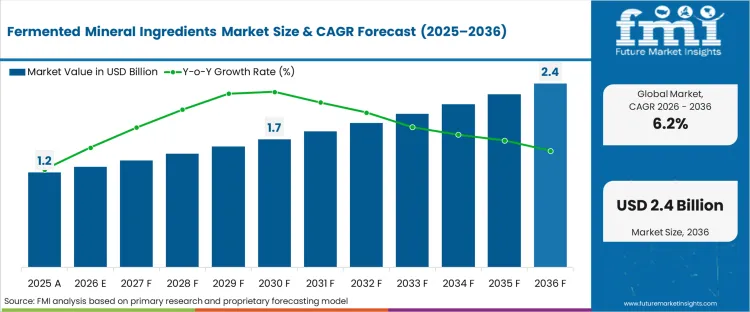
The fermented mineral ingredients market was valued at USD 1.3 billion in 2025. The industry is expected to reach USD 1.3 billion in 2026 at a CAGR of 6.2% during the forecast period. Sustained investment carries the valuation to USD 2.5 billion through 2036 as formulators across dietary supplement and functional food channels shift baseline specifications from synthetic inorganic salts to bio-transformed complexes to bypass gastrointestinal absorption barriers.
Formulation engineers are currently being forced to choose whether to continue managing the poor solubility and harsh sensory profiles of standard mineral oxides with artificial masking agents, or to migrate to inherently bio-compatible fermented complexes. Consumer tolerance for heavy, synthetic-tasting ingredients has collapsed, placing brands that rely on legacy mineral fortification at risk of immediate retail delisting. Switching to biologically transformed sources allows brands to drastically shorten their ingredient decks, which has become a baseline requirement for securing space within premium functional food ingredients retail networks. Brands that delay this ingredient transition lose their preferred supplier status and forfeit the pricing premium associated with whole-food nutrition labeling.
Before this transition becomes absolute, one specific operational gate must be crossed. Contract manufacturing organizations (CMOs) must standardize the tableting and encapsulation behavior of these highly hygroscopic fermented powders. Once major CMOs adjust their tooling and excipient ratios to handle moisture-sensitive biological complexes without requiring specialized low-humidity cleanrooms, the cost barrier drops sharply. Large-scale production environments that currently default to cheap carbonates flip to fermented alternatives purely for processing ease once this operational threshold is cleared.
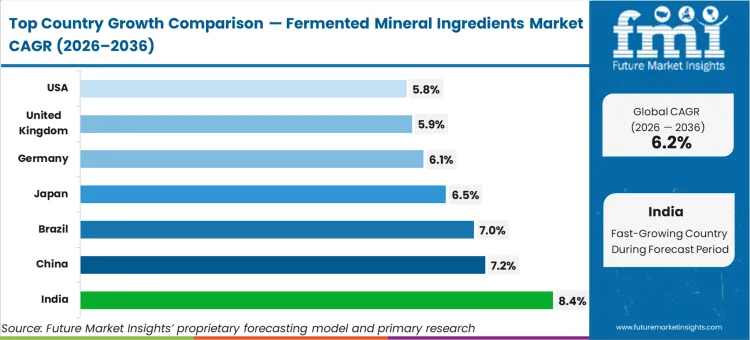
India is anticipated to advance at 8.4%, while China is set to register a 7.2% rate. Brazil follows at 7.0%, and Japan tracks closely at 6.5%. Germany is projected to grow at 6.1%, the United Kingdom at 5.9%, and the United States will expand at 5.8%. The divergence across this growth range is dictated entirely by how quickly domestic contract manufacturing infrastructure in each country can transition from handling stable, inert chemical salts to managing moisture-sensitive biological matrices efficiently.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.3 billion |
| Industry Value (2036) | USD 2.5 billion |
| CAGR (2026–2036) | 6.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The fermented mineral ingredients market encompasses organically bound mineral complexes produced through controlled biological fermentation. It represents the specific commercial intersection where inorganic mineral substrates are fed to microorganisms, typically yeast, fungi, or bacteria, which incorporate the target minerals into their cellular structure as peptides and amino acid chelates. This market is functionally distinct from basic chemical chelation, defined strictly by the use of a live biological conversion process to achieve bioavailability.
