Automated High Level Disinfectant Market
Automated High Level Disinfectant Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Automated High Level Disinfectant Market Forecast and Outlook 2026 to 2036
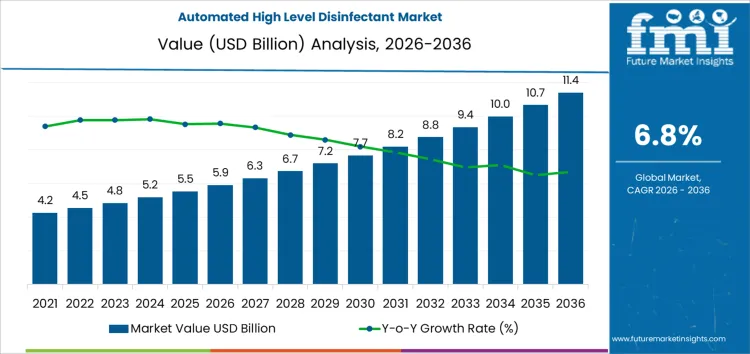
The automated high level disinfectant market is forecast to grow from USD 5.9 billion in 2026 to USD 11.4 billion by 2036, expanding at a CAGR of 6.8%. Between 2026 and 2036, market performance is closely influenced by production concentration and controlled manufacturing ecosystems rather than demand expansion alone. Manufacturing of automated disinfection systems and compatible chemistries remains concentrated among a limited group of validated suppliers, reinforcing supply discipline and standardization. Dependence on regulated chemical inputs, precision dosing components, and compliance-tested hardware constrains rapid capacity scaling.
Regional manufacturing control plays a critical role, as localized production supports regulatory alignment, service responsiveness, and supply continuity for healthcare facilities. Supply reliability is further shaped by validation requirements, documentation obligations, and quality audits, which restrict supplier substitution. Disruptions in chemical sourcing, component availability, or certification timelines can directly affect delivery schedules. Operational resilience, controlled production processes, and secured upstream dependencies remain central determinants of competitive stability in the automated high level disinfectant market over the forecast period.
Quick Stats for Automated High Level Disinfectant Market
- Automated High Level Disinfectant Market Value (2026): USD 5.9 Billion
- Automated High Level Disinfectant Market Forecast Value (2036): USD 11.4 Billion
- Automated High Level Disinfectant Market Forecast CAGR 2026 to 2036: 6.8%
- Leading Type of Automated High Level Disinfectant: Glutaraldehyde (35% share)
- Key Growth Regions in Automated High Level Disinfectant Market: North America, Asia Pacific, Europe, Latin America, Middle East & Africa
- Top Key Players in Automated High Level Disinfectant Market: Ecolab Inc., STERIS Corporation, 3M Company, Johnson & Johnson, Getinge AB, Metrex Research, LLC, Cantel Medical
Automated High Level Disinfectant Market Key Takeaways
| Metric | Value |
|---|---|
| Automated High Level Disinfectant Market Value (2026) | USD 5.9 Billion |
| Automated High Level Disinfectant Market Forecast Value (2036) | USD 11.4 Billion |
| Automated High Level Disinfectant Market Forecast CAGR 2026 to 2036 | 6.8% |
What Factors Are Driving Growth in the Automated High Level Disinfectant Market?
Growth of the automated high level disinfectant market is driven by increasing emphasis on infection prevention and patient safety across healthcare facilities. Hospitals, diagnostic centers, and outpatient clinics are adopting automated disinfection systems to ensure consistent, standardized, and traceable reprocessing of medical devices. Rising procedural volumes, particularly in endoscopy and minimally invasive diagnostics, are increasing demand for high-level disinfection solutions that reduce human error and improve compliance with hygiene protocols.
Regulatory enforcement and accreditation requirements are further supporting market expansion. Healthcare authorities are strengthening guidelines related to medical device reprocessing, documentation, and validation of disinfection cycles. Automated high level disinfectant systems offer controlled exposure, precise dosing, and digital monitoring, which helps facilities meet compliance requirements efficiently. These systems also reduce occupational exposure to chemicals and lower variability in manual disinfection processes, improving operational safety and workflow efficiency.
