Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market is segmented by Product Format (Powder, Liquid, Capsules, Gummies), Application (Dietary Supplements, Functional Foods, Animal Feed, Pharmaceuticals), Consumer Group (Adults, Pediatrics, Geriatrics), Distribution Channel (Online Retail, Pharmacies, Supermarkets, Specialty Stores), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Size, Market Forecast and Outlook By FMI
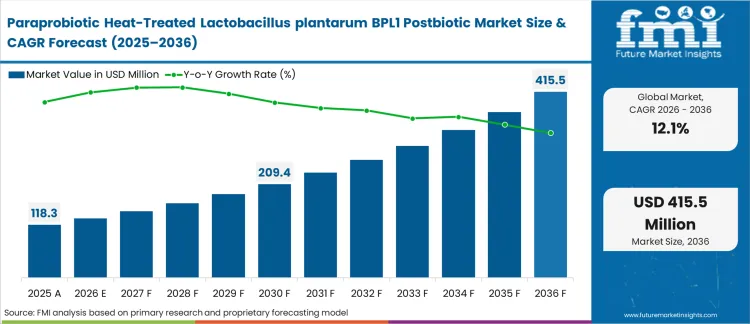
In 2025, the Paraprobiotic heat-treated lactobacillus plantarum BPL1 postbiotic market was valued at USD 118.4 million. Based on Future Market Insights’ analysis, demand is estimated to grow to USD 132.6 million in 2026 and USD 412.9 million by 2036. FMI projects a CAGR of 12.1% during the forecast period.
Brand owners reprioritize formulation stability to address escalating metabolic conditions, shifting specifications toward inanimate biomass capable of surviving high-temperature gummy and extrusion manufacturing. Live cultures suffer viability degradation during transit, forcing manufacturers toward heat-treated BPL1 to secure consistent, CFU-free positioning without cold-chain overhead. Procurement teams mandate these stable inputs to compress logistics costs and guarantee label compliance upon retail shelving. In September 2024, United States authorities reported adult obesity prevalence at 40.3%, escalating the clinical requirement for targeted, mass-market metabolic interventions [1].
Summary of Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic
- Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Definition
- Industry encompasses non-viable, thermally processed microbial biomass utilized across commercial nutrition and health applications.
- Demand Drivers in the Market
- Escalating global diabetes and obesity rates compel preventative nutrition adoption.
- Manufacturers demand ambient-stable ingredients to eliminate cold-chain freight expenses.
- Consumer preference shifts toward science-backed, transparently labeled postbiotic ingredients.
- Key Segments Analyzed in the FMI Report
- Product Format: Dominant powder formats (44% share in 2026) versus liquid suspensions, capsules, and gummies.
- Application: Dietary supplements (39% of total volume) versus functional foods, animal feed, and pharmaceutical applications.
- Geography: High-growth Asia-Pacific (India 15.1%, China 14.5%) versus regulatory-intensive mature North American and European markets.
- Analyst Opinion at FMI
- Nandini Roy Choudhury, Principal Consultant for Food & Beverage at Future Market Insights, opines, “Through our recent analysis, we observed that tightening probiotic labeling scrutiny is accelerating the pivot toward postbiotics and paraprobiotics. Heat-treated BPL1 offers defined composition, simplified storage logistics, and clearer compliance pathways across global markets. Companies that integrate documented strain specificity with transparent health-claim substantiation will lead the next generation of microbiome-driven product innovation.”
- Strategic Implications/Executive Takeaways
- Formulators should pivot away from live cultures to minimize shelf-life degradation.
- Suppliers must secure regional distribution partnerships to penetrate fragmented geographies.
- Investors view documented thermal stability as a critical valuation multiplier.
- Methodology
- Validated through first-hand corporate production and capacity data.
- Zero reliance on speculative third-party market research reports.
- Based on verifiable industrial benchmarks and verified sources.
Localized distribution networks de-risk the procurement of advanced inanimate microbial strains by eliminating complex cross-border cold-chain logistics. Doug Lynch, Co-founding Partner and Chief Commercial Officer of Postbiotics, Inc. stated “We are pleased to be working with Sabinsa, and believe their global sales organization and manufacturing prowess are perfect complements to our world-class research. This distribution partnership with Sabinsa for will ensure consumer brands of supplements and foods in the United States and beyond will experience the many benefits from postbiotics on improving health and lifespan.” [13]. Access to established manufacturing prowess compresses ingredient lead times and lowers formulation barriers for contract manufacturers. Buyers shift volume toward these vertically integrated distribution partnerships to guarantee consistent ambient-stable supply and protect retail margins.
