At-Home Syphilis Testing Market
At-Home Syphilis Testing Market Analysis - Size, Share, and Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
At-Home Syphilis Testing Market Forecast and Outlook 2025 to 2035
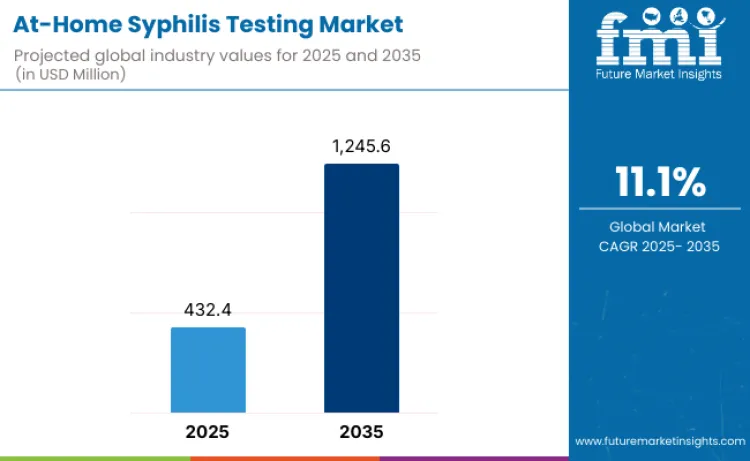
At-home syphilis testing market is projected to reach USD 1,245.6 million by 2035, recording an absolute increase of USD 813.2 million over the forecast period. This market is valued at USD 432.4 million in 2025 and is set to rise at a CAGR of 11.16% during the assessment period. Overall size is expected to grow by nearly 2.88x times, supported by rising STI burden globally, increasing emphasis on discreet and convenient sexual-health diagnostics, and expansion of digital health ecosystems enabling remote screening, counseling, and treatment pathways.
Expansion reflects fundamental shifts in sexual-health seeking behavior, where at-home syphilis test kits enable individuals to conduct private screening, enhance early detection rates, and eliminate clinic-visit barriers while maintaining seamless linkage to confirmatory testing and tele-consultation services. Sexually active individuals, high-risk cohorts, and users of preventive sexual-health programs face growing pressure to adopt regular screening practices, with self-testing solutions typically providing 30-50% improvement in testing adherence and accessibility compared with traditional clinic-based diagnostics.
Quick Stats for At-Home Syphilis Testing Market
- At-Home Syphilis Testing Market Value (2025): USD 432.4 million
- At-Home Syphilis Testing Market Forecast Value (2035): USD 1,245.6 million
- At-Home Syphilis Testing Market Forecast CAGR: 11.16%
- Leading Market Segment in At-Home Syphilis Testing Market: Blood sample tests 63.2% share driven by superior accuracy for treponemal screening and suitability for rapid & mail-in kits
- Key Growth Regions in At-Home Syphilis Testing Market: North America, Europe, and Asia Pacific
- Top Players in At-Home Syphilis Testing Market: LetsGetChecked, Everlywell, myLAB Box, Mylab Discovery Solutions, OraSure Technologies
At-Home Syphilis Testing Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 432.4 million |
| Market Forecast Value (2035) | USD 1,245.6 million |
| Forecast CAGR (2025 to 2035) | 11.16% |
At-Home Syphilis Testing Market Year-over-Year Forecast (2025-2035)
Technological advancements in rapid test sensitivity, digital result interpretation, and sample-to-lab logistics are reshaping the landscape. Modern platforms incorporate improved treponemal or non-treponemal antibody detection, app-based guidance to minimize user error, and encrypted tele-health reporting features that streamline treatment initiation and partner-notification workflows. Seamless integration with virtual sexual-health clinics, electronic prescriptions, and automated follow-up systems supports continuous screening cycles and enhances disease management outcomes.
Between 2025 and 2030, at-home syphilis testing market is projected to expand from USD 432.4 million to USD 733.9 million, resulting in a value increase of USD 301.5 million, which represents 37.1% of total forecast growth for the decade. This phase will be shaped by rising STI incidence rates, increasing preference for private and convenient self-testing options, and rapid adoption of digital sexual-health platforms enabling seamless testing-to-treatment pathways.
From 2030 to 2035, growth is forecast to expand from USD 733.9 million to USD 1,245.6 million, adding another USD 511.7 million, which constitutes 62.9% of overall ten-year expansion. This period is expected to be defined by wider deployment of digitally integrated screening kits, automated test interpretation tools, and bundled STI self-testing portfolios offering multi-pathogen home screening solutions.
Why Is the At-Home Syphilis Testing Market Growing Worldwide?
At-home syphilis testing market grows by enabling individuals to access confidential, rapid, and convenient STI screening without the need for clinic visits, helping overcome stigma, accessibility barriers, and healthcare-system bottlenecks. Rising syphilis incidence globally, particularly among sexually active adults, high-risk populations, and prenatal segments, is increasing demand for routine and early-stage detection, with self-testing typically improving screening compliance by 30-50% compared to clinic-based pathways.
Growing awareness of sexual wellness, increased frequency of multi-partner dynamics, and expanding digital health ecosystems are driving adoption of clinically reliable self-collection kits and rapid finger-prick tests that support early diagnosis and treatment linkage. Government- and NGO-backed initiatives promoting STI prevention, maternal health protection, and decentralized testing access are accelerating adoption across underserved geographies and youth populations.
Public-health agencies and telehealth platforms are integrating at-home testing into virtual sexual-health programs to improve disease surveillance, reduce transmission risk, and support PrEP programs where frequent syphilis monitoring is essential. Global shift toward privacy-oriented, user-directed healthcare and rise of online sexual-wellness services expand self-testing uptake as consumers prioritize discreet, on-demand diagnostic solutions.
How Are the At-Home Syphilis Testing Market Segmented by Product Type, Sample Type, End User and Region?
At-home syphilis testing market is segmented by product type, sample type, end user, and region. By product type, it is divided into rapid test kits, digital test kits, laboratory mail-in kits, and others. Based on sample type, it is categorized into blood, urine, saliva, and others. By end user, it is segmented into individual consumers, pharmacies & retail, and online channels. Regionally, it is divided into Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
By Product Type, Rapid Test Kits Account for 46.7% Share

Rapid test kits dominate the at-home syphilis testing landscape with a 46.7% share in 2025, driven by instant finger-prick formats that offer fast, private, and affordable screening without clinic visits. Their convenience supports early detection, routine STI testing, and broad access through pharmacies, public-health programs, and digital sexual-wellness platforms. Digital test kits hold 22.9% share, appealing to users seeking smartphone-guided interpretation. Laboratory mail-in kits represent 21.4% share, providing higher diagnostic accuracy, while the remaining 9.0% includes bundled STI panels and emerging biotech-based formats.
Key advantages driving the rapid-test dominance include:
- Instant screening outcomes enabling faster linkage to treatment and partner notification
- High privacy and convenience supporting routine self-testing without clinic interaction
- Ideal fit for community distribution programs and first-time testers with low learning barriers
By Sample Type, Blood Sample Tests Account for 63.2% Share
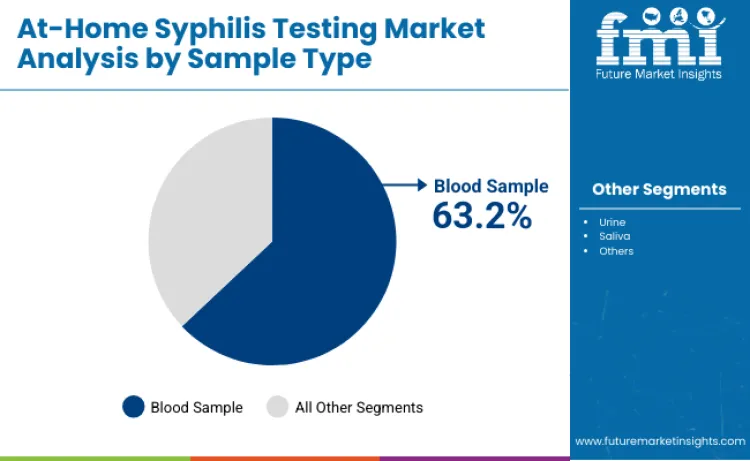
Blood-based tests lead the at-home syphilis testing segment with 63.2% share in 2025, reflecting their high diagnostic sensitivity and strong clinical acceptance for detecting both active and past infections. Their reliability supports use in rapid self-tests, mail-in kits, and structured screening programs across maternal health, MSM communities, and digital sexual-health platforms. Urine-based tests hold 18.6% share, offering a non-invasive collection option, while saliva-based formats represent 11.7% as emerging tools for broader STI self-screening. The remaining 6.5% includes hybrid and experimental sample-collection technologies.
