical, regulatory, and operational drivers are contributing to sustained demand growth in the toxicology screening systems market.
Demand for toxicology screening systems is shaped by regulatory oversight, rapid decision-making needs, and expansion of compliance-driven testing programs. Laboratories and care settings prioritize accuracy, turnaround time, and defensible results across screening and confirmation workflows. Adoption reflects growth in substance monitoring, automation of sample handling, and integration with laboratory information systems. Selection decisions emphasize analytical specificity, throughput scalability, and data traceability. Segmentation clarifies how technology format, testing purpose, and service environment influence utilization patterns across clinical, occupational, and forensic toxicology operations.
LC-MS/MS confirmatory systems represent 24.0%, reflecting reliance on high-specificity methods for definitive identification and quantification. These systems support confirmatory workflows following initial screens and withstand legal and clinical scrutiny. Immunoassay analyzers at 22.0% enable rapid, high-throughput screening for common drug classes. Immunoassay reagents and consumables at 20.0% sustain routine testing volumes and recurring demand. Sample preparation and automation systems at 14.0% improve consistency and reduce manual error. Point-of-care devices and LIS or analytics software each at 10.0% address decentralized testing and data management. Where rapid triage is required, point-of-care diagnostics are used as a front-line layer before samples move into centralized confirmation workflows. Device segmentation highlights balance between rapid screening, confirmatory accuracy, and workflow efficiency.
Key Points
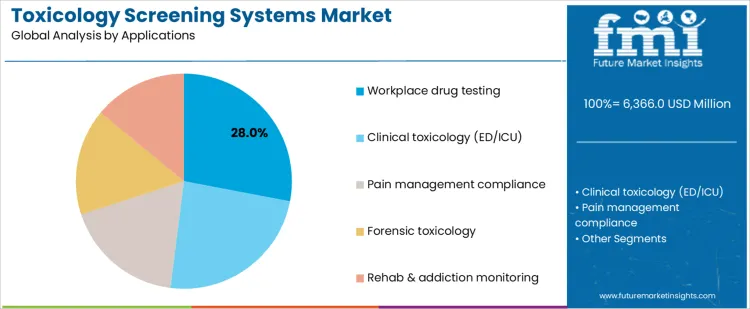
Workplace drug testing accounts for 28.0%, driven by compliance requirements and routine monitoring programs. Clinical toxicology in emergency and intensive care settings at 24.0% requires rapid results to guide acute treatment decisions. Pain management compliance at 18.0% supports monitoring of prescribed substance use. Forensic toxicology at 16.0% applies robust methods for legal investigations. Rehabilitation and addiction monitoring at 14.0% focuses on longitudinal assessment. Application-based segmentation reflects higher utilization where testing frequency, legal defensibility, and clinical urgency determine system selection.
Key Points
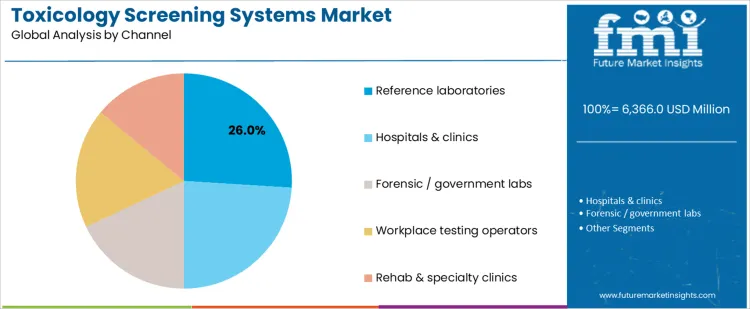
Reference laboratories account for 26.0%, reflecting centralized testing capacity, method validation, and economies of scale. Hospitals and clinics at 24.0% integrate screening into patient care workflows. Forensic and government laboratories at 18.0% manage legally sensitive analyses. Workplace testing operators at 18.0% support employer compliance programs with standardized protocols. Rehabilitation and specialty clinics at 14.0% conduct ongoing monitoring. Channel-based segmentation indicates concentration where infrastructure, regulatory alignment, and testing volume support consistent toxicology service delivery.
Key Points
Demand for toxicology screening systems reflects analytical requirements to detect drugs, poisons, and metabolites in clinical, forensic, workplace, and research settings. Adoption concentrates in hospital laboratories, forensic institutes, emergency departments, and occupational testing facilities. Global scope aligns with regulatory testing mandates and public safety programs. Usage centers on immunoassay platforms, chromatography coupled with mass spectrometry, and integrated screening workflows delivering rapid qualitative and confirmatory results.
