Critical Care Biomarker Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Critical Care Biomarker Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Critical Care Biomarker Market Forecast and Outlook 2026 to 2036
The critical care biomarker market is projected to reach USD 5.3 billion in 2026 and grow to USD 18.6 billion by 2036, reflecting a CAGR of 13.4%. Procurement decisions in this market are heavily influenced by the need for accuracy and speed in diagnostic tools, with a growing preference for proven solutions. Buyer risk aversion is significant due to the high stakes of critical care, leading to a tendency to favor well-established suppliers with a history of reliable performance. Vendor lock-in is often seen in this market, as long-term contracts and specific regulatory requirements make switching to new suppliers challenging. Additionally, high switching costs associated with specialized equipment and integration complexities contribute to long-term supplier relationships. As the demand for personalized and precise diagnostics increases, procurement strategies will be shaped by the balance of cost, reliability, and technological advancements, while ensuring the adaptability of solutions to meet evolving healthcare needs.
Quick Stats for Critical Care Biomarker Market
- Critical Care Biomarker Market Value (2026): USD 5.3 Billion
- Critical Care Biomarker Market Forecast Value (2036): USD 18.6 Billion
- Critical Care Biomarker Market Forecast CAGR 2026 to 2036: 13.4%
- Leading Product Type in Critical Care Biomarker Market: Cardiac Biomarkers
- Leading Application in Critical Care Biomarker Market: Early Diagnosis
- Key Growth Regions in Critical Care Biomarker Market: North America, Asia Pacific, Europe
- Top Key Players in Critical Care Biomarker Market: Abbott, Roche Diagnostics, Siemens Healthineers, Beckman Coulter, bioMérieux
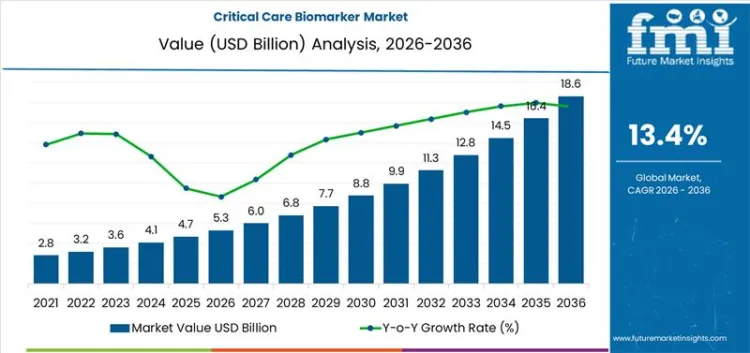
Critical Care Biomarker Market Key Takeaways
| Metric | Value |
|---|---|
| Critical Care Biomarker Market Value (2026) | USD 5.3 Billion |
| Critical Care Biomarker Market Forecast Value (2036) | USD 18.6 Billion |
| Critical Care Biomarker Market Forecast CAGR 2026 to 2036 | 13.4% |
Why is the Critical Care Biomarker Market Growing?
The critical care biomarker market is experiencing growth due to the increasing demand for biomarkers that can help in the early diagnosis, monitoring, and treatment of patients in critical care settings. Biomarkers play a crucial role in assessing the severity of diseases, determining prognosis, and guiding therapeutic interventions, especially in conditions like sepsis, heart failure, and organ dysfunction. As healthcare providers seek more accurate and efficient diagnostic tools, the demand for critical care biomarkers continues to rise.
Technological advancements in diagnostic tools and biomarker discovery are driving the expansion of the critical care biomarker market. Innovations in high-throughput screening, proteomics, genomics, and liquid biopsy technologies are enhancing the ability to identify and validate new biomarkers with greater sensitivity and specificity. These advancements help in better disease management, faster patient recovery, and more personalized treatment plans, making biomarkers increasingly essential in critical care environments.
The growing prevalence of chronic diseases, rising healthcare expenditure, and the shift toward personalized medicine are reinforcing the need for critical care biomarkers. As healthcare systems prioritize precision medicine and more efficient patient care, biomarkers are becoming indispensable in clinical decision-making. These factors are contributing to the sustained growth of the critical care biomarker market over the forecast period.
What Does The Segment Overview Indicate About The Critical Care Biomarker Market?
