Antibacterial Film Market
Antibacterial Film Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Antibacterial Film Market Forecast and Outlook By FMI
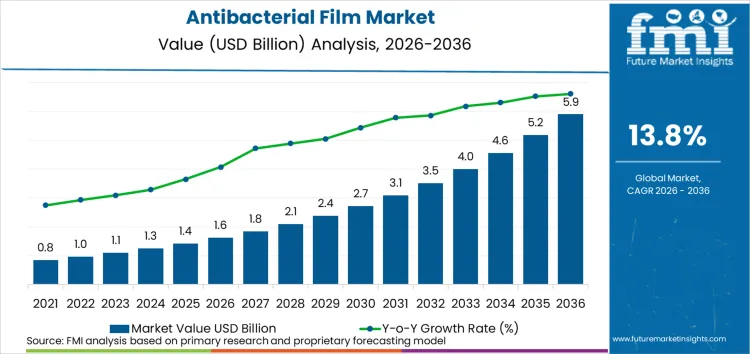
The antibacterial film market is forecasted to total USD 1,620 million in 2026 and is expected to increase to USD 5,880 million by 2036, expanding at a 13.8% CAGR over the forecast period. The market is transitioning from a specialized infection-control packaging category into a multi-vertical compliance and accreditation enabler, driven by heightened accountability for contamination prevention across food, healthcare, and high-touch consumer products.
Food packaging is becoming the strongest conversion engine as sanitary transport enforcement and retailer supplier scorecards converge. Full enforcement of the U.S. FDA FSMA sanitary transportation final rule in 2026 is elevating packaging requirements for high-risk perishables by formalizing temperature control and contamination prevention expectations throughout distribution. This enforcement climate increases litigation exposure for foodborne illness incidents and pushes converters and protein processors toward validated antibacterial film architectures (silver-ion coatings and zinc-based additives) that provide auditable pathogen suppression through multi-day cold-chain cycles.
Healthcare and medical device packaging represent the second structural growth pillar. HAIs are framed as an ongoing cost and mortality crisis, with infection prevalence cited at 7% of hospitalized patients in developed nations and over 15% in developing countries, and annual attributable deaths exceeding 4.95 million globally. This burden intensifies demand for antibacterial films across sterile barrier systems, medical device packaging, and high-touch surfaces. Parallel regulatory pressure comes from the EU Medical Device Regulation, which requires validated antimicrobial efficacy documentation for packaging claims for Class II-a and above device categories, increasing adoption of antibacterial films with established efficacy test files and compliance documentation.
Summary of Antibacterial Film Market
- The antibacterial film market comprises polymeric and coated film systems engineered to inhibit microbial growth on surfaces through active agent release (e.g., silver-ion, zinc-based additives, organic antimicrobials), contact-based killing mechanisms, or physicomechanical membrane disruption (e.g., plasma-treated nanostructures).
- The scope is structured under FMI taxonomy by end-use sector, active technology, base polymer, and region, focusing exclusively on films with intentionally incorporated antibacterial functionality. Exclusions include passive barrier films without antibacterial functionality, post-production sprayed/dipped antimicrobial coatings applied after film manufacture, non-film antimicrobial formats (paper, nonwovens, rigid containers), and downstream sterilization/logistics services.
- The market is projected to grow at a 13.8% CAGR from 2026 to 2036, expanding from USD 1,620 million in 2026 to USD 5,880 million by 2036, supported by sanitary-transport enforcement for food, antimicrobial efficacy documentation requirements in medical device supply chains, and the persistent economic burden of healthcare-associated infections.
- Competition concentrates among specialty film and materials-science corporations with proprietary antimicrobial formulations, multi-jurisdiction regulatory master files, and global manufacturing footprints, including Berry Global, Amcor, Mondi, Wipak, Kuraray, and Toray.
