Beadlets Capsule Market
Beadlets Capsule Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Beadlets Capsule Market Forecast and Outlook 2026 to 2036
The beadlets capsule market is projected to grow from USD 477 million in 2026 to USD 854.3 million by 2036, at a CAGR of 6.0%. Microencapsulated beadlets dominate product forms with a 50% share, primarily serving nutraceutical applications at 55%. Capsules and sachets cater to pharmaceutical and specialty ingredient sectors. CDMOs and B2B nutraceutical brands represent key end users. Retail channels and ingredient suppliers further support demand. Rising interest in convenience, targeted nutrient delivery, and improved bioavailability are key growth drivers.
Market expansion is fueled by ongoing innovations in encapsulation technologies and strategic investments by leading companies like Lonza Capsugel, Colorcon, and Roquette. Multi-channel distribution, capacity enhancements, and adoption of advanced formulations strengthen market penetration. Consumer preference for functional nutrition and wellness products globally sustains consistent demand. Growth is expected across regions, with nutraceutical applications driving adoption. Focus on stability, shelf-life, and precise dosing ensures continued relevance of beadlets in health-oriented formulations.
Quick Stats of the Beadlets Capsule Market:
- Beadlets Capsule Market Value (2026): USD 477 million
- Beadlets Capsule Forecast Value (2036): USD 854.3 million
- Beadlets Capsule Market Forecast CAGR (2026 to 2036): 6.0%
- Leading Product Form in the Market: Microencapsulated Beadlets (50%)
- Key Growth Regions in the Market: India, China, Brazil, USA, Germany
- Top Players in the Market: Lonza Capsugel, Colorcon, Roquette, Evonik, BASF, Others
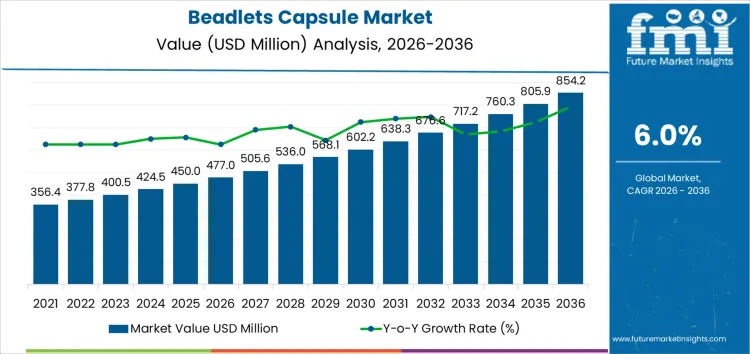
What is the Growth Forecast for the Beadlets Capsule Market through 2036?
From 2026 to 2031, the beadlets capsule market grows from USD 477 million to USD 602 million. Annual increments start at USD 27 million in 2026 and reach USD 50 million by 2031. Growth is concentrated in nutraceuticals and dietary supplements, with vitamin/mineral beadlets representing roughly 40% of demand, omega-3 and lipid-soluble formulations around 25%, and probiotics 15-20%. North America leads early adoption, contributing 35-40% of market value, driven by established dietary supplement channels, regulatory compliance for encapsulation, and consumer preference for stable, controlled-release formulations. Europe and Asia Pacific collectively account for 50-55% of early uptake, mainly in high-value premium capsules.
Between 2031 and 2036, the market expands from USD 602 million to USD 854.3 million. Annual increments accelerate to USD 94 million by 2036, reflecting broader commercialization across mass-market and specialized nutraceuticals. Functional food and pharmaceutical-grade beadlets uptake rises sharply, representing 30-35% of incremental growth. Growth is reinforced by repeat procurement, scaling in contract manufacturing, and adoption across emerging markets in Asia and Latin America. Early growth focused on pilot projects and high-margin products, while later expansion reflects full-scale integration in global supplement supply chains, higher per-unit utilization, and multi-SKU portfolio deployment.
Beadlets Capsule Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 477 million |
| Forecast Value (2036) | USD 854.3 million |
| Forecast CAGR (2026 to 2036) | 6.0% |
What Is Driving Demand in the Beadlets Capsule Market?
