CellSync Creams Market
CellSync Creams Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
CellSync Creams Market Forecast and Outlook 2026 to 2036
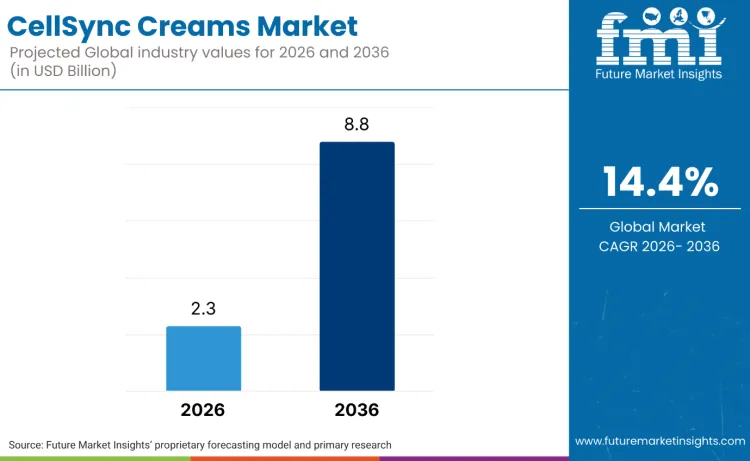
The CellSync creams market is valued at USD 2.3 billion in 2026 and is projected to reach USD 8.8 billion by 2036, registering a 14.4% CAGR. Growth is anchored in governance over safety and claims, which is reshaping premium skin repair routines. In the United States, the Modernization of Cosmetics Regulation Act of 2022 (MoCRA) introduced federal requirements such as serious adverse event reporting, pushing responsible persons and manufacturers to run more formal post-market controls and documentation.
Physician-dispensed brands are using portfolio cadence to defend clinical channels. In April 2024, Allergan Aesthetics, an AbbVie company, launched new SkinMedica acne products positioned around provider-led care. Jasson Gilmore, Senior Vice President, USA Aesthetics, stated: ‘For 25 years, SkinMedica has been dedicated to perfecting scientifically proven skincare that yields visible results.’ This links repeat purchase to office recommendation and post-procedure recovery protocols.
Dermatological skincare scale is now visible in financial disclosures that validate category investment. Galderma reported 2024 Dermatological Skincare net sales of USD 1,331 million and identifies Cetaphil and Alastin as flagship brands, with Alastin continuing to grow beyond the USA In China, the regulator issued the Standard for the Evaluation of Efficacy Claims of Cosmetics effective in 2021, raising the cost of substantiation and favouring platforms that can fund testing at launch.
CellSync Creams Market Key Takeaways
| Items | Values |
|---|---|
| Industry Size (2026) | USD 2.3 Billion |
| Forecast Value (2036) | USD 8.8 Billion |
| CAGR (2026 to 2036) | 14.4% |
Summary of CellSync Creams Market
What Is Growth Outlook for CellSync Creams Market as per Future Market Insights Projection?
Future Market Insights projects the CellSync creams market to expand from USD 2.3 Billion in 2026 to USD 8.8 Billion by 2036, reflecting a 14.4% CAGR.
FMI Research Approach: Forecasting anchored in protocol-based aftercare demand, compliance-led premiumisation, and channel governance across clinics and controlled e-commerce.
How Do FMI Analysts Perceive the CellSync Creams Market to Evolve?
FMI sees the category moving toward renewal-led claims that can be substantiated as cosmetics, while frontier regenerative narratives face tighter enforcement and higher documentation cost.
FMI Research Approach: Tracking claim substantiation rules, post-market obligations, and repeated portfolio refresh actions by physician-dispensed brands.
Which Country Holds Largest Growth Opportunity for CellSync Creams Market?
China leads growth outlook with a 19.44% CAGR, supported by efficacy-claim evaluation requirements that reward test-backed platforms and disciplined dossier execution.