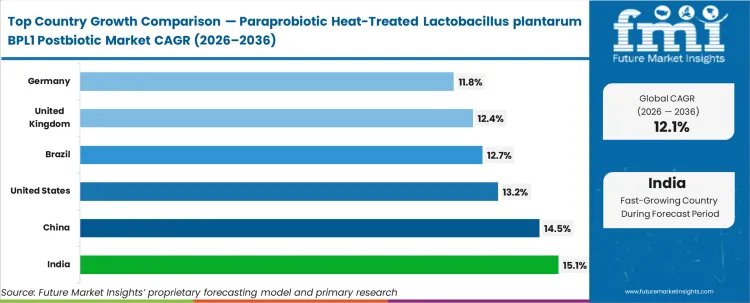
Growth trends differ from country to country and are shaped by a mix of chronic disease burden, healthcare infrastructure, and how willing consumers are to spend on preventive health products. The United States (13.2%), United Kingdom (12.4%), Germany (11.8%), China (14.5%), India (15.1%), and Brazil (12.7%) are all growing at different rates, with stronger momentum usually seen in markets where metabolic health concerns are increasing and clinical awareness is relatively high. In response, brand owners are focusing more on strains supported by credible clinical trials so they can better match local health needs and consumer expectations.
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 132.6 million |
| Industry Value (2036) | USD 412.9 million |
| CAGR (2026-2036) | 12.1% |
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Definition
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic represents inanimate microbial cells and associated metabolites utilized for health benefits. Material undergoes specific thermal processing to deactivate live cells while preserving structural integrity. Applications span human nutrition, animal feed, and topical formulations. Output measures total commercial value generated from business-to-business raw material sales and specialized consumer-facing bulk formats.
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Inclusions
The Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 postbiotic scope includes commercially produced, heat-inactivated BPL1 biomass manufactured to ensure non-viability while preserving structural bioactive components. It covers tyndallized or pasteurized cellular material, including intact cell walls and exopolysaccharides, supplied in standardized powder or liquid formats for dietary supplements and functional food applications.
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Exclusions
The scope excludes live or viable Lactobacillus plantarum BPL1 probiotic cultures, other Lactobacillus strains, and unrelated postbiotic variants. It does not include commodity prebiotics, fiber ingredients, synbiotic blends, or finished consumer product value. Experimental materials without established commercial-scale production are also excluded from the defined category.
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Research Methodology
- Primary Research: Interviews with ingredient suppliers, formulation chemists, and regulatory consultants confirm capacity outputs.
- Desk Research: UNCTAD trade statistics, CDC prevalence data, and International Probiotics Association bulletins inform baseline models.
- Market-Sizing and Forecasting: Bottom-up volume assessment reconciles against top-down metabolic disease prevalence curves.
- Data Validation and Update Cycle: Analyst panels cross-verify corporate expansion disclosures against actual plant commissioning timelines.
Segmental Analysis
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Analysis by Product Format
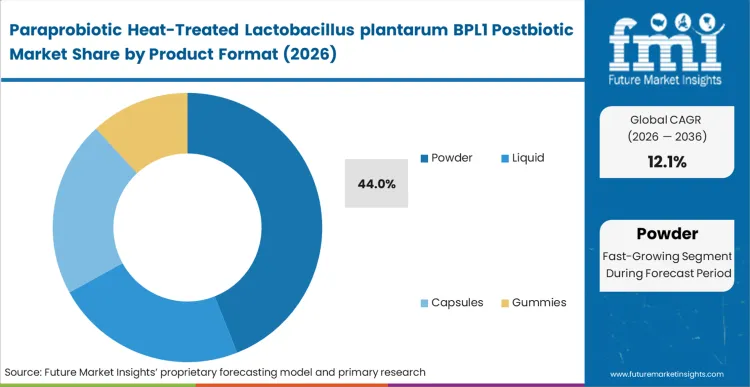
Manufacturers are steadily shifting capital toward dry matrices as they look to reduce process instability and protect ingredient performance during scale-up. Dry systems are increasingly favored in high-shear encapsulation environments because they lower the risk of moisture-induced degradation and support more consistent throughput. At the same time, suppliers are moving away from liquid suspensions to reduce freight costs and remove the need for specialized temperature controls across distribution networks. This structural preference is reflected in market concentration, with powder grades accounting for 44% of global volume in 2026.
Sirio Pharma’s February 2024 launch of postbiotic gummies featuring EpiCor also signals broader confidence in resilient formats that can withstand thermal processing and commercial manufacturing demands [3]. For brand owners, powder stability improves margin protection and expands formulation options in delivery formats such as confectionery and baked goods. According to FMI's estimates, formulation scientists increasingly require finely milled powder profiles to maintain homogeneous dispersion in continuous blending systems. Ingredient suppliers are responding by adding spray-drying capacity and locking in longer-term supply agreements with contract manufacturers. Repeated investment in drying infrastructure reinforces dry powder as the commercial standard for inanimate microbial deployment, while cold-chain-dependent liquid suspensions continue to lose buyer preference.
