Demand for Postbiotic Supplements in EU
This report delivers insights into the Demand for Postbiotic Supplements in EU through market size analysis, revenue forecasting, competitor evaluation, demand assessment, growth opportunities, industry restraints, nutraceutical developments, supply chain trends, and strategic recommendations.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Demand for Postbiotic Supplements in EU Size, Market Forecast and Outlook By FMI
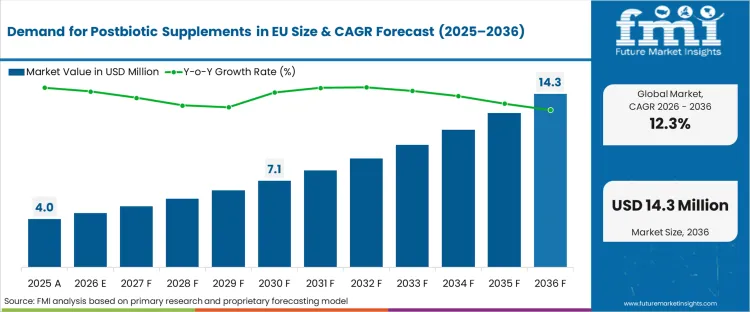
The demand for postbiotic supplements in eu was valued at USD 1.7 million in 2025, projected to reach USD 4.5 million in 2026, and is forecast to expand to USD 14.3 million by 2036 at a 12.3% CAGR. The absolute dollar expansion over the forecast period, at 12.3% CAGR from a small USD 4.5 million base, reflects the early-stage growth dynamics of a category where consumer awareness is low but clinical evidence is accumulating rapidly. Capsules and tablets lead product form at 46% share, as the most familiar supplement delivery format for consumers accustomed to probiotic capsule purchasing. Drug stores dominate distribution at 67% share, reflecting the pharmacist-mediated purchasing pathway that characterizes early-stage health supplement categories before mainstream retail adoption.
As per FMI, postbiotic supplements operate in a regulatory grey zone in the EU where the term postbiotic lacks a formal definition under food supplement or health claim regulations. The International Scientific Association for Probiotics and Prebiotics (ISAPP) defined postbiotics in 2021 as preparations of inanimate microorganisms and/or their components that confer a health benefit on the host. EU manufacturers must navigate this definitional gap by marketing postbiotic products under existing food supplement frameworks (Directive 2002/46/EC) without making unauthorized health claims.
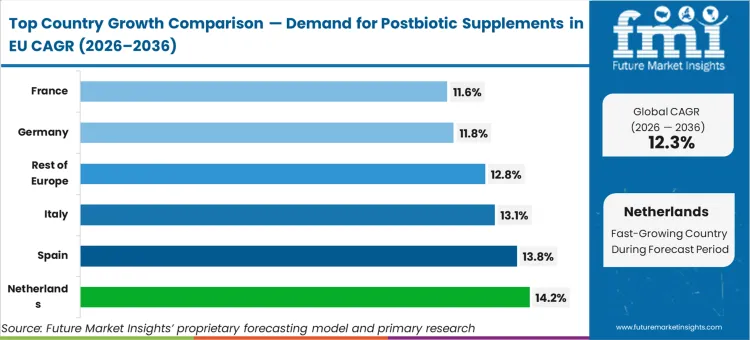
All major EU markets reflect differentiated growth. Netherlands at 14.2%, Spain at 13.8%, Italy at 13.1%, Rest of Europe at 12.8%, Germany at 11.8%, France at 11.6%. Netherlands sets the pace, while France registers steadier growth.
Summary of Demand for Postbiotic Supplements in EU
- Demand for Postbiotic Supplements in EU Definition
- Postbiotic supplements are dietary supplement products containing preparations of inanimate (non-living) microorganisms and/or their cellular components and metabolites that confer health benefits. Unlike probiotics, postbiotics do not contain live organisms, providing shelf stability, heat resistance, and formulation versatility.
- Demand Drivers in the Market
- Stability advantage over live probiotics: Postbiotic preparations do not require refrigeration, maintain consistent potency throughout shelf life, and are compatible with food processing conditions (heat, acidity) that kill live probiotic organisms.