FMI Research Approach: Country CAGR modelling aligned to claim substantiation rules and the ability of brands to fund evidence packages at launch.
Which Segment Leads the CellSync Creams Market in 2026?
Cell Renewal Creams lead with a 47.5% share, reflecting their compatibility with measurable cosmetic endpoints and repeatable daily-use regimen placement.
FMI Research Approach: Segment share modelling based on channel conversion logic and regulatory risk tolerance by active positioning.
Which Forces Accelerate CellSync Creams' Adoption?
Adoption is being propelled by the conversion of aesthetic procedure volume into structured post-procedure recovery regimens where dermatologist offices and medical spas can standardise repeatable aftercare. ISAPS reports that total procedures performed in 2024 exceeded 37.9 million, with the United States leading non-surgical procedure volume and Brazil leading surgical volume, reinforcing a large installed base of recovery-touchpoint settings. MoCRA requires serious adverse event reporting for cosmetics in the USA, pushing brands to formalise surveillance and documentation. In China, the NMPA efficacy claim evaluation standard effective May 2021 raises the cost of performance substantiation, advantaging peptide-led renewal platforms that can fund testing and data disclosure at launch.
How is the CellSync Creams Market Segmented?
The CellSync creams market is segmented by product type, active technology, skin type, distribution channel, and end user to capture how purchase decisions are governed by risk tolerance, claim supportability, and channel economics. Product type separates cell renewal, DNA repair, anti-ageing regeneration, and barrier repair formats because each maps to different evidence expectations and post-procedure usage patterns. Active technology segmentation matters because peptides, growth factors, stem cell derived actives, and exosome positioning sit on different sides of regulatory scrutiny, especially where authorities are explicit that regenerative medicine claims are not cosmetic workarounds. Skin type segmentation reflects where barrier compromise is most acute, while distribution channel segmentation isolates the economics of physician recommendation versus direct digital acquisition. End user segmentation captures the procedure-led demand skew toward women that clinics monetise through bundled aftercare.
Why do Cell Renewal Creams Lead the Product Type Mix?
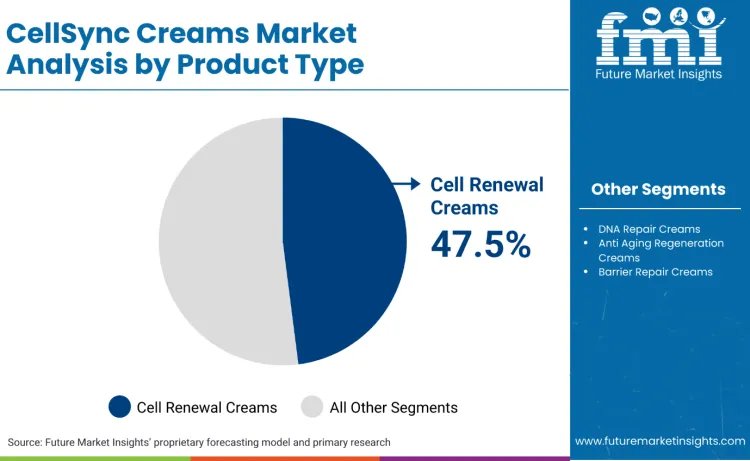
Cell renewal creams lead because they align with the strictest repeatability requirement in this category: performance claims that can be evidenced without triggering drug or biologics positioning. China’s regulator issued the Standard for the Evaluation of Efficacy Claims of Cosmetics effective May 2021, pushing brands toward measurable endpoints supported through testing and disclosure. In the USA, MoCRA increases compliance burden around post-market safety reporting, which favours renewal formats built around widely used cosmetic actives and documented tolerability rather than frontier regenerative positioning. In the EU, Regulation (EC) No 1223/2009 obliges a safety assessment and a product information file, and scaled brands can reuse that governance stack across renewal SKUs, compressing time-to-market. Commercially, renewal creams occupy the daily use slot following in-clinic procedures, making them the easiest unit to bundle into protocols.