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Analysis by Application
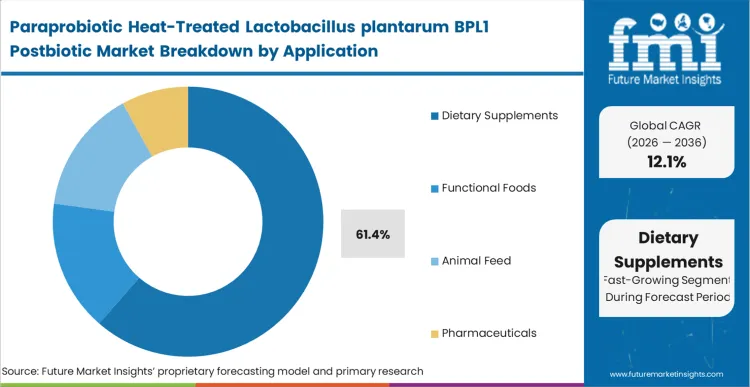
The application landscape is being reshaped by preventive health spending, which is pulling demand away from broad digestive aids and toward clinically positioned metabolic formulations. Supplement brands are redesigning product portfolios to capture higher price points linked to measurable physiological outcomes, especially where product claims can be anchored in clearer evidence. A strong adoption base already exists: dietary supplement use reached 61.4% among United States adults during the 2021 to 2023 period, showing how deeply nutritional interventions are embedded in routine health behavior [4]. Within this environment, dietary supplements capture 39% of total consumption in 2026, making them the leading application channel for BPL1 postbiotic commercialization.
Companies are also replacing viable probiotics with heat-treated alternatives to avoid overage costs and reduce recall risks tied to bacterial die-off. FMI analysts opine that supplement-specific compliance pathways can favor highly characterized microbial ingredients, particularly when stability and composition are easier to verify. Brand messaging increasingly emphasizes ambient stability and precise dosage delivery as core product advantages. Larger players are gradually redirecting capital from legacy live-culture portfolios toward stable postbiotic ferment filtrates that ensure active ingredient presence through expiry.
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Analysis by Consumer Group
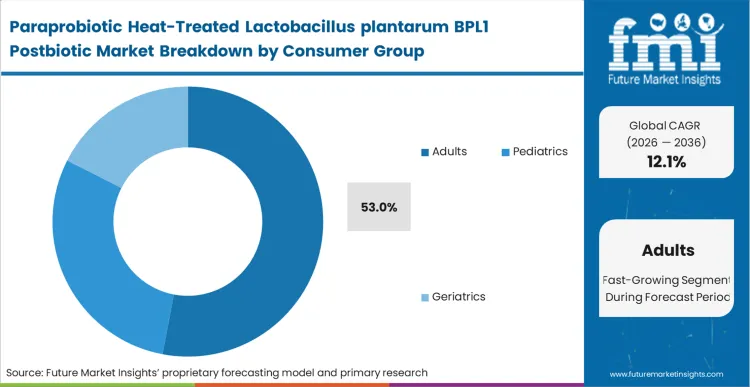
Rising metabolic health concerns among working-age populations are making adults the central demand engine for BPL1-based paraprobiotic products. Many consumers in this group are looking for non-prescription tools that fit daily routines, while employers, insurers, and healthcare platforms are placing greater emphasis on preventive interventions that may help delay long-term chronic disease costs. The scale of the opportunity is significant: global data for 2025 shows 589 million adults living with diabetes, creating a large addressable base for formulations tied to glucose management and metabolic support [5]. Against this backdrop, adults account for 53% of segment share in 2026, supported by both purchasing power and a stronger willingness to pay for clinically validated products.
Leading nutrition companies are aligning marketing and innovation budgets around adult lifestyle and metabolic outcomes rather than broad-spectrum wellness messaging. This segment also allows premium pricing for BPL1 formulations backed by stronger efficacy narratives. Buying behavior increasingly favors specialized, evidence-supported microbial solutions over generic vitamin products. Strategic portfolio expansion across major brands continues to confirm adult metabolic health as the primary route for scaling inanimate bacterial commercialization.