Scope includes all nutritionally relevant minerals that have undergone a verified fermentation step prior to extraction or drying. This covers yeast-derived selenium, lactic acid-fermented calcium, fungi-bound zinc, and biologically transformed iron matrices. Secondary metabolites naturally co-produced during the mineral-enrichment process remain within scope. Powders, aqueous dispersions, and stabilized liquid formats intended for human or animal ingestion, particularly within dietary supplements, fall strictly within the measurement boundary.
The market explicitly excludes synthetically manufactured amino acid chelates that use chemical reactions rather than live microorganism metabolism to bind the mineral. Simple mineral salts physically mixed with inactive yeast or probiotics post-production are omitted. These exclusions enforce the functional boundary: the economic value of this market is derived entirely from the biological transformation of the mineral, not from simple blending or chemical synthesis.
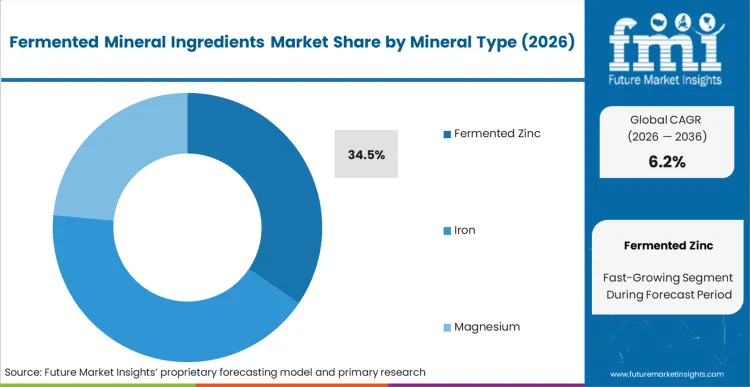
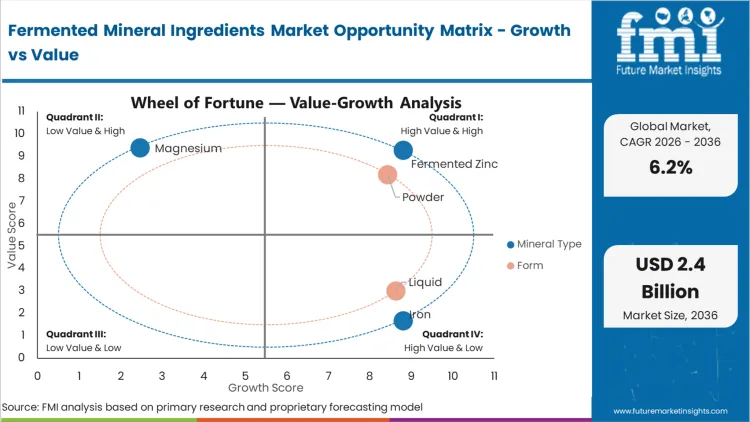
The commercial outcome for buyers specifying zinc hinges on a single physiological reaction: standard inorganic zinc salts consistently trigger acute nausea when taken on an empty stomach. According to FMI's estimates, this failure mode explains why fermented zinc secures a 34.5% share in 2026. Formulation teams do not choose fermented zinc merely to inch up absorption metrics; they specify it to eliminate the absolute highest cause of brand abandonment within the zinc supplements category. By binding the element to yeast peptides, the mineral bypasses gastric acid receptors entirely. R&D directors attempting to solve this with enteric coatings simply add complex excipients without addressing the root tolerability flaw. Brands refusing to adopt bio-transformed zinc find themselves permanently locked out of the premium health-food retail channels that actively screen out poorly tolerated formulations.