- Clinical evidence accumulation: Published clinical trials on heat-killed Lactobacillus paracasei, Lactobacillus plantarum, and Bifidobacterium preparations are demonstrating gut barrier improvement, immune modulation, and metabolic benefits that build the evidence base for EFSA health claim submissions and healthcare professional recommendation.
- Pharmacist and healthcare professional recommendation channel: Drug stores and pharmacies account for 67% of EU postbiotic supplement distribution, reflecting the healthcare professional mediation that characterizes early-stage supplement categories.
- Key Segments Analyzed in the FMI Report
- Drug Stores: 67.0% share in 2026 by Distribution Channel.
- Capsules & Tablets: 46.0% share in 2026 by Product Form.
- Netherlands: 14.2% compound growth, the fastest among tracked EU markets.
- Analyst Opinion at FMI
- Nandini Roy Choudhury, Principal Consultant for Food & Beverage analysts observe that, the EU postbiotic supplements market is the smallest category in this report by absolute value but represents the highest-conviction emerging opportunity for supplement brands willing to invest ahead of the regulatory curve. The category will inflect when EFSA authorizes the first postbiotic-specific health claim, which will trigger mainstream retail adoption and consumer awareness growth that the current pharmacist-mediated channel cannot deliver at scale. Brands investing now in clinical trial sponsorship, EFSA dossier preparation, and pharmacist education programmes are building the foundation for first-mover advantage when the regulatory authorization event occurs.
- Strategic Implications / Executive Takeaways
- Procurement teams must map EU regulatory pathways as classification, labelling, and health claim rules vary by product type and application.
- Product development should evaluate premium positioning opportunities where quality differentiation captures higher retail pricing.
- Supply chain managers must diversify sourcing and maintain inventory buffers to mitigate origin-concentration and seasonal risks.
Demand for Postbiotic Supplements in EU Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 4.5 million |
| Industry Value (2036) | USD 14.3 million |
| CAGR (2026 to 2036) | 12.3% |
Source: Future Market Insights, 2026
Demand for Postbiotic Supplements in EU Definition
Postbiotic supplements are dietary supplement products containing preparations of inanimate (non-living) microorganisms and/or their cellular components and metabolites that confer health benefits. Unlike probiotics, postbiotics do not contain live organisms, providing shelf stability, heat resistance, and formulation versatility. Products are supplied in capsule, tablet, soft-gel, chew, powder, and granule formats.
Demand for Postbiotic Supplements in EU Inclusions
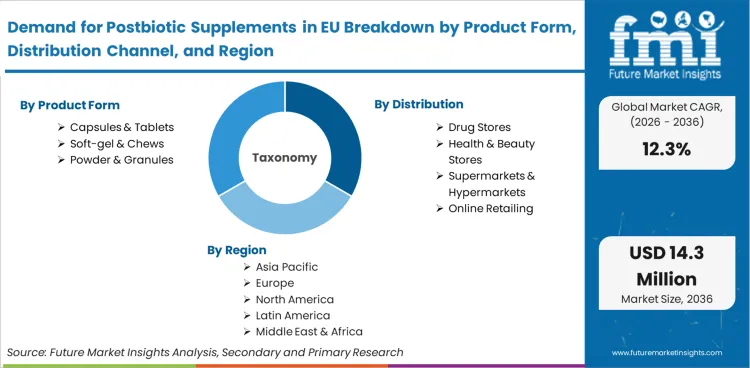
The report covers EU regional market sizes for postbiotic supplements from 2026 to 2036. Segment analysis includes product form (capsules and tablets, soft-gel and chews, powder and granules) and distribution channel (drug stores, health and beauty stores, supermarkets and hypermarkets, online retailing). Country coverage includes Netherlands, Spain, Italy, Germany, France, and rest of Europe.
Demand for Postbiotic Supplements in EU Exclusions
The scope excludes live probiotic supplements and prebiotic fibre supplements as separate categories. Postbiotic ingredients sold as bulk raw materials to supplement manufacturers are excluded at the B2B ingredient level. Fermented foods (yoghurt, kefir, kimchi) containing postbiotic compounds as part of the whole food matrix fall outside the analytical boundary.
Demand for Postbiotic Supplements in EU Research Methodology
- Primary Research: Structured interviews with procurement directors, formulation engineers, and regulatory managers.
- Desk Research: Aggregated EU regulatory filings, trade publications, company reports, and Eurostat data.
