Postbiotic Fermented Barley Extract Market
Postbiotic Fermented Barley Extract Market is segmented by Product Form (Powdered Extracts, Liquid Fermented Concentrates, Encapsulated/Functional Blend Formats), Application (Dietary Supplements, Functional Foods & Beverages, Animal & Pet Nutrition, Cosmetics & Personal Care, Pharmaceuticals/Therapeutic Products), Distribution Channel (Specialty Ingredient Distributors, Direct OEM Sales, Online Retail, Institutional/B2B Contracts), End User (Adults, Elderly, Athletes/Active Lifestyle, Others), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Postbiotic Fermented Barley Extract Market Size, Market Forecast and Outlook By FMI
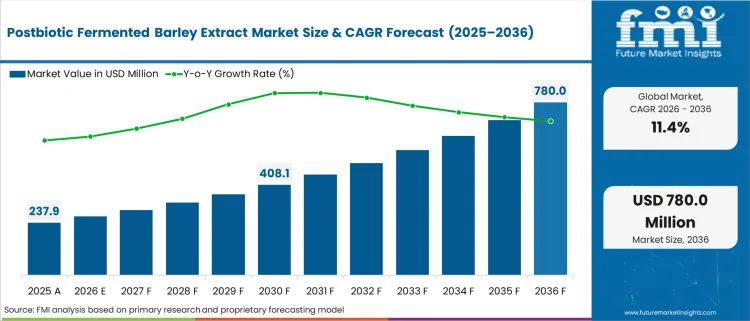
In 2025, the postbiotic fermented barley extract market was valued at USD 240.0 million and is projected to reach USD 265.0 million in 2026 and USD 840.0 million by 2036, reflecting a CAGR of 11.4% as regulatory committees mandate clinical efficacy over generic symptom management. Ingredient standardization resets minimum capability across postbiotic supplements pipelines. Procurement teams require verifiable stability data before authorizing formulation upgrades.
Absolute dollar growth of USD 575.0 Million over decade signals incremental expansion rather than transformational opportunity. Supply networks shift dependence toward resilient cereal crops, responding to February 2026 global harvest projections of 153.74 million metric tons [1]. As per FMI's projection, facilities possessing audited extraction capabilities bypass seasonal volatility. Volume contracts secure dedicated capacity ahead of peak purchasing cycles.
Summary of Postbiotic Fermented Barley Extract
- Postbiotic Fermented Barley Extract Market Definition
- Controlled microbial digestion of agricultural substrates yields stabilized bacterial metabolites for commercial nutrition integration.
- Demand Drivers in Market
- Clinical documentation forces specification upgrades across commercial nutrition.
- Formulators replace live probiotics with shelf-stable metabolite compounds.
- Skincare brands demand validated anti-inflammatory compounds.
- Key Segments Analyzed in FMI Report
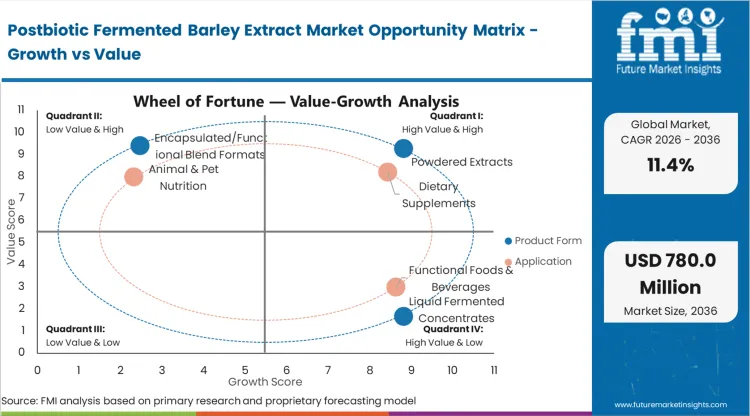
- Product Form: Powdered extracts (48%) versus liquid fermented concentrates and encapsulated/functional blend formats.
- Application: Dietary supplements (37% of total volume) versus functional foods, cosmetics, pharmaceuticals, and animal nutrition.