The critical care biomarker market is segmented by product type and application, highlighting the importance of biomarkers in diagnosing, monitoring, and managing critical care conditions. Cardiac biomarkers lead the product segment with 28% share, primarily due to their role in diagnosing acute myocardial infarction and heart failure. Sepsis, coagulation, renal injury, and respiratory biomarkers cater to specific critical care needs. On the application side, early diagnosis accounts for 36% of the market share, as biomarkers are crucial for identifying diseases at an early stage, enabling timely intervention. Other applications, including risk stratification, treatment monitoring, and prognosis assessment, reflect the wide range of diagnostic and therapeutic uses in intensive care units (ICUs) and emergency settings.
Why Do Cardiac Biomarkers Hold The Leading Product Share In The Critical Care Biomarker Market?
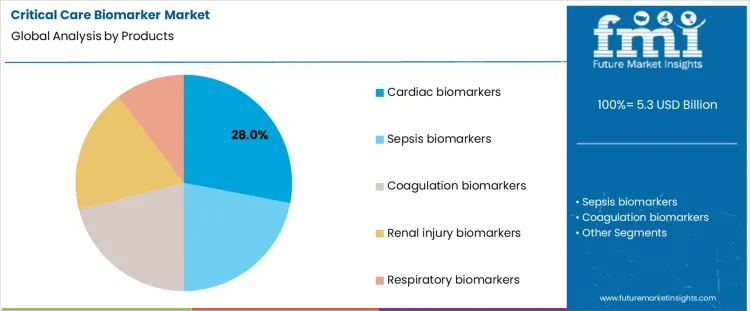
Cardiac biomarkers hold the largest 28% share in the critical care biomarker market due to their essential role in diagnosing and monitoring heart-related conditions such as acute myocardial infarction, heart failure, and arrhythmias. These biomarkers, such as troponin, BNP (B-type natriuretic peptide), and CK-MB (creatine kinase-MB), are crucial for rapid diagnosis in emergency and ICU settings. The high demand for accurate, early detection of cardiovascular events, along with the increasing prevalence of heart disease, continues to drive the dominance of cardiac biomarkers. Advances in biomarker discovery, improved sensitivity, and their ability to guide treatment decisions further strengthen their position in the market.
How Does Early Diagnosis Drive Application Demand In The Critical Care Biomarker Market?
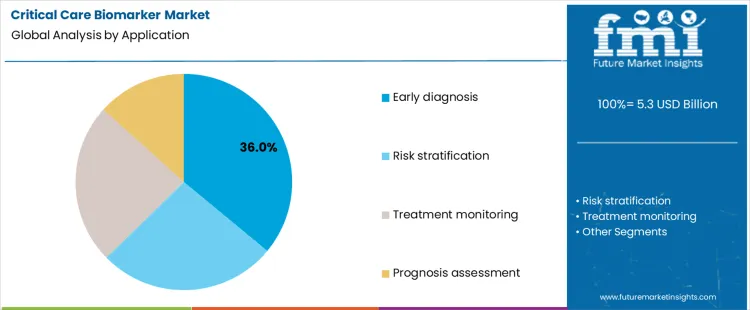
Early diagnosis is the leading application in the critical care biomarker market, accounting for 36% share, as biomarkers are essential in the rapid identification of life-threatening conditions. Early detection allows for timely interventions that can significantly improve patient outcomes, especially in critical care environments such as ICUs and emergency departments. For instance, cardiac biomarkers enable the early diagnosis of myocardial infarction, while sepsis biomarkers help identify sepsis before it progresses to organ failure. Early diagnosis also plays a key role in triaging patients, prioritizing those who need immediate treatment. The growing emphasis on preventive healthcare and early intervention strategies further fuels the demand for biomarkers in early diagnosis applications.
What Are the Key Dynamics for the Critical Care Biomarker Market?
The critical care biomarker market is shaped by growing demand for faster, more accurate diagnostics in intensive care settings. Biomarkers are crucial in detecting and monitoring life-threatening conditions like sepsis, organ failure, and cardiac events, driving their adoption in hospitals and emergency care units. Advances in biomarker technology, such as point-of-care devices and AI-driven analysis, are improving diagnostic capabilities and reducing healthcare costs. However, challenges related to standardization, regulatory approval, and high costs remain obstacles to wider implementation. As demand for personalized medicine and early-stage diagnostics rises, the market is expected to continue growing, especially in developed healthcare markets where advanced diagnostic tools are in higher demand.
What Are the Key Challenges in the Critical Care Biomarker Market?