- Market-Sizing and Forecasting: Bottom-up aggregation of production, trade, and consumption with country-level pricing adjustments.
- Data Validation: Cross-validated against company disclosures and EU trade statistics.
Why is the Demand for Postbiotic Supplements in EU Growing
Industry expansion is being supported by the rapid advancement in microbiome science across European research institutions and the corresponding demand for next-generation gut health solutions that overcome traditional probiotic limitations including stability challenges, colonization uncertainty, and manufacturing complexity. Modern consumers increasingly understand that beneficial effects of probiotics often derive from their metabolites rather than live bacteria, driving demand for postbiotic products that deliver consistent bioactive compounds without viability concerns, standardized therapeutic molecules with batch-to-batch consistency, and enhanced safety profiles suitable for immunocompromised populations unable to consume live microorganisms.
The growing body of mechanistic research elucidating postbiotic modes of action, including short-chain fatty acid production, antimicrobial peptide activity, and immune modulation pathways, is driving demand for scientifically-substantiated postbiotic supplements from manufacturers with appropriate research credentials and quality standards. Regulatory authorities are increasingly recognizing postbiotics as distinct supplement category requiring specific guidelines for characterization, safety assessment, and efficacy substantiation to ensure consumer protection and product quality. Scientific publications and clinical trials are providing evidence supporting postbiotic benefits in gut barrier function, inflammatory regulation, and pathogen inhibition, requiring specialized production methods and analytical protocols for metabolite identification, potency standardization, and stability optimization throughout shelf life.
Segmental Analysis
Sales are segmented by product form, distribution channel, and country. By product form, demand is divided into soft-gel & chews, capsules & tablets, and powder & granules. Based on distribution channel, sales are categorized into drug stores, health & beauty stores, supermarkets & hypermarkets, and online retailing. Regionally, demand is focused on Germany, France, Italy, Spain, the Netherlands, and the Rest of Europe.
By Product Form, Capsules & Tablets Segment Accounts for 46.0% Share
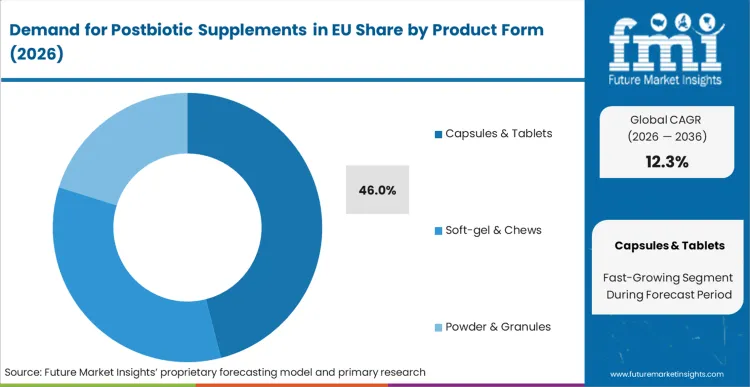
The capsules & tablets segment is projected to account for 46.0% of EU postbiotic supplements sales in 2025, declining slightly to 42.0% by 2035, establishing itself as the preferred format across pharmaceutical and nutraceutical channels. This commanding position is fundamentally supported by capsules' superior protection of sensitive metabolites, convenient dosing for daily supplementation, and established consumer familiarity from traditional supplement consumption. The capsule format delivers exceptional versatility, providing manufacturers with reliable encapsulation technologies that preserve postbiotic activity, enable targeted intestinal delivery, and facilitate combination formulations essential for comprehensive gut health protocols.
This segment benefits from advanced encapsulation technologies including enteric coating, delayed-release mechanisms, and moisture-barrier systems that maintain postbiotic stability throughout distribution and storage. The capsules & tablets offer advantages in precise dosing standardization, quality control implementation, and regulatory compliance, supported by established pharmaceutical manufacturing infrastructure that ensures consistent product quality and therapeutic efficacy.