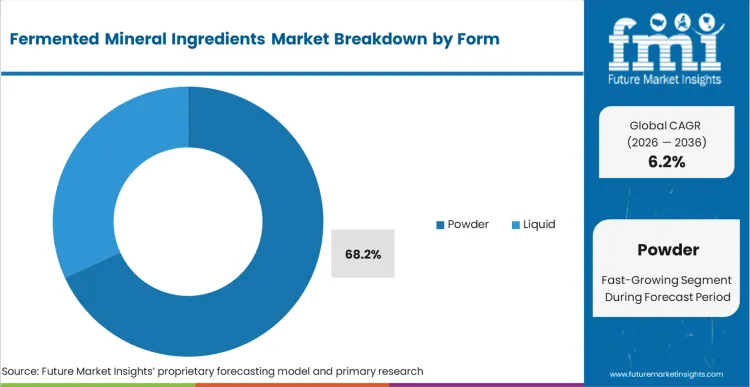
The reason powdered forms maintain a commanding 68.2% share in 2026 is grounded in global distribution realities: spray-drying is the only commercially viable method to stabilize liquid-grown biological biomass for international transit. Based on FMI's assessment, liquid formats carry prohibitive weight-to-value shipping ratios and restrictive shelf-life parameters that multinational supply chains cannot accommodate. Procurement directors require the 24-month stability that dehydrated matrices deliver. However, handling these inputs forces contract manufacturers to heavily manage ambient plant humidity, as biological powders are aggressively hygroscopic. Buyers who fail to mandate strict moisture-barrier packaging during transit experience rapid material caking, leading to total batch loss before encapsulation lines even commence operations.
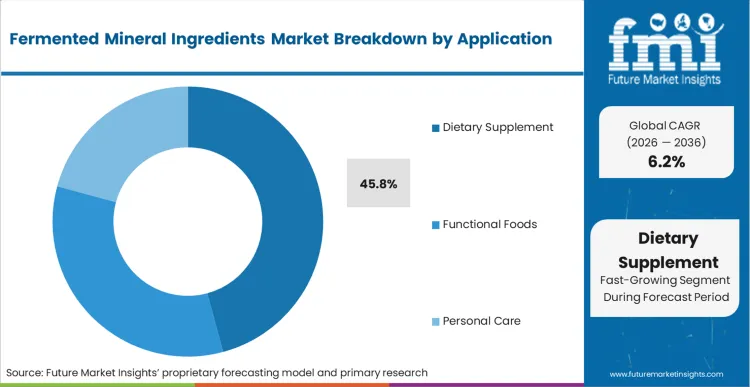
Product development directors in the dietary supplement space are currently deciding whether to defend legacy synthetic mineral formulations or absorb the cost of upgrading to whole-food matrices. This sector accounts for 45.8% of total demand in 2026. The shift is mandated by sophisticated retail buyers stripping shelves of products containing chemical excipients and harsh mineral oxides. In FMI's view, the margin structures in premium sports nutrition easily absorb the elevated per-kilogram cost of fermented inputs. Upgrading a label to declare food-state fermented iron instantly repositions a commoditized product into a premium tier. Brands refusing to authorize this specification change are rapidly relegated to discount channels, competing strictly on margin against synthetic generics.
Through 2036, the reliance on standard Saccharomyces cerevisiae will fracture as specialized bacterial and fungal platforms demonstrate superior uptake capacities for difficult elements like magnesium. Currently, yeast-based processes command a 52.1% share in 2026, driven largely by decades of established global regulatory familiarity. FMI analysts opine that specifying bodies are already drafting distinct quality parameters to separate fungal-mycelial mineral binding from traditional yeast outputs. While yeast pathways allow rapid compliance clearance globally, medical nutrition brands requiring higher concentration thresholds are pushing suppliers toward novel fungal bio-reactors. Ingredient suppliers failing to diversify their fermentation platforms beyond baker's yeast will find their product portfolios functionally obsolete as high-density medical requirements outpace yeast capabilities.
The structural shift toward clean-label nutrition forces product formulation teams to abandon chemically synthesized mineral salts across high-value categories. This transition represents a rigid procurement requirement dictated by major retail buyers who refuse to stock formulations relying on harsh inorganic oxides. Formulation engineers must evaluate biologically transformed alternatives that inherently eliminate the need for artificial flavor maskers and chemical buffers. The commercial stakes are definitive: brands qualifying fermented ingredients secure sustained placement in premium retail networks, while those clinging to legacy synthetic formulations face systemic exclusion from high-margin distribution channels.