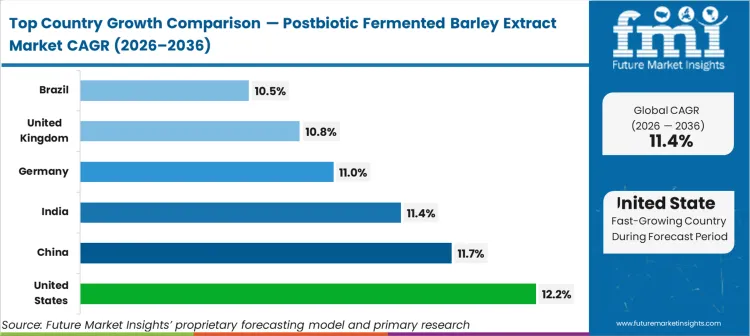
- Geography: High-growth North America and Asia-Pacific (United States 12.2%, China 11.7%, India 11.4%) versus regulation-driven Europe (Germany 11.0%, United Kingdom 10.8%).
- Analyst Opinion at FMI
- Nandini Roy Choudhury, Principal Consultant for Food & Beverage at Future Market Insights, opines, “In our latest Postbiotic Fermented Barley Extract Market analysis, my team and I have identified metabolite transparency as the next trust anchor in this category. As postbiotics move into mainstream functional foods, brands must clearly define which bioactive compounds, short-chain fatty acids, peptides, or cell wall fragments, drive the claimed benefits. Generic ‘fermented’ positioning will not sustain premium pricing. Companies investing in advanced fermentation control, analytical profiling, and clinically backed communication will shape the long-term credibility of barley-derived postbiotic solutions.”
- Strategic Implications/Executive Takeaways
- Secure long-term grain procurement contracts to mitigate harvest volatility.
- Invest in clinical trials to validate specific functional claims.
- Transition production toward dehydrated formats to reduce logistics overhead.
- Methodology
- Validated through first-hand corporate production and capacity data.
- Zero reliance on speculative third-party market research reports.
- Based on verifiable industrial benchmarks and verified sources.
Documented clinical outcomes compress hospital procurement reviews. As noted by CJ BIO, "This study provides scientific evidence that postbiotics can contribute to skin health. CJ BIO will continue expanding its portfolio of science-backed biotics and functional ingredients to deliver differentiated solutions in inner beauty market." [2] Buyers mandate certified indications to clear budgeting committees within single fiscal cycles.
United States registers 12.2% CAGR, China expands at 11.7% CAGR, India tracks 11.4% CAGR, Germany shows 11.0% CAGR, United Kingdom hits 10.8% CAGR, and Brazil achieves 10.5% CAGR. Dispersion mirrors regional regulatory adoption curves; nations possessing established medical food frameworks clear novel formulations faster than regions requiring primary trials.
Postbiotic Fermented Barley Extract Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 265.0 Million |
| Industry Value (2036) | USD 840.0 Million |
| CAGR (2026-2036) | 11.4% |
Postbiotic Fermented Barley Extract Market Definition
Postbiotic fermented barley extract designates non-viable bacterial products or metabolic byproducts derived via controlled microbial digestion of agricultural substrates. Industrial processing stabilizes beneficial metabolites for direct integration into commercial nutrition products. Extract parameters rely on verifiable beta-glucan content and metabolite density across postbiotic ingredients.
Postbiotic Fermented Barley Extract Market Inclusions
Commercial tracking counts powdered extracts, liquid concentrates, and encapsulated blends derived explicitly from controlled fermentation. Revenue calculations aggregate sales directed toward dietary supplements, functional foods, animal nutrition, and cosmetic formulations. Facility capacity expansions and strategic sourcing contracts directly inform valuation models.
Postbiotic Fermented Barley Extract Market Exclusions
Calculations exclude live probiotic strains, unfermented malt derivatives, and generic spent grain flours lacking deliberate postbiotic metabolite concentration. Adjacent therapeutic categories relying on synthetic analogues remain outside tracked valuations. Unprocessed agricultural commodity sales register zero impact on final extract estimations.
Postbiotic Fermented Barley Extract Market Research Methodology
- Primary Research: Analysts conducted direct interviews with facility managers and clinical researchers.