Demand for beadlets capsules developed from practical formulation challenges in food, pharmaceutical, and consumer products rather than from broad consumer preference alone. Traditional powders, liquids, and bulk granules often exhibited instability, volatility, or poor flow properties during manufacturing and storage. Ingredients such as vitamins, flavours, probiotics, and fragrance compounds can degrade, oxidise, or volatilise when exposed to moisture, heat, or air, complicating dosing and reducing product shelf life. Beadlet encapsulation was introduced to protect sensitive actives by enclosing them in discrete, stabilised units that release under defined conditions. Historical uptake reflected the need to improve handling, control release profiles, and maintain potency in complex formulations rather than general market trends.
Future demand for beadlets capsules is expected to reflect structural shifts in product formulation strategies and regulatory quality expectations rather than functional protection needs alone. Food and pharmaceutical manufacturers are increasingly specifying encapsulated formats to meet precise dosing tolerances and ensure consistent delivery of active ingredients across batches. Regulatory frameworks are tightening on stability documentation, ingredient performance, and moisture control, encouraging the use of beadlets to achieve defined shelf life outcomes. Advances in encapsulation technologies are enabling a wider range of release profiles, target environments, and compatibility with diverse formulation platforms. Demand will expand as encapsulation becomes part of standard formulation toolkits and aligns with quality specifications rather than only addressing stability failures during product development.
What Factors Are Affecting the Demand for Beadlets Capsule Market in Terms of Product Form and Application?
Demand for beadlets capsules is segmented by product form and application across nutrition, pharmaceutical, and specialty ingredient delivery systems. Microencapsulated beadlets account for about 50% of total demand, making them the leading product form. Capsules and sachets or other formats represent additional forms without disclosed share splits. These forms differ in stability protection, release control, and formulation compatibility. Application segmentation reflects end market requirements and regulatory pathways. Nutraceuticals account for about 55% of demand, followed by pharmaceutical and specialty ingredient applications.
What Drives Demand by Product Form in the Beadlets Capsule Market?
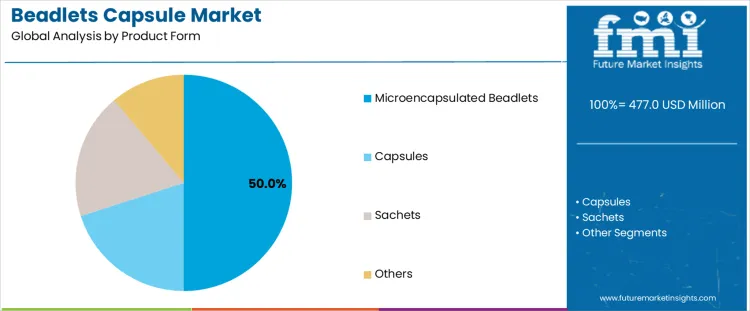
Microencapsulated beadlets lead demand with a 50% share because they protect sensitive actives from oxidation, heat, and moisture during processing and storage. This form supports controlled release, improved bioavailability, and masking of undesirable taste or odor. Capsules remain relevant where formulation simplicity and direct dosing are sufficient. Sachets and other formats serve niche applications requiring single dose delivery or blending flexibility. Product form selection reflects technical performance requirements rather than consumer convenience alone. Demand therefore aligns with formulation stability and functional delivery objectives rather than marketing driven format selection or packaging differentiation strategies across health and nutrition products.
Product form demand remains stable because delivery format decisions are fixed during formulation development. Beadlets are embedded in products requiring enhanced protection and release control. Capsules persist in less complex formulations with lower stability risk. Alternative formats remain limited to specific use cases. These patterns restrict rapid substitution across forms. Product form segmentation therefore highlights reliance on validated delivery technologies rather than short term innovation cycles or frequent reformulation within regulated or performance sensitive product categories.
How Does Application Shape Demand for Beadlets Capsule Market?