The primary organizational friction slowing this transition is the severe cost-in-use differential inherent to biological processing. Fermented mineral complexes require capital-intensive bioreactor cultivation, rendering them significantly more expensive per kilogram than standard mined carbonates. This builds a structural wall within procurement departments, which are traditionally incentivized to reduce raw material expenditures. Although secondary extraction efficiencies continue to improve, they cannot erase the fundamental biological pricing gap. Formulation teams must navigate prolonged internal negotiations against their own finance departments to justify margin compression, systematically delaying premium product launch timelines.
Based on the regional analysis, the Fermented Mineral Ingredients market is segmented into North America, Europe, Asia Pacific, and Latin America across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 8.4% |
| China | 7.2% |
| Brazil | 7.0% |
| Japan | 6.5% |
| Germany | 6.1% |
| United Kingdom | 5.9% |
| United States | 5.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
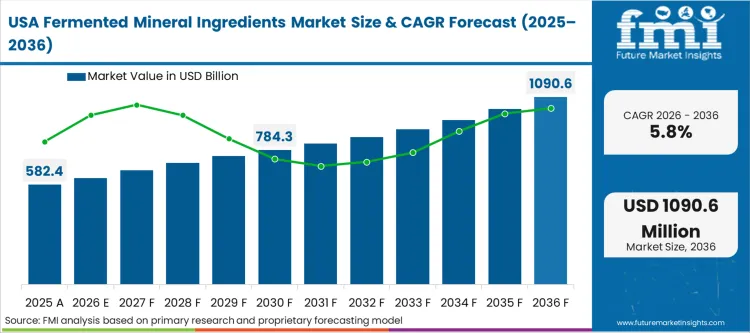
Procurement practices among tier-1 supplement brands define the adoption curve across North America. The domestic dietary space is saturated, forcing product development teams to hunt for specific, defensible differentiation rather than competing purely on dose concentration. According to FMI's estimates, North American buyers demonstrate a unique willingness to completely overhaul flagship products simply to upgrade the ingredient panel language from generic oxides to fermented cultures. The integration of cross-border supply chains ensures that Canadian and Mexican contract manufacturers must rapidly mirror these clean label ingredients sourcing standards to retain their North American client base.
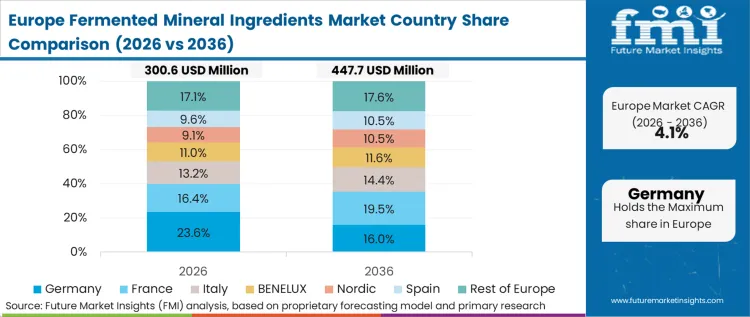
The regulatory environment dictates the pace and nature of mineral transition across Europe. The European Food Safety Authority sets the world's most rigid parameters for health claims, meaning formulators cannot market bioavailability without explicit, peer-reviewed clinical dossiers. In FMI's view, European buyers specify fermented minerals not for marketing appeal, but because ingredient suppliers have successfully navigated the novel food framework to secure specific health claims that standard mineral salts lack. Regulatory harmonization ensures that an approved fermented strain in one member state immediately triggers formulation updates across the bloc.
Capital availability and domestic infrastructure goals drive the structural reality in Asia Pacific. The region is not primarily focused on premium capsule supplements; it is fortifying massive volumes of foundational dairy and beverage products without altering taste profiles. As per FMI's projection, procurement directors evaluate fermented minerals purely as a functional solution to the sensory failures of standard fortification. The defining pattern across the region is the rapid localization of fermentation capabilities, shifting nations from importing finished active ingredients to cultivating specialized mineral-binding strains domestically.