What is Driving Online Direct-to-Consumer Leadership in Distribution?
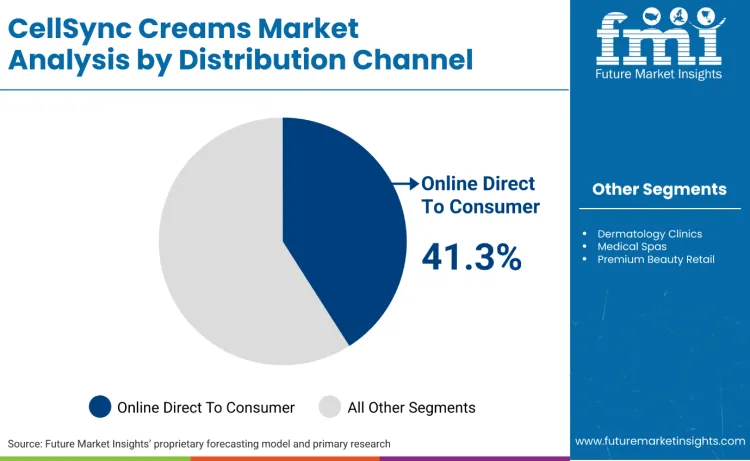
Online direct-to-consumer leads because compliance, education, and replenishment can be centralised in a single operating model, which is critical when claims and safety oversight are tightening. MoCRA adds obligations such as serious adverse event reporting for cosmetics in the USA, and this pushes brands to maintain tighter traceability over what is sold, in which format, and to whom. Direct channels reduce third-party relabelling risk and make it easier to manage label changes and consumer communications at scale. In Great Britain, companies must notify products through the Submit Cosmetic Product Notifications service administered by the Office for Product Safety and Standards, reinforcing the need for accurate product dossiers and rapid update capability. The channel also matches regimen demand, where predictable restocking is central to protocol adherence.
What are the Trends and Restraints in the CellSync Creams Market?
The primary trend is compliance-driven channel consolidation. MoCRA requires serious adverse event reporting for cosmetics in the USA, and it is pulling premium regeneration creams toward systems that resemble regulated health products in how they track complaints, maintain labels, and document safety. In Great Britain, brands must notify products through the Submit Cosmetic Product Notifications service, strengthening expectations for complete product dossiers and rapid update capability when formulations or labels change. China’s efficacy-claim evaluation standard effective in 2021 reinforces the same direction by tying marketing language to test evidence. Large operators are responding by prioritising renewal-oriented platforms and controlled digital channels that allow faster dossier governance and consistent education across markets.
The key restraint is the regulatory boundary between cosmetics and regenerative medicine, which constrains the most aggressive active narratives in this category. FDA has issued public safety communications stating that there are currently no FDA-approved exosome products and warning that firms can mislead patients by treating exosomes as outside drug and biologics controls. This posture limits on-pack and advertising claims, increases legal exposure for stem cell derived and exosome-positioned creams, and forces clinics to be conservative in protocol selection. The result is slower adoption for high-priced regenerative formats and a capital shift toward peptide-based renewal and barrier repair that can be defended as cosmetic positioning.