Key market dynamics supporting blood-sample leadership include:
- Highest accuracy and sensitivity for treponemal and non-treponemal screening
- Strong clinical validation and regulatory acceptance in global markets
- Preferred format for confirmatory self-testing and repeat-screening users
By End User, Individual Consumers Account for 59.4% Share
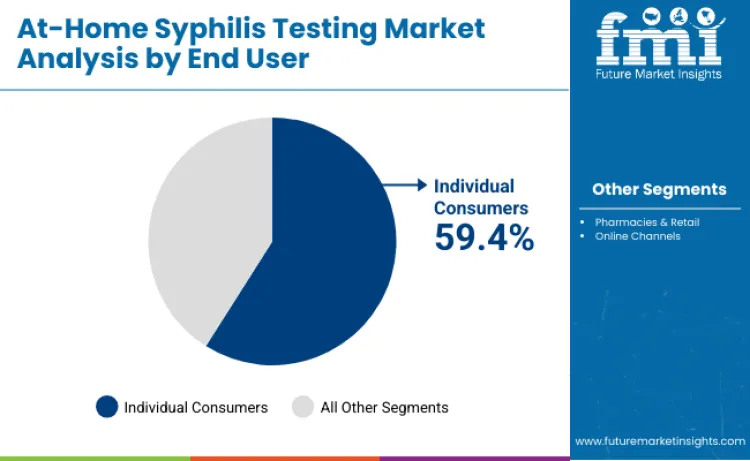
Individual consumers lead the at-home syphilis testing segment with 59.4% share in 2025, driven by rising self-directed sexual-health practices, growing demand for convenient private testing, and widespread comfort with home diagnostics. Discreet online purchasing, subscription-based sexual-wellness services, and telehealth ecosystems that enable testing, consultation, and treatment remotely further reinforce this segment’s dominance. Awareness campaigns and digital health adoption among young adults continue to accelerate uptake. Pharmacies and retail channels hold 23.8% share, while online platforms represent 16.8% through e-commerce, telehealth providers, and digitally enabled STI testing programs.
Key drivers supporting individual-consumer dominance include:
- Growing preference for private, stigma-free testing without clinic visits
- Digital health platforms encouraging direct-to-consumer self-testing culture
- Increasing subscription-based sexual-wellness services with home-testing bundles
What Are the Major Drivers, Restraints, and Emerging Trends Influencing the At-Home Syphilis Testing Market?
The market is driven by rising global syphilis incidence, particularly among sexually active adults, PrEP users, and maternal-care populations, creating sustained demand for frequent, discreet screening. Increasing STI caseloads and expansion of sexual-health programs support rapid uptake of at-home diagnostics. Consumer preference for private, self-directed health management further accelerates adoption as telehealth access, stigma reduction, and digital result-linkage platforms make routine screening easier outside clinical settings. Growth is also reinforced by direct-to-consumer sexual-wellness platforms offering subscription-based self-testing programs that support ongoing monitoring and early detection.
What Are the Major Restraints Limiting Adoption of At-Home Syphilis Testing?
Market restraints include limited awareness and affordability barriers in low-income regions, where clinic-based testing is often subsidized. User error concerns and anxiety around self-collection or result interpretation hinder adoption among first-time testers. Regulatory variability and inconsistent healthcare or insurance coverage slow penetration across several geographies. Misconceptions regarding accuracy relative to laboratory testing reduce confidence among some consumers.
What Emerging Trends Are Shaping the Future of the At-Home Syphilis Testing Market?
Key trends highlight rapid adoption in North America, Europe, and Asia Pacific, where digital sexual-health networks, public-health campaigns, and NGO distribution programs expand access. Innovation focuses on AI-assisted interpretation, app-guided sample collection, and bundled multipanel STI test kits combining syphilis, HIV, chlamydia, and gonorrhea screening. Telehealth integration enables seamless test-to-treatment pathways with digital prescriptions and partner-notification tools. Maternal-care programs and PrEP-aligned protocols strengthen structural demand. Market evolution will be shaped by convergence of rapid self-tests, lab-connected platforms, and hybrid community screening models forming a decentralized diagnostic ecosystem.
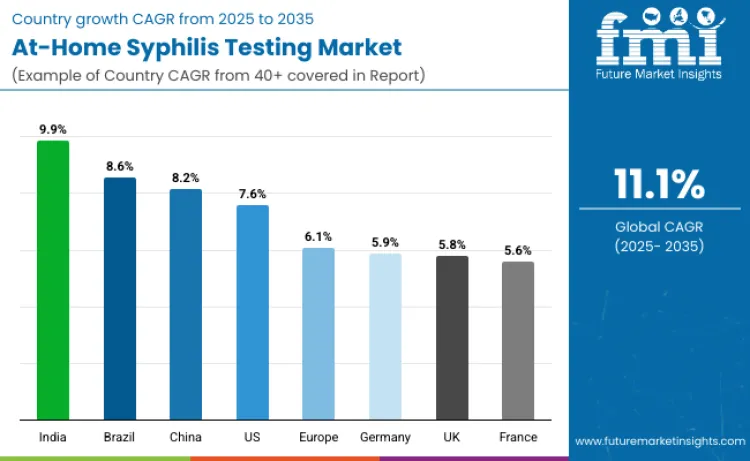
How Are Leading Countries Driving Growth in the At-Home Syphilis Testing Market?
| Country/Region | CAGR (2025-2035) |
|---|---|
| India | 9.9% |
| Brazil | 8.6% |
| China | 8.2% |
| USA | 7.6% |
| Europe | 6.1% |
| Germany | 5.9% |
| UK | 5.8% |
| France | 5.6% |
The at-home syphilis testing landscape is expanding rapidly, led by India (9.9%), where rising sexual-health awareness, digital health adoption, and maternal-care screening programs drive strong growth. Brazil (8.6%) follows through STI prevention campaigns and telehealth-enabled community testing, while China (8.2%) scales quickly via e-commerce channels and technology-driven wellness adoption. The USA (7.6%) maintains momentum through widespread home-diagnostic acceptance and telehealth-linked STI subscriptions. Europe (6.1%), including Germany (5.9%), the UK (5.8%), and France (5.6%), shows steady uptake through national programs, NGO outreach, and expanding pharmacy and online channels.
The report covers an in-depth analysis of 40+ countries top-performing countries are highlighted below.
How Is Digital Health Infrastructure Accelerating Growth in China?
China shows strong growth potential in at-home syphilis testing with a CAGR of 8.2% through 2035, supported by rapid digital-health expansion, nationwide STI prevention efforts, and rising adoption of home diagnostics across metropolitan and tier-2 cities. Growth is concentrated in Beijing, Shanghai, Guangzhou, and Shenzhen, where digitally engaged consumers and sexual-wellness campaigns drive demand for discreet self-testing. E-pharmacies, government-backed teleconsultation, and widespread mobile-health use enhance access to kits and digital reporting. Healthy China 2030 and provincial digital-health programs promote preventive care and early detection, strengthening consumer trust in self-testing pathways.