- Country-level expansion is led by China (14.6% CAGR) driven by food-contact positive list enforcement and cold-chain e-commerce protein growth, followed by the United States (12.4%) supported by sanitary transport enforcement and pathogen reduction frameworks, and Germany (11.0%) shaped by MDR efficacy documentation requirements and BfR-linked expectations.
- India advances at 9.8% CAGR on antimicrobial packaging guidelines and medical device manufacturing expansion, while Japan grows at 5.5% CAGR reflecting a mature base and specialization in healthcare and high-clarity electronics films.
Competitive advantage is increasingly determined by regulatory readiness and compliance velocity. Registration for new antimicrobial formulations is described as requiring 18–24 months and testing costs exceeding USD 500,000 per product variation, creating a structural barrier to entry. Suppliers with established EPA/FDA/BfR master files and pre-qualified antimicrobial technologies are securing multi-year supply agreements as end users prioritize solutions that minimize qualification timelines and regulatory uncertainty.
A defining strategic theme is the “hygiene permanence + regulatory accountability” convergence. Retailers are codifying antibacterial packaging expectations into supplier compliance scorecards, regulators are embedding efficacy substantiation into frameworks, and electronics OEMs are mainstreaming antimicrobial touch-surface films as baseline features. Together, these forces convert antibacterial film from a value-add differentiator into a market-access prerequisite in several end-use chains.
Antibacterial Film Market Key Takeaways
| Metric | Value |
|---|---|
| Expected Value (2026E) | USD 1,620 million |
| Projected Value (2036F) | USD 5,880 million |
| CAGR (2026 to 2036) | 13.8% |
Source: Future Market Insights - analysis driven by proprietary forecasting models and primary research
Why is the Antibacterial Film Market Growing?
Growth is propelled by food safety enforcement, healthcare infection-control economics, medical device validation requirements, and hygiene permanence across consumer categories. Institutional procurement specifications from protein processors, hospital purchasing organizations, and electronics OEMs are increasingly forcing adoption of antibacterial films as a baseline packaging and surface-protection requirement.
The strongest driver is food safety accountability. Full enforcement of sanitary transportation requirements in 2026 elevates contamination prevention from internal QA practice to enforceable compliance, prompting retailers to require verified antibacterial packaging performance for high-risk perishables. Efficacy claims are reinforced by cited performance outcomes where antibacterial packaging can reduce bacterial contamination in fresh produce by up to 99% compared with conventional packaging. For poultry processors operating under pathogen reduction expectations, recall risk economics are material-average direct costs of a major recall are cited as exceeding USD 3.5 million, excluding downstream brand and litigation impacts.
Healthcare adoption is sustained by the persistent HAI burden and the economic costs associated with infections, cited at USD 280-450 billion. Antibacterial films provide continuous pathogen reduction independent of cleaning compliance, supporting sterilization packaging, device protection, and high-touch surface hygiene across clinical environments. Medical device manufacturers face additional pressure to maintain validated documentation for antimicrobial efficacy claims, further embedding antibacterial film solutions into regulated packaging qualification workflows.
How is the Antibacterial Film Market Segmented?
The antibacterial film market is segmented by end-use sector, active technology, and base polymer, reflecting that performance requirements vary significantly across food packaging, healthcare sterile barrier systems, consumer product protection, and electronics surface applications. Segmentation in this market is less about film gauge alone and more about efficacy durability, regulatory claim substantiation, and compatibility with high-speed converting and sealing lines. As antibacterial functionality becomes tied to compliance and procurement scorecards, segments with validated test files and stable antimicrobial performance over typical shelf-life periods increasingly command share.
Market structure also reflects a transition from additive supply to solution supply. In higher-risk end uses such as protein packaging and medical devices, buyers specify not only antimicrobial agent type but also dispersion uniformity, migration behavior, and compatibility with food-contact or medical regulatory frameworks. This elevates segment value where suppliers can offer complete packages of formulation + film + compliance documentation.