By Distribution Channel, Drug Stores Account for 67.0% Share
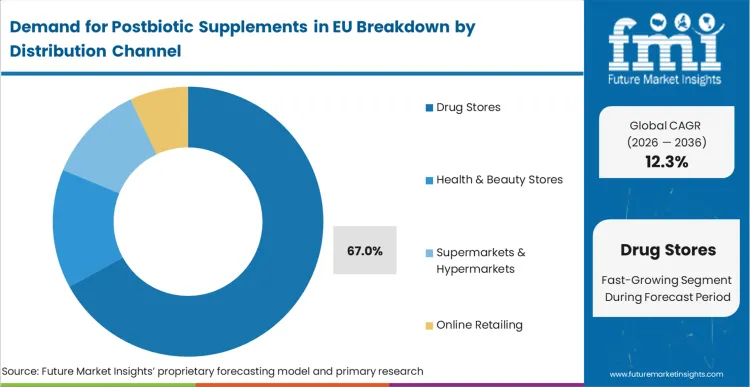
Drug stores (pharmacies) are positioned to represent 67.0% of total postbiotic supplements demand across European operations in 2025, moderating to 59.0% by 2035, reflecting the channel's dominance as the primary access point for health supplements requiring professional guidance. This considerable share directly demonstrates that drug stores represent the trusted distribution channel, with consumers preferring pharmacy purchases for novel supplement categories where pharmacist consultation provides valuable education, product selection assistance, and safety assurance for emerging ingredients like postbiotics.
European pharmacies increasingly position postbiotics within comprehensive gut health categories, driving demand through professional recommendation systems emphasizing evidence-based products, quality certification from reputable manufacturers, and integration with conventional digestive health treatments. The segment benefits from pharmacist education programs sponsored by manufacturers, scientific literature distribution supporting product credibility, and point-of-sale materials explaining postbiotic mechanisms and benefits to curious consumers.
What are the Drivers, Restraints, and Key Trends
EU postbiotic supplements sales are advancing rapidly due to increasing scientific validation of metabolite-based therapeutics, growing consumer awareness of microbiome health importance, and rising demand for stable alternatives to probiotics. The industry faces challenges, including limited consumer understanding of postbiotic concepts requiring extensive education, higher production costs for standardized metabolite preparations, and regulatory uncertainties regarding health claims and category definitions. Continued innovation in production technologies and clinical validation remains central to industry development.
Advancement of Metabolite Standardization Technologies
The rapidly accelerating development of metabolite standardization technologies is fundamentally transforming postbiotic supplements from variable fermentation products to precisely characterized therapeutic preparations, enabling consistent potency previously unattainable through traditional fermentation approaches. Advanced analytical platforms featuring mass spectrometry, metabolomics profiling, and bioactivity assays allow manufacturers to create postbiotic products with defined metabolite compositions, verified biological activity, and batch-to-batch consistency comparable to pharmaceutical standards. These standardization innovations prove particularly transformative for clinical applications, healthcare practitioner adoption, and regulatory approval processes, where reproducible composition directly determines therapeutic reliability.
Major postbiotic manufacturers invest heavily in analytical method development, metabolite library establishment, and quality control systems, recognizing that standardization represents breakthrough solutions for efficacy variability challenges limiting category expansion. Manufacturers collaborate with analytical laboratories, research institutions, and regulatory consultants to develop validated methods that characterize postbiotic composition while maintaining commercial viability and scalability supporting market growth.
Integration of Precision Fermentation Technologies
Modern postbiotic producers systematically implement precision fermentation technologies, including controlled bioreactor systems, optimized growth media, and targeted metabolite production that deliver specific bioactive compounds, enhanced therapeutic potency, and reduced production costs compared to traditional fermentation methods. Strategic integration of precision fermentation enables manufacturers to produce designer postbiotics with tailored metabolite profiles where specificity directly determines health benefits and clinical applications. These production improvements prove essential for pharmaceutical development, as drug companies demand consistent composition, validated production processes, and scalable manufacturing supporting commercial requirements.
Companies implement extensive fermentation optimization programs, strain selection protocols targeting specific metabolite production, and downstream processing innovations maximizing yield and purity, including filtration technologies, concentration methods, and stabilization processes. Manufacturers leverage precision fermentation advantages in scientific communications, highlighting controlled production environments, sustainability benefits through reduced resource consumption, and quality advantages positioning precision-fermented postbiotics as superior alternatives to traditional preparations.