Key market factors:
- Growing STI prevention priority supported by national health campaigns, maternal care policies, and urban sexual-wellness programs promoting early and routine screening
- Strong telehealth and e-pharmacy penetration via platforms such as JD Health and Alibaba Health enabling seamless kit delivery, online guidance, and tele-consultation access
Why Are Public Health Programs Enhancing Adoption in India?
India’s at-home syphilis testing uptake is accelerating across metropolitan healthcare hubs and digital-first consumer markets, driven by Digital India initiatives and expanding national screening efforts. The segment shows strong momentum with a CAGR of 9.9% through 2035, supported by rising STI burden, growing sexual-wellness awareness, and wider access to home diagnostics via smartphone-enabled care platforms. Consumers increasingly use self-testing kits to maintain privacy, avoid stigma, and access teleconsultation-based treatment. Government programs, maternal screening initiatives, and NGO outreach strengthen early diagnosis, while telemedicine and e-pharmacy fulfillment encourage routine self-screening across urban and tier-2 regions.
Key market factors:
- Leading adoption across major metro clusters including Bengaluru, Mumbai, Delhi-NCR, and Hyderabad through digital clinics, e-pharmacies, and sexual-wellness healthtech platforms
- Government and NGO-driven STI prevention and maternal health programs strengthening early screening and at-home test kit distribution in high-risk and underserved populations
How Do Sexual-Wellness Standards Sustain Leadership in Germany?
Germany’s advanced healthcare system supports strong adoption of at-home syphilis testing, with growing consumer trust in certified diagnostics and increasing use of self-tests to complement routine sexual-health screening across urban and university populations. Major regions such as Berlin, Hamburg, Munich, and Frankfurt enable integrated digital-care pathways through regulated e-pharmacies and telemedicine platforms offering test kits, teleconsultation, and digital prescriptions. Emphasis on accuracy, CE-marked validation, and data privacy drives demand for premium products. With a CAGR of 5.9% through 2035, growth is reinforced by public-health STI initiatives, education programs, and strong pharmacy-retail access.
Key development areas:
- Widespread adoption across university health networks and young urban adults supported by confidential home-testing solutions and clinical-grade accuracy standards
- Healthcare partnerships with insurers, pharmacies, and digital-health providers enabling reimbursement pilots and integrated online care navigation for positive cases
Why Is Expanding Digital Health Fueling Demand in Brazil?
Brazil leads Latin American adoption of at-home syphilis testing, supported by rising STI prevalence, expanding public-health screening initiatives, and growing use of digital healthcare services that enable private, remote sexual-health testing. With a CAGR of 8.6% through 2035, momentum is driven by increased awareness, rapid telemedicine uptake, and strong demand for discreet home-testing options across São Paulo, Rio de Janeiro, Brasília, and Porto Alegre. Consumers use self-testing to maintain privacy, reduce stigma, and support early detection among high-risk groups. Expanding e-pharmacy, retail drugstore, and community-health distribution improves nationwide accessibility.
Leading market drivers:
- Independent users and young adults adopting self-testing to avoid stigma and ensure privacy in sexual-health screening routines
- Retail pharmacy and digital-health partnerships expanding access through subsidized pricing, installment-payment options, and discreet online ordering
How Do Established Digital Health Ecosystems Strengthen Performance in United States?
The USA at-home syphilis testing market shows mature adoption supported by advanced digital-health infrastructure, high STI awareness, and strong acceptance of self-directed sexual-wellness services across major hubs including California, New York, Texas, and Florida. With a CAGR of 7.6% through 2035, growth is reinforced by rising syphilis incidence, insurance-backed telemedicine pathways, and broad consumer readiness for discreet home testing among more than 30 million digital-health users annually. Leading diagnostic and health-tech companies enable seamless integration of test kits with app-based guidance, lab networks, and electronic prescriptions, strengthening accuracy, privacy, and treatment connectivity.
Key market characteristics:
- Strong sexual-health adoption culture supported by national telehealth operators, pharmacy networks, and clinical oversight systems ensuring accurate test use and follow-up care
- Established distribution infrastructure through large pharmacy chains, e-commerce channels, and digital clinic platforms offering discreet delivery and round-the-clock support
How Is Digital Sexual-Health Expansion Supporting Growth in United Kingdom?
In London, Manchester, Birmingham, and Edinburgh, sexual-health providers, NHS digital pathways, and independent telehealth platforms are expanding at-home syphilis testing to improve routine screening, early diagnosis, and access for users seeking privacy and shorter wait times. With a CAGR of 5.8% through 2035, growth is supported by the UK’s strong public-health infrastructure, digital-first health strategy, and emphasis on STI prevention. Consumers increasingly adopt home kits and teleconsultation-based diagnostics to maintain discretion, strengthen regular screening habits, and enable rapid treatment linkage while easing NHS capacity pressures across urban and university populations.
Market development factors:
- Nationwide integration across NHS sexual-health programs and pharmacy-supported home-testing initiatives improving routine screening and early treatment access
- University and youth-focused sexual-wellness outreach enabling discreet testing adoption through mail-order programs, student health services, and digital guidance tools
How Are Public Health Investments Advancing Market Expansion Across Europe?
Europe’s at-home syphilis testing market is set to grow from USD 88.6 million in 2025 to USD 247.9 million by 2035 at a CAGR of 10.84%, driven by strong sexual-health policies, expanding telehealth infrastructure, and rising integration of self-testing into national STI programs. Germany leads with 28.1% share, supported by advanced healthcare systems and high trust in certified diagnostics.
France holds 22.4% through national prevention strategies and pharmacy-led services, while the UK maintains 19.2% with NHS-supported pathways. Italy accounts for 13.0%, Spain 9.7%, and the Netherlands 6.8%. The remaining 7.5% comes from Nordic and Central & Eastern European regions advancing digital self-testing initiatives.
Privacy-Centric Testing and Clinical Reliability Dominate Requirements in Japan
Japan’s at-home syphilis testing landscape reflects a mature, trust-driven ecosystem enabled by clinically validated self-testing kits and physician-supervised digital workflows integrated across leading telehealth platforms, pharmacy networks, and urban wellness programs. Strong emphasis on medical precision, regulatory rigor, and confidentiality fuels demand for highly accurate, premium self-testing solutions that align with national quality standards and strict diagnostic reliability expectations.
Collaboration between established diagnostic companies, government health agencies, and major telemedicine providers enables seamless test-to-care pathways prioritizing discretion, clinical accuracy, and secure digital record management. Consumers in Tokyo, Osaka, and other metropolitan regions show advanced adoption, valuing privacy and medically supervised follow-up. Growth remains steady, supported by a CAGR of 11.38% through 2035 and rising preference for certified, trustworthy preventive-care tools.
International Diagnostic Platforms Lead Digital Sexual-Health Services in South Korea
South Korea’s at-home syphilis testing market is expanding through increasing involvement from global digital-health and diagnostic providers, which strengthen their presence via localized telemedicine support, multilingual testing guidance, and integrated digital prescription pathways. Demand is rising as consumers favor private, discreet sexual-health solutions aligned with cultural preferences for confidentiality.
Digital healthcare platforms and pharmacy-delivery services enable access to certified kits, secure app-based result interpretation, and physician-supervised treatment through major telehealth ecosystems such as Naver and Kakao. Retail pharmacies and online marketplaces gain share by partnering with international brands to offer authenticated products, discreet delivery, and Korean-language counseling. Collaboration between multinational health-tech firms and medical associations supports hybrid service models. Growth remains steady with a CAGR of 11.71% through 2035.
How Competitive Is the Global At-Home Syphilis Testing Market and What Defines Its Structure?