Why does Food Packaging dominate antibacterial film demand?
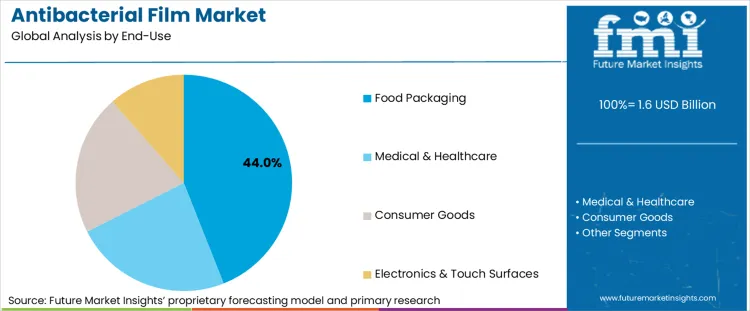
Food packaging accounts for 44% of market demand, making it the largest end-use segment. This dominance is structurally tied to contamination-risk economics and retailer accountability. High-risk perishables-including fresh produce, poultry, and processed protein-are increasingly governed by sanitary transport enforcement and stricter supplier performance requirements. Antibacterial films mitigate microbial growth on packaging-contact surfaces during cold-chain movement, improving shelf life and reducing contamination probabilities in distribution environments where temperature excursions and handling variability remain persistent risks.
Food packaging leadership is reinforced by scale. Even small increments of adoption translate into large film volumes due to the sheer throughput of fresh food and protein supply chains. Furthermore, antibacterial packaging is viewed as a risk-reduction mechanism that supports traceability and compliance, strengthening its position relative to conventional barrier films that do not offer active microbial inhibition.
How are Silver-Ion Coatings sustaining technology leadership?
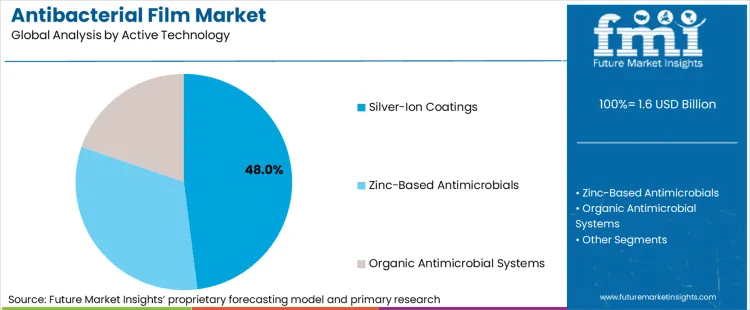
Silver-ion coatings represent 48% of active technology adoption, reflecting their broad-spectrum antimicrobial performance, relatively stable efficacy across time, and established presence across regulated and non-regulated packaging environments. Silver-based systems have long been used in antimicrobial applications, enabling smoother claim substantiation pathways than newer chemistries in many jurisdictions. This advantage matters as antibacterial films are increasingly specified in compliance-bound contexts where substantiated efficacy claims and validated testing are mandatory.
In operational terms, silver-ion coatings support durable antibacterial activity while maintaining film clarity, seal integrity, and converting compatibility-attributes that are crucial for high-speed food packaging lines and medical packaging manufacturing. The scale of adoption also supports supplier investment in optimized coating and dispersion methods, reinforcing silver-ion technology leadership through proven manufacturability and standardized qualification pathways.
Why do PE / PET films remain the dominant base polymer platform?