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Analysis by Distribution Channel
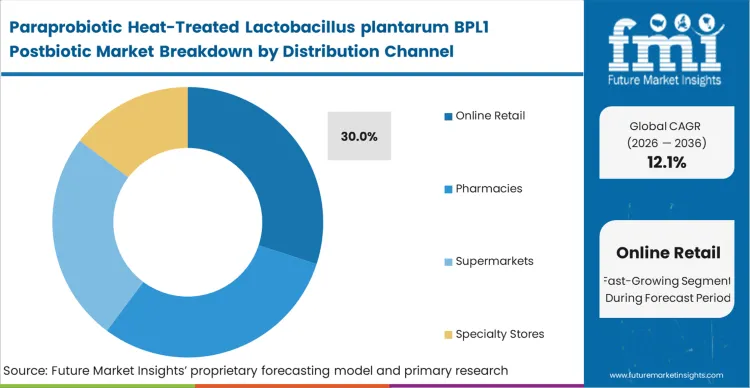
Digital commerce has become the preferred commercialization route for technically complex nutrition products because it gives brands more room to educate consumers before purchase. For paraprobiotic formulations, that educational layer matters, since customer conversion often depends on explaining mechanism, stability, and usage benefits in a way that traditional retail shelves cannot support. Direct-to-consumer platforms also improve launch economics by enabling faster testing, tighter feedback loops, and better control over messaging. In June 2024, online retail represented roughly 25% to 30% of total retail share in China and the United Kingdom, confirming the strength of digital purchasing behavior in large consumer markets [6]. This channel momentum is reflected in category structure, with online retail accounting for 40% of distribution volume in 2026.
Subscription models further strengthen the channel by supporting recurring revenue and more accurate inventory planning. As per FMI's projection, demand for postbiotic supplements in Europe is scaling through targeted digital campaigns that connect metabolic health searches with relevant BPL1 products. Many emerging brands now use e-commerce-first launches to validate formulation acceptance before absorbing brick-and-mortar slotting costs. This shift is forcing legacy retailers to adapt category management strategies and reinforces online retail as the leading launch platform for advanced postbiotic and paraprobiotic products.
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Drivers, Restraints, and Opportunities
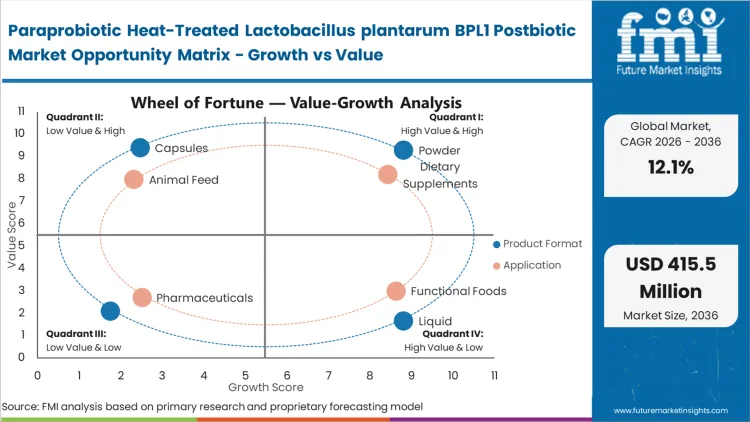
Rising metabolic disease rates are reshaping nutrition procurement strategies worldwide, as preventive health moves further into mainstream FMCG channels. Manufacturers are increasingly seeking active ingredients that can address specific metabolic markers while remaining outside prescription-drug classification frameworks. In 2025, health authorities reported that 81% of adults living with diabetes were in low- and middle-income countries, highlighting the need for scalable and accessible nutritional interventions [7]. In response, formulators are incorporating inanimate microbial biomass into everyday consumption formats. Heat-treated variants are especially attractive because they overcome many stability limitations, allowing brands to extend functional products across large, non-refrigerated distribution networks.
Regulatory compliance structures restrain rapid global expansion timelines. Health authorities strictly monitor physiological claims associated with metabolic improvements or weight management. Companies face prolonged audit periods when translating clinical trial data into consumer-facing packaging language. Brands lacking rigorous, strain-specific dossiers face immediate market exclusion or forced product recalls. Substantiation costs elevate barrier to entry for smaller manufacturers. Differing regional standards complicate single-formula global rollouts, forcing companies to maintain multiple compliance profiles and localized manufacturing partnerships to avoid border rejections.
- Format Integration Capability: Thermal stability permits active ingredient inclusion in previously inaccessible food matrices, expanding revenue potential. Fonterra collaborated with Superbrewed Food in August 2024 to advance novel protein applications, signaling broad sector acceptance [8]. Buyers capitalize on this durability to launch functional baked goods and hot beverages.
- Veterinary Applications: Livestock producers seek stable feed additives to improve herd immunity and conversion ratios without antibiotics. Agricultural conglomerates require ambient-stable postbiotic feed additives capable of surviving pelleting processes. Implementation immediately reduces livestock mortality rates and improves overall yield economics.
- Topical Formulations: Skincare brands incorporate microbial metabolites to balance epidermal flora and reduce localized inflammation. Formulators utilize postbiotic skincare derivatives to create premium anti-aging serums without risking bacterial contamination. Cosmetic manufacturers secure higher retail margins through specialized microbiome positioning.