Cost structures and regional supply availability shape the adoption pace across Latin America. Domestic supplement brands operate within tighter margin environments, requiring explicit commercial justification before upgrading formulation inputs. Based on FMI's assessment, the shift occurs primarily in high-value animal nutrition and premium export-focused nutraceutical lines, where the biological transformation of trace minerals yields measurable reductions in environmental excretion rates.
The competitive structure of this market is heavily concentrated at the raw biological processing tier. Global yeast and fermentation specialists dominate because the barrier to entry is not simply purchasing raw minerals; it requires owning and operating massive, precisely controlled bioreactor infrastructure. Procurement directors at major dietary supplement brands differentiate qualified from unqualified vendors based entirely on strain documentation. They require suppliers to prove analytically that the mineral is genuinely incorporated into the microbial cellular matrix, rather than just physically dry-blended with inactive yeast post-production.
To command premium pricing, incumbents rely on deep clinical dossiers proving their specific bacterial or yeast strains enhance human cellular uptake. Challengers attempting to enter this space must build comprehensive safety and bioavailability data packages, a category of regulatory and clinical capability that requires years and significant capital to replicate. An ingredient supplier lacking a proprietary, validated fermented strain cannot compete for tier-1 immunity boosting ingredients contracts, regardless of how aggressively they discount their generic products.
Large formulation houses resist locking themselves into single-supplier proprietary strains by demanding standard specifications that multiple vendors can fulfill. The structural tension, however, heavily favors the dominant fermentation specialists. Because changing an active microbial strain forces a brand to rewrite product labels and repeat complex stability testing, vendor switching costs remain aggressively high. FMI analysts opine that the market will likely see legacy chemical ingredient companies acquiring specialized biotechnology firms to secure the bioreactor capacity necessary to compete in this transitioning landscape through 2036.

| Metric | Value |
|---|---|
| Quantitative Units | USD 1.3 billion to USD 2.5 billion, at a CAGR of 6.2% |
| Market Definition | The commercial application of mineral elements biologically digested and incorporated into the cellular matrix of microorganisms, transitioning away from chemically synthesized salts. |
| Mineral Type Segmentation | Zinc, Iron, Magnesium, Calcium, Selenium |
| Form Segmentation | Powder, Liquid |
| Application Segmentation | Dietary Supplements, Functional Foods, Personal Care, Animal Feed |
| Fermentation Process Segmentation | Yeast-based, Fungi-based, Bacteria-based |
| Regions Covered | North America, Europe, Asia Pacific, Latin America |
| Countries Covered | India, China, Brazil, Japan, Germany, United Kingdom, United States, and 40 plus countries |
| Key Companies Profiled | Novonesis, Kerry Group plc, Alltech Inc., Lallemand Inc., Balchem Corporation, Novus International, Lesaffre Group, Angel Yeast Co., Ltd., Corbion N.V., DSM-Firmenich |
| Forecast Period | 2026 to 2036 |
| Approach | FMI interviewed formulation scientists and procurement directors within the nutraceutical sectors. The baseline anchors to traceable volumes of active yeast strains procured specifically for mineral enrichment. Forecasts were cross-validated against global import-export data for specialty yeast derivatives and retail velocity metrics. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
How large is the Fermented Mineral Ingredients market in 2026?
The market reaches USD 1.3 billion in 2026. This figure signals the exact scale at which major supplement brands have completely abandoned cheap synthetic salts in favor of premium, biologically compatible active inputs.
What will it be valued at by 2036?
The valuation advances to USD 2.5 billion by 2036. This cumulative buildup indicates that biological mineral binding is transitioning from a niche premium feature to the mandatory baseline specification for human nutrition globally.
What CAGR is projected?
A 6.2% CAGR is expected during the forecast period. This rate reflects the speed at which contract manufacturers can physically upgrade their plant humidity controls to handle sensitive fermented powders, not a lack of end-consumer demand.
Which Mineral Type segment leads?
Fermented zinc commands the segment. By binding zinc to yeast peptides, formulators entirely eliminate the severe acute nausea that causes massive consumer abandonment in standard immunity supplements.