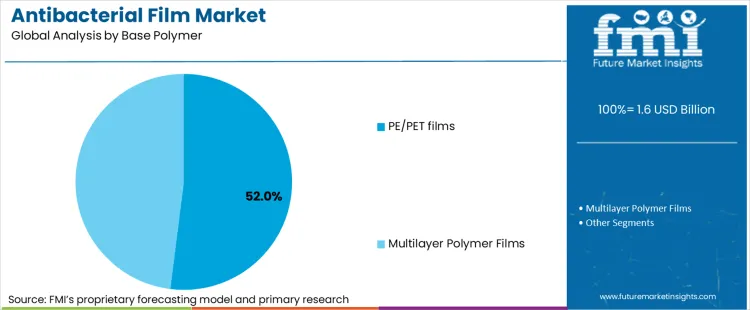
PE/PET films account for 52% of base polymer share, supported by their wide adoption across food and medical packaging, strong supply-chain availability, and well-understood performance characteristics in sealing, lamination, and barrier design. These polymers offer the flexibility needed for diverse packaging architectures-from tray lidding to pouches-while enabling integration of antimicrobial masterbatches or coatings without destabilizing processing behavior.
Material dominance is also linked to compatibility with regulatory frameworks and test methods. PE and PET systems are already widely validated for food-contact applications and are common substrates for sterile barrier systems. As antibacterial performance becomes an additional layer of qualification, buyers prefer polymer platforms with established compliance and performance baselines, sustaining PE/PET dominance while antimicrobial functionality is layered onto existing packaging infrastructures.
What Market Forces Are Shaping the Antibacterial Film Industry?
The antibacterial film market is being shaped by an enduring shift from reactive hygiene response to permanent contamination-prevention infrastructure across food, healthcare, and high-touch consumer ecosystems. Food supply chains are among the strongest accelerators as sanitary transportation enforcement and retailer supplier accountability mechanisms tighten. When contamination prevention is evaluated through audits, scorecards, and traceability scrutiny, antibacterial films become a defensive procurement decision that reduces exposure to recalls, litigation, and reputational damage. This logic is particularly strong in high-risk perishables, where even small improvements in microbial suppression translate into reduced spoilage, longer shelf life, and lower rejection rates across distribution nodes.
Healthcare demand is reinforced by the persistent burden of healthcare-associated infections and the operational reality that cleaning compliance is variable. Antibacterial films offer continuous suppression benefits independent of human behavior, which supports adoption in sterile barrier packaging, medical device protection, and high-touch surface applications. Medical device ecosystems also require stricter substantiation for antimicrobial claims, increasing preference for antibacterial film solutions with validated efficacy documentation and predictable performance across sterilization, storage, and handling cycles.
At the same time, the market faces structural constraints linked to regulatory complexity and cost. Developing and registering new antimicrobial formulations requires long qualification windows and high testing expenditures, creating barriers for smaller firms and slowing technology turnover. These constraints also increase buyer preference for established antimicrobial chemistries and suppliers with pre-existing compliance master files, reinforcing concentration among large specialty film and materials-science providers. In addition, antimicrobial performance must be balanced against material compatibility; additives and coatings must not compromise sealing behavior, clarity, or barrier performance, especially in high-speed converting environments.
Opportunities continue to expand as antibacterial films extend beyond food and healthcare into electronics and consumer goods where hygiene permanence is becoming a standard expectation. Electronics OEMs increasingly incorporate antimicrobial surface films into touch interfaces, while consumer brands use antibacterial protection to differentiate premium packaging and reduce odor or contamination concerns during extended product handling. Innovation is also moving toward lower-migration, longer-lasting antimicrobial mechanisms and physicomechanical surface treatments that reduce reliance on additive release, improving claim defensibility and durability while supporting safety expectations.
Analysis of Antibacterial Film Market by Key Countries
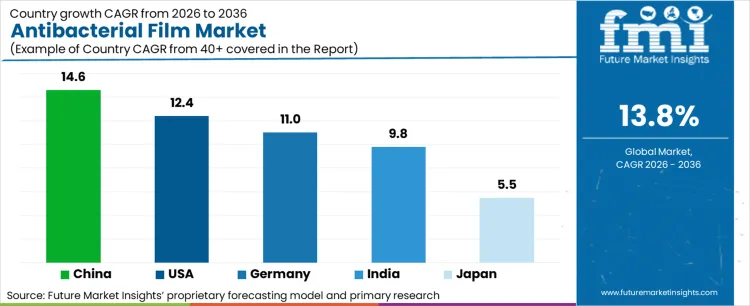
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 14.6% |
| United States | 12.4% |
| Germany | 11.0% |
| India | 9.8% |
| Japan | 5.5% |
Source: Future Market Insights’ proprietary forecasting model and primary research
How is food-contact positive-list enforcement accelerating China at 14.6% CAGR?