The critical care biomarker market faces challenges related to the complexity of biomarker discovery, regulatory hurdles, and issues with standardization. Developing accurate and reliable biomarkers for use in critical care settings requires extensive research and validation, which can be time-consuming and expensive. Regulatory approval processes for critical care diagnostics are stringent, leading to delays in market entry. Furthermore, variability in patient populations and the complexity of multi-organ conditions make it difficult to develop biomarkers that are universally applicable across different demographic groups. Additionally, the high cost of advanced biomarker tests and the need for specialized equipment can limit widespread adoption, particularly in resource-limited settings.
How Are Technological Advancements Shaping the Critical Care Biomarker Market?
Technological advancements are transforming the critical care biomarker market by improving the speed, accuracy, and accessibility of diagnostic tests. Innovations in point-of-care testing devices, microfluidics, and biosensors are enabling rapid biomarker detection at the patient’s bedside, reducing wait times and improving treatment outcomes. The integration of artificial intelligence (AI) and machine learning (ML) is enhancing the analysis of biomarker data, enabling more precise predictions of patient prognosis and treatment response. Advanced multiplexing techniques are allowing for the simultaneous detection of multiple biomarkers, improving diagnostic accuracy and facilitating comprehensive patient assessments. These advancements are making critical care biomarkers more efficient and accessible for healthcare providers.
What is the Country-Wise Growth Outlook for the Critical Care Biomarker Market?
The critical care biomarker market is expanding due to advancements in diagnostic technologies, the increasing prevalence of chronic diseases, and the rising demand for personalized medicine. India leads with a CAGR of 11.2%, driven by a growing healthcare infrastructure, rising awareness of critical care diagnostics, and an increasing need for biomarkers in disease monitoring. China follows closely with a CAGR of 11%, supported by significant investments in healthcare and medical research. Brazil shows a CAGR of 10.6%, driven by improvements in healthcare access and government initiatives for advanced diagnostic technologies. The USA records a CAGR of 9.4%, with increasing adoption of biomarkers in critical care settings and advancements in personalized medicine. France grows at a CAGR of 9.3%, benefiting from strong healthcare research, an aging population, and increasing demand for early diagnosis and treatment of critical diseases.
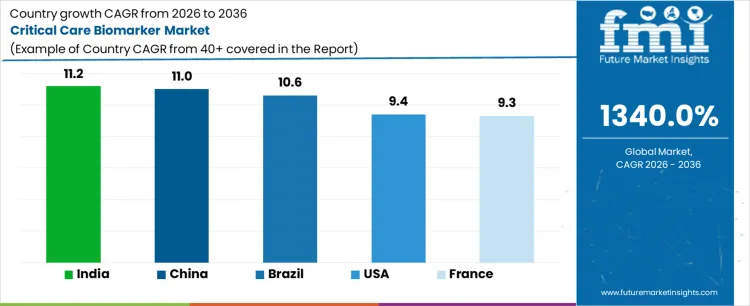
| Country | CAGR |
|---|---|
| India | 11.2% |
| China | 11% |
| Brazil | 10.6% |
| USA | 9.4% |
| France | 9.3% |
Why Is India Leading in the Critical Care Biomarker Market?
Demand for critical care biomarkers in India is growing at a CAGR of 11.2%, driven by increasing healthcare infrastructure, rising awareness of critical care diagnostics, and an expanding need for biomarkers in disease monitoring. India’s rapidly expanding healthcare sector is placing greater emphasis on early diagnosis and personalized medicine, fueling the demand for advanced diagnostic tools. The rising burden of chronic diseases, such as cardiovascular conditions, diabetes, and infections, is driving the need for biomarkers that can aid in early detection, treatment planning, and monitoring. The Indian government’s initiatives to improve access to healthcare and increase investments in medical research further contribute to the growth of the biomarker market. The increasing adoption of point-of-care diagnostic solutions in hospitals and clinics is enabling faster and more accurate biomarker testing, contributing to the market’s expansion. As India’s healthcare system continues to evolve and improve, the demand for critical care biomarkers is expected to continue its strong growth.
What Is Driving China’s Growth in the Critical Care Biomarker Market?