Screening programs require rapid turnaround to support clinical decision-making, legal processes, and compliance actions. Demand increases where emergency care depends on timely identification of intoxication to guide treatment. High-throughput immunoassays enable initial screening across large sample volumes, while confirmatory methods ensure specificity and defensibility. Workplace programs value standardized panels aligned with regulatory thresholds. Forensic applications prioritize chain-of-custody integrity and analytical accuracy. Expanding drug classes, including synthetic opioids and novel psychoactives, drive platform flexibility requirements. Automation reduces manual handling and variability. Adoption reflects operational need to balance speed, accuracy, and evidentiary reliability across diverse testing contexts. Scaling programs typically depends on lab automation maturity, since handling steps and batching discipline determine achievable turnaround under accreditation.
Advanced confirmatory systems require skilled personnel, rigorous calibration, and controlled environments, increasing operational burden. Demand sensitivity rises where staffing constraints limit method availability. Regulatory accreditation imposes documentation, proficiency testing, and audit requirements that elevate overhead. Assay cross-reactivity risks necessitate confirmatory testing, extending workflows. Instrument capital cost and consumables affect budget allocation, particularly in public laboratories. Sample matrix variability complicates standardization. Data management and result reporting integration add IT complexity. Scalability remains constrained by expertise requirements, compliance obligations, and total cost of ownership relative to testing volume and criticality.
Demand for toxicology screening systems is expanding globally as clinical diagnostics, forensic testing, and workplace safety programs intensify analytical rigor. Hospitals and laboratories increase rapid screening to support emergency medicine, substance misuse management, and therapeutic monitoring. Regulatory frameworks and accreditation standards reinforce standardized testing workflows. Automation and throughput improvements support scalability across centralized and decentralized settings. Growth rates in India at 10.4%, Brazil at 9.9%, China at 9.4%, the USA at 6.5%, and Germany at 6.4% indicate sustained expansion driven by public health priorities, laboratory modernization, and compliance requirements rather than episodic enforcement activity.
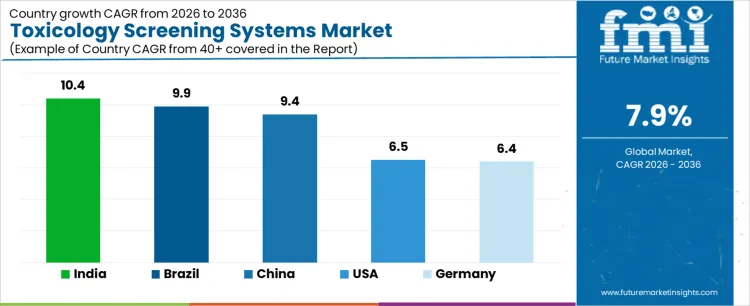
| Country | CAGR (%) |
|---|---|
| India | 10.4% |
| Brazil | 9.9% |
| China | 9.4% |
| USA | 6.5% |
| Germany | 6.4% |
Demand for toxicology screening systems in India is growing at a CAGR of 10.4%, supported by expanding emergency care networks and diagnostic laboratory capacity. Urban hospitals increase rapid drug screening to manage poisoning cases and substance exposure. Forensic laboratories scale screening to address caseload growth and turnaround expectations. Occupational health testing expands across industrial corridors. Private diagnostic chains invest in automated platforms to standardize workflows. Growth reflects healthcare access expansion, forensic throughput needs, and operational efficiency priorities rather than changes in substance prevalence.
Toxicology screening demand in Brazil is expanding at a CAGR of 9.9%, driven by public health surveillance and forensic modernization. Hospitals integrate toxicology panels to support acute care and mental health services. Forensic institutes adopt higher-throughput systems to reduce backlog. Workplace drug testing increases in regulated industries. Government funding supports laboratory upgrades at state levels. Growth reflects institutional investment and standardized testing mandates rather than episodic enforcement campaigns.
Demand for toxicology screening systems in China is growing at a CAGR of 9.4%, aligned with healthcare system scale and regulatory oversight. Large hospitals deploy automated analyzers to manage high patient volumes. Public security and forensic agencies expand screening capacity. Drug safety monitoring programs reinforce routine testing. Domestic manufacturers provide cost-competitive platforms at scale. Growth reflects centralized deployment, compliance-driven utilization, and capacity scaling rather than decentralized point testing proliferation.
Toxicology screening demand in the United States is expanding at a CAGR of 6.5%, supported by guideline-driven clinical care and payer coverage. Emergency departments rely on rapid screening for overdose management. Pain management and addiction treatment programs require routine monitoring. Workplace testing remains stable across regulated sectors. Advanced laboratory automation supports consistency and throughput. Growth reflects protocol adherence and replacement cycles rather than expansion of testing indications.