China leads growth at 14.6% CAGR, with adoption shaped by mandatory food-contact compliance mechanics that directly influence which antibacterial additives can be used in packaging. Enforcement of GB 9685 to 2016 establishes a positive-list system in which antibacterial agents (including silver-ion, zinc-based, and organic systems) require pre-market authorization, forcing converters and brand owners to standardize around compliant masterbatch and coating pathways.
This regulatory structure reshapes supplier selection and favors organizations that can maintain localized dossiers, in-country testing, and regulatory liaison capability-making compliance execution a competitive differentiator rather than a back-office function. China’s demand profile is further intensified by the scale and speed of cold-chain e-commerce protein delivery, where last-mile temperature variability raises spoilage and pathogen-control risk; antibacterial films are increasingly specified for vacuum-packed chilled meats moving across long geographies on compressed delivery windows.
What compliance triggers are driving the United States at 12.4% CAGR?
The United States expands at 12.4% CAGR, shaped by enforceable food-safety accountability across transit and poultry pathogen management. Full enforcement of the FSMA sanitary transportation rule effective 2026 elevates shipper and carrier liability for temperature control and contamination prevention, pushing validated antibacterial packaging adoption in case-ready meat, poultry, and high-risk produce supply chains. Retailers have operationalized this into supplier packaging requirements, with non-compliance linked to chargebacks, delisting, and liability assignment in the event of contamination incidents.
In parallel, USDA-FSIS enforcement pressure around Salmonella risk has increased urgency for packaging formats that suppress pathogen populations through storage and handling cycles. Antibacterial liner films achieving multi-log reductions are increasingly treated as baseline requirements for suppliers serving highly scrutinized channels. A structural advantage in the U.S. market is the relatively efficient regulatory review pathway for new additives (via established notification processes), supporting earlier commercialization of next-generation antibacterial film technologies compared with slower authorization environments elsewhere.
Why is MDR-backed efficacy documentation sustaining Germany at 11.0% CAGR?
Germany grows at 11.0% CAGR, with demand reinforced by medical device compliance expectations and procurement practices that treat validated antimicrobial packaging as part of quality management. Enforcement of EU Medical Device Regulation (MDR) Article 10(9) requires manufacturers to substantiate antimicrobial efficacy claims for Class IIa and above devices, converting antibacterial film use from a discretionary feature into a documentation-linked compliance tool.
Notified-body expectations extend to technical files with ISO 22196 efficacy reporting, evidence of efficacy retention through sterilization cycles, and biocompatibility documentation under ISO 10993, which increases adoption of antibacterial films that already have mature test libraries and technical dossiers.
Adoption extends beyond minimum compliance as infection prevention is embedded into hospital procurement logic, especially for high-value implant categories. The market also reflects engineering-led specialization, including antibacterial film formulations designed to maintain efficacy through radiation sterilization conditions used in implant packaging, strengthening the premiumization pathway and sustaining growth even from an already mature European base.
What healthcare and food regulatory signals are lifting India at 9.8% CAGR?
India advances at a 9.8% CAGR, driven by healthcare infrastructure scaling and a parallel tightening of packaging hygiene expectations. Expansion of national health coverage programs and policy support for domestic medical device production is increasing demand for sterile packaging formats, including antibacterial film applications across wound care, surgical kits, and device packaging. Fiscal incentives supporting local manufacturing and packaging capability are strengthening domestic supply availability, while converter investment into compounding and coating capacity improves access to cost-optimized antibacterial film structures.