Technological advancements and healthcare infrastructure expansion are creating additional growth opportunities. Integration of automation, smart sensors, and data logging capabilities is enhancing system reliability and audit readiness. Emerging economies are investing in hospital infrastructure, diagnostic capacity, and ambulatory care centers, increasing adoption of automated disinfection equipment. At the same time, rising awareness of healthcare-associated infections is prompting facilities to upgrade legacy systems. Regulatory pressure, procedural growth, and technology adoption are driving sustained expansion of the automated high level disinfectant market over the forecast period.
What Is The Segment Overview Of The Automated High Level Disinfectant Market?
The automated high level disinfectant market is segmented by type and application, with clear dominance in select categories. By type, glutaraldehyde holds the leading position, supported by its long-standing use in automated disinfection systems. Other types in the automated high level disinfectant market include ortho-phthalaldehyde (OPA), hydrogen peroxide, peracetic acid, and other chemical formulations designed for specialized disinfection needs. By application, medical devices represent the dominant segment, driven by stringent infection control protocols in healthcare facilities. Additional applications such as endoscopes, surgical instruments, other hospital equipment, and miscellaneous uses contribute to diversified demand across clinical environments.
Why Does Glutaraldehyde Lead The Automated High Level Disinfectant Market By Type?
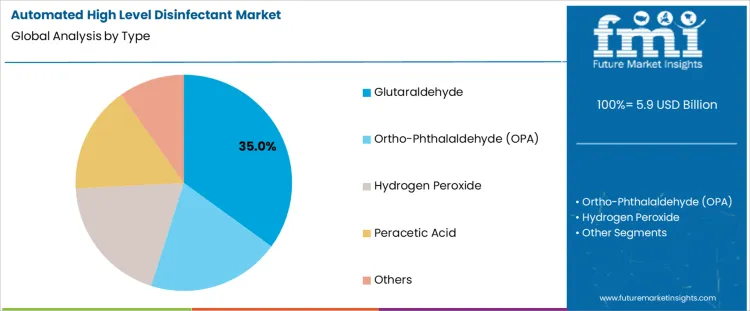
Glutaraldehyde leads the automated high level disinfectant market by type with 35% share because of its proven efficacy against a broad spectrum of microorganisms, including bacteria, viruses, and mycobacteria. Healthcare facilities rely on glutaraldehyde-based systems for consistent high-level disinfection of heat-sensitive medical equipment. Its chemical stability supports integration into automated reprocessing units used in hospitals and diagnostic centers. Compatibility with complex device geometries ensures effective penetration into lumens and narrow channels. Cost efficiency compared to newer formulations supports continued adoption in high-throughput settings. Regulatory familiarity and established usage protocols further reinforce reliance on glutaraldehyde, explaining its sustained leadership in automated disinfection applications.
Why Do Medical Devices Dominate The Automated High Level Disinfectant Market By Application?
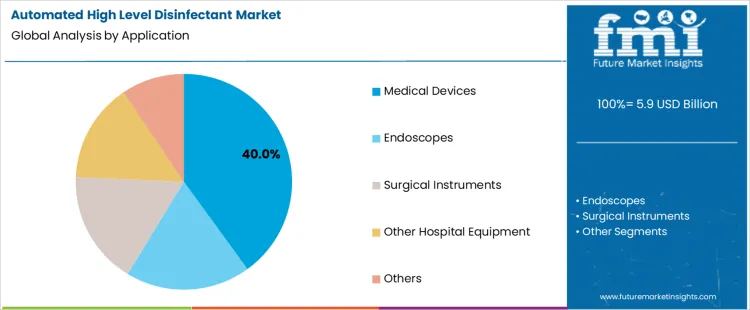
Medical devices dominate the automated high level disinfectant market by application with 40% share due to the critical need for infection prevention across reusable clinical equipment. Devices such as catheters, probes, and diagnostic tools require validated high-level disinfection between patient uses. Automated systems provide controlled exposure times, chemical concentration accuracy, and repeatable outcomes, reducing human error. Growing procedural volumes in hospitals and outpatient facilities increase reprocessing frequency. Regulatory guidelines mandate strict compliance with sterilization and disinfection standards for reusable devices. This consistent operational requirement explains why medical devices remain the largest application segment within the automated high level disinfectant market.
What Are The Key Dynamics Of The Automated High Level Disinfectant Market?