Growing Emphasis on Condition-Specific Formulations
European supplement manufacturers increasingly develop condition-specific postbiotic formulations targeting particular health conditions through tailored metabolite combinations, synergistic ingredient additions, and clinically-validated protocols that differentiate products through therapeutic specificity. This specialization trend enables manufacturers to command premium pricing through targeted efficacy claims, create defensible market positions through proprietary formulations, and build brand loyalty among consumers seeking solutions for specific health concerns. Condition-specific development proves particularly important for healthcare practitioner channels where evidence-based protocols drive recommendation patterns and patient outcomes determine continued usage.
The development of targeted postbiotic systems, including formulations for irritable bowel syndrome, immune support protocols, and metabolic health combinations expands manufacturers' abilities to address diverse consumer needs delivering measurable health improvements without broad-spectrum approaches. Brands collaborate with clinical researchers, healthcare practitioners, and patient organizations to develop formulations balancing therapeutic efficacy with safety profiles, supporting medical positioning and professional endorsement while maintaining supplement regulatory status.
Europe Market Split by Country
EU postbiotic supplements sales are projected to grow from USD 4.5 million in 2026 to USD 14.3 million by 2036, registering a robust CAGR of 12.3% over the forecast period. The Netherlands is expected to demonstrate the strongest growth trajectory with a 14.2% CAGR, supported by innovative biotechnology sector, advanced microbiome research infrastructure, and early adoption of novel supplement categories. Spain follows with 13.8% CAGR, attributed to rapidly expanding wellness market and increasing health consciousness driving supplement adoption.
Germany maintains the largest share at 37.5% in 2026, driven by established pharmaceutical infrastructure and strong practitioner channels, while growing at 11.8% CAGR. France follows with 37.5% share and 11.6% CAGR growth reflecting market maturity. Italy demonstrates 13.1% CAGR, benefiting from growing wellness culture and pharmacy channel strength.
Demand Analysis by Key Countries
.webp)
| Country | CAGR % |
|---|---|
| Netherlands | 14.2% |
| Spain | 13.8% |
| Italy | 13.1% |
| Rest of Europe | 12.8% |
| Germany | 11.8% |
| France | 11.6% |
EU postbiotic supplements sales demonstrate differentiated growth trajectories across major European economies, with emerging markets significantly outpacing established ones through 2036, driven by varying market maturity levels, healthcare system dynamics, and consumer education stages. Netherlands shows exceptional growth from USD 0.2 million in 2026 to USD 0.7 million by 2036 at 14.2% CAGR. Spain expands rapidly from USD 0.2 million to USD 0.7 million at 13.8% CAGR. Italy records strong growth from USD 0.3 million to USD 1.1 million at 13.1% CAGR. Rest of Europe demonstrates USD 0.3 million to USD 1.1 million at 12.8% CAGR. Germany shows steady expansion from USD 1.5 million to USD 4.6 million at 11.8% CAGR. France maintains consistent growth from USD 1.5 million to USD 4.6 million at 11.6% CAGR. Overall, sales show varied regional development reflecting different healthcare practitioner adoption rates, consumer education levels, and distribution channel evolution across European countries.
Germany Maintains Leadership with Established Infrastructure
Revenue from postbiotic supplements in Germany is projected to exhibit steady growth with a CAGR of 11.8% through 2036, driven by exceptionally well-developed pharmaceutical distribution networks, comprehensive healthcare practitioner education programs, and strong consumer trust in pharmacy-recommended supplements throughout the country. Germany's sophisticated understanding of microbiome science and internationally recognized leadership in biotechnology research are creating substantial demand for evidence-based postbiotic products across therapeutic applications.
Major pharmacy chains, including DocMorris, Europa Apotheek, and traditional Apotheken networks, systematically expand postbiotic selections, often positioning them prominently within digestive health categories and supporting sales through pharmacist education and recommendation protocols. German demand benefits from high healthcare expenditure, substantial insurance coverage for preventive health products, and cultural acceptance of supplement usage that naturally supports postbiotic adoption across mainstream consumers seeking digestive wellness solutions.
The relatively moderate growth rate reflects Germany's mature supplement market where established probiotic usage creates both opportunity and competition for postbiotic positioning, requiring extensive education to differentiate metabolite-based benefits from traditional live bacteria supplements.