Sales for critical care biomarkers in China are growing at a CAGR of 11%, fueled by significant investments in healthcare infrastructure, medical research, and the demand for advanced diagnostic technologies. China’s healthcare system is expanding rapidly, driven by urbanization, an aging population, and a rising incidence of chronic diseases. As the country focuses on improving healthcare access and adopting innovative medical technologies, there is increasing demand for critical care biomarkers that can aid in the early detection and management of diseases such as cancer, heart disease, and diabetes. China’s large population and growing healthcare spending provide a solid foundation for the continued growth of the biomarker market. The Chinese government is actively supporting the development of diagnostic technologies and clinical research, which is further driving the demand for critical care biomarkers. The increased use of biomarkers for personalized medicine and treatment plans is contributing to the country’s rapidly growing market, positioning China as a leading player in the global critical care biomarker market.
What Is Contributing to the Growth of the Critical Care Biomarker Market in Brazil?
Demand for critical care biomarkers in Brazil is growing at a CAGR of 10.6%, driven by the country’s healthcare modernization, government support for advanced diagnostics, and increasing prevalence of chronic diseases. Brazil’s expanding healthcare system is improving access to diagnostic tools, allowing more patients to benefit from the early detection and management of critical conditions. The growing burden of diseases like cardiovascular disorders, cancer, and diabetes is increasing the need for biomarkers to aid in diagnosis and treatment. Brazil’s public and private healthcare sectors are increasingly investing in advanced diagnostic technologies, including critical care biomarkers, to improve disease outcomes and healthcare efficiency. The government’s initiatives to strengthen healthcare infrastructure, coupled with increasing collaborations between healthcare providers and research institutions, are further driving market growth. Brazil’s large and diverse population provides ample opportunities for the widespread adoption of biomarkers in critical care settings, positioning the country as a key growth region in Latin America.
What Is Supporting Growth in the USA’s Critical Care Biomarker Market?
Sales for critical care biomarkers in the USA are growing at a CAGR of 9.4%, driven by the strong adoption of advanced diagnostic tools, rising demand for personalized medicine, and increasing prevalence of chronic and critical diseases. The USA has a well-established healthcare infrastructure and a robust clinical research environment, making it a leading market for biomarkers used in critical care settings. The rising incidence of chronic diseases, particularly cardiovascular conditions and diabetes, is increasing the need for biomarkers that can aid in diagnosis, risk stratification, and treatment monitoring. The USA’s focus on precision medicine is fueling the demand for biomarkers that enable individualized treatment approaches. Advancements in biotechnology and diagnostics, including the development of point-of-care solutions, are further supporting market growth. The growing adoption of novel biomarkers in clinical trials, healthcare practices, and research institutions ensures that the USA remains a key player in the global critical care biomarker market.
What Is Contributing to the Growth of the Critical Care Biomarker Market in France?
Demand for critical care biomarkers in France is increasing at a CAGR of 9.3%, supported by advancements in medical research, healthcare system improvements, and the rising need for early diagnosis of critical conditions. France is a leader in medical research and biotechnology, with numerous academic and private institutions conducting research on new biomarkers for disease detection and management. The country’s growing focus on personalized medicine and the adoption of innovative diagnostic technologies are driving the demand for critical care biomarkers in hospitals and healthcare centers. France’s aging population, coupled with an increasing prevalence of cardiovascular and metabolic diseases, is further fueling the need for biomarkers to aid in diagnosis, treatment, and monitoring. The country’s well-established healthcare system, supported by government initiatives and public-private partnerships, is expanding access to biomarker testing and treatment options. As France continues to prioritize innovation in medical diagnostics, the demand for critical care biomarkers is expected to grow steadily.
How Are Leading Companies Competing And Differentiating In The Critical Care Biomarker Market?

Competition in the critical care biomarker market is driven by innovation in diagnostic tools, accuracy, and the ability to provide real-time, actionable insights for patient care. Abbott is positioned as a leader with its comprehensive range of diagnostic tests and platforms tailored for critical care, offering high sensitivity and rapid results for biomarkers related to sepsis, cardiac events, and renal function. Their product brochures emphasize the ability to deliver reliable diagnostics at the point of care, improving patient outcomes by facilitating early detection and timely interventions. Roche Diagnostics differentiates itself by providing advanced biomarker assays that can be integrated with its comprehensive diagnostic systems, enhancing clinical decision-making through precise biomarker measurement in various critical care settings.