- Desk Research: Verification processes examined USDA crop circulars, NIH clinical databases, and corporate patent filings.
- Market-Sizing and Forecasting: Models triangulated raw agricultural output against patented extraction capacities.
- Data Validation and Update Cycle: Internal peer reviews cross-checked assumptions against disclosed vendor capacity investments.
Segmental Analysis
Postbiotic Fermented Barley Extract Market Analysis by Product Form
Manufacturers continue to favor dried extract formats as they simplify storage, remove cold-chain requirements, and offer longer shelf stability. Formulation teams can incorporate powdered extracts directly into capsules and functional blends without introducing moisture-related challenges. Scientific validation has confirmed barley beta-glucan concentrations ranging from 2.5% to 11.3%, supporting use in premium applications [3]. These advantages position powdered extracts to command 48% share in 2026.
- Capacity expansion: Facilities expand commercial-scale production through strategic partnerships formed in February 2026 to meet global demand surges [4]. Dehydrated postbiotic ferment filtrates integrate directly into high-speed encapsulation lines without secondary drying phases.
- Format compatibility: Solid dose manufacturing requires strict moisture control to prevent caking during encapsulation. Formulation teams select powdered extracts to maintain active compound integrity across prolonged retail cycles.
- Logistics advantage: Dehydrated formats neutralize ambient temperature degradation risks during transit. Freight operations execute prolonged shipments without refrigerated containers, lowering transport overhead by measurable margins.
Postbiotic Fermented Barley Extract Market Analysis by Application
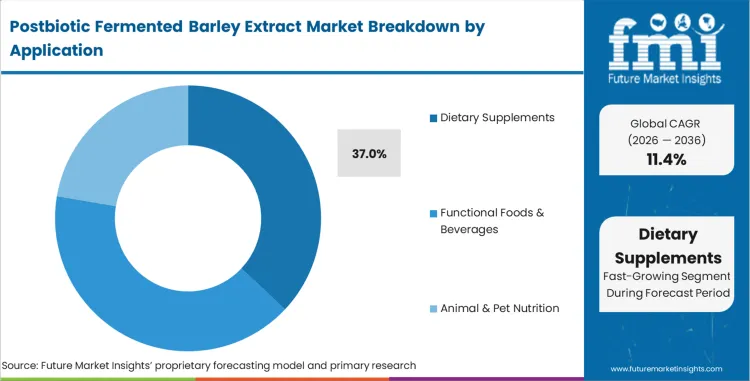
Dietary supplements represent 37% share in 2026. Health brands reformulate existing product lines to include stabilized immunity compounds. Active ingredients survive stomach acid processing better than live cultures. FMI analysts opine that medical research cites projected growth to USD 28.3 Million by 2032 for related postbiotic skincare and supplement categories [5].
- Therapeutic substitution: Medical food manufacturers require stable ingredients to formulate therapeutic diets targeting hormonal balance. October 2025 formula launches introduce specific perimenopause formulations to retail channels [6].
- Clinical validation: Documented outcomes compress hospital procurement reviews for institutional buyers. Buyers mandate certified indications to clear budgeting committees within fiscal cycles.
- Format versatility: Extract stability allows inclusion in gummies, capsules, and pressed tablets without viability losses. Formulators achieve desired dosing accuracy without complex overage calculations.
Postbiotic Fermented Barley Extract Market Drivers, Restraints, and Opportunities
Clinical documentation forces specification upgrades across commercial nutrition. Formulators abandon generic fermented extracts in favor of concentrated, stable metabolite profiles. Extensive documentation spanning over 2,215 recorded patents validates specific functional claims [7]. Procurement teams mandate precise concentrations to satisfy strict labeling requirements. Medical food manufacturers incorporate validated strains to secure institutional reimbursement codes.
Agricultural raw material dependencies constrain pure-play extraction scalability. Regional droughts disrupt core cereal yields, exposing ingredient buyers to severe pricing volatility. United States harvest volumes dropped 2% to 141 million bushels in September 2025, forcing extractors to secure forward contracts at elevated premiums [8]. Facilities lacking integrated supply networks face margin compression during poor harvest cycles. Formulators seeking prebiotic ingredient substitutes encounter temporary shortages.
