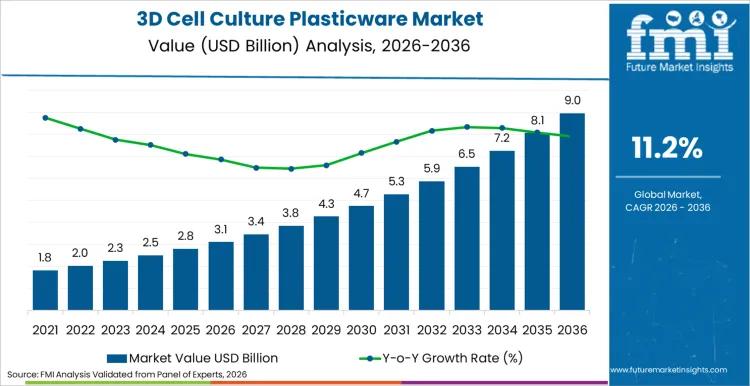
Rising investments in oncology research and regenerative medicine are propelling the valuation of advanced laboratory consumables to unprecedented heights. Global revenue for the 3D cell culture plasticware market is estimated to reach USD 3.1 billion in 2026, reflecting a robust trajectory driven by the shift from traditional 2D models to more physiologically relevant 3D systems. Anticipated to expand at a CAGR of 11.2% through 2036, the industry is poised to generate USD 9.0 billion by the end of the forecast period.
Surge is largely attributed to the critical need for reliable in vitro models that accurately mimic in vivo environments for drug discovery and toxicology testing. As pharmaceutical entities intensify their focus on reducing drug attrition rates, the adoption of specialized plasticware that supports spheroid and organoid growth is becoming indispensable, creating a sustained avenue for high-value expansion across developed and emerging regions.
Advancements in biotechnology and increasing regulatory support for alternative testing methods are acting as primary catalysts for the 3D cell culture plasticware market. Manufacturers are witnessing a significant uptick in demand as academic institutes and contract research organizations (CROs) prioritize reproducibility and scalability in their experimental workflows. The transition towards high-throughput screening (HTS) in drug development has further necessitated the use of micro-patterned plates and scaffold-based systems, which offer superior cell-to-cell interaction capabilities compared to conventional flat surfaces.
Burgeoning field of personalized medicine is creating lucrative opportunities for industry players to develop customized solutions that cater to specific patient-derived cell lines. These drivers, combined with the growing prevalence of chronic diseases requiring complex tissue engineering solutions, are establishing a solid foundation for long-term commercial growth, encouraging stakeholders to invest heavily in expanding their manufacturing capacities and product portfolios.
Technological integration within laboratory workflows is revolutionizing how researchers utilize consumable platforms for complex biological studies. Innovations in 3D cell culture are directly influencing the design and functionality of plasticware, leading to the development of surfaces that enhance cell attachment and proliferation without introducing artificial variables. Researchers are increasingly leveraging these advanced tools alongside 3D ready organoid expansion service providers to streamline the generation of consistent and high-quality organoid models. This synergy between service providers and consumable manufacturers is fostering an ecosystem where standardized plasticware becomes critical for the reproducibility of outsourced research projects.
Incorporation of smart sensors and biocompatible materials into plasticware designs is allowing for real-time monitoring of cell health, thereby reducing experimental errors and accelerating the pace of discovery in life sciences. Such technological convergence ensures that the market remains dynamic and responsive to the evolving needs of modern science.
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 3.1 Billion |
| Industry Value (2036) | USD 9.0 Billion |
| CAGR (2026–2036) | 11.2% |
Sophisticated material engineering and surface modifications are serving as the primary technological engines driving the widespread adoption of specialized plasticware in life sciences. The evolution of scaffold technology has necessitated complementary advancements in plasticware design to support complex structural matrices used in tissue engineering. These innovations allow for better nutrient diffusion and waste removal, which are critical for maintaining long-term culture viability. Concurrently, broader cell culture trends are witnessing a shift towards automated workflows, prompting manufacturers to produce plasticware that is compatible with robotic handling systems. This compatibility reduces human error and increases throughput, making 3D cultures more accessible for large-scale pharmaceutical screening. As researchers demand more physiological relevance in their in vitro models, the ability of modern plasticware to simulate microenvironments found in the human body becomes a decisive factor in purchasing decisions, fueling a continuous cycle of innovation and adoption across the industry.