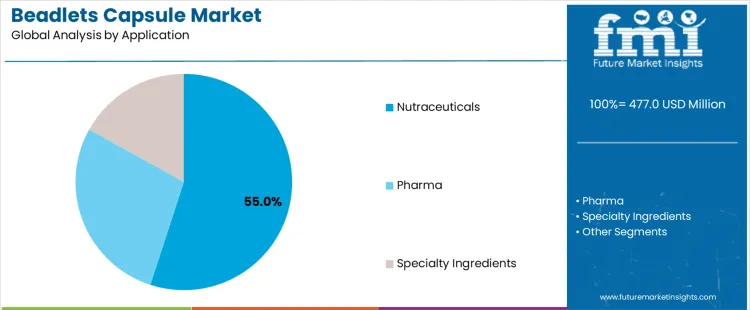
Nutraceuticals account for about 55% of total demand, making them the dominant application segment. This reflects extensive use of vitamins, minerals, probiotics, and functional ingredients requiring stability and controlled release. Pharmaceutical applications adopt beadlets where modified release or protection is required but face longer validation timelines. Specialty ingredient applications focus on targeted performance outcomes in functional foods or supplements. Application driven demand reflects regulatory burden, formulation complexity, and development speed rather than absolute consumption volume.
Nutraceutical applications maintain leadership due to faster product development cycles and flexible formulation requirements. Pharmaceutical use remains selective and tightly controlled. Specialty ingredient applications remain limited to defined performance needs. Once selected, delivery systems are rarely changed. This stabilizes demand distribution by application.
How Are Beadlets Capsules Being Applied Across Nutraceuticals and Pharmaceutical Products?
Use appears in formulations where precise dosing, stability, and controlled release of active ingredients are required. Nutritional supplements, vitamins, and herbal extracts adopt beadlets to maintain potency, mask taste, and prevent ingredient interaction during storage. Pharmaceutical companies integrate beadlet capsules for encapsulating sensitive compounds such as enzymes, probiotics, or oils that degrade under heat, moisture, or oxygen exposure. These applications reflect operational and functional priorities rather than decorative purposes, with adoption driven by ingredient stability, bioavailability, and manufacturing efficiency.
What Operational and Material Conditions Support Adoption of Beadlets Capsules?
Selection aligns with formulations requiring uniform particle size, encapsulation efficiency, and controlled release profiles. Encapsulation materials, coatings, and excipients are optimized to withstand processing conditions such as mixing, granulation, or compression. Manufacturing lines rely on consistent beadlet flow for accurate dosing and capsule filling. Quality control ensures potency retention, uniformity, and regulatory compliance for nutraceutical or pharmaceutical standards. These conditions emerge from operational reliability, product stability, and formulation precision priorities in structured production workflows.
Which Practical Considerations Limit Wider Use of Beadlets Capsules?
Higher production cost compared with conventional powders or tablets may limit adoption in cost-sensitive markets. Compatibility with active ingredients, excipients, and capsule shells must be verified. Process parameters such as temperature, humidity, and shear must be carefully controlled to maintain beadlet integrity. Regulatory approvals for food, nutraceutical, or pharmaceutical applications are required. These factors result in selective deployment where ingredient stability, controlled release, and dosage precision justify incremental cost and process management.
What is the Demand for Beadlets Capsules by Country?
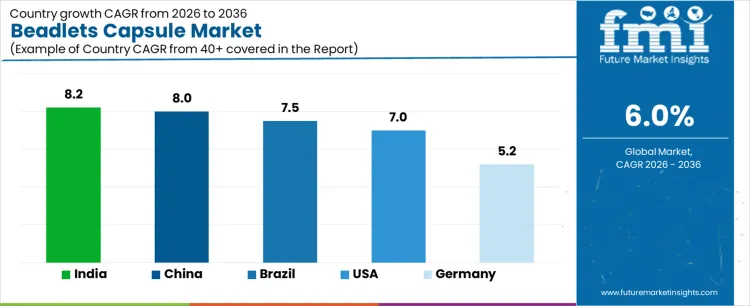
| Country | CAGR (%) |
|---|---|
| USA | 7.0% |
| Brazil | 7.5% |
| China | 8.0% |
| India | 8.2% |
| Germany | 5.2% |
The demand for beadlets capsules varies across countries, driven by pharmaceutical and nutraceutical production, dietary supplement consumption, and manufacturing capabilities. India leads with an 8.2% CAGR, supported by growing nutraceutical demand, expanding pharmaceutical manufacturing, and increased consumer health awareness. China follows at 8.0%, driven by large scale production of dietary supplements and pharmaceutical ingredients. Brazil records 7.5%, shaped by growing domestic consumption and supplement adoption. The USA grows at 7.0%, reflecting steady use in vitamins, supplements, and functional food applications. Germany posts 5.2%, supported by mature pharmaceutical markets and stable demand for capsule formulations.