The at-home syphilis testing landscape is moderately consolidated, with a small group of leading companies shaping global performance. Competition is driven by diagnostic accuracy, regulatory approvals, digital-health integration, confidential logistics, and seamless linkage to clinical care rather than pricing. LetsGetChecked holds a leading position due to its extensive home-diagnostics portfolio, certified laboratory capabilities, strong telehealth networks, and high consumer trust across major regions.
Everlywell and myLAB Box strengthen their positions through omnichannel distribution, subscription-based sexual-wellness programs, national retail partnerships, and rapid physician follow-up pathways. These companies continue investing in secure data systems, optimized logistics, and clinician-supervised testing workflows to expand their reach.
Challengers such as SelfDiagnostics, Healthy.io, and DetectaChem compete through smartphone-assisted diagnostics, AI-enhanced interpretation tools, and real-time teleconsultation models appealing to digitally engaged consumers and health-system partners. Product specialists like Mylab Discovery Solutions and OraSure Technologies focus on regulatory rigor and integrated manufacturing to deliver validated test kits for consumer use and public-health programs. Market momentum increasingly favors companies that pair clinically validated diagnostics with privacy-compliant digital ecosystems, discreet fulfillment models, automated patient support, and streamlined care-coordination pathways.
Key Players in the At-Home Syphilis Testing Market
- LetsGetChecked
- Everlywell
- myLAB Box
- SelfDiagnostics
- Healthy.io
- DetectaChem
- Mylab Discovery Solutions
- OraSure Technologies
- Others
Scope of the Report
| Item | Value |
|---|---|
| Quantitative Units | USD 432.4 million |
| Product Type | Rapid Test Kits, Digital Test Kits, Laboratory Mail-in Kits, Others |
| Sample Type | Blood, Urine, Saliva, Others |
| End User | Individual Consumers, Pharmacies & Retail, Online Channels |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Country Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40+ countries |
| Key Companies Profiled | LetsGetChecked, Everlywell, myLAB Box, SelfDiagnostics, Healthy.io, DetectaChem, Mylab Discovery Solutions, OraSure Technologies |
| Additional Attributes | Dollar sales by product type and sample format, regional trends across North America, Europe, Asia Pacific, and Latin America, competitive landscape of DTC health platforms, regulatory requirements, telehealth integration, innovations in rapid immunoassays, mail-in logistics, smartphone workflows, and privacy-focused home-testing with automated treatment linkage and prevention programs. |
At-Home Syphilis Testing Market by Segments
By Product Type:
- Rapid Test Kits
- Digital Test Kits
- Laboratory Mail-in Kits
- Others
By Sample Type:
- Blood
- Urine
- Saliva
- Others
By End User:
- Individual Consumers
- Pharmacies & Retail
- Online Channels
By Region:
- Asia Pacific
- China
- India
- Japan
- South Korea
- Vietnam
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the at-home syphilis testing market in 2025?
The global at-home syphilis testing market is valued at USD 432.4 million in 2025.
What will be the size of the at-home syphilis testing market in 2035?
The market is projected to reach USD 1,245.6 million by 2035.
How fast will the at-home syphilis testing market grow between 2025 and 2035?
The market will grow at a CAGR of 11.16% from 2025 to 2035.
Which product type leads the at-home syphilis testing market in 2025?
Rapid test kits lead the market with a 46.7% share in 2025.
Who are the major players in the at-home syphilis testing market?
Key players include LetsGetChecked, Everlywell, myLAB Box, Mylab Discovery Solutions, and OraSure Technologies.
Table of Content
- Executive Summary
- Regional Level Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Assessment
- Investment Feasibility Matrix
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- By Key Regions
- By Key Countries
- Regional Parent Market Outlook
- Technological Innovation
- Market Dynamics
- Regional Level At-Home Syphilis Testing Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size US$ Mn Analysis, 2020-2024
- Current and Future Market Size US$ Mn Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Regional Level At-Home Syphilis Testing Market Analysis By Product Type
- Introduction / Key Findings
- Historical Market Size US$ Mn Analysis By Product Type, 2020-2024
- Current & Future Market Size US$ Mn Forecast By Product Type, 2025-2035
- Rapid Test Kits
- Digital Test Kits
- Laboratory Mail-in Kits
- Others
- Y-o-Y Growth Trend Analysis By Product Type, 2020-2024
- Absolute $ Opportunity Analysis By Product Type, 2025-2035
- Regional Level At-Home Syphilis Testing Market Analysis By Sample Type
- Introduction / Key Findings
- Historical Market Size US$ Mn Analysis By Sample Type, 2020-2024
- Current & Future Market Size US$ Mn Forecast By Sample Type, 2025-2035
- Blood
- Urine
- Saliva
- Others
- Y-o-Y Growth Trend Analysis By Sample Type, 2020-2024
- Absolute $ Opportunity Analysis By Sample Type, 2025-2035
- Regional Level At-Home Syphilis Testing Market Analysis By End User
- Introduction / Key Findings
- Historical Market Size US$ Mn Analysis By End User, 2020-2024
- Current & Future Market Size US$ Mn Forecast By End User, 2025-2035
- Individual Consumers
- Pharmacies & Retail
- Online Channels
- Y-o-Y Growth Trend Analysis By End User, 2020-2024
- Absolute $ Opportunity Analysis By End User, 2025-2035
- Regional Level At-Home Syphilis Testing Market Analysis By Region
- Introduction
- Historical Market Size US$ Mn Analysis By Region, 2020-2024
- Current Market Size US$ Mn Forecast By Region, 2025-2035
- North America
- Latin America
- East Asia
- South Asia
- Western Europe
- Eastern Europe
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America At-Home Syphilis Testing Market
- Historical Market Size US$ Mn Trend Analysis, 2020-2024
- Market Size US$ Mn Forecast, 2025-2035
- By Country
- U.S.
- Canada
- Mexico
- By Product Type
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sample Type
- By End User
- Key Takeaways
- Latin America At-Home Syphilis Testing Market
- Historical Market Size US$ Mn Trend Analysis, 2020-2024
- Market Size US$ Mn Forecast, 2025-2035
- By Country
- Brazil
- Argentina
- Rest of Latin America
- By Product Type
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sample Type
- By End User
- Key Takeaways
- East Asia At-Home Syphilis Testing Market
- Historical Market Size US$ Mn Trend Analysis, 2020-2024
- Market Size US$ Mn Forecast, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sample Type
- By End User
- Key Takeaways
- South Asia At-Home Syphilis Testing Market
- Historical Market Size US$ Mn Trend Analysis, 2020-2024
- Market Size US$ Mn Forecast, 2025-2035
- By Country
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- By Product Type
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sample Type
- By End User
- Key Takeaways
- Western Europe At-Home Syphilis Testing Market
- Historical Market Size US$ Mn Trend Analysis, 2020-2024
- Market Size US$ Mn Forecast, 2025-2035
- By Country
- Germany
- U.K.