- Trade diversification: Manufacturers introduce novel delivery formats to capture younger demographics. Expo West 2026 debuts showcase concentrated formulations designed for direct consumer integration [9].
- Mental wellness: Formulators target emotional well-being pathways using precision fermentation techniques. Keystone postbiotic launches in 2025 establish new product categories combining immunity and stress relief [10].
- Ingredient bioavailability: Supply chains prioritize extracts enhancing nutrient absorption. Product launches demonstrate increased bioavailability metrics, capturing premium pricing tiers.
Regional Analysis
.webp)
Based on regional analysis, Postbiotic Fermented Barley Extract market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. Full report also offers market attractiveness analysis based on regional trends.
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 12.2% |
| China | 11.7% |
| India | 11.4% |
| Germany | 11.0% |
| United Kingdom | 10.8% |
| Brazil | 10.5% |
North America Postbiotic Fermented Barley Extract Market Analysis
North America drives demand via aggressive functional food reformulations and advanced medical nutrition frameworks. Facilities upgrade extraction hardware to process domestic agricultural outputs efficiently. According to FMI's estimates, formulators prioritize dehydrated formats to bypass complex cold-chain logistics across continental distribution networks. Regulatory clarity surrounding non-viable bacterial products accelerates new product approvals. Medical food applications expand rapidly as clinical evidence links specific metabolites to systemic health improvements. Local university partnerships secure exclusive commercialization rights for patented fermentation processes utilizing spent grain flour.
- United States: In the United States, demand for postbiotic fermented barley extract is gaining momentum as structured clinical validation protocols strengthen confidence before procurement and budgeting decisions are finalized. Clinical study announcements in June 2025 further reinforced the efficacy of cultured proteins in targeted applications [11], encouraging broader commercial deployment and supporting expansion at a 12.2% CAGR through 2036.
FMI's report includes comprehensive assessment spanning North American jurisdictions. Canada and Mexico present secondary expansion targets. Formulators watch cross-border regulatory harmonization efforts closely to scale regional distribution models.
Europe Postbiotic Fermented Barley Extract Market Analysis
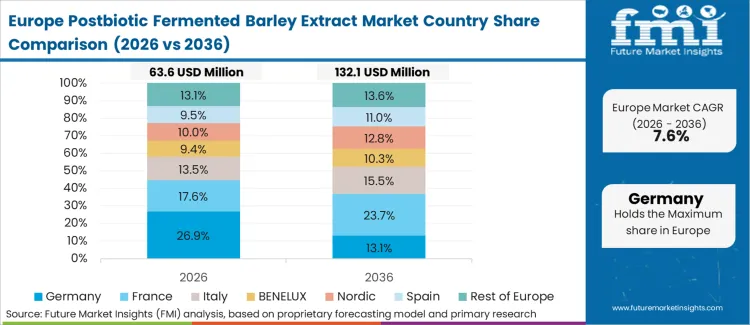
Europe represents a critical processing hub due to extensive agricultural infrastructure and strict food safety regulations. Food safety authorities mandate extensive documentation proving active compound stability before granting retail access. Extractors invest heavily in precision drying equipment to meet demanding pharmaceutical-grade specifications. Brands substitute live cultures with stable metabolites to extend retail shelf life and reduce spoilage waste. Clinical studies validating skin barrier improvements prompt widespread adoption across premium cosmetic segments. Local processing plants scale capabilities to service domestic and export requirements.
- Germany: Postbiotic Fermented Barley Extract sales in Germany are projected to rise at 11.0% CAGR from 2026 to 2036 as rigorous quality certifications advance, creating a clearer pathway from policy or spending intent to purchase orders. Award-winning ES1 postbiotic integrations in 2025 highlight domestic innovation leadership [12].
- United Kingdom: United Kingdom is expected to grow at 10.8% CAGR in Postbiotic Fermented Barley Extract demand over 2026 to 2036 as local manufacturing policies tighten, tightening link between operational constraints and adoption-led investment. Distribution agreements signed in May 2025 secure proprietary scientific strains for regional markets [13].
FMI's report includes comprehensive assessment across European states. France and Italy show strong adoption potential. Procurement teams monitor European Union regulatory updates regarding non-viable fermented meal bases classification, noting region commands 35% of global production share [14].