CellSync Creams Market Analysis by Key Countries
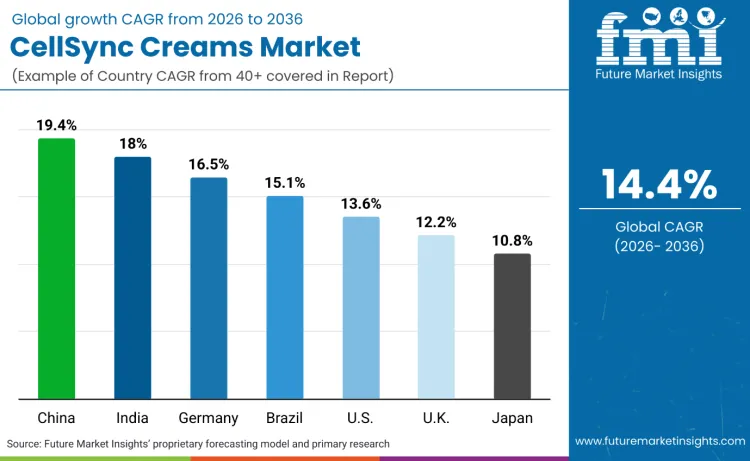
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 19.44% |
| India | 18.00% |
| Germany | 16.56% |
| Brazil | 15.12% |
| USA | 13.68% |
| UK | 12.24% |
| Japan | 10.80% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
How Is Claims Governance Converting Digital Scale into CellSync Creams Demand in China?
China’s 19.44% CAGR is tied to regulatory architecture that rewards substantiated performance positioning and to the speed at which compliant brands can scale through digital channels. The NMPA issued the Standard for the Evaluation of Efficacy Claims of Cosmetics, effective May 1, 2021, which formalises how efficacy claims must be supported and disclosed. That requirement pushes CellSync brands toward measurable renewal outcomes and away from loosely framed regenerative language, making peptide-led renewal creams structurally advantaged. The same governance shift increases the value of firms that can fund testing and maintain documentation across frequent SKU refresh cycles. In parallel, clinic-led aftercare expands the addressable base for recovery creams, and controlled online reordering converts visits into repeatable regimens, sustaining growth.
How Are Cosmetics Rules and Import Discipline Expanding Premium CellSync Creams Uptake in India?
India’s 18.0% CAGR reflects a shift from fragmented import and manufacturing compliance to a more systematised cosmetics regime that reduces friction for premium actives while increasing accountability. The Central Drugs Standard Control Organisation hosts the Cosmetics Rules, 2020 framework and subsequent amendments, which set licensing, import registration, and inspection expectations for manufacturers and importers. For CellSync creams, this governance matters because premium regeneration positioning depends on consistent labelling, traceable sourcing, and controlled claims across batch variation. As compliance infrastructure matures, dermatology clinics gain confidence in prescribing higher priced home-use recovery products, while brands scale direct digital fulfilment beyond tier one cities. These mechanisms support sustained adoption across 2026 to 2036.
Why Do EU Compliance Obligations Push CellSync Creams Demand into Dermatology and Pharmacy Gatekeepers in Germany?
Germany’s 16.56% CAGR is driven by the way European compliance obligations concentrate premium cosmeceutical purchasing into trusted medical and pharmacy-adjacent channels. Regulation (EC) No 1223/2009 requires a responsible person, a safety assessment, and a product information file for cosmetics placed on the EU market, raising documentation costs and favouring suppliers with established regulatory operations. This environment rewards CellSync brands that can defend renewal and barrier claims within cosmetic boundaries while maintaining conservative language around DNA repair and regenerative positioning. Demand is also supported by high utilisation of specialist skin care. Peer reviewed German evidence shows a large dermatology case burden requiring specialist care each year, reinforcing dermatologists as gatekeepers for regimen decisions.
How Do Procedure Density and ANVISA Pathways Translate into Protocol Aftercare Pull for CellSync Creams in Brazil?
Brazil’s 15.12% CAGR is supported by a procedure-dense aesthetics market and by Anvisa’s clearer compliance pathways for cosmetics that stabilise premium import and local manufacturing decisions. ISAPS reports that Brazil performed the highest number of surgical procedures in 2024, creating a large installed base of patients who typically require structured skin recovery routines after interventions. This procedure volume feeds demand for CellSync creams sold through medical spas and dermatology practices, where aftercare is monetised as a protocol rather than a discretionary add-on. On the governance side, Anvisa states that the general regulation for marketing authorisation of personal hygiene products, cosmetics and fragrances in Brazil is Resolution RDC 907/2024, specifying which products require pre-marketing approval and strengthening traceability expectations in premium categories.