Regional Analysis
Based on the regional analysis, the Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 15.1% |
| China | 14.5% |
| United States | 13.2% |
| Brazil | 12.7% |
| United Kingdom | 12.4% |
| Germany | 11.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
North America Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Analysis
North America exhibits high penetration of advanced nutritional formats driven by sophisticated consumer awareness. Mature dietary supplement infrastructure facilitates rapid deployment of next-generation inanimate ingredients. Strict FDA compliance parameters filter out unsubstantiated claims, leaving clear pathways for clinically validated formulas. Companies leverage massive existing user bases to upsell premium, condition-specific paraprobiotic solutions. Formulators replace live cultures with BPL1 to ensure label claim consistency, reducing legal exposure and retailer chargebacks.
- United States: Demand for paraprobiotic ingredients in United States is set to grow at 13.2% CAGR through 2036, supported by severe metabolic health burdens before procurement cycles translate into higher deployment volumes. By December 2025, 100% of states and territories reported adult obesity rates exceeding 25%, establishing a permanent baseline for preventative nutrition spending [9]. Formulators prioritize ambient-stable ingredients to scale metabolic support products across diverse retail environments.
FMI's report includes granular assessment of cross-border supply dynamics impacting North American operations. Canada and Mexico present secondary expansion targets for domestic producers seeking broader footprint. Analysts monitor regulatory harmonization efforts between FDA and Health Canada regarding gut health supplement definitions. Harmonized guidelines will reduce compliance expenditures for multinational brand owners.
Europe Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Analysis
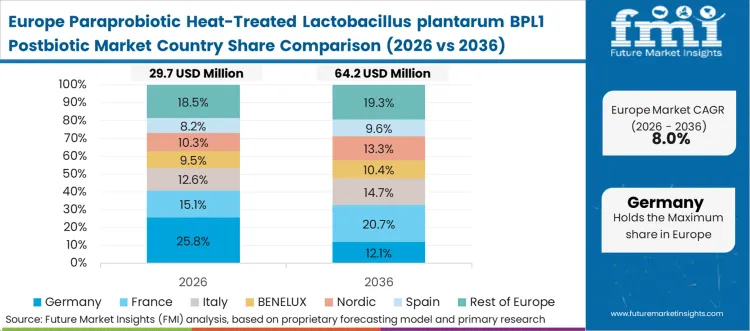
Europe represents a highly regulated environment demanding exceptional clinical substantiation prior to commercial launch. European Food Safety Authority parameters force manufacturers to invest heavily in strain-specific dossiers. According to FMI's estimates, stringent requirements eliminate opportunistic competitors, allowing established players to command significant margins. Consolidation among biotechnology leaders accelerates the commercialization of specialized metabolic interventions.
- United Kingdom: United Kingdom paraprobiotic sector is poised to expand at 12.4% CAGR during 2026 to 2036 because robust corporate investments lift utilization rates and justify technology refresh decisions. In June 2025, Danone acquired The Akkermansia Company, signaling aggressive strategic intent to dominate next-generation microbial therapeutics within the region [10]. Digital channels strongly support brands capable of direct-to-consumer shipping.
- Germany: Germany is expected to record 11.8% CAGR in paraprobiotic demand over 2026 to 2036 as evidence-based positioning tightens links between operational constraints and adoption-led investment. Pharmaceutical-grade consistency requirements drive preference for stable biomass, evidenced by peak studies showing 145,151 probiotic supplement products sold within relevant EU datasets [11]. Apoteke channels demand rigorous quality controls achievable only through thermal deactivation.
FMI's report includes detailed analysis of EFSA novel food applications shaping European commercialization. France and Italy lead adjacent research initiatives targeting microbiome therapeutics. Clearer guidelines will unlock immediate capital expenditure for regional production capacity.
Asia Pacific Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Analysis
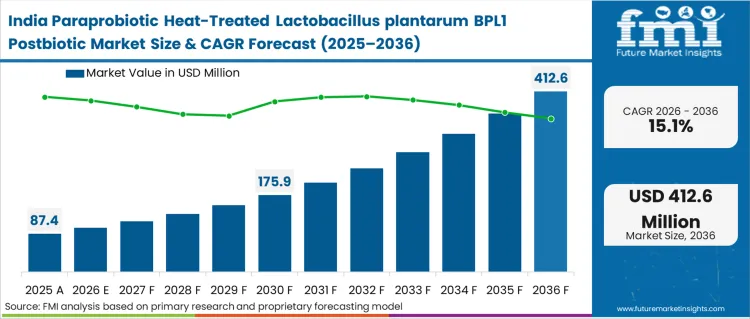
Asia Pacific exhibits explosive growth fueled by unprecedented urbanization and shifting dietary patterns. Massive population centers face severe metabolic crises, prompting government-led preventative health initiatives. Multinational ingredient suppliers aggressively target regional manufacturers to capture first-mover advantages. Localized blending facilities streamline distribution and reduce exposure to international shipping bottlenecks.