Asia Pacific Postbiotic Fermented Barley Extract Market Analysis
Asia Pacific exhibits rapid adoption scaling, felled by expanding functional beverage sectors and rising preventative health awareness. Regional food manufacturers integrate stabilized extracts into traditional product formats. FMI analysts opine that clinical validation regarding skin health accelerates inclusion in inner beauty formulations. Facilities automate extraction processes to lower per-unit production costs while maintaining strict quality controls. Agricultural commodity pricing volatility pushes buyers toward negotiated volume contracts to protect operating margins utilizing local malt.
- China: China is likely to witness Postbiotic Fermented Barley Extract revenues increase at 11.7% CAGR during 2026 to 2036, given rising functional ingredient demand and resulting push for capacity and workflow upgrades. May 2025 hypoglycemic patented series launches capture high-value therapeutic categories [15].
- India: Growth in Postbiotic Fermented Barley Extract across India is estimated at 11.4% CAGR through 2036, driven by rising preventative healthcare spending that increases value of reliability and compliance in buyer criteria. May 2025 afternoon snack launches introduce accessible postbiotic formats to retail consumers [16].
FMI's report includes detailed evaluation of Asia Pacific markets. Japan and South Korea represent critical innovation centers. Buyers track clinical trial outcomes linking specific derivatives to specific health markers.
Latin America Postbiotic Fermented Barley Extract Market Analysis
Latin America presents emerging opportunities as regional food producers upgrade ingredient profiles to target premium wellness demographics. Facilities import concentrated powders to formulate locally manufactured supplements and cosmetics. Livestock operators adopt specific postbiotic feed additives to replace preventative antibiotics in industrial farming environments. Procurement teams navigate fluctuating currency exchange rates by establishing long-term volume agreements with global suppliers.
- Brazil: Outlook for Postbiotic Fermented Barley Extract in Brazil indicates a 10.5% CAGR from 2026 to 2036 as agricultural modernization scales, strengthening demand from both public programs and private operators. March 2025 innovation awards recognize regional companies pioneering advanced gut health technologies [17].
FMI's report includes analysis spanning Latin American territories. Argentina and Chile offer prospective agricultural integration opportunities. Manufacturers monitor local health ministry guidelines regarding novel ingredient registration procedures.
Competitive Aligners for Market Players
Extractors prioritize process standardization to guarantee consistent beta-glucan and metabolite concentrations across commercial batches. As per FMI's projection, facilities possessing advanced analytical testing equipment secure exclusive contracts with pharmaceutical-grade buyers. Documented batch consistency removes institutional procurement barriers, allowing rapid integration into medical food product lines.
Vertical supply chain integration shields manufacturers from raw material pricing volatility. Extractors establishing direct purchasing agreements with agricultural cooperatives secure predictable input costs despite global harvest fluctuations. Stable cost structures enable aggressive pricing strategies during competitive tender negotiations.
Strategic partnerships with clinical research institutions accelerate product commercialization timelines. Companies licensing patented extraction techniques from university laboratories bypass costly internal development phases. Clinical efficacy data generated through independent trials provides critical marketing leverage against generic unfermented ingredient alternatives.
Recent Developments
Report includes full coverage of key trends from competitive benchmarking. Some of recent developments covered in reports:
- In November 2025, Cargill Animal Nutrition & Health completed a 50% capacity expansion at its Engerwitzdorf, Austria facility, boosting micronutrition production to 30,000 metric tons annually. [18]
Key Players in Postbiotic Fermented Barley Extract Market
- Cargill, Incorporated
- Archer Daniels Midland (ADM)
- Kerry Group PLC
- DSM-Firmenich AG
- Sami-Sabinsa Group
- Postbiotica S.r.l.
- MCLS Europe B.V.