Why Are MoCRA Compliance and Aesthetics Throughput Industrialising the USA CellSync Creams Category?
The United States grows at a 13.68% CAGR because the market is being industrialised through regulatory uplift and through the scale of aesthetic procedure ecosystems that convert visits into recurring at-home consumption. ISAPS reports the USA as the country with the most non-surgical procedures performed in 2024, expanding the base of patients placed on structured aftercare regimens. MoCRA adds federal requirements for cosmetic facility registration and product listing and raises expectations around safety oversight, increasing the strategic value of large, compliance-ready professional skincare brands. AbbVie’s Allergan Aesthetics is refreshing SkinMedica’s portfolio, including an April 2024 acne-focused launch framed around provider-led skin health, reinforcing clinic channels as repeat purchase engines through 2036.
How Does Post-Brexit Notification Governance Concentrate CellSync Creams Growth in Traceable UK Channels?
The UK at 12.24% CAGR is being shaped by post-Brexit compliance execution that rewards brands able to run parallel product governance for Great Britain alongside EU dossiers. GOV.UK guidance requires companies to submit cosmetic product notifications to the Office for Product Safety and Standards through the Submit Cosmetic Product Notifications service before products are made available to consumers in Great Britain. For CellSync creams, this reinforces disciplined formulation control, label accuracy, and rapid document updates across frequent line extensions typical of premium renewal platforms. As clinics expand protocol-based aftercare, providers recommend a narrow set of compliant products and consumers reorder online directly from the brand or authorised partners, concentrating growth in traceable channels.
Why Does Cosmetics Versus Quasi-Drug Classification Keep Japan’s CellSync Creams Growth More Measured?
Japan’s 10.8% CAGR reflects a regulatory structure that separates mild cosmetics from quasi-drugs, shaping which CellSync claims can be commercialised and through which channels they scale. Japan’s Ministry of Health, Labour and Welfare describes classification and examination pathways for drugs, quasi-drugs and cosmetics, and the PMDA provides guidance on product classification and procedures. This division matters because stronger functional positioning can push products toward quasi-drug frameworks, raising evidence and notification burdens and favouring established operators with Japanese regulatory capability. Imported regeneration creams therefore compete on renewal and barrier support within cosmetics boundaries, while controlled distribution through clinics and authorised online partners maintains the documentation trail for label and safety updates.
How is the Competition in the CellSync Creams Market?
Competition is concentrated in physician-dispensed and premium protocol brands that can defend cosmetic positioning while borrowing clinical credibility from in-office ecosystems. Scope in this analysis includes cell renewal, DNA repair, anti-ageing regeneration, and barrier repair creams positioned around peptides, growth factors, stem cell derived actives, or exosome-led communication. It excludes injectables, devices, and prescription dermatology drugs. FMI estimates SkinMedica as the largest global player in 2026, supported by AbbVie’s Allergan Aesthetics platform and clinic-first portfolio cadence. North America is the operational centre of this category, where SkinMedica, Alastin and ZO Skin Health compete through dermatologist offices and medical spas, then extend retention through controlled online replenishment.
Europe’s competitive set skews toward premium brands that can operate under Regulation (EC) No 1223/2009 compliance expectations, with positioning anchored in conservative claim language and high-touch specialist channels. Asia is structurally led by China’s claim substantiation and filing discipline, which favours players able to fund efficacy testing, while Japan’s cosmetics versus quasi-drug boundary means global leaders do not automatically translate into regional leadership if claims push products into quasi-drug pathways. Galderma skincare scale supports access, but protocol sellthrough is decisive regionally.
Recent Developments:
- In April 2024, AbbVie’s Allergan Aesthetics launched new SkinMedica acne products positioned around provider-led care pathways.