Siemens Healthineers leverages its expertise in imaging and laboratory diagnostics, offering innovative solutions in biomarker testing that integrate seamlessly with its broader healthcare portfolio. Their products focus on enhancing diagnostic accuracy and efficiency, particularly for patients in intensive care, and aim to reduce turnaround time for test results. Beckman Coulter provides a broad array of biomarker testing solutions, particularly for the detection of biomarkers related to inflammation, infection, and cardiac conditions. Their systems are known for delivering reliable results quickly, facilitating better clinical management in critical care environments.
bioMérieux focuses on providing high-quality diagnostics for infectious diseases and critical conditions in intensive care units. The company is known for its expertise in microbiological testing and biomarker identification, which helps clinicians address sepsis and other life-threatening conditions. Across the market, differentiation is driven by the integration of diagnostic platforms, rapid results, and the ability to provide actionable insights that support timely clinical interventions, with a strong emphasis on improving patient care in critical settings.
Key Players in Critical Care Biomarker Market
- Abbott
- Roche Diagnostics
- Siemens Healthineers
- Beckman Coulter
- bioMérieux
- Others
Scope of Report
| Attributes | Description |
|---|---|
| Quantitative Unit | USD Billion |
| Product Types | Cardiac biomarkers, Sepsis biomarkers, Coagulation biomarkers, Renal injury biomarkers, Respiratory biomarkers |
| Applications | Early diagnosis, Risk stratification, Treatment monitoring, Prognosis assessment |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Key Companies Profiled | Abbott, Roche Diagnostics, Siemens Healthineers, Beckman Coulter, bioMérieux |
| Additional Attributes | Market revenue by product and application; adoption trends for critical care biomarkers; regional growth analysis; projected market size and CAGR for 2026-2036; technological advancements in diagnostic systems; competitive landscape with strategy and portfolio analysis of leading players; emerging applications in critical care, prognosis assessment, and treatment monitoring. |
Critical Care Biomarker Market by Key Segments
Products
- Cardiac biomarkers
- Sepsis biomarkers
- Coagulation biomarkers
- Renal injury biomarkers
- Respiratory biomarkers
Application
- Early diagnosis
- Risk stratification
- Treatment monitoring
- Prognosis assessment
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- Food and Drug Administration, Center for Drug Evaluation and Research. (2024). Biomarker qualification and use in drug development for serious or life-threatening illnesses: Critical care contexts (Guidance for Industry). U.S. Department of Health and Human Services.
- National Institutes of Health, National Heart, Lung, and Blood Institute. (2024). Cardiovascular biomarkers: Clinical and research applications in acute and critical care. National Institutes of Health.
- European Medicines Agency. (2024). Scientific guidance on biomarkers in therapeutic development: Implications for emergency and intensive care medicine. European Medicines Agency.
Frequently Asked Questions
How big is the critical care biomarker market in 2026?
The global critical care biomarker market is estimated to be valued at USD 5.3 billion in 2026.
What will be the size of critical care biomarker market in 2036?
The market size for the critical care biomarker market is projected to reach USD 18.6 billion by 2036.
How much will be the critical care biomarker market growth between 2026 and 2036?
The critical care biomarker market is expected to grow at a 13.4% CAGR between 2026 and 2036.
What are the key product types in the critical care biomarker market?
The key product types in critical care biomarker market are cardiac biomarkers, sepsis biomarkers, coagulation biomarkers, renal injury biomarkers and respiratory biomarkers.
Which application segment to contribute significant share in the critical care biomarker market in 2026?
In terms of application, early diagnosis segment to command 36.0% share in the critical care biomarker market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Products
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Products, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Products, 2026 to 2036
- Cardiac biomarkers
- Sepsis biomarkers
- Coagulation biomarkers
- Renal injury biomarkers
- Respiratory biomarkers
- Cardiac biomarkers
- Y to o to Y Growth Trend Analysis By Products, 2021 to 2025
- Absolute $ Opportunity Analysis By Products, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Early diagnosis
- Risk stratification
- Treatment monitoring
- Prognosis assessment
- Early diagnosis
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Products
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Products
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Products
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Products
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Products
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Products
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Products
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Products
- By Application
- Competition Analysis
- Competition Deep Dive
- Abbott
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Roche Diagnostics
- Siemens Healthineers
- Beckman Coulter
- bioMérieux
- Others
- Abbott
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Products, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Products, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Products, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Products, 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Products, 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Products, 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Products, 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Products, 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Products, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Products, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Products
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Products, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Products, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Products
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Products, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Products, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Products
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Products, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Products, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Products
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Products, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Products, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Products
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Products, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Products, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Products
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Products, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Products, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Products
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Products, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Products, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Products
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