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- By Product Type
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sample Type
- By End User
- Key Takeaways
- Eastern Europe At-Home Syphilis Testing Market
- Historical Market Size US$ Mn Trend Analysis, 2020-2024
- Market Size US$ Mn Forecast, 2025-2035
- By Country
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- By Product Type
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sample Type
- By End User
- Key Takeaways
- Middle East & Africa At-Home Syphilis Testing Market
- Historical Market Size US$ Mn Trend Analysis, 2020-2024
- Market Size US$ Mn Forecast, 2025-2035
- By Country
- Saudi Arabia
- Other GCC Countries
- Türkiye
- South Africa
- Rest of MEA
- By Product Type
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sample Type
- By End User
- Key Takeaways
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Sample Type
- By End User
- Competition Analysis
- Competition Deep Dive
- LetsGetChecked
- Overview
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Everlywell
- Overview
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- myLAB Box
- Overview
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- SelfDiagnostics
- Overview
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Healthy.io
- Overview
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- DetectaChem
- Overview
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Mylab Discovery Solutions
- Overview
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- OraSure Technologies
- Overview
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- LetsGetChecked
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Size (USD Million), by Product Type, 2020-2035
- Table 2: Global Market Size (USD Million), by Rapid Test Kits, 2020-2035
- Table 3: Global Market Size (USD Million), by Digital Test Kits, 2020-2035
- Table 4: Global Market Size (USD Million), by Laboratory Mail-in Kits, 2020-2035
- Table 5: Global Market Size (USD Million), by Others, 2020-2035
- Table 6: Global Market Size (USD Million), by Sample Type, 2020-2035
- Table 7: Global Market Size (USD Million), by Blood, 2020-2035
- Table 8: Global Market Size (USD Million), by Urine, 2020-2035
- Table 9: Global Market Size (USD Million), by Saliva, 2020-2035
- Table 10: Global Market Size (USD Million), by Others, 2020-2035
- Table 11: Global Market Size (USD Million), by End User, 2020-2035
- Table 12: Global Market Size (USD Million), by Individual Consumers, 2020-2035
- Table 13: Global Market Size (USD Million), by Pharmacies & Retail, 2020-2035
- Table 14: Global Market Size (USD Million), by Online Channels, 2020-2035
- Table 15: North America Market Size (USD Million), by Product Type, 2020-2035
- Table 16: North America Market Size (USD Million), by Rapid Test Kits, 2020-2035
- Table 17: North America Market Size (USD Million), by Digital Test Kits, 2020-2035
- Table 18: North America Market Size (USD Million), by Laboratory Mail-in Kits, 2020-2035
- Table 19: North America Market Size (USD Million), by Others, 2020-2035
- Table 20: North America Market Size (USD Million), by Sample Type, 2020-2035
- Table 21: North America Market Size (USD Million), by Blood, 2020-2035
- Table 22: North America Market Size (USD Million), by Urine, 2020-2035
- Table 23: North America Market Size (USD Million), by Saliva, 2020-2035
- Table 24: North America Market Size (USD Million), by Others, 2020-2035
- Table 25: North America Market Size (USD Million), by End User, 2020-2035
- Table 26: North America Market Size (USD Million), by Individual Consumers, 2020-2035
- Table 27: North America Market Size (USD Million), by Pharmacies & Retail, 2020-2035
- Table 28: North America Market Size (USD Million), by Online Channels, 2020-2035
- Table 29: USA Market Size (USD Million), by Product Type, 2020-2035
- Table 30: USA Market Size (USD Million), by Sample Type, 2020-2035
- Table 31: USA Market Size (USD Million), by End User, 2020-2035
- Table 32: Canada Market Size (USD Million), by Product Type, 2020-2035
- Table 33: Canada Market Size (USD Million), by Sample Type, 2020-2035
- Table 34: Canada Market Size (USD Million), by End User, 2020-2035
- Table 35: Mexico Market Size (USD Million), by Product Type, 2020-2035
- Table 36: Mexico Market Size (USD Million), by Sample Type, 2020-2035
- Table 37: Mexico Market Size (USD Million), by End User, 2020-2035
- Table 38: Latin America Market Size (USD Million), by Product Type, 2020-2035
- Table 39: Latin America Market Size (USD Million), by Rapid Test Kits, 2020-2035
- Table 40: Latin America Market Size (USD Million), by Digital Test Kits, 2020-2035
- Table 41: Latin America Market Size (USD Million), by Laboratory Mail-in Kits, 2020-2035
- Table 42: Latin America Market Size (USD Million), by Others, 2020-2035
- Table 43: Latin America Market Size (USD Million), by Sample Type, 2020-2035
- Table 44: Latin America Market Size (USD Million), by Blood, 2020-2035
- Table 45: Latin America Market Size (USD Million), by Urine, 2020-2035
- Table 46: Latin America Market Size (USD Million), by Saliva, 2020-2035
- Table 47: Latin America Market Size (USD Million), by Others, 2020-2035
- Table 48: Latin America Market Size (USD Million), by End User, 2020-2035
- Table 49: Latin America Market Size (USD Million), by Individual Consumers, 2020-2035
- Table 50: Latin America Market Size (USD Million), by Pharmacies & Retail, 2020-2035
- Table 51: Latin America Market Size (USD Million), by Online Channels, 2020-2035
- Table 52: Brazil Market Size (USD Million), by Product Type, 2020-2035
- Table 53: Brazil Market Size (USD Million), by Sample Type, 2020-2035
- Table 54: Brazil Market Size (USD Million), by End User, 2020-2035
- Table 55: Argentina Market Size (USD Million), by Product Type, 2020-2035
- Table 56: Argentina Market Size (USD Million), by Sample Type, 2020-2035
- Table 57: Argentina Market Size (USD Million), by End User, 2020-2035
- Table 58: Rest of Latin America Market Size (USD Million), by Product Type, 2020-2035
- Table 59: Rest of Latin America Market Size (USD Million), by Sample Type, 2020-2035
- Table 60: Rest of Latin America Market Size (USD Million), by End User, 2020-2035
- Table 61: East Asia Market Size (USD Million), by Product Type, 2020-2035
- Table 62: East Asia Market Size (USD Million), by Rapid Test Kits, 2020-2035
- Table 63: East Asia Market Size (USD Million), by Digital Test Kits, 2020-2035
- Table 64: East Asia Market Size (USD Million), by Laboratory Mail-in Kits, 2020-2035
- Table 65: East Asia Market Size (USD Million), by Others, 2020-2035
- Table 66: East Asia Market Size (USD Million), by Sample Type, 2020-2035
- Table 67: East Asia Market Size (USD Million), by Blood, 2020-2035
- Table 68: East Asia Market Size (USD Million), by Urine, 2020-2035
- Table 69: East Asia Market Size (USD Million), by Saliva, 2020-2035
- Table 70: East Asia Market Size (USD Million), by Others, 2020-2035
- Table 71: East Asia Market Size (USD Million), by End User, 2020-2035
- Table 72: East Asia Market Size (USD Million), by Individual Consumers, 2020-2035
- Table 73: East Asia Market Size (USD Million), by Pharmacies & Retail, 2020-2035
- Table 74: East Asia Market Size (USD Million), by Online Channels, 2020-2035
- Table 75: China Market Size (USD Million), by Product Type, 2020-2035
- Table 76: China Market Size (USD Million), by Sample Type, 2020-2035
- Table 77: China Market Size (USD Million), by End User, 2020-2035
- Table 78: Japan Market Size (USD Million), by Product Type, 2020-2035
- Table 79: Japan Market Size (USD Million), by Sample Type, 2020-2035
- Table 80: Japan Market Size (USD Million), by End User, 2020-2035
- Table 81: South Korea Market Size (USD Million), by Product Type, 2020-2035
- Table 82: South Korea Market Size (USD Million), by Sample Type, 2020-2035
- Table 83: South Korea Market Size (USD Million), by End User, 2020-2035
- Table 84: South Asia Market Size (USD Million), by Product Type, 2020-2035
- Table 85: South Asia Market Size (USD Million), by Rapid Test Kits, 2020-2035
- Table 86: South Asia Market Size (USD Million), by Digital Test Kits, 2020-2035
- Table 87: South Asia Market Size (USD Million), by Laboratory Mail-in Kits, 2020-2035
- Table 88: South Asia Market Size (USD Million), by Others, 2020-2035
- Table 89: South Asia Market Size (USD Million), by Sample Type, 2020-2035
- Table 90: South Asia Market Size (USD Million), by Blood, 2020-2035
- Table 91: South Asia Market Size (USD Million), by Urine, 2020-2035
- Table 92: South Asia Market Size (USD Million), by Saliva, 2020-2035
- Table 93: South Asia Market Size (USD Million), by Others, 2020-2035
- Table 94: South Asia Market Size (USD Million), by End User, 2020-2035
- Table 95: South Asia Market Size (USD Million), by Individual Consumers, 2020-2035
- Table 96: South Asia Market Size (USD Million), by Pharmacies & Retail, 2020-2035
- Table 97: South Asia Market Size (USD Million), by Online Channels, 2020-2035
- Table 98: India Market Size (USD Million), by Product Type, 2020-2035
- Table 99: India Market Size (USD Million), by Sample Type, 2020-2035
- Table 100: India Market Size (USD Million), by End User, 2020-2035
- Table 101: ASEAN Countries Market Size (USD Million), by Product Type, 2020-2035
- Table 102: ASEAN Countries Market Size (USD Million), by Sample Type, 2020-2035
- Table 103: ASEAN Countries Market Size (USD Million), by End User, 2020-2035
- Table 104: Australia & New Zealand Market Size (USD Million), by Product Type, 2020-2035
- Table 105: Australia & New Zealand Market Size (USD Million), by Sample Type, 2020-2035
- Table 106: Australia & New Zealand Market Size (USD Million), by End User, 2020-2035
- Table 107: Rest of South Asia Market Size (USD Million), by Product Type, 2020-2035
- Table 108: Rest of South Asia Market Size (USD Million), by Sample Type, 2020-2035