Growth drivers:
- Pharmaceutical infrastructure supporting professional distribution
- Healthcare practitioner influence driving evidence-based adoption
- Insurance coverage potential expanding accessibility
France Demonstrates Steady Expansion with Pharmacy Dominance
Revenue from postbiotic supplements in France is expanding at a CAGR of 11.6%, supported by strong pharmacy distribution networks, growing microbiome awareness among healthcare professionals, and increasing consumer sophistication regarding gut health supplementation. France's centralized healthcare system and pharmacy-centric supplement distribution create unique market dynamics favoring professionally-endorsed products with scientific substantiation like postbiotics.
Major pharmacy cooperatives, including Groupement Pharmaceutique de l'Union Européenne and Alliance Healthcare France, facilitate postbiotic distribution through comprehensive pharmacy networks reaching urban and rural populations. French sales particularly benefit from medical community engagement, with gastroenterologists and general practitioners increasingly recognizing postbiotic benefits for patients unable to tolerate live probiotics. Consumer education initiatives through pharmacy consultations and health magazines significantly enhance understanding of postbiotic mechanisms and advantages over traditional probiotics.
The moderate growth rate reflects France's established probiotic market requiring careful positioning to differentiate postbiotics while avoiding confusion with existing gut health supplements consumers already understand and utilize regularly.
Success factors:
- Pharmacy channel dominance ensuring professional credibility
- Medical community engagement supporting clinical adoption
- Consumer sophistication enabling advanced supplement acceptance
Italy Achieves Above-Average Growth Through Wellness Integration
Revenue from postbiotic supplements in Italy is growing at a robust CAGR of 13.1%, fundamentally driven by expanding wellness consciousness, strong pharmacy tradition supporting supplement recommendations, and growing integration of gut health into holistic wellness approaches. Italy's evolving healthcare landscape increasingly emphasizes preventive nutrition, creating favorable conditions for innovative supplement categories addressing fundamental health through microbiome optimization.
Major Italian pharmacy chains, including Farmacia Loreto Gallo, Hippocrates Holding, and independent farmacias, actively promote postbiotic supplements through professional consultation services and educational initiatives explaining metabolite benefits. Italian sales particularly benefit from Mediterranean diet culture emphasizing fermented foods, creating natural consumer receptivity to fermentation-derived health products. The country's growing sports nutrition sector also drives postbiotic adoption for performance and recovery applications beyond traditional digestive health positioning.
Development factors:
- Wellness culture evolution driving preventive supplementation
- Pharmacy tradition supporting professional supplement guidance
- Mediterranean heritage enhancing fermentation product acceptance
Spain Shows Rapid Market Development from Low Base
Demand for postbiotic supplements in Spain is projected to grow at an impressive CAGR of 13.8%, substantially supported by rapidly evolving health consciousness, expanding distribution beyond traditional pharmacies, and increasing consumer education about microbiome health importance. Spanish market transformation from traditional medicine toward preventive supplementation positions postbiotics as aligned with contemporary wellness trends, particularly among younger demographics embracing scientific nutrition approaches.
Major Spanish retailers, including El Corte Inglés pharmacy sections, Farmacias Trébol network, and emerging online platforms like PromoFarma, systematically introduce postbiotic offerings with competitive pricing and consumer education supporting category development. Spain's growing fitness culture and increasing inflammatory bowel disease prevalence drive specific postbiotic applications for gut health restoration and maintenance. The country's relatively low baseline enables rapid growth as consumer awareness increases and distribution expands beyond specialized channels.
Growth enablers:
- Health consciousness acceleration from traditional baseline
- Distribution expansion improving product accessibility
- Younger demographic receptivity to innovation
Netherlands Leads Growth Through Innovation Excellence
Demand for postbiotic supplements in the Netherlands is expanding at a leading CAGR of 14.2%, fundamentally driven by exceptional microbiome research capabilities, progressive regulatory environment for novel ingredients, and sophisticated consumer base with high scientific literacy regarding gut health. Dutch companies demonstrate particular strength in fermentation technology and metabolite characterization, positioning the Netherlands as European innovation hub for advanced postbiotic development.
Netherlands sales significantly benefit from world-class research institutions, including Wageningen University's microbiome research center and TNO's gut health programs, conducting cutting-edge postbiotic studies advancing scientific understanding. Dutch consumers' early adoption tendencies and willingness to try innovative health products support rapid market penetration for next-generation postbiotic formulations, particularly those featuring validated metabolite profiles or novel delivery systems. The country's leadership in precision fermentation and biotechnology creates unique advantages for local postbiotic producers developing proprietary strains and production methods.