Automated high level disinfectant systems are increasingly adopted in hospitals, diagnostic centers, and ambulatory surgical facilities to reduce manual handling risks and improve workflow efficiency. Growth in endoscope procedures, infection control protocols, and patient safety regulations is strengthening demand. Automation supports consistent exposure time, chemical concentration control, and traceability. Capital investment capability, regulatory compliance requirements, and healthcare facility sterilization volumes remain central factors shaping adoption patterns within the automated high level disinfectant market.
How Are Trends Shaping The Automated High Level Disinfectant Market?
Key trends in the automated high level disinfectant market focus on automation, standardization, and compliance-driven workflow integration. Healthcare facilities are increasingly adopting fully enclosed automated systems to ensure consistent disinfection of endoscopes and heat-sensitive medical devices. Integration of digital monitoring, cycle validation, and electronic record-keeping is improving traceability and audit readiness. Demand for systems compatible with multiple disinfectant chemistries is increasing to support flexible procurement. Compact, point-of-use automated disinfectors are gaining traction in ambulatory surgical centers. Emphasis on reducing staff exposure to hazardous chemicals is accelerating replacement of manual disinfection processes.
What Opportunities Are Supporting Expansion Of The Automated High Level Disinfectant Market?
Expansion opportunities in the automated high level disinfectant market are supported by rising volumes of endoscopic and minimally invasive procedures worldwide. Growth of outpatient care, ambulatory surgical centers, and specialty diagnostic clinics is increasing demand for compact automated disinfection systems. Strengthening infection prevention policies and hospital accreditation requirements are encouraging upgrades from manual methods. Emerging markets are investing in healthcare infrastructure, creating new installation opportunities. Development of environmentally safer disinfectant chemistries compatible with automated systems is opening new product avenues. Service contracts, software upgrades, and compliance support services are further enhancing long-term revenue potential.
What Is the Country-Wise Outlook of the Automated High Level Disinfectant Market?
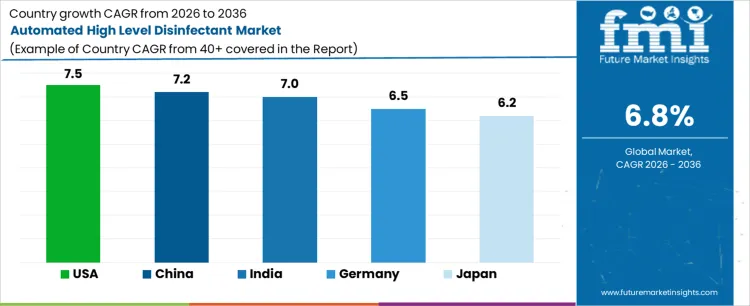
Demand for automated high level disinfectant systems differs across countries based on healthcare infrastructure maturity, procedural volumes, and infection control regulations. North America leads adoption due to strong compliance requirements, while Asia Pacific shows rapid expansion supported by hospital capacity growth. The USA is projected to grow at a CAGR of 7.5% during 2026 to 2036, followed by China at 7.2% and India at 7.0%, driven by rising endoscopy and surgical procedure volumes. Germany and Japan are expected to expand at CAGRs of 6.5% and 6.2%, respectively, reflecting stable but regulation-driven demand in developed healthcare systems.
| Country | CAGR (%) |
|---|---|
| USA | 7.5% |
| China | 7.2% |
| India | 7.0% |
| Germany | 6.5% |
| Japan | 6.2% |
How Is Demand for Automated High Level Disinfectant Systems Shaping Growth in the USA?
Demand for automated high level disinfectant systems in the USA, expanding at a CAGR of 7.5% during 2026 to 2036, is driven by high procedural volumes and stringent infection prevention requirements. The automated high level disinfectant market in the USA benefits from widespread use of reusable endoscopes and surgical instruments across hospitals and ambulatory surgical centers. Healthcare providers emphasize automation to improve consistency, reduce manual handling risks, and meet regulatory compliance standards. Replacement of semi-automated and manual disinfection processes continues to support recurring demand. Accreditation requirements and hospital quality benchmarks further reinforce procurement of validated disinfection systems. Investments are also supported by focus on workflow efficiency and staff safety. These factors collectively sustain strong value-driven expansion in the automated high level disinfectant market in the USA over the forecast period.
Why Are Sales of Automated High Level Disinfectant Systems Increasing in China?