The global landscape is intricately segmented to address the diverse requirements of modern biological research, categorizing the market by product types, applications, and end-users. This structural division allows stakeholders to identify specific growth pockets, such as the rising demand for scaffold-based systems in tissue engineering or the preference for spheroid plates in cancer studies. By analyzing these segments, manufacturers can tailor their production lines to meet the distinct needs of pharmaceutical giants versus academic laboratories. The segmentation also highlights the shift from general-purpose consumables to highly specialized tools designed for niche applications like stem cell differentiation and toxicity screening.
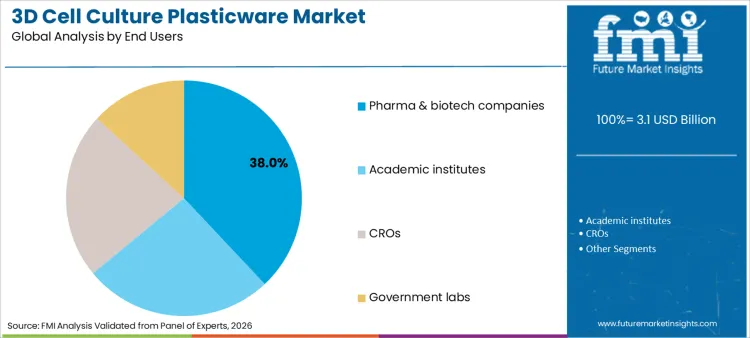
Pharma & biotech companies are projected to capture 38% of the revenue share in 2026, establishing themselves as the dominant end-users in this sector. This segmentation reveals a landscape heavily influenced by the rigorous demands of drug discovery and development pipelines where 3D models are increasingly replacing animal testing.
Academic institutes follow closely, driven by fundamental research in cancer biology and stem cells. In terms of product types, spheroid plates hold the largest share due to their ease of use and applicability in high-throughput screening environments. The structure highlights a clear preference for products that simplify the transition from 2D to 3D protocols. Understanding these segmental dynamics is crucial for stakeholders, as cell culture media continues to evolve, necessitating plasticware that is chemically compatible with novel nutrient formulations designed for specific 3D applications.
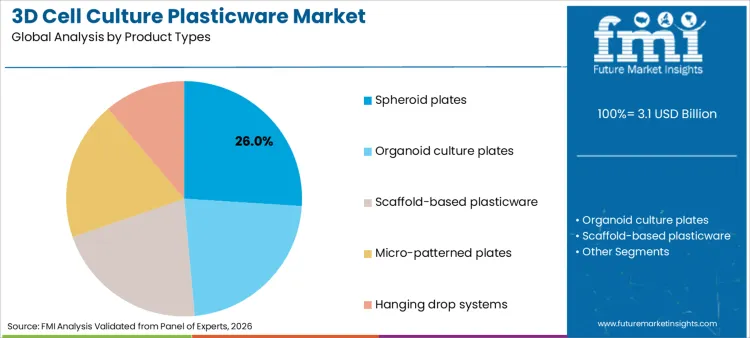
Spheroid plates account for 26% of the share in 2026, driven by their proven efficacy in generating uniform multicellular spheroids for oncology and toxicology studies. Their dominance is underpinned by design features that utilize ultra-low attachment surfaces to promote cell aggregation, a critical requirement for mimicking tumor microenvironments.
This segment benefits significantly from the synergistic growth in cell culture media cell lines, where specialized media formulations are optimized for use with these specific plate types to enhance cell viability and functionality. The simplicity of spheroid formation in these plates allows researchers to conduct reproducible experiments without the need for complex scaffolding matrices, making them a cost-effective choice for large-scale screening campaigns. As high-throughput screening becomes standard in pharmaceutical research, the demand for standardized spheroid plates is expected to remain robust, sustaining their leadership position throughout the forecast period.