Food safety provides a second growth lane. Draft antimicrobial packaging guidelines published in 2025 establish testing and migration expectations for antibacterial systems and signal the direction of regulatory tightening, encouraging early adoption among export-oriented meat and seafood processors that require certification-aligned packaging. A distinctive feature of India’s adoption curve is cost-performance optimization, lower-concentration silver-ion approaches are positioned to deliver pragmatic log-reduction levels at meaningfully lower cost, expanding adoption feasibility in price-sensitive domestic supply chains while still improving hygiene assurance.
How is demographic pressure shaping Japan’s trajectory at 5.5% CAGR?
Japan expands at 5.5% CAGR, reflecting a mature adoption base and a market structure shaped by a super-aging demographic profile. With a high share of the population aged 65+, infection prevention and hygiene permanence remain structurally embedded in healthcare operations, driving continued specification of antibacterial films for medical device packaging and high-touch protection in long-term care environments. Product development focus leans toward high-performance, application-specific films such as high-barrier variants designed for retort-sterilized nutrition and healthcare products used heavily by elderly populations.
Electronics also contributes through premium positioning strategies in consumer devices, where antibacterial surface films must maintain optical clarity while delivering sustained pathogen reduction. Japan’s growth is comparatively slower because penetration for critical perishables and priority healthcare applications is already high; incremental expansion is increasingly value-driven enhanced sustained-release kinetics, recycled-content antibacterial films, and multifunctional structures (e.g., antioxidant + antibacterial) that support premium pricing rather than large unit-volume increases.
What Is the Competition Outlook for the Antibacterial Film Market?
Competition in the antibacterial film market is increasingly defined by regulatory readiness, efficacy validation depth, and speed of qualification, rather than by film production capacity alone. As antibacterial functionality shifts from a differentiator to a procurement requirement in food packaging, medical devices, and high-touch consumer applications, buyers prioritize suppliers with established antimicrobial master files, validated test libraries, and multi-jurisdiction compliance capability.
A critical competitive axis is time-to-approval. New antimicrobial formulations typically require long registration cycles and significant testing expenditure, creating structural advantages for suppliers with pre-approved chemistries and documented efficacy across food-contact, medical, and consumer safety frameworks. This dynamic favors large specialty materials and packaging companies that can amortize regulatory investment across global volumes, while smaller or regional players face barriers to entry or are pushed toward niche applications.
Another dimension of competition is integration with high-speed converting and sealing environments. Antibacterial films must deliver consistent microbial suppression without compromising seal strength, clarity, or machinability. Suppliers that can demonstrate performance stability under industrial processing conditions are increasingly preferred, particularly in protein packaging and medical sterile barrier systems where failure risk is unacceptable. Over the forecast period, competitive intensity is expected to concentrate market share among a limited set of global suppliers capable of combining antimicrobial science, scalable film manufacturing, and compliance documentation into turnkey solutions.
Key Players Profiled in the Antibacterial Film Market
- Berry Global Group, Inc.
- Amcor plc
- Mondi Group
- Wipak Group
- Kuraray Co., Ltd.
- Toray Industries, Inc.
Market Definition
The antibacterial film market comprises polymeric film structures engineered with active antimicrobial functionality designed to inhibit or suppress microbial growth on packaging and surface-contact interfaces. These films operate through mechanisms such as metal-ion release (e.g., silver, zinc), organic antimicrobial additives, or contact-based microbial disruption, providing continuous hygiene protection during storage, transport, and use.
Within FMI’s scope, the market includes antibacterial films used in food packaging, medical device and sterile barrier systems, consumer goods protection, and electronics surface applications. The scope excludes passive barrier films without antimicrobial agents, non-film antimicrobial formats (paper, nonwovens, rigid plastics), and post-packaging sterilization services.