Innovation drivers:
- Microbiome research excellence advancing scientific validation
- Biotechnology capabilities enabling advanced production
- Early adopter consumers driving premium innovation uptake
Rest of Europe Demonstrates Emerging Market Potential
Revenue from postbiotic supplements in Rest of Europe is expanding at 12.8% CAGR, representing diverse smaller markets including Belgium, Austria, Scandinavian countries, and Eastern European nations experiencing varied development stages. These markets collectively demonstrate growing potential as consumer education increases, distribution channels develop, and regulatory frameworks clarify supporting postbiotic commercialization across previously underserved regions.
Emerging European markets benefit from increasing health consciousness, expanding modern retail infrastructure, and growing access to international supplement brands introducing postbiotic products. Nordic countries particularly show strong adoption potential given high health awareness and supplement usage rates, while Eastern European markets represent longer-term opportunities as economic development enables premium supplement purchases.
Competitive Landscape
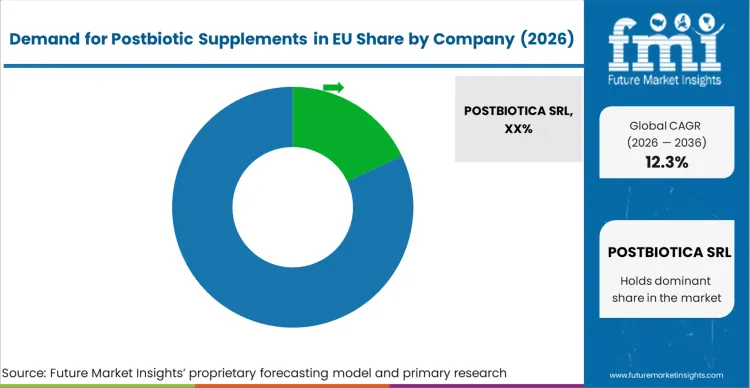
EU postbiotic supplements sales are defined by competition among specialized postbiotic companies, established probiotic manufacturers, and emerging biotechnology firms. Companies are investing in metabolite characterization, clinical validation, production optimization, and educational initiatives to deliver high-quality, scientifically-substantiated, and commercially viable postbiotic solutions. Strategic partnerships with research institutions, healthcare practitioners, and distribution networks emphasizing scientific credibility and professional endorsement are central to strengthening competitive position.
Major participants include POSTBIOTICA SRL with an estimated 18.0% share, leveraging its specialized focus on postbiotic development, European headquarters location, and comprehensive research program through proprietary technology platforms. POSTBIOTICA benefits from first-mover advantages, scientific publication record, and established relationships with European distributors supporting market penetration.
Designs for Health holds approximately 12.0% share, emphasizing practitioner channel expertise, evidence-based formulation approach, and professional education programs supporting healthcare provider engagement. Designs for Health's success in developing clinically-oriented postbiotic products with detailed protocols creates strong positioning among healthcare practitioners, supported by educational resources and scientific substantiation materials.
Other companies collectively hold 70.0% share, reflecting the nascent nature of European postbiotic supplements sales, where numerous specialized manufacturers, probiotic companies expanding into postbiotics, ingredient suppliers developing proprietary metabolites, and emerging startups serve specific applications, distribution channels, and consumer segments. This competitive environment provides opportunities for differentiation through unique metabolite profiles, condition-specific formulations, innovative delivery systems, and scientific validation supporting therapeutic positioning.
Key Players
- POSTBIOTICA SRL
- Designs for Health
- Cerbios-Pharma SA
- ADM Protexin
- Morinaga Milk Industry Co., Ltd.