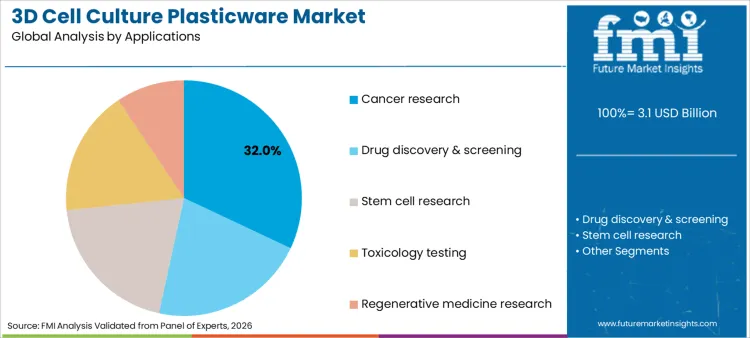
Cancer research applications command a 32% share of the market in 2026, reflecting the intense global focus on understanding tumor biology and developing effective oncological therapies. This dominance is inextricably linked to the ability of 3D cultures to replicate the hypoxic cores and drug resistance patterns observed in solid tumors, capabilities that traditional monolayers lack. The sector relies heavily on cell culture supplements to provide the necessary growth factors and proteins that, when combined with 3D plasticware, create physiologically accurate tumor models.
These models are essential for screening novel compounds and personalized medicine approaches, where patient-derived cells are cultured to predict treatment responses. Consequently, the relentless drive to cure cancer ensures that plasticware designed for tumoroid formation and analysis remains the highest priority for funding and procurement in both academic and commercial research settings.
Automation is set to fundamentally reshape the sales landscape by necessitating plasticware that meets precise dimensional tolerances for robotic manipulation. As high-throughput laboratories integrate solutions from cell culture media bags and automated liquid handlers, the plasticware used must ensure seamless compatibility to prevent jamming or spillage during automated processes. This shift towards "automation-ready" consumables is creating a sub-market for microplates with reinforced frames and standardized footprints that adhere to ANSI/SLAS guidelines.
Manufacturers who fail to align their product specifications with these automated systems risk losing market share to competitors who prioritize interoperability. Furthermore, automation drives the consumption volume of consumables, as robotic systems can process significantly more plates per day than manual workflows, thereby amplifying the revenue potential for suppliers who can guarantee supply chain consistency and product uniformity for automated environments.
How Will Regulatory Changes Impact Market Growth?
Regulatory bodies globally are increasingly advocating for the reduction of animal testing, directly boosting the validity and necessity of 3D cell culture systems as alternative testing methods. This regulatory shift forces pharmaceutical companies to validate their safety and efficacy data using robust in vitro models, increasing the dependency on high-quality plasticware. However, this also introduces stricter quality control standards for consumables, impacting the production of cell culture sampling devices and related plasticware.
Manufacturers must now ensure their products are certified free of pyrogens, DNase, and RNase, and are sterilized to levels that meet clinical research standards. While these regulations drive the adoption of premium plasticware, they also impose higher compliance costs on manufacturers. Companies that proactively align with FDA and EMA guidelines regarding in vitro diagnostic devices will gain a competitive edge, positioning their products as the gold standard for regulatory-compliant research and development activities.
The industry in Europe is projected to demonstrate significant expansion from 2026 to 2036, while emerging economies like India and China are demonstrating even faster acceleration. Regional evolution is characterized by distinct growth drivers: established markets in the West are pivoting towards high-end precision medicine and automated workflows, whereas Asian nations are expanding their biomanufacturing capabilities and academic research infrastructure.
The disparity in growth rates highlights a shifting center of gravity, where the sheer scale of pharmaceutical manufacturing in the Asia-Pacific region is creating massive volume demand. Conversely, the North American and European sectors are focusing on innovation and specialized applications, such as organ-on-chip compatible plasticware. Government grants and private funding in these major nations are heavily slanted towards biotechnology, ensuring a sustained influx of capital to support the procurement of advanced laboratory consumables.