How Is Beadlets Capsule Demand Expanding in the United States?
In the United States, revenue from the beadlets capsule market is growing at a CAGR of 7% through 2036, supported by rising consumer focus on dietary supplements and precision-dosed nutraceuticals. Manufacturers are increasingly adopting beadlet capsules to ensure ingredient stability, controlled release, and improved bioavailability for vitamins, minerals, and functional compounds. Demand is concentrated in nutraceutical production, pharmaceutical formulations, and specialty functional foods. Domestic suppliers are providing high-performance, formulation-compatible capsules that integrate easily with soft gels, hard capsules, and powder-in-capsule designs. Regulatory requirements for safety and quality, combined with repeated production cycles, are driving consistent adoption nationwide.
- Nutraceutical, pharmaceutical, and functional food sectors drive adoption
- Capsules ensure ingredient stability, controlled release, and bioavailability
- Domestic suppliers provide formulation-compatible, high-performance capsules
- Recurring production cycles sustain adoption
How Is Beadlets Capsule Growth Shaping Brazil?
In Brazil, the market is expanding at a CAGR of 7.5% through 2036, fueled by increasing health awareness and demand for dietary supplements in urban populations. Manufacturers are using beadlet capsules to improve active ingredient stability and deliver consistent dosing in vitamins and herbal formulations. Demand is concentrated among nutraceutical producers, pharmaceutical firms, and functional food companies. Local suppliers provide reliable, high-quality beadlets suitable for soft gels, hard capsules, and blended formulations. Growth in consumer spending on health products and recurring manufacturing cycles is sustaining steady adoption across the country.
- Urban nutraceutical and pharmaceutical producers drive demand
- Capsules improve ingredient stability and dosing consistency
- Local suppliers provide high-quality, versatile beadlets
- Recurring production cycles sustain steady adoption
How Is Beadlets Capsule Adoption Accelerating in China?
China is witnessing the highest growth, with a CAGR of 8% through 2036, supported by rapid expansion in nutraceutical and pharmaceutical manufacturing. Companies are integrating beadlet capsules to protect sensitive actives from degradation, achieve controlled release, and maintain bioavailability in vitamins, minerals, and functional food products. Demand is strong across industrial production facilities and high-volume supplement brands. Domestic manufacturers are scaling high-performance beadlets compatible with soft gels, hard capsules, and powder-in-capsule technologies. Expanding consumer health awareness, growing production infrastructure, and recurring formulation cycles are driving rapid adoption nationwide.
- Nutraceutical and pharmaceutical manufacturers drive demand
- Capsules protect actives and maintain controlled release
- Domestic suppliers scale high-performance, multi-compatible beadlets
- Recurring formulation cycles sustain rapid adoption
How Is Beadlets Capsule Use Growing in India?
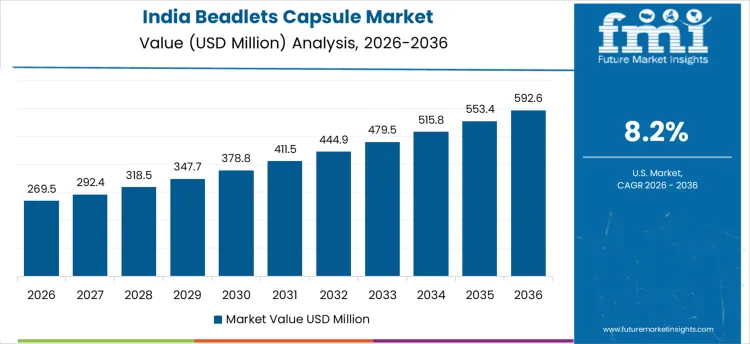
In India, the market is expanding at a CAGR of 8.2% through 2036, driven by increased consumption of dietary supplements and functional foods. Manufacturers are adopting beadlet capsules to enhance stability, precise dosing, and bioavailability of vitamins, minerals, and herbal extracts. Demand is concentrated in nutraceutical companies, pharmaceutical manufacturers, and specialty functional food producers. Domestic suppliers provide high-performance, formulation-compatible beadlets suited for various capsule types. Rising health-consciousness, regulatory standards, and recurring production operations are sustaining robust adoption across India.