Toxicology screening demand in Germany is growing at a CAGR of 6.4%, shaped by evidence-based clinical practice and forensic standards. Hospitals integrate screening within emergency and psychiatric care pathways. Certified forensic laboratories maintain standardized toxicology workflows. Insurance reimbursement supports clinically indicated testing. Emphasis on quality assurance governs system selection. Growth remains steady, reflecting structured utilization and compliance discipline rather than rapid testing volume escalation.
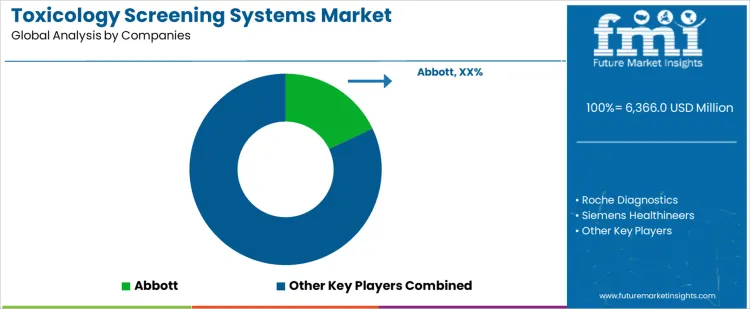
Demand for toxicology screening systems is driven by emergency care requirements, workplace drug testing programs, and forensic laboratory expansion. Clinical laboratories evaluate assay sensitivity, cross-reactivity control, throughput capacity, turnaround time, and integration with laboratory information systems. Buyer assessment includes menu breadth for drug of abuse testing, confirmatory testing compatibility, reagent stability, and regulatory compliance. Procurement behavior reflects hospital tendering cycles, analyzer standardization strategies, and preference for platforms supporting both screening and confirmation workflows. Trend in the toxicology screening systems market shows sustained use in acute care and occupational testing with gradual automation upgrades.
Abbott leads competitive positioning through immunoassay-based toxicology platforms embedded within high-volume clinical chemistry systems. Roche Diagnostics competes with integrated screening solutions offering standardized performance across centralized laboratories. Siemens Healthineers supports demand with toxicology assays aligned with automation and workflow efficiency in hospital settings. Thermo Fisher Scientific maintains relevance through chromatography and mass spectrometry-linked screening pathways for advanced toxicology laboratories. Beckman Coulter participates by supplying screening systems optimized for reliability and scalability in regulated clinical environments. Competitive differentiation centers on assay reliability, throughput scalability, system integration, and service coverage strength.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Devices | Immunoassay analyzers; Immunoassay reagents & consumables; LC-MS/MS confirmatory systems; Sample prep & automation systems; POC toxicology devices; LIS / analytics software |
| Applications | Workplace drug testing; Clinical toxicology (ED/ICU); Pain management compliance; Forensic toxicology; Rehab & addiction monitoring |
| Channel | Reference laboratories; Hospitals & clinics; Forensic / government labs; Workplace testing operators; Rehab & specialty clinics |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Brazil, China, USA, Germany, and 40+ countries |
| Key Companies Profiled | Abbott; Roche Diagnostics; Siemens Healthineers; Thermo Fisher Scientific; Beckman Coulter; Others |
| Additional Attributes | Dollar sales by device category, application, and channel; screening throughput and analytical performance across immunoassay and LC-MS/MS platforms; role of confirmatory testing and chain-of-custody compliance in forensic and workplace programs; adoption of automation and LIS integration to reduce turnaround time and errors; POC deployment trends in emergency and rehabilitation settings; procurement dynamics shaped by laboratory consolidation, regulatory mandates, and employer-led testing programs. |
How big is the toxicology screening systems market in 2026?
The global toxicology screening systems market is estimated to be valued at USD 6,366.0 million in 2026.
What will be the size of toxicology screening systems market in 2036?
The market size for the toxicology screening systems market is projected to reach USD 13,617.0 million by 2036.
How much will be the toxicology screening systems market growth between 2026 and 2036?
The toxicology screening systems market is expected to grow at a 7.9% CAGR between 2026 and 2036.
What are the key product types in the toxicology screening systems market?
The key product types in toxicology screening systems market are immunoassay analyzers, immunoassay reagents & consumables, lc-ms/ms confirmatory systems, sample prep & automation systems, poc toxicology devices and lis / analytics software.
Which applications segment to contribute significant share in the toxicology screening systems market in 2026?
In terms of applications, workplace drug testing segment to command 28.0% share in the toxicology screening systems market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.