Scope of the Report
| Attribute | Details |
|---|---|
| Base Year | 2026 |
| Forecast Period | 2026 to 2036 |
| Market Size (2026E) | USD 1,620 million |
| Market Size (2036F) | USD 5,880 million |
| CAGR (2026 to 2036) | 13.8% |
| End-Use Sectors | Food Packaging; Medical & Healthcare; Consumer Goods; Electronics |
| Active Technologies | Silver-Ion Systems; Zinc-Based Systems; Organic Antimicrobials |
| Base Polymers | PE; PET; Multilayer Structures |
| Regions Covered | North America; Europe; East Asia; South Asia; Japan; Rest of the World |
| Key Countries | China; United States; Germany; India; Japan |
Source: Future Market Insights (FMI)
Antibacterial Film Market Key Segments
By End-Use Sector:
- Food Packaging
- Medical & Healthcare
- Consumer Goods
- Electronics & Touch Surfaces
By Active Technology:
- Silver-Ion Coatings
- Zinc-Based Antimicrobials
- Organic Antimicrobial Systems
By Base Polymer:
- PE
- PET
- Multilayer Polymer Films
By Region:
- North America
- Europe
- East Asia
- South Asia
- Japan
- Rest of the World
Bibliography
- USA Food and Drug Administration. (2026). Food Safety Modernization Act: Sanitary transportation of human and animal food-final rule enforcement guidance. FDA.
- European Commission. (2024). Medical Device Regulation (EU) 2017/745: Guidance on antimicrobial claims and efficacy documentation. Official Journal of the European Union.
- International Organization for Standardization. (2024). ISO 22196: Measurement of antibacterial activity on plastics and other non-porous surfaces. ISO.
- Organisation for Economic Co-operation and Development. (2025). Antimicrobial resistance and infection prevention in food and healthcare systems. OECD Publishing.
- China National Health Commission. (2024). GB 9685: National food safety standard-Use of additives in food contact materials. NHC China.
Frequently Asked Questions
What is the long-term growth outlook for the antibacterial film market?
FMI projects the market to expand at a 13.8% CAGR from 2026 to 2036, driven by food safety enforcement, healthcare infection-control economics, and hygiene requirements across consumer products.
How large is the antibacterial film market expected to be by 2036?
The market is expected to reach USD 5,880 million by 2036, up from USD 1,620 million in 2026.
Which end-use sector dominates demand?
Food packaging dominates due to contamination-risk economics and enforceable sanitary transport requirements.
Which country leads growth?
China leads growth at 14.6% CAGR, supported by food-contact compliance enforcement and rapid cold-chain expansion.
Why are antibacterial films becoming a procurement requirement rather than a premium feature?
Because contamination prevention is increasingly codified into regulations, audits, and supplier scorecards, making antibacterial functionality essential for market access in multiple end-use sectors.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-Use , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-Use , 2026 to 2036
- Food Packaging
- Medical & Healthcare
- Consumer Goods
- Electronics & Touch Surfaces
- Food Packaging
- Y to o to Y Growth Trend Analysis By End-Use , 2021 to 2025
- Absolute $ Opportunity Analysis By End-Use , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Active Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Active Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Active Technology, 2026 to 2036
- Silver-Ion Coatings
- Zinc-Based Antimicrobials
- Organic Antimicrobial Systems
- Silver-Ion Coatings
- Y to o to Y Growth Trend Analysis By Active Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Active Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Base Polymer
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Base Polymer, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Base Polymer, 2026 to 2036
- PE/PET films
- Multilayer Polymer Films
- PE/PET films
- Y to o to Y Growth Trend Analysis By Base Polymer, 2021 to 2025
- Absolute $ Opportunity Analysis By Base Polymer, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By End-Use
- By Active Technology
- By Base Polymer
- By Country
- Market Attractiveness Analysis
- By Country
- By End-Use
- By Active Technology
- By Base Polymer
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By End-Use
- By Active Technology
- By Base Polymer
- By Country
- Market Attractiveness Analysis
- By Country
- By End-Use
- By Active Technology
- By Base Polymer
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By End-Use
- By Active Technology
- By Base Polymer
- By Country
- Market Attractiveness Analysis
- By Country
- By End-Use
- By Active Technology
- By Base Polymer
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By End-Use
- By Active Technology
- By Base Polymer
- By Country
- Market Attractiveness Analysis
- By Country
- By End-Use
- By Active Technology
- By Base Polymer
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By End-Use
- By Active Technology
- By Base Polymer
- By Country
- Market Attractiveness Analysis
- By Country
- By End-Use
- By Active Technology
- By Base Polymer
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By End-Use
- By Active Technology
- By Base Polymer
- By Country
- Market Attractiveness Analysis
- By Country
- By End-Use
- By Active Technology
- By Base Polymer
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By End-Use
- By Active Technology
- By Base Polymer
- By Country
- Market Attractiveness Analysis
- By Country
- By End-Use
- By Active Technology
- By Base Polymer
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By End-Use
- By Active Technology
- By Base Polymer
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By End-Use
- By Active Technology
- By Base Polymer
- Competition Analysis
- Competition Deep Dive
- Berry Global Group, Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Amcor plc
- Mondi Group
- Wipak Group
- Kuraray Co., Ltd.