- Nutraceutical, pharmaceutical, and functional food companies drive adoption
- Capsules enhance stability, dosing precision, and bioavailability
- Domestic suppliers provide versatile, high-performance beadlets
- Recurring production operations sustain robust adoption
How Is Beadlets Capsule Market Growing in Germany?
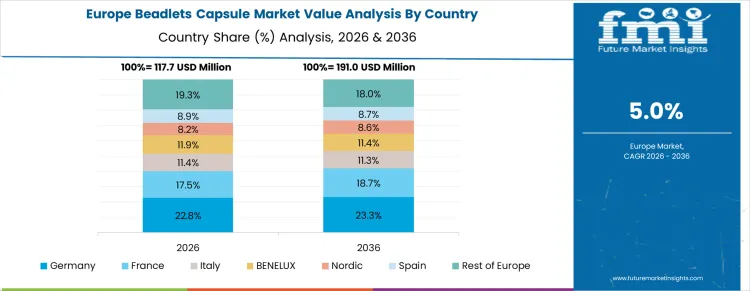
Germany is seeing moderate growth at a CAGR of 5.2% through 2036, supported by steady demand for dietary supplements and nutraceutical products among aging populations. Manufacturers are integrating beadlet capsules to protect sensitive actives, ensure controlled release, and improve stability in vitamin and herbal formulations. Demand is concentrated in nutraceutical producers, pharmaceutical companies, and functional food manufacturers. Domestic suppliers provide high-quality, formulation-compatible beadlets suitable for hard capsules, soft gels, and powder blends. Consumer awareness, regulatory requirements, and recurring manufacturing cycles are sustaining consistent adoption nationwide.
- Nutraceutical, pharmaceutical, and functional food producers anchor demand
- Capsules protect actives and maintain stability and controlled release
- Domestic suppliers provide high-quality, formulation-compatible beadlets
- Recurring manufacturing cycles sustain consistent adoption
Which Companies Shape Competition in The Beadlets Capsule Market Through Formulation Function and Manufacturing Control?
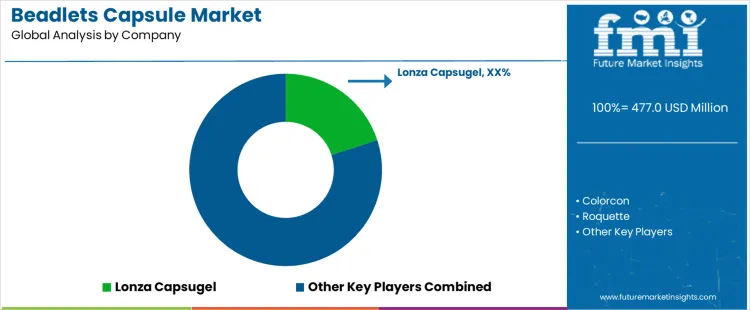
Competition in the Beadlets Capsule Market is defined by how effectively beadlet technologies deliver controlled release, stability, and uniform dosing within oral solid formulations. Lonza Capsugel influences this segment through capsule platforms designed to accommodate multiparticulate beadlets used in modified release and taste masking applications. Colorcon and Evonik participate by supplying functional coatings and excipient systems that govern beadlet performance once encapsulated. BASF contributes polymer and excipient chemistries used in beadlet formation and protection rather than finished dosage formats. Roquette supports this market through carbohydrate based excipients and inert cores used in beadlet production. Selection is driven by flow behavior, encapsulation efficiency, and compatibility with high speed filling operations rather than capsule aesthetics or branding.