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 265.0 Million (2026) to USD 840.0 Million (2036), at a CAGR of 11.4% |
| Market Definition | Postbiotic fermented barley extract designates non-viable bacterial products or metabolic byproducts derived via controlled microbial digestion of agricultural substrates. |
| Product Form Segmentation | Powdered Extracts, Liquid Fermented Concentrates, Encapsulated/Functional Blend Formats |
| Application Segmentation | Dietary Supplements, Functional Foods & Beverages, Animal & Pet Nutrition, Cosmetics & Personal Care, Pharmaceuticals/Therapeutic Products |
| Distribution Channel Segmentation | Specialty Ingredient Distributors, Direct OEM Sales, Online Retail, Institutional/B2B Contracts |
| End User Segmentation | Adults, Elderly, Athletes/Active Lifestyle, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, China, India, Germany, United Kingdom, Brazil and 40 plus countries |
| Key Companies Profiled | Cargill, Incorporated, Archer Daniels Midland (ADM), Kerry Group PLC, DSM-Firmenich AG, Sami-Sabinsa Group, Postbiotica S.r.l., MCLS Europe B.V. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling, validated through primary interviews with postbiotic producers and fermentation technology providers, supported by trade data benchmarking and production capacity analysis for fermented barley-based extracts |
Postbiotic Fermented Barley Extract Market Analysis by Segments
Product Form
- Powdered Extracts
- Liquid Fermented Concentrates
- Encapsulated/Functional Blend Formats
Application
- Dietary Supplements
- Functional Foods & Beverages
- Animal & Pet Nutrition
- Cosmetics & Personal Care
- Pharmaceuticals/Therapeutic Products
Distribution Channel
- Specialty Ingredient Distributors
- Direct OEM Sales
- Online Retail
- Institutional/B2B Contracts
End User
- Adults
- Elderly
- Athletes/Active Lifestyle
- Others
Region
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- [1] USDA FAS WASDE (Feb 2026): World barley production 153.74 million metric tons.
- [2] Korea Herald (Feb 2026): CJ BIO Statement.
- [3] PMC/NIH (2025): Barley beta-glucan content 2.5-11.3%.
- [4] Nutrition Insight (Feb 2026): Döhler + Superbrewed Food partnership.
- [5] PMC/NIH (2025): Postbiotic supplements market projected growth to $28.3M by 2032.
- [6] GlobeNewswire (Oct 2025): resbiotic resW Perimenopause Postbiotic launch.
- [7] PMC/NIH Frontiers (2025): Postbiotic research trends >2,215 patents.
- [8] USDA NASS (Sep 2025): US barley production 141 million bushels.
- [9] WholeFoods Magazine (2026): Essential Formulas Expo West 2026 debut.
- [10] SupplySide SJ (2025): Verb Biotics Keystone Postbiotic launch.
- [11] Superbrewed Food (June 2025): SB1 postbiotic cultured protein clinical study.
- [12] ADM (2025): ES1 postbiotic wins Vitafoods Europe Innovation Award.
- [13] Lallemand Health Solutions (May 2025): Kirin Holdings distribution agreement.
- [14] USDA FAS (2025): European Union barley production share 35% of global.
- [15] BioSpace (May 2025): BON postbiotic hypoglycemic patented ingredient series.
- [16] GlobeNewswire (May 2025): 3pm LLC Afternoon Bites launch.
- [17] Future Food-Tech (Mar 2025): Postbiotics Inc. wins Puratos Innovation Challenge.
- [18] Cargill Animal Nutrition & Health. (2025, November 4). Cargill Animal Nutrition & Health increases micronutrition production capacity to meet growing customer demand.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence enabling structured strategic decision making across mature and emerging nutrition economies
- Market size estimation and 10 year revenue forecasts from 2026 to 2036, supported by validated capacity and trade benchmarks
- Growth opportunity mapping across powder, liquid, and encapsulated formats with emphasis on clinical validation transitions
- Segment and regional revenue forecasts covering dietary supplements, functional foods, cosmetics, and therapeutic applications
- Competition strategy assessment including integration models, cost survivability positioning, and compliance capability benchmarking
- Product reformulation and active compound tracking including verifiable beta-glucan and metabolite parameters
- Regulatory impact analysis covering medical food mandates, quality assurance thresholds, and export driven specification upgrades
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is demand for Postbiotic Fermented Barley Extract in global market in 2026?
Demand registers USD 265.0 Million in 2026.
What will be market size of Postbiotic Fermented Barley Extract in global market by 2036?