- China: In China, paraprobiotic market is forecast to advance at 14.5% CAGR through 2036, with growth anchored in mass-market functional nutrition that converts into measurable budget allocation. Domestic diabetes counts reached 148.0 million adults in 2024, creating intense pressure for accessible metabolic modulators [12]. Formulators secure large-scale ingredient contracts to feed aggressive domestic distribution networks.
- India: Paraprobiotic uptake in India is anticipated to climb at 15.1% CAGR over forecast period as premiumization in supplements helps suppliers align positioning with local buying triggers. The country recorded 89.8 million adults with diabetes during 2024, necessitating highly scalable preventative interventions [13]. Heat-treated stability allows brands to bypass fragmented cold-chain infrastructure across sub-continent logistics routes.
FMI's report includes strategic assessment of intra-regional trade partnerships accelerating commercialization. Japan and South Korea export critical fermentation technologies into emerging ASEAN production hubs. Manufacturers evaluate probiotic supplements market share analysis data to benchmark competitive positioning. Regional compliance harmonization will dictate speed of cross-border product launches.
Latin America Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Analysis
Latin America transitions toward preventative nutrition as healthcare systems face escalating chronic disease costs. Rising disposable income within urban centers fuels premium supplement acquisition. Supply chains historically struggled with live probiotic viability due to severe temperature fluctuations across regional logistics networks. Local pharmacy chains act as primary educators, driving consumer transition from basic vitamins to advanced metabolic modulators.
- Brazil: Paraprobiotic sales in Brazil are projected to rise at 12.7% CAGR from 2026 to 2036 as localized distribution agreements create clearer pathways from policy intent to purchase orders. In February 2025, ADM signed an exclusive agreement with Asahi Group Foods to market clinically supported postbiotics globally, directly enhancing Latin American supply reliability [14]. Warm supply chains mandate heat-stable inputs to prevent complete ingredient degradation prior to retail shelving.
FMI's report includes comprehensive evaluation of distribution bottlenecks impacting Latin American supply. Argentina and Chile represent logical next steps for regional brand expansion following successful Brazilian deployment. Currency volatility mandates localized blending facilities to hedge against animal intestinal health import costs. Domestic manufacturing capacity will dictate long-term competitive positioning.
Competitive Aligners for Market Players
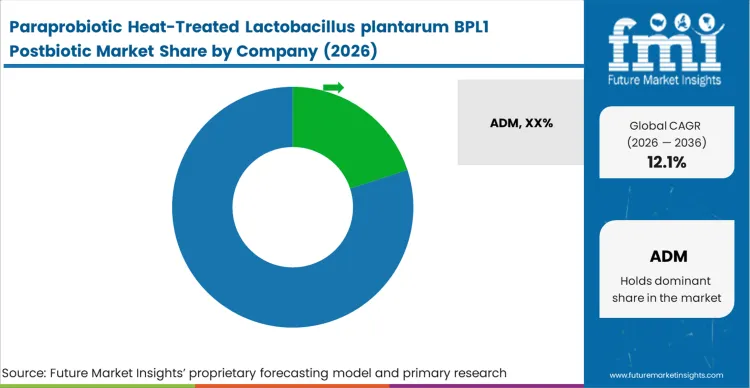
Distribution partnerships serve as primary expansion vehicles for proprietary strain owners. Companies lacking global sales infrastructure partner with established chemical and ingredient distributors to penetrate fragmented regional territories. In May 2025, BON Natural Life signed an exclusive cooperation agreement valued at USD 32 million for postbiotic hypoglycemic ingredients, demonstrating the massive capital deployed to secure regional market share [15]. Distributors handle localized regulatory compliance and import logistics, allowing researchers to focus entirely on strain discovery and clinical validation.
Corporate consolidation reshapes global supply capacity and research bandwidth. Mega-mergers create entities capable of dominating raw material production and patent portfolios. In January 2024, Novozymes and Chr. Hansen completed their combination to form Novonesis, creating an unprecedented biosolutions platform that structurally alters microbial ingredient supply chains [16]. FMI analysts opine that consolidated entities leverage vast distribution networks to push proprietary strains into localized manufacturing facilities, effectively boxing out smaller, unintegrated competitors.