- Table 109: Rest of South Asia Market Size (USD Million), by End User, 2020-2035
- Table 110: Western Europe Market Size (USD Million), by Product Type, 2020-2035
- Table 111: Western Europe Market Size (USD Million), by Rapid Test Kits, 2020-2035
- Table 112: Western Europe Market Size (USD Million), by Digital Test Kits, 2020-2035
- Table 113: Western Europe Market Size (USD Million), by Laboratory Mail-in Kits, 2020-2035
- Table 114: Western Europe Market Size (USD Million), by Others, 2020-2035
- Table 115: Western Europe Market Size (USD Million), by Sample Type, 2020-2035
- Table 116: Western Europe Market Size (USD Million), by Blood, 2020-2035
- Table 117: Western Europe Market Size (USD Million), by Urine, 2020-2035
- Table 118: Western Europe Market Size (USD Million), by Saliva, 2020-2035
- Table 119: Western Europe Market Size (USD Million), by Others, 2020-2035
- Table 120: Western Europe Market Size (USD Million), by End User, 2020-2035
- Table 121: Western Europe Market Size (USD Million), by Individual Consumers, 2020-2035
- Table 122: Western Europe Market Size (USD Million), by Pharmacies & Retail, 2020-2035
- Table 123: Western Europe Market Size (USD Million), by Online Channels, 2020-2035
- Table 124: Germany Market Size (USD Million), by Product Type, 2020-2035
- Table 125: Germany Market Size (USD Million), by Sample Type, 2020-2035
- Table 126: Germany Market Size (USD Million), by End User, 2020-2035
- Table 127: UK Market Size (USD Million), by Product Type, 2020-2035
- Table 128: UK Market Size (USD Million), by Sample Type, 2020-2035
- Table 129: UK Market Size (USD Million), by End User, 2020-2035
- Table 130: France Market Size (USD Million), by Product Type, 2020-2035
- Table 131: France Market Size (USD Million), by Sample Type, 2020-2035
- Table 132: France Market Size (USD Million), by End User, 2020-2035
- Table 133: Italy Market Size (USD Million), by Product Type, 2020-2035
- Table 134: Italy Market Size (USD Million), by Sample Type, 2020-2035
- Table 135: Italy Market Size (USD Million), by End User, 2020-2035
- Table 136: Spain Market Size (USD Million), by Product Type, 2020-2035
- Table 137: Spain Market Size (USD Million), by Sample Type, 2020-2035
- Table 138: Spain Market Size (USD Million), by End User, 2020-2035
- Table 139: BENELUX Market Size (USD Million), by Product Type, 2020-2035
- Table 140: BENELUX Market Size (USD Million), by Sample Type, 2020-2035
- Table 141: BENELUX Market Size (USD Million), by End User, 2020-2035
- Table 142: Nordic Countries Market Size (USD Million), by Product Type, 2020-2035
- Table 143: Nordic Countries Market Size (USD Million), by Sample Type, 2020-2035
- Table 144: Nordic Countries Market Size (USD Million), by End User, 2020-2035
- Table 145: Rest of Western Europe Market Size (USD Million), by Product Type, 2020-2035
- Table 146: Rest of Western Europe Market Size (USD Million), by Sample Type, 2020-2035
- Table 147: Rest of Western Europe Market Size (USD Million), by End User, 2020-2035
- Table 148: Eastern Europe Market Size (USD Million), by Product Type, 2020-2035
- Table 149: Eastern Europe Market Size (USD Million), by Rapid Test Kits, 2020-2035
- Table 150: Eastern Europe Market Size (USD Million), by Digital Test Kits, 2020-2035
- Table 151: Eastern Europe Market Size (USD Million), by Laboratory Mail-in Kits, 2020-2035
- Table 152: Eastern Europe Market Size (USD Million), by Others, 2020-2035
- Table 153: Eastern Europe Market Size (USD Million), by Sample Type, 2020-2035
- Table 154: Eastern Europe Market Size (USD Million), by Blood, 2020-2035
- Table 155: Eastern Europe Market Size (USD Million), by Urine, 2020-2035
- Table 156: Eastern Europe Market Size (USD Million), by Saliva, 2020-2035
- Table 157: Eastern Europe Market Size (USD Million), by Others, 2020-2035
- Table 158: Eastern Europe Market Size (USD Million), by End User, 2020-2035
- Table 159: Eastern Europe Market Size (USD Million), by Individual Consumers, 2020-2035
- Table 160: Eastern Europe Market Size (USD Million), by Pharmacies & Retail, 2020-2035
- Table 161: Eastern Europe Market Size (USD Million), by Online Channels, 2020-2035
- Table 162: Russia Market Size (USD Million), by Product Type, 2020-2035
- Table 163: Russia Market Size (USD Million), by Sample Type, 2020-2035
- Table 164: Russia Market Size (USD Million), by End User, 2020-2035
- Table 165: Hungary Market Size (USD Million), by Product Type, 2020-2035
- Table 166: Hungary Market Size (USD Million), by Sample Type, 2020-2035
- Table 167: Hungary Market Size (USD Million), by End User, 2020-2035
- Table 168: Poland Market Size (USD Million), by Product Type, 2020-2035
- Table 169: Poland Market Size (USD Million), by Sample Type, 2020-2035
- Table 170: Poland Market Size (USD Million), by End User, 2020-2035
- Table 171: Rest of Eastern Europe Market Size (USD Million), by Product Type, 2020-2035
- Table 172: Rest of Eastern Europe Market Size (USD Million), by Sample Type, 2020-2035
- Table 173: Rest of Eastern Europe Market Size (USD Million), by End User, 2020-2035
- Table 174: Middle East & Africa Market Size (USD Million), by Product Type, 2020-2035
- Table 175: Middle East & Africa Market Size (USD Million), by Rapid Test Kits, 2020-2035
- Table 176: Middle East & Africa Market Size (USD Million), by Digital Test Kits, 2020-2035
- Table 177: Middle East & Africa Market Size (USD Million), by Laboratory Mail-in Kits, 2020-2035
- Table 178: Middle East & Africa Market Size (USD Million), by Others, 2020-2035
- Table 179: Middle East & Africa Market Size (USD Million), by Sample Type, 2020-2035
- Table 180: Middle East & Africa Market Size (USD Million), by Blood, 2020-2035
- Table 181: Middle East & Africa Market Size (USD Million), by Urine, 2020-2035
- Table 182: Middle East & Africa Market Size (USD Million), by Saliva, 2020-2035
- Table 183: Middle East & Africa Market Size (USD Million), by Others, 2020-2035
- Table 184: Middle East & Africa Market Size (USD Million), by End User, 2020-2035
- Table 185: Middle East & Africa Market Size (USD Million), by Individual Consumers, 2020-2035
- Table 186: Middle East & Africa Market Size (USD Million), by Pharmacies & Retail, 2020-2035
- Table 187: Middle East & Africa Market Size (USD Million), by Online Channels, 2020-2035
- Table 188: Saudi Arabia Market Size (USD Million), by Product Type, 2020-2035
- Table 189: Saudi Arabia Market Size (USD Million), by Sample Type, 2020-2035
- Table 190: Saudi Arabia Market Size (USD Million), by End User, 2020-2035
- Table 191: Other GCC Countries Market Size (USD Million), by Product Type, 2020-2035
- Table 192: Other GCC Countries Market Size (USD Million), by Sample Type, 2020-2035
- Table 193: Other GCC Countries Market Size (USD Million), by End User, 2020-2035
- Table 194: Türkiye Market Size (USD Million), by Product Type, 2020-2035
- Table 195: Türkiye Market Size (USD Million), by Sample Type, 2020-2035
- Table 196: Türkiye Market Size (USD Million), by End User, 2020-2035
- Table 197: South Africa Market Size (USD Million), by Product Type, 2020-2035
- Table 198: South Africa Market Size (USD Million), by Sample Type, 2020-2035
- Table 199: South Africa Market Size (USD Million), by End User, 2020-2035
- Table 200: Rest of MEA Market Size (USD Million), by Product Type, 2020-2035
- Table 201: Rest of MEA Market Size (USD Million), by Sample Type, 2020-2035
- Table 202: Rest of MEA Market Size (USD Million), by End User, 2020-2035
List of Figures
- Figure 1: Global Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 2: Global Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 3: Global Market (USD Million) Forecast, by Digital Test Kits, 2020-2035
- Figure 4: Global Market (USD Million) Forecast, by Laboratory Mail-in Kits, 2020-2035
- Figure 5: Global Market (USD Million) Forecast, by Others, 2020-2035
- Figure 6: Global Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 7: Global Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 8: Global Market (USD Million) Forecast, by Urine, 2020-2035
- Figure 9: Global Market (USD Million) Forecast, by Saliva, 2020-2035
- Figure 10: Global Market (USD Million) Forecast, by Others, 2020-2035
- Figure 11: Global Market (USD Million) Forecast, by End User, 2020-2035
- Figure 12: Global Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 13: Global Market (USD Million) Forecast, by Pharmacies & Retail, 2020-2035
- Figure 14: Global Market (USD Million) Forecast, by Online Channels, 2020-2035
- Figure 15: North America Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 16: North America Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 17: North America Market (USD Million) Forecast, by Digital Test Kits, 2020-2035
- Figure 18: North America Market (USD Million) Forecast, by Laboratory Mail-in Kits, 2020-2035
- Figure 19: North America Market (USD Million) Forecast, by Others, 2020-2035
- Figure 20: North America Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 21: North America Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 22: North America Market (USD Million) Forecast, by Urine, 2020-2035
- Figure 23: North America Market (USD Million) Forecast, by Saliva, 2020-2035
- Figure 24: North America Market (USD Million) Forecast, by Others, 2020-2035
- Figure 25: North America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 26: North America Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 27: North America Market (USD Million) Forecast, by Pharmacies & Retail, 2020-2035
- Figure 28: North America Market (USD Million) Forecast, by Online Channels, 2020-2035
- Figure 29: USA Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 30: USA Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 31: USA Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 32: USA Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 33: USA Market (USD Million) Forecast, by End User, 2020-2035