- In August 2025, AbbVie announced an extension to the SkinMedica HA5 Hydra Collagen portfolio with a new hydrating cleanser.
- In March 2025, Galderma reported record 2024 results and disclosed Dermatological Skincare net sales performance, sustaining investment visibility for protocol skincare platforms.
Key Players in the CellSync Creams Market
- SkinMedica
- Alastin Skincare
- ZO Skin Health
- Revision Skincare
- Neocutis
- PCA Skin
- iS Clinical
- Dr. Barbara Sturm
- Augustinus Bader
- Allies of Skin
Market Definition
CellSync creams are premium topical leave-on formulations positioned to support visible skin recovery and renewal following ageing-related decline or clinic-led aesthetic interventions. The category includes moisturising creams and treatment creams marketed around cellular communication, renewal signalling, or barrier restoration using cosmetic-compliant actives such as peptides, growth factors, or botanical complexes. Products are typically sold through dermatologist offices, medical spas, premium beauty retail, and authorised e-commerce where education and regimen adherence can be controlled. Market value captures manufacturer-level sales of finished creams, excluding in-clinic services and bundled sample-only giveaways.
Market Inclusion
Included revenues cover finished cell renewal creams, DNA repair-positioned creams, anti-ageing regeneration creams, and barrier repair creams sold as cosmetics for facial use. Actives included span peptide-based systems, growth factor complexes, stem cell conditioned media claims where sold as cosmetics, and exosome-positioned actives that remain within cosmetic boundaries. Channel coverage includes dermatology clinics, medical spas, premium beauty retail, and online direct-to-consumer. FMI estimates SkinMedica as the largest global brand in 2026, while Europe is more fragmented and Asia scale is led by China regulated operators. Japan differs due to quasi-drug boundaries.
Market Exclusion
Excluded from this market are injectable aesthetics products, dermal fillers, neurotoxins, energy-based devices, microneedling tools, and professional in-clinic consumables. Prescription dermatology drugs and OTC drug creams regulated as medicines are excluded, even when used for skin renewal outcomes. Oral supplements and ingestible beauty products are excluded. Products marketed with regenerative medicine claims that require biologics or drug approval are excluded where they cannot be legally sold as cosmetics in the stated country. Revenue from clinic services and procedure fees is excluded, even when bundled with aftercare kits. This prevents overstating leadership where local regulatory pathways differ.
Scope of Report Table
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 2.3 billion |
| Forecast Value (2036) | USD 8.8 billion |
| CAGR (2026 to 2036) | 14.4% |
| Product Type Segments | Cell Renewal Creams; DNA Repair Creams; Anti Aging Regeneration Creams; Barrier Repair Creams |
| Active Technology Segments | Peptide Based; Growth Factor Based; Stem Cell Derived; Exosome Based |
| Skin Type Segments | Dry Skin; Normal Skin; Oily Skin; Sensitive Skin |
| Distribution Channel Segments | Online Direct To Consumer; Dermatology Clinics; Medical Spas; Premium Beauty Retail |
| End User Segments | Women; Men; Unisex |
| Countries Covered | China; India; Germany; Brazil; USA; UK; Japan |
| Key Companies Profiled | SkinMedica; Alastin Skincare; ZO Skin Health; Revision Skincare; Neocutis; PCA Skin; iS Clinical; Dr. Barbara Sturm; Augustinus Bader; Allies of Skin |
| Additional Attributes | Dollar sales by product type, active technology, skin type, distribution channel, and end user; protocol mix across dermatologist offices and medical spas versus retail-led regimens; compliance uplift impacts under MoCRA, EU Regulation (EC) No 1223/2009, Great Britain SCPN notification, and China efficacy-claim evaluation; claim defensibility dynamics for peptide and growth factor systems versus stem cell derived and exosome-positioned narratives under regulator enforcement; channel governance requirements including traceability, label change control, adverse event handling, and authorised online replenishment; competition intensity by region with explicit Japan differentiation under cosmetics versus quasi-drug boundaries; supply chain priorities covering ingredient traceability, batch documentation, and quality systems to support post-market obligations. |
CellSync Creams Market by Key Segments
By Product Type:
Cell Renewal Creams; DNA Repair Creams; Anti Aging Regeneration Creams; Barrier Repair Creams
By Active Technology:
Peptide Based; Growth Factor Based; Stem Cell Derived; Exosome Based
By Skin Type:
Dry Skin; Normal Skin; Oily Skin; Sensitive Skin
By Distribution Channel:
Online Direct To Consumer; Dermatology Clinics; Medical Spas; Premium Beauty Retail
By End User:
Women; Men; Unisex
Bibliography
- Agência Nacional de Vigilância Sanitária. (2024).Personal hygiene products, cosmetics and fragrances regulation (RDC 907/2024) overview.