Size is projected to reach USD 840.0 Million by 2036.
What is expected demand growth for Postbiotic Fermented Barley Extract in global market between 2026 and 2036?
Demand expands at 11.4% CAGR between 2026 and 2036.
Which Product Form is poised to lead global sales by 2026?
Powdered Extracts command 48% share in 2026 due to extended shelf stability and direct formulation compatibility.
How significant is role of Dietary Supplements in driving Postbiotic Fermented Barley Extract adoption in 2026?
Dietary Supplements represent 37% share in 2026 as health brands reformulate existing product lines to include stabilized immunity compounds.
What is driving demand in United States?
Aggressive functional food reformulations and advanced medical nutrition frameworks push procurement teams to establish stringent supplier contracts.
What compliance standards or regulations are referenced for United States?
Medical food application frameworks and non-viable bacterial product approvals are referenced for United States.
What is United States growth outlook in this report?
United States expands at 12.2% CAGR from 2026 to 2036.
Why is Europe described as a priority region in this report?
Europe provides 35% of global raw material production, offering extractors secure, localized supply chains alongside rigorous food safety mandates.
What type of demand dominates in Europe?
Demand centers around pharmaceutical-grade extracts demanding extensive documentation proving active compound stability before retail access.
What is China growth outlook in this report?
China expands at 11.7% CAGR from 2026 to 2036.
Does report cover India in its regional analysis?
Yes, India falls under Asia Pacific regional coverage.
What are sources referred to for analyzing India?
Sources include facility capacity investments, functional ingredient demand metrics, and preventative healthcare spending data.
What is main demand theme linked to India in its region coverage?
Demand relates to incorporating stable metabolites into ambient-temperature nutritional products to lower final acquisition costs.
Does report cover Germany in its regional analysis?
Yes, Germany falls under Europe regional coverage.
What is main Germany related demand theme in its region coverage?
Demand connects to rigorous quality certifications advancing standardized extraction protocols and premium raw material sourcing.
Which product formats or configurations are strategically important for North America supply chains?
Dehydrated powder formats prove critical to bypass complex cold-chain logistics across continental distribution networks.
What is Postbiotic Fermented Barley Extract and what is it mainly used for?
It designates non-viable bacterial products derived via controlled digestion of agricultural substrates, used primarily in dietary supplements and functional foods.
What does Postbiotic Fermented Barley Extract mean in this report?
It refers to commercial tracking of powdered extracts, liquid concentrates, and encapsulated blends derived explicitly from controlled fermentation.
What is included in scope of this Postbiotic Fermented Barley Extract report?
Scope aggregates sales directed toward dietary supplements, functional foods, animal nutrition, and cosmetic formulations.
What is excluded from scope of this report?
Calculations exclude live probiotic strains, unfermented malt derivatives, and generic spent grain flours lacking deliberate postbiotic metabolite concentration.
What does market forecast mean on this page?
Market forecast represents a model based projection built on defined assumptions for strategic planning purposes.
How does FMI build and validate Postbiotic Fermented Barley Extract forecast?
Models triangulate raw agricultural output against patented extraction capacities, validated through corporate production data verification.
What does zero reliance on speculative third party market research mean here?
Primary interviews and verifiable public datasets inform calculations instead of unverified syndicated market estimates.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Form , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Form , 2026 to 2036
- Powdered Extracts
- Liquid Fermented Concentrates
- Encapsulated/Functional Blend Formats
- Powdered Extracts
- Y to o to Y Growth Trend Analysis By Product Form , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Form , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Dietary Supplements
- Functional Foods & Beverages
- Animal & Pet Nutrition
- Dietary Supplements
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Form
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Form
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Form
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Form
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Form
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Form
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Form
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Form
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Form
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Form
- By Application
- Competition Analysis
- Competition Deep Dive
- Cargill, Incorporated
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Archer Daniels Midland (ADM)
- Kerry Group PLC
- DSM-Firmenich AG
- Sami-Sabinsa Group
- Postbiotica S.r.l.
- MCLS Europe B.V.
- Cargill, Incorporated
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Form , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Form
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Form
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Form
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Form
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Form
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Form
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Form
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Form , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Form , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Form
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