Format innovation dictates immediate commercial success within saturated consumer markets. Ingredient suppliers must engineer paraprobiotics capable of surviving extreme manufacturing stressors associated with novel delivery systems. Suppliers capable of providing comprehensive formulation support and stability data capture dominant market share among contract manufacturers. Brands rapidly adopt advanced delivery formats to differentiate product lines and maintain premium retail pricing power.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In June 2025, dsm-firmenich launched GutServ Biotics postbiotic solution to provide heat-tolerant operational flexibility [2].
- In June 2025, Danone acquired The Akkermansia Company to strategically invest in next-generation microbial technologies [10].
Key Players in Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic
- ADM
- dsm-firmenich
- Novonesis
- Lallemand Health Solutions
- Sabinsa
- Danone
- Sirio Pharma
- BON Natural Life
- Postbiotics Inc.
- HealthBiome
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 132.6 million (2026) to USD 412.9 million (2036), at a CAGR of 12.1% |
| Market Definition | Industry encompasses non-viable, thermally processed microbial biomass utilized across commercial nutrition and health applications. |
| Product Format Segmentation | Powder, Liquid, Capsules, Gummies |
| Application Segmentation | Dietary Supplements, Functional Foods, Animal Feed, Pharmaceuticals |
| Application Coverage | Dietary Supplements, Functional Foods, Animal Feed, Pharmaceuticals |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40 plus countries |
| Key Companies Profiled | ADM, dsm-firmenich, Novonesis, Lallemand Health Solutions, Sabinsa, Danone, Sirio Pharma, BON Natural Life, Postbiotics Inc., HealthBiome |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with ingredient suppliers, supported by trade data benchmarking |
Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic Market Analysis by Segments
Product Format:
- Powder
- Liquid
- Capsules
- Gummies
Application:
- Dietary Supplements
- Functional Foods
- Animal Feed
- Pharmaceuticals
Consumer Group:
- Adults
- Pediatrics
- Geriatrics
Distribution Channel:
- Online Retail
- Pharmacies
- Supermarkets
- Specialty Stores
Region:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- [1] CDC, 40.3% adult obesity prevalence in United States, Sep 2024.
- [2] dsm-firmenich, Press Release on GutServ Biotics heat tolerance, Jun 2025.
- [3] Nutraceuticals World, Sirio Pharma launches BloomDays postbiotic gummies with EpiCor, Feb 2024.
- [4] PubMed, 61.4% overall dietary supplement use among adults in 2021-2023, 2025.
- [5] Diabetes Atlas, 589 million adults living with diabetes globally, 2025.
- [6] UNCTAD, 25-30% online retail share in China and UK, Jun 2024.
- [7] Diabetes Atlas, 81% of adults with diabetes live in LMICs, 2025.
- [8] AgFunderNews, Fonterra and Superbrewed Food collaboration on postbiotic protein, Aug 2024.
- [9] CDC, 100% of US states and territories report 25% or higher adult obesity, Dec 2025.
- [10] NutraIngredients.com, Danone acquires The Akkermansia Company, Jun 2025.
- [11] PMC, 145,151 probiotic supplement products sold in EU dataset, 2024.
- [12] Diabetes Atlas, 148.0 million adults with diabetes in China, 2025.
- [13] Diabetes Atlas, 89.8 million adults with diabetes in India, 2025.
- [14] ADM, Exclusive distribution agreement with Asahi Group Foods, Feb 2025.
- [15] BioSpace, BON Natural Life USD 32 million cooperation agreement for hypoglycemic ingredients, May 2025.
- [16] Investegate, Combination of Novozymes and Chr. Hansen into Novonesis completed, Jan 2024.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence to enable structured strategic decision making across mature and emerging nutritional ingredient economies
- Market size estimation and 10 year revenue forecasts from 2026 to 2036, supported by validated capacity and trade benchmarks
- Growth opportunity mapping across powder, liquid, and encapsulated forms with emphasis on heat-treated transition dynamics
- Segment and regional revenue forecasts covering dietary supplements, functional foods, and pharmaceutical applications
- Competition strategy assessment including integration models, cost survivability positioning, and compliance capability benchmarking
- Product reformulation tracking including EFSA and FDA aligned regulatory grades
- Regulatory impact analysis covering metabolic claims mandates and cross border ingredient specifications
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic in the global market in 2026?
Demand is estimated to be valued at USD 132.6 million in 2026.
What will be the market size of Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic in the global market by 2036?
Market size is projected to reach USD 412.9 million by 2036.
What is the expected demand growth for Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic in the global market between 2026 and 2036?
Demand is expected to grow at a CAGR of 12.1% between 2026 and 2036.
Which Product Format is poised to lead global sales by 2026?
Powder is expected to be dominant, capturing 44% share due to its superior stability against moisture degradation during manufacturing.
How significant is the role of Dietary Supplements in driving Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic adoption in 2026?