- Berry Global Group, Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by End-Use , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Active Technology, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Base Polymer, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End-Use , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Active Technology, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Base Polymer, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by End-Use , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Active Technology, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Base Polymer, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by End-Use , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Active Technology, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Base Polymer, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by End-Use , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Active Technology, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Base Polymer, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by End-Use , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Active Technology, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Base Polymer, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by End-Use , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Active Technology, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Base Polymer, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by End-Use , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Active Technology, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Base Polymer, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by End-Use , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by End-Use , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by End-Use
- Figure 6: Global Market Value Share and BPS Analysis by Active Technology, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Active Technology, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Active Technology
- Figure 9: Global Market Value Share and BPS Analysis by Base Polymer, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Base Polymer, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Base Polymer
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by End-Use , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End-Use , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End-Use
- Figure 26: North America Market Value Share and BPS Analysis by Active Technology, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Active Technology, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Active Technology
- Figure 29: North America Market Value Share and BPS Analysis by Base Polymer, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Base Polymer, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Base Polymer
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by End-Use , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by End-Use , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by End-Use
- Figure 36: Latin America Market Value Share and BPS Analysis by Active Technology, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Active Technology, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Active Technology
- Figure 39: Latin America Market Value Share and BPS Analysis by Base Polymer, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Base Polymer, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Base Polymer
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by End-Use , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by End-Use , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by End-Use
- Figure 46: Western Europe Market Value Share and BPS Analysis by Active Technology, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Active Technology, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Active Technology
- Figure 49: Western Europe Market Value Share and BPS Analysis by Base Polymer, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Base Polymer, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Base Polymer
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by End-Use , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by End-Use , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by End-Use
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Active Technology, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Active Technology, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Active Technology
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Base Polymer, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Base Polymer, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Base Polymer
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by End-Use , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by End-Use , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by End-Use
- Figure 66: East Asia Market Value Share and BPS Analysis by Active Technology, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Active Technology, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Active Technology
- Figure 69: East Asia Market Value Share and BPS Analysis by Base Polymer, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Base Polymer, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Base Polymer
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by End-Use , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by End-Use , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by End-Use
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Active Technology, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Active Technology, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Active Technology
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Base Polymer, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Base Polymer, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Base Polymer
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by End-Use , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by End-Use , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by End-Use
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Active Technology, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Active Technology, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Active Technology
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Base Polymer, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Base Polymer, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Base Polymer
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