Sales of automated high level disinfectant systems in China, growing at a CAGR of 7.2% from 2026 to 2036, are supported by rapid expansion of hospital infrastructure and diagnostic service capacity. The automated high level disinfectant market in China benefits from rising volumes of endoscopic and minimally invasive procedures. Public hospital modernization programs and private healthcare investment are accelerating adoption of automated reprocessing solutions. Healthcare facilities are prioritizing standardized disinfection workflows to improve infection control outcomes. Local manufacturing capabilities improve system affordability and supply availability. Increasing regulatory focus on hygiene compliance further supports system adoption. These factors position China as a high-growth country within the automated high level disinfectant market during the forecast period.
What Is Driving Demand for Automated High Level Disinfectant Systems in India?
Demand for automated high level disinfectant systems in India, expanding at a CAGR of 7.0% during 2026 to 2036, is driven by rising surgical volumes and strengthening infection control practices. The automated high level disinfectant market in India is supported by expansion of multi-specialty hospitals, diagnostic centers, and private healthcare networks. Providers are shifting toward automated systems to improve consistency and reduce manual reprocessing variability. Accreditation requirements and quality assurance initiatives are increasing awareness of validated disinfection protocols. Government health programs and private investment in hospital infrastructure further support adoption. Cost sensitivity remains, but growing emphasis on patient safety sustains demand. These factors collectively support steady growth of the automated high level disinfectant market in India.
How Does Germany Sustain Demand for Automated High Level Disinfectant Systems?
Demand for automated high level disinfectant systems in Germany, growing at a CAGR of 6.5% over the forecast period, is sustained by strict regulatory compliance and structured healthcare procurement practices. The automated high level disinfectant market in Germany is driven primarily by replacement demand and technology upgrades rather than facility expansion. Hospitals and clinics prioritize validated disinfection processes, traceability, and operational reliability. Emphasis on infection prevention standards supports consistent system adoption across clinical settings. Investments focus on improving workflow efficiency and ensuring compliance with evolving hygiene regulations. Limited expansion in healthcare facilities is offset by recurring refurbishment cycles. These dynamics result in stable and predictable growth in the automated high level disinfectant market in Germany.
Why Is Demand for Automated High Level Disinfectant Systems Stable in Japan?
Demand for automated high level disinfectant systems in Japan, expanding at a CAGR of 6.2% during 2026 to 2036, reflects a mature healthcare system with strong emphasis on patient safety. The automated high level disinfectant market in Japan is driven largely by refurbishment and replacement of existing systems. Healthcare providers prioritize precision, reliability, and long equipment lifecycles when investing in disinfection technologies. Procedural volumes remain steady, supporting baseline system demand. Limited hospital expansion constrains rapid volume growth, while strict hygiene protocols ensure continued adoption. Manufacturers focus on compact designs and process stability to meet facility requirements. These factors contribute to steady, controlled expansion of the automated high level disinfectant market in Japan.
How Are Leading Companies Competing in the Automated High Level Disinfectant Market?
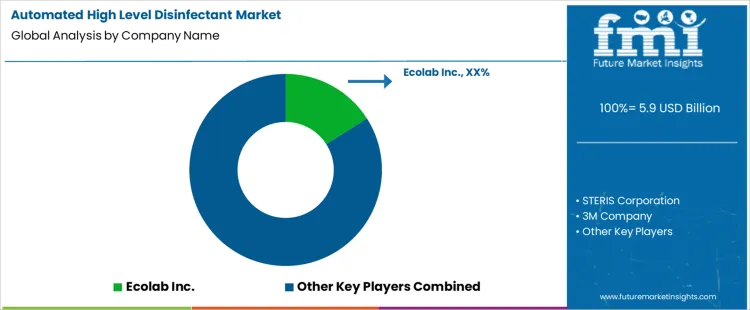
Competition in the automated high level disinfectant market is shaped by automation reliability, disinfectant efficacy, and breadth of validated device workflows as presented in official product brochures. Ecolab Inc. has positioned its automated disinfectant systems with an emphasis on integrated environmental hygiene platforms. Brochures describe high-level disinfectants and automated delivery systems validated for endoscope and critical instrument reprocessing, with performance claims supported by controlled test data. STERIS Corporation competes through automated high-level disinfection solutions that are promoted for repeatable cycle parameters, traceability reporting, and compliance support. Its product literature highlights process automation and validated disinfectant chemistries for critical care assets.
Strategic differentiation is reinforced through portfolio depth and cross-platform integration. 3M Company markets automated disinfectant applicators alongside high-efficacy chemistries with technical brochures emphasizing compatibility with workflow software and environmental monitoring. Johnson & Johnson has leveraged its medical device cleaning and disinfection segment to offer automated high-level disinfectant systems with branded chemistry, with brochures detailing pathogen kill spectra and material compatibility data. Automated solutions are presented as part of complete reprocessing cycles rather than stand-alone products.
Mid-sized competitors target niche clinical segments through validated automation and service support. Getinge AB has positioned its automated disinfectant systems with emphasis on ergonomic design and detailed process documentation in official catalogs. Metrex Research, LLC focuses on automated chemistries validated for high-level disinfection of semi-critical devices, with brochures stressing rapid action and residue control. Cantel Medical has competed through automated high-level disinfection platforms that are promoted for consistent cycle controls and integrated quality assurance reporting.
Key Players in Automated High Level Disinfectant Market
- Ecolab Inc.
- STERIS Corporation
- 3M Company
- Johnson & Johnson
- Getinge AB
- Metrex Research, LLC
- Cantel Medical
Scope of Report
| Attribute | Description |
|---|---|
| Quantitative Unit | USD Billion |
| Type | Glutaraldehyde, Ortho-Phthalaldehyde (OPA), Hydrogen Peroxide, Peracetic Acid, Others |
| Application | Medical Devices, Endoscopes, Surgical Instruments, Other Hospital Equipment, Others |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Companies Profiled | Ecolab Inc., STERIS Corporation, 3M Company, Johnson & Johnson, Getinge AB, Metrex Research, LLC, Cantel Medical |
| Additional Attributes | Dollar sales by type and application are analyzed across hospital sterilization workflows and device categories. The report includes country-level demand assessment, evaluation of regulatory compliance and safety standards, assessment of automation adoption in reprocessing units, competitive benchmarking of leading suppliers, and analysis of procurement trends across healthcare facilities. |
Automated High Level Disinfectant Market by Key Segments
Type
- Glutaraldehyde
- Ortho-Phthalaldehyde (OPA)
- Hydrogen Peroxide
- Peracetic Acid
- Others
Application
- Medical Devices
- Endoscopes
- Surgical Instruments
- Other Hospital Equipment
- Others
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- Centers for Disease Control and Prevention. (2024). Guideline for disinfection and sterilization in healthcare facilities (2024 update). U.S. Department of Health and Human Services.
- U.S. Food and Drug Administration. (2025). Reprocessing medical devices in health care settings: Validation methods and safety considerations (2025 revision). U.S. Department of Health and Human Services.
- World Health Organization. (2025). Decontamination and reprocessing of medical devices for health-care facilities: Updated implementation guidance. World Health Organization.
Frequently Asked Questions
How big is the automated high level disinfectant market in 2026?
The global automated high level disinfectant market is estimated to be valued at USD 5.9 billion in 2026.
What will be the size of automated high level disinfectant market in 2036?
The market size for the automated high level disinfectant market is projected to reach USD 11.4 billion by 2036.
How much will be the automated high level disinfectant market growth between 2026 and 2036?
The automated high level disinfectant market is expected to grow at a 6.8% CAGR between 2026 and 2036.
What are the key product types in the automated high level disinfectant market?
The key product types in automated high level disinfectant market are glutaraldehyde, ortho-phthalaldehyde (opa), hydrogen peroxide, peracetic acid and others.
Which application segment to contribute significant share in the automated high level disinfectant market in 2026?
In terms of application, medical devices segment to command 40.0% share in the automated high level disinfectant market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2026 to 2036
- Glutaraldehyde
- Ortho-Phthalaldehyde (OPA)
- Hydrogen Peroxide
- Peracetic Acid
- Others
- Glutaraldehyde
- Y to o to Y Growth Trend Analysis By Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Medical Devices
- Endoscopes
- Surgical Instruments
- Other Hospital Equipment
- Others
- Medical Devices
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Ecolab Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- STERIS Corporation
- 3M Company
- Johnson & Johnson
- Getinge AB
- Metrex Research, LLC
- Cantel Medical
- Ecolab Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Type , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Type , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Type , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Type , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Type , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Type , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Type , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