Dietary supplements represent 39% share, driven by a structural pivot toward preventative metabolic healthcare.
What is driving demand in India?
Unprecedented diabetes prevalence and rapid expansion of preventative supplement channels drive immediate deployment.
What compliance standards or regulations are referenced for United States?
FDA compliance parameters regarding specific metabolic health claims act as primary regulatory benchmarks.
What is the United States growth outlook in this report?
United States is projected to grow at a CAGR of 13.2% during 2026 to 2036.
Why is Europe described as a priority region in this report?
Stringent EFSA clinical substantiation requirements eliminate low-quality alternatives, creating high-margin environments for validated strains.
What type of demand dominates in Europe?
Evidence-based positioning targeting apoteke channels and clinical formulations dictates regional purchasing behavior.
What is United Kingdom growth outlook in this report?
United Kingdom is projected to expand at a CAGR of 12.4% during 2026 to 2036.
Does the report cover Germany in its regional analysis?
Yes, Germany is included within Europe.
What are the sources referred to for analyzing Germany?
PMC dataset studies tracking probiotic sales trajectories inform the regional analysis.
What is the main demand theme linked to Germany in its region coverage?
Strict pharmaceutical-grade consistency requirements drive preference for stable, thermally deactivated biomass.
Does the report cover China in its regional analysis?
Yes, China is included within Asia Pacific.
What is the main China related demand theme in its region coverage?
Mass-market functional nutrition expansion targeting immense domestic metabolic disease populations dictates strategy.
Which product formats or configurations are strategically important for Latin America supply chains?
Powder formulations remain critical due to severe ambient temperature fluctuations across local logistics networks.
What is Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic and what is it mainly used for?
It is an inanimate microbial biomass utilized primarily in supplements and functional foods to support metabolic health without cold-chain requirements.
What does Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic mean in this report?
It refers to commercial trade and utilization of thermally deactivated BPL1 bacterial cells and associated cellular fragments.
What is included in the scope of this Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic report?
Scope includes heat-killed biomass, tyndallized fragments, and standardized powders intended for nutritional applications.
What is excluded from the scope of this report?
Live probiotic cultures, unrelated bacterial strains, and basic commodity dietary fibers are excluded from measurement.
What does market forecast mean on this page?
Market forecast represents a model-based projection built on defined demographic and capacity assumptions for strategic planning.
How does FMI build and validate the Paraprobiotic Heat-Treated Lactobacillus plantarum BPL1 Postbiotic forecast?
Forecast relies on hybrid bottom-up capacity modeling validated through primary interviews and top-down disease prevalence statistics.
What does zero reliance on speculative third party market research mean here?
Primary interviews and verifiable public datasets dictate modeling, explicitly excluding unverified syndicated market estimates.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Format , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Format , 2026 to 2036
- Powder
- Liquid
- Capsules
- Gummies
- Powder
- Y to o to Y Growth Trend Analysis By Product Format , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Format , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Dietary Supplements
- Functional Foods
- Animal Feed
- Pharmaceuticals
- Dietary Supplements
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Consumer Group
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Consumer Group, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Consumer Group, 2026 to 2036
- Adults
- Pediatrics
- Geriatrics
- Adults
- Y to o to Y Growth Trend Analysis By Consumer Group, 2021 to 2025
- Absolute $ Opportunity Analysis By Consumer Group, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Online Retail
- Pharmacies
- Supermarkets
- Specialty Stores
- Online Retail
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Format
- By Application
- By Consumer Group
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- ADM
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- dsm-firmenich
- Novonesis
- Lallemand Health Solutions
- Sabinsa
- Danone
- Sirio Pharma
- BON Natural Life
- Postbiotics Inc.
- ADM
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Format , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Product Format , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Product Format , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Product Format , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Product Format , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Product Format , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Product Format , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Product Format , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Format , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Format , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Format
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Consumer Group
- Figure 12: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Distribution Channel
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Product Format , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Product Format , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Product Format
- Figure 29: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Application
- Figure 32: North America Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Consumer Group
- Figure 35: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Distribution Channel
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Product Format , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Product Format , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Product Format
- Figure 42: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Application
- Figure 45: Latin America Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Consumer Group
- Figure 48: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Product Format , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Product Format , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Product Format
- Figure 55: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Application
- Figure 58: Western Europe Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Consumer Group
- Figure 61: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Product Format , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Product Format , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Product Format
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Application
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Consumer Group
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Product Format , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Product Format , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Product Format
- Figure 81: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Application
- Figure 84: East Asia Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Consumer Group
- Figure 87: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Product Format , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Format , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Product Format
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Consumer Group
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Product Format , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Product Format , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Product Format
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Consumer Group
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis