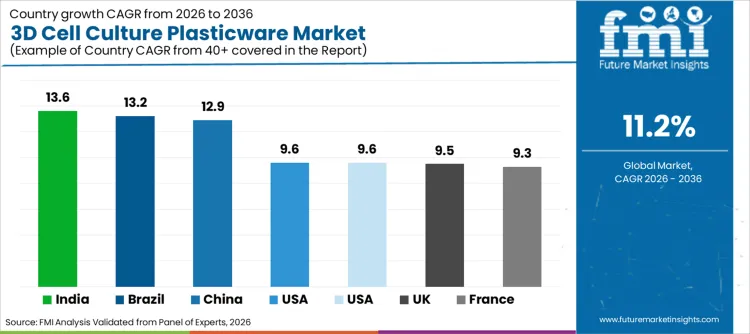
| Region | CAGR (2026 to 2036) |
|---|---|
| USA | 9.6% |
| Brazil | 13.2% |
| China | 12.9% |
| India | 13.6% |
| Germany | 9.6% |
| France | 9.3% |
| UK | 9.5% |
The 3D cell culture plasticware sector in the USA is expanding at a CAGR of 9.6%, underpinned by the robust presence of global pharmaceutical giants and a dense network of biotechnology hubs in cities like Boston and San Francisco. This growth is fueled by substantial federal funding from the NIH for cancer and stem cell research, which directly translates into high procurement volumes for advanced laboratory consumables.
Rapid adoption of precision medicine initiatives in the USA healthcare system necessitates the use of patient-derived organoids, increasing the reliance on specialized plasticware. The presence of key industry players headquartered within the country also streamlines the supply chain, ensuring rapid access to the latest innovations. The emphasis on high-throughput screening for drug discovery continues to be a primary driver, maintaining the USA position as a critical revenue generator in the global landscape.
3D cell culture plasticware sales in China is growing at a CAGR of 12.9%, driven by aggressive government initiatives to transform the nation into a global biotechnology powerhouse. The "Made in China 2025" policy emphasizes the development of high-tech medical devices and pharmaceuticals, fostering a favorable environment for domestic and international manufacturers of laboratory supplies. Increasing investments in contract research organizations (CROs) within China are creating a massive demand for cost-effective yet high-quality cell culture tools to support outsourced drug discovery projects.
Rising prevalence of cancer and chronic diseases in the region is prompting local research institutes to adopt advanced 3D modeling techniques. This shift is further supported by the repatriation of foreign-trained scientists who are bringing expertise in advanced cell culture methodologies, thereby accelerating the modernization of Chinese laboratories and fueling sector expansion.
The demand for 3D cell culture plasticware in India is expanding at a CAGR of 13.6%, representing the highest growth rate among the analyzed nations due to a rapidly maturing biopharmaceutical industry. This surge is largely attributed to the country's strengthening position as a global hub for vaccine manufacturing and generic drug production, which is now moving up the value chain into novel biologics discovery. The Indian government's focus on "Biotechnology Industry Research Assistance Council" (BIRAC) initiatives is providing essential funding for startups and academic labs, enabling them to upgrade from traditional glass and flat plasticware to advanced 3D systems.
Increasing number of partnerships between Indian CROs and global pharma companies necessitates adherence to international research standards, driving the adoption of premium, certified plasticware. The cost advantage of conducting research in India, combined with improving regulatory frameworks, is attracting significant foreign investment into the life sciences ecosystem.
Competition in this industry is intensifying as major players leverage mergers and acquisitions to consolidate their portfolios and expand their geographic footprint. Leading companies like Corning and Thermo Fisher Scientific are actively acquiring smaller firms with niche technologies to integrate advanced surface chemistries into their mass-production lines. Strategies are increasingly focused on bundling plasticware with instruments from microplate handling instruments and microplate instrumentation and systems to offer comprehensive workflow solutions.
The "razor and blade" business model ensures long-term customer retention. Additionally, strategic partnerships with academic centers of excellence allow companies to co-develop products that address specific research pain points, such as organoid culture stability. The competitive landscape is also witnessing a rise in sustainability initiatives, where manufacturers are differentiating themselves by offering eco-friendly, biodegradable, or reduced-plastic packaging options to appeal to environmentally conscious institutions who buy laboratory supplies.
| Items | Values |
|---|---|
| Quantitative Units | USD Million, USD Billion |
| Product Types | Spheroid plates, Organoid culture plates, Scaffold-based plasticware, Micro-patterned plates, Hanging drop systems |
| Applications | Cancer research, Drug discovery & screening, Stem cell research, Toxicology testing, Regenerative medicine research |
| End Users | Pharma & biotech companies, Academic institutes, CROs, Government labs |
| Regions | North America, Europe, East Asia, South Asia, Latin America, MEA |
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.