Which Form segment leads?
Powder formats dominate the market infrastructure. Global yeast producers rely entirely on spray-drying to arrest biological activity and guarantee the 24-month transit stability that international procurement directors demand.
Which Application segment leads?
Dietary supplements maintain the lead position. The retail margin structures in the premium vitamin channel easily absorb the higher per-kilogram cost of fermented inputs, whereas high-volume feed applications struggle with the price premium.
What drives rapid growth?
Clean-label mandates at the retail buyer level compel brands to drop chemical flavor maskers. Formulators use naturally bio-compatible fermented complexes to clean up ingredient panels and secure premium shelf placements.
What is the primary restraint?
The severe cost-in-use differential creates deep internal friction. R&D engineers must wage internal battles against their own finance departments to justify the margin compression required to utilize capital-intensive bioreactor ingredients.
Which country grows fastest?
India expands rapidly at an 8.4% pace, structurally outpacing China. Indian formulators are aggressively updating maternal anemia lines to bypass the gastric distress of standard iron oxides, leapfrogging intermediate chelate technologies entirely.
How do EFSA regulations shape the European trajectory?
The European Food Safety Authority requires localized, peer-reviewed clinical dossiers for any absorption claims. Formulators buy fermented minerals in Europe specifically because major suppliers have already funded and secured these difficult novel food approvals.
Why is moisture management critical for this market?
Fermented biological powders are intensely hygroscopic. Plant managers must implement absolute humidity controls during blending; failing to do so results in rapid caking and total batch loss before tableting can occur.
How do incumbent suppliers defend their market position?
Global fermentation specialists lock in formulation contracts through deep safety dossiers and proprietary strain validation. Competitors cannot simply buy standard yeast; they must replicate years of clinical bioavailability data to win tier-1 brand accounts.
What role do contract manufacturers play in adoption?
CMOs act as the structural gatekeeper. Until a contract manufacturer validates its tooling to handle sticky, fermented powder formats without jamming, the brand relying on that CMO cannot launch a fermented SKU.
Why are fungal fermentation platforms emerging?
Traditional baker's yeast has absolute biological ceilings for mineral uptake. Medical nutrition formulation teams requiring extremely high-dose magnesium or calcium are forcing suppliers to develop novel fungal-mycelial vectors.
What is the functional difference between fermented minerals and amino acid chelates?
Chelates use chemical reactions to synthesize a bond, while fermented complexes use live microorganism metabolism. Clean-label retailers actively distinguish between the two, increasingly rejecting chemical chelates in favor of the biological approach.
Why are functional beverage makers adopting these ingredients?
Standard mineral salts precipitate out of solution and introduce heavy metallic off-notes. Beverage scientists deploy biologically bound minerals to maintain suspension stability during UHT pasteurization without adding artificial emulsifiers.
How does supplier switching work in this sector?
Switching costs are punitively high. Because a brand's label claims are tied to the specific clinical data of a supplier's proprietary yeast strain, changing vendors requires rewriting labels and repeating stability testing.
What prevents agricultural feed producers from adopting fermented minerals completely?
While fermented minerals vastly reduce environmental heavy-metal excretion, the high capital cost of bioreactor processing destroys the tight margin structures of commercial poultry and swine production, limiting use to premium breeder lines.
Why is standard zinc oxide losing market share?
Zinc oxide interacts harshly with stomach acid on an empty stomach. Premium brands are abandoning it entirely because this specific physiological failure directly causes high consumer churn rates.
What data anchors the FMI market sizing model?
The baseline forecast anchors directly to the measurable procurement volume of live yeast and bacterial biomass designated specifically for trace mineral enrichment protocols at the industrial scale.
Which ingredient category is excluded from this report scope?
Synthetically manufactured bisglycinates and other chemical chelates are explicitly excluded. The economic boundary of this report is defined entirely by the use of a live biological conversion step.
Why do North American brands reformulate so quickly?
The domestic direct-to-consumer supplement space is violently saturated. Marketing departments demand fermented specifications purely to differentiate their product language from generic competitors, forcing rapid ingredient turnover.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.