Manufacturing execution and regulatory alignment shape competitive positioning beyond core materials. Suppliers are evaluated on batch to batch consistency, particle size control, and the ability to support scale up from development to commercial production. Contract manufacturers and formulation developers often influence supplier choice by standardizing beadlet and capsule combinations validated in earlier programs. Smaller and regional players participate through niche beadlet systems or custom encapsulation support for specific therapeutic profiles. Competitive differentiation rests on technical service depth, documentation readiness, and reliability during long production runs. Market dynamics are governed by pharmaceutical development timelines, qualification requirements, and risk management practices rather than rapid product turnover or volume driven competition.
Key Players in the Beadlets Capsule Market
- Lonza Capsugel
- Colorcon
- Roquette
- Evonik
- BASF
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Product Form | Microencapsulated Beadlets, Capsules, Sachets, Others |
| Application | Nutraceuticals, Pharma, Specialty Ingredients |
| End-Use Industry | CDMOs, B2B Brands, Ingredient Suppliers, Retail |
| Region | Asia Pacific (China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific), Europe (Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe), North America (United States, Canada, Mexico), Latin America (Brazil, Chile, Rest of Latin America), Middle East & Africa (KSA, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa) |
| Key Companies Profiled | Lonza Capsugel, Colorcon, Roquette, Evonik, BASF, Others |
| Additional Attributes | Dollar by sales by product form, Dollar by sales by application, Dollar by sales by end-use, Dollar by sales by region, Ingredient stability and protection, Controlled release performance, Bioavailability enhancement, Flow and fill consistency, Capsule and beadlet compatibility, Regulatory compliance for nutraceutical and pharmaceutical use, Recurring production cycles, Multi-SKU portfolio integration, Scale-up capability across commercial manufacturing |
Beadlets Capsule Market Segmentation
Product Form:
- Microencapsulated Beadlets
- Capsules
- Sachets
- Others
Application:
- Nutraceuticals
- Pharma
- Specialty Ingredients
End User:
- CDMOs
- B2B Brands
- Ingredient Suppliers
- Retail
Region
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- International Society for Nutraceuticals and Functional Foods. (2023). Beadlet formulation and encapsulation technologies for dietary supplements. ISNFF Technical Publications.
- United States Pharmacopeia. (2023). Standards for capsules, beadlets, and modified release formulations. USP Monographs and Standards.
- World Health Organization. (2023). Guidelines on quality assurance and safety for pharmaceutical and nutraceutical excipients including beadlet technologies. WHO Technical Guidance Documents.
- European Pharmacopoeia Commission. (2023). Monographs on beadlet capsules and excipient specifications for nutraceutical products. European Pharmacopoeia Publications.
Frequently Asked Questions
How big is the beadlets capsule market in 2026?
The global beadlets capsule market is estimated to be valued at USD 477.0 million in 2026.
What will be the size of beadlets capsule market in 2036?
The market size for the beadlets capsule market is projected to reach USD 854.2 million by 2036.
How much will be the beadlets capsule market growth between 2026 and 2036?
The beadlets capsule market is expected to grow at a 6.0% CAGR between 2026 and 2036.
What are the key product types in the beadlets capsule market?
The key product types in beadlets capsule market are microencapsulated beadlets, capsules, sachets and others.
Which application segment to contribute significant share in the beadlets capsule market in 2026?
In terms of application, nutraceuticals segment to command 55.0% share in the beadlets capsule market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Form , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Form , 2026 to 2036
- Microencapsulated Beadlets
- Capsules
- Sachets
- Others
- Microencapsulated Beadlets
- Y to o to Y Growth Trend Analysis By Product Form , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Form , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Nutraceuticals
- Pharma
- Specialty Ingredients
- Nutraceuticals
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- CDMOs
- B2B Brands
- Ingredient Suppliers
- Retail
- CDMOs
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Form
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Form
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Form
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Form
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Form
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Form
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Form
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Form
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Lonza Capsugel
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Colorcon
- Roquette
- Evonik
- BASF
- Others
- Lonza Capsugel
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Form
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product Form
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Form
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Form
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Form
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Form
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Form
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Form
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