- Central Drugs Standard Control Organisation. (n.d.).Cosmetics rules. Government of India.
- European Parliament and Council of the European Union. (2009).Regulation (EC) No 1223/2009 on cosmetic products. EUR-Lex.eur-lex.europa.eu
- International Society of Aesthetic Plastic Surgery. (2025).2024 international survey on aesthetic/cosmetic procedures performed in 2024[Report].
- National Medical Products Administration. (2021, April 9).Announcement of the standard for the evaluation of efficacy claims of cosmetics(China).
- USA Food and Drug Administration. (2019, December 6).Public safety notification on exosome products.
- USA Food and Drug Administration. (2026, January 21).MoCRA: Modernization of Cosmetics Regulation Act of 2022.
Frequently Asked Questions
How big is the CellSync creams market in 2026 and 2036?
The CellSync creams market is valued at USD 2.3 billion in 2026 and is projected to reach USD 8.8 billion by 2036, expanding at a 14.4% CAGR.
Which product type leads the CellSync creams market?
Cell Renewal Creams lead with a 47.5% share, supported by defensible cosmetic claims and daily protocol placement.
Which active technology leads the CellSync creams market?
Peptide-based platforms lead with a 38.6% share, reflecting lower regulatory exposure versus frontier regenerative positioning.
Which distribution channel leads adoption?
Online direct-to-consumer leads with a 41.3% share, supported by traceability, rapid label governance, and replenishment economics.
Which country has the strongest growth outlook?
China leads with a 19.44% CAGR, driven by efficacy-claim evaluation requirements and disciplined documentation-led scaling.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Product Type, 2026 to 2036
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Active Technology
- Y to o to Y Growth Trend Analysis By Active Technology
- Absolute $ Opportunity Analysis By Active Technology
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Skin Type
- Y to o to Y Growth Trend Analysis By Skin Type
- Absolute $ Opportunity Analysis By Skin Type
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Y to o to Y Growth Trend Analysis By Distribution Channel
- Absolute $ Opportunity Analysis By Distribution Channel
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Y to o to Y Growth Trend Analysis By End User
- Absolute $ Opportunity Analysis By End User
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- Key Countries Market Analysis
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- Competition Analysis
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- CellSync Creams Market Key Takeaways
- Global CellSync Creams Market Size (2021 to 2036)
- CellSync Creams Market Size by Product Type
- CellSync Creams Market Size by Active Technology
- CellSync Creams Market Size by Skin Type
- CellSync Creams Market Size by Distribution Channel
- CellSync Creams Market Size by End User
- CellSync Creams Market Size by Region
- CellSync Creams Market CAGR by Country
- Scope of Report
List of Figures
- CellSync Creams Market Value Growth Trajectory (2026 to 2036)
- Governance-led Market Evolution Framework
- Product Type Share Analysis
- Active Technology Share Distribution
- Distribution Channel Governance Model
- Regional Market Attractiveness Map
- Competitive Landscape Overview
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