- Figure 34: USA Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 35: Canada Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 36: Canada Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 37: Canada Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 38: Canada Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 39: Canada Market (USD Million) Forecast, by End User, 2020-2035
- Figure 40: Canada Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 41: Mexico Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 42: Mexico Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 43: Mexico Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 44: Mexico Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 45: Mexico Market (USD Million) Forecast, by End User, 2020-2035
- Figure 46: Mexico Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 47: Latin America Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 48: Latin America Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 49: Latin America Market (USD Million) Forecast, by Digital Test Kits, 2020-2035
- Figure 50: Latin America Market (USD Million) Forecast, by Laboratory Mail-in Kits, 2020-2035
- Figure 51: Latin America Market (USD Million) Forecast, by Others, 2020-2035
- Figure 52: Latin America Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 53: Latin America Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 54: Latin America Market (USD Million) Forecast, by Urine, 2020-2035
- Figure 55: Latin America Market (USD Million) Forecast, by Saliva, 2020-2035
- Figure 56: Latin America Market (USD Million) Forecast, by Others, 2020-2035
- Figure 57: Latin America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 58: Latin America Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 59: Latin America Market (USD Million) Forecast, by Pharmacies & Retail, 2020-2035
- Figure 60: Latin America Market (USD Million) Forecast, by Online Channels, 2020-2035
- Figure 61: Brazil Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 62: Brazil Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 63: Brazil Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 64: Brazil Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 65: Brazil Market (USD Million) Forecast, by End User, 2020-2035
- Figure 66: Brazil Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 67: Argentina Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 68: Argentina Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 69: Argentina Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 70: Argentina Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 71: Argentina Market (USD Million) Forecast, by End User, 2020-2035
- Figure 72: Argentina Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 73: Rest of Latin America Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 74: Rest of Latin America Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 75: Rest of Latin America Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 76: Rest of Latin America Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 77: Rest of Latin America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 78: Rest of Latin America Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 79: East Asia Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 80: East Asia Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 81: East Asia Market (USD Million) Forecast, by Digital Test Kits, 2020-2035
- Figure 82: East Asia Market (USD Million) Forecast, by Laboratory Mail-in Kits, 2020-2035
- Figure 83: East Asia Market (USD Million) Forecast, by Others, 2020-2035
- Figure 84: East Asia Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 85: East Asia Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 86: East Asia Market (USD Million) Forecast, by Urine, 2020-2035
- Figure 87: East Asia Market (USD Million) Forecast, by Saliva, 2020-2035
- Figure 88: East Asia Market (USD Million) Forecast, by Others, 2020-2035
- Figure 89: East Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 90: East Asia Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 91: East Asia Market (USD Million) Forecast, by Pharmacies & Retail, 2020-2035
- Figure 92: East Asia Market (USD Million) Forecast, by Online Channels, 2020-2035
- Figure 93: China Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 94: China Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 95: China Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 96: China Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 97: China Market (USD Million) Forecast, by End User, 2020-2035
- Figure 98: China Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 99: Japan Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 100: Japan Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 101: Japan Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 102: Japan Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 103: Japan Market (USD Million) Forecast, by End User, 2020-2035
- Figure 104: Japan Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 105: South Korea Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 106: South Korea Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 107: South Korea Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 108: South Korea Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 109: South Korea Market (USD Million) Forecast, by End User, 2020-2035
- Figure 110: South Korea Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 111: South Asia Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 112: South Asia Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 113: South Asia Market (USD Million) Forecast, by Digital Test Kits, 2020-2035
- Figure 114: South Asia Market (USD Million) Forecast, by Laboratory Mail-in Kits, 2020-2035
- Figure 115: South Asia Market (USD Million) Forecast, by Others, 2020-2035
- Figure 116: South Asia Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 117: South Asia Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 118: South Asia Market (USD Million) Forecast, by Urine, 2020-2035
- Figure 119: South Asia Market (USD Million) Forecast, by Saliva, 2020-2035
- Figure 120: South Asia Market (USD Million) Forecast, by Others, 2020-2035
- Figure 121: South Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 122: South Asia Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 123: South Asia Market (USD Million) Forecast, by Pharmacies & Retail, 2020-2035
- Figure 124: South Asia Market (USD Million) Forecast, by Online Channels, 2020-2035
- Figure 125: India Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 126: India Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 127: India Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 128: India Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 129: India Market (USD Million) Forecast, by End User, 2020-2035
- Figure 130: India Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 131: ASEAN Countries Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 132: ASEAN Countries Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 133: ASEAN Countries Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 134: ASEAN Countries Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 135: ASEAN Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 136: ASEAN Countries Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 137: Australia & New Zealand Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 138: Australia & New Zealand Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 139: Australia & New Zealand Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 140: Australia & New Zealand Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 141: Australia & New Zealand Market (USD Million) Forecast, by End User, 2020-2035
- Figure 142: Australia & New Zealand Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 143: Rest of South Asia Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 144: Rest of South Asia Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 145: Rest of South Asia Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 146: Rest of South Asia Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 147: Rest of South Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 148: Rest of South Asia Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 149: Western Europe Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 150: Western Europe Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 151: Western Europe Market (USD Million) Forecast, by Digital Test Kits, 2020-2035
- Figure 152: Western Europe Market (USD Million) Forecast, by Laboratory Mail-in Kits, 2020-2035
- Figure 153: Western Europe Market (USD Million) Forecast, by Others, 2020-2035
- Figure 154: Western Europe Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 155: Western Europe Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 156: Western Europe Market (USD Million) Forecast, by Urine, 2020-2035
- Figure 157: Western Europe Market (USD Million) Forecast, by Saliva, 2020-2035
- Figure 158: Western Europe Market (USD Million) Forecast, by Others, 2020-2035
- Figure 159: Western Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 160: Western Europe Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 161: Western Europe Market (USD Million) Forecast, by Pharmacies & Retail, 2020-2035
- Figure 162: Western Europe Market (USD Million) Forecast, by Online Channels, 2020-2035
- Figure 163: Germany Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 164: Germany Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 165: Germany Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 166: Germany Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 167: Germany Market (USD Million) Forecast, by End User, 2020-2035