- Mycology Research Laboratories
- Deerland Probiotics & Enzymes
- Sabinsa Corporation
- IFF Health
- Chr. Hansen
- Others
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 4.5 million to USD 14.3 million, CAGR 12.3% |
| Market Definition | Postbiotic supplements are dietary supplement products containing preparations of inanimate (non-living) microorganisms and/or their cellular components and metabolites that confer health benefits. Unlike probiotics, postbiotics do not contain live organisms, providing shelf stability, heat resistance, and formulation versatility. |
| Segmentation | Product Form (Capsules & Tablets, Soft-gel & Chews, Powder & Granules), Distribution Channel (Drug Stores, Health & Beauty Stores, Supermarkets & Hypermarkets, Online Retailing) |
| Regions Covered | Europe (EU member states) |
| Countries Covered | Netherlands, Spain, Italy, Rest of Europe, Germany, France |
| Key Companies Profiled | POSTBIOTICA SRL, Designs for Health, Cerbios-Pharma SA, ADM Protexin, Morinaga Milk Industry Co., Ltd., Mycology Research Laboratories, Deerland Probiotics & Enzymes, Sabinsa Corporation, IFF Health, Chr. Hansen |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up methodology using EU production, trade, and consumption data. |
Key Segments
Product Form
- Capsules & Tablets
- Soft-gel & Chews
- Powder & Granules
Distribution Channel
- Drug Stores
- Health & Beauty Stores
- Supermarkets & Hypermarkets
- Online Retailing
Countries
- Germany
- France
- Italy
- Spain
- Netherlands
- Rest of Europe
Bibliography
- International Scientific Association for Probiotics and Prebiotics (ISAPP). (2024). Postbiotics: Consensus Definition and Scientific Framework. Nature Reviews Gastroenterology.
- European Food Safety Authority (EFSA). (2024). Guidance on Health Claims Related to Gut and Immune Function: Postbiotic Evidence Requirements. EFSA Journal.
- European Commission. (2024). Directive 2002/46/EC on Food Supplements: Permitted Ingredients and Labelling. EUR-Lex.
- Morinaga Milk Industry Co. (2024). Annual Report 2024: Functional Ingredients Division, Postbiotic Research. Morinaga.
- World Gastroenterology Organisation (WGO). (2024). Global Guidelines on Probiotics and Prebiotics: Postbiotic Section Update. WGO.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
This Report Addresses
- Market sizing and forecast metrics for postbiotic supplements across EU end-use sectors through 2036.
- Segmentation analysis mapping adoption velocity and regulatory factors.
- Country-level intelligence comparing growth across major EU markets.
- Regulatory compliance assessment analyzing EU classification and labelling standards.
- Competitive posture evaluation tracking supplier dynamics and margins.
- Strategic procurement guidance defining supply agreements and inventory parameters.
- Supply chain vulnerability analysis identifying origin and seasonal risks.
- Custom data delivery: dashboards, Excel datasets, and PDF reports.
Frequently Asked Questions
How large is EU demand for postbiotic supplements in 2026?
EU demand is estimated at USD 4.5 million in 2026.
What will be the EU market size by 2036?
Projected to reach USD 14.3 million by 2036.
What is the expected CAGR?
Expected CAGR of 12.3% between 2026 and 2036.
Which Distribution Channel leads by 2026?
Drug Stores accounts for 67.0% share in 2026.
How is Capsules & Tablets positioned?
Capsules & Tablets holds 46.0% segment share in 2026.
Which country leads growth?
Netherlands at 14.2% CAGR through 2036.
What is the France outlook?
France at 11.6% CAGR during 2026 to 2036.
What does the forecast represent?
Model-based projection on EU production, trade, and consumption, cross-validated by FMI against company disclosures and Eurostat data.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Form , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Form , 2026 to 2036
- Capsules & Tablets
- Soft-gel & Chews
- Powder & Granules
- Capsules & Tablets
- Y to o to Y Growth Trend Analysis By Product Form , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Form , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Drug Stores
- Health & Beauty Stores
- Supermarkets & Hypermarkets
- Online Retailing
- Drug Stores
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Form
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- POSTBIOTICA SRL
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Designs for Health
- Cerbios-Pharma SA
- ADM Protexin
- Morinaga Milk Industry Co., Ltd.
- Mycology Research Laboratories
- Deerland Probiotics & Enzymes
- Sabinsa Corporation
- IFF Health
- Chr. Hansen
- Others
- POSTBIOTICA SRL
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Form
- Figure 6: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Distribution Channel
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Form
- Figure 23: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Distribution Channel
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Form
- Figure 30: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Form
- Figure 37: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Form
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Form
- Figure 51: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Form
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Form
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