- Figure 168: Germany Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 169: UK Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 170: UK Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 171: UK Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 172: UK Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 173: UK Market (USD Million) Forecast, by End User, 2020-2035
- Figure 174: UK Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 175: France Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 176: France Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 177: France Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 178: France Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 179: France Market (USD Million) Forecast, by End User, 2020-2035
- Figure 180: France Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 181: Italy Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 182: Italy Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 183: Italy Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 184: Italy Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 185: Italy Market (USD Million) Forecast, by End User, 2020-2035
- Figure 186: Italy Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 187: Spain Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 188: Spain Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 189: Spain Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 190: Spain Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 191: Spain Market (USD Million) Forecast, by End User, 2020-2035
- Figure 192: Spain Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 193: BENELUX Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 194: BENELUX Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 195: BENELUX Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 196: BENELUX Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 197: BENELUX Market (USD Million) Forecast, by End User, 2020-2035
- Figure 198: BENELUX Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 199: Nordic Countries Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 200: Nordic Countries Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 201: Nordic Countries Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 202: Nordic Countries Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 203: Nordic Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 204: Nordic Countries Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 205: Rest of Western Europe Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 206: Rest of Western Europe Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 207: Rest of Western Europe Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 208: Rest of Western Europe Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 209: Rest of Western Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 210: Rest of Western Europe Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 211: Eastern Europe Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 212: Eastern Europe Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 213: Eastern Europe Market (USD Million) Forecast, by Digital Test Kits, 2020-2035
- Figure 214: Eastern Europe Market (USD Million) Forecast, by Laboratory Mail-in Kits, 2020-2035
- Figure 215: Eastern Europe Market (USD Million) Forecast, by Others, 2020-2035
- Figure 216: Eastern Europe Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 217: Eastern Europe Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 218: Eastern Europe Market (USD Million) Forecast, by Urine, 2020-2035
- Figure 219: Eastern Europe Market (USD Million) Forecast, by Saliva, 2020-2035
- Figure 220: Eastern Europe Market (USD Million) Forecast, by Others, 2020-2035
- Figure 221: Eastern Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 222: Eastern Europe Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 223: Eastern Europe Market (USD Million) Forecast, by Pharmacies & Retail, 2020-2035
- Figure 224: Eastern Europe Market (USD Million) Forecast, by Online Channels, 2020-2035
- Figure 225: Russia Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 226: Russia Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 227: Russia Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 228: Russia Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 229: Russia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 230: Russia Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 231: Hungary Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 232: Hungary Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 233: Hungary Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 234: Hungary Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 235: Hungary Market (USD Million) Forecast, by End User, 2020-2035
- Figure 236: Hungary Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 237: Poland Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 238: Poland Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 239: Poland Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 240: Poland Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 241: Poland Market (USD Million) Forecast, by End User, 2020-2035
- Figure 242: Poland Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 243: Rest of Eastern Europe Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 244: Rest of Eastern Europe Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 245: Rest of Eastern Europe Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 246: Rest of Eastern Europe Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 247: Rest of Eastern Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 248: Rest of Eastern Europe Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 249: Middle East & Africa Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 250: Middle East & Africa Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 251: Middle East & Africa Market (USD Million) Forecast, by Digital Test Kits, 2020-2035
- Figure 252: Middle East & Africa Market (USD Million) Forecast, by Laboratory Mail-in Kits, 2020-2035
- Figure 253: Middle East & Africa Market (USD Million) Forecast, by Others, 2020-2035
- Figure 254: Middle East & Africa Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 255: Middle East & Africa Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 256: Middle East & Africa Market (USD Million) Forecast, by Urine, 2020-2035
- Figure 257: Middle East & Africa Market (USD Million) Forecast, by Saliva, 2020-2035
- Figure 258: Middle East & Africa Market (USD Million) Forecast, by Others, 2020-2035
- Figure 259: Middle East & Africa Market (USD Million) Forecast, by End User, 2020-2035
- Figure 260: Middle East & Africa Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 261: Middle East & Africa Market (USD Million) Forecast, by Pharmacies & Retail, 2020-2035
- Figure 262: Middle East & Africa Market (USD Million) Forecast, by Online Channels, 2020-2035
- Figure 263: Saudi Arabia Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 264: Saudi Arabia Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 265: Saudi Arabia Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 266: Saudi Arabia Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 267: Saudi Arabia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 268: Saudi Arabia Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 269: Other GCC Countries Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 270: Other GCC Countries Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 271: Other GCC Countries Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 272: Other GCC Countries Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 273: Other GCC Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 274: Other GCC Countries Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 275: Türkiye Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 276: Türkiye Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 277: Türkiye Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 278: Türkiye Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 279: Türkiye Market (USD Million) Forecast, by End User, 2020-2035
- Figure 280: Türkiye Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 281: South Africa Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 282: South Africa Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 283: South Africa Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 284: South Africa Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 285: South Africa Market (USD Million) Forecast, by End User, 2020-2035
- Figure 286: South Africa Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 287: Rest of MEA Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 288: Rest of MEA Market (USD Million) Forecast, by Rapid Test Kits, 2020-2035
- Figure 289: Rest of MEA Market (USD Million) Forecast, by Sample Type, 2020-2035
- Figure 290: Rest of MEA Market (USD Million) Forecast, by Blood, 2020-2035
- Figure 291: Rest of MEA Market (USD Million) Forecast, by End User, 2020-2035
- Figure 292: Rest of MEA Market (USD Million) Forecast, by Individual Consumers, 2020-2035
- Figure 293: LetsGetChecked - Company Snapshot
- Figure 294: LetsGetChecked - SWOT Analysis
- Figure 295: Everlywell - Company Snapshot
- Figure 296: Everlywell - SWOT Analysis
- Figure 297: myLAB Box - Company Snapshot
- Figure 298: myLAB Box - SWOT Analysis
- Figure 299: SelfDiagnostics - Company Snapshot
- Figure 300: SelfDiagnostics - SWOT Analysis
- Figure 301: Healthy.io - Company Snapshot
- Figure 302: Healthy.io - SWOT Analysis
- Figure 303: DetectaChem - Company Snapshot
- Figure 304: DetectaChem - SWOT Analysis
- Figure 305: Mylab Discovery Solutions - Company Snapshot
- Figure 306: Mylab Discovery Solutions - SWOT Analysis
- Figure 307: OraSure Technologies - Company Snapshot
- Figure 308: OraSure Technologies - SWOT Analysis
- Figure 309: Others - Company Snapshot
- Figure 310: Others - SWOT Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

