The Pharmerging Contract Manufacturing Market is estimated to be valued at USD 195.2 billion in 2025 and is projected to reach USD 375.5 billion by 2035, registering a compound annual growth rate (CAGR) of 6.8% over the forecast period.
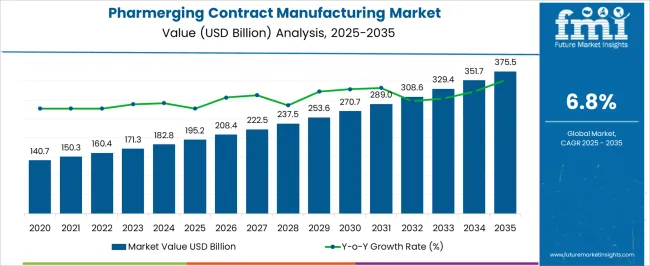
| Metric | Value |
|---|---|
| Pharmerging Contract Manufacturing Market Estimated Value in (2025 E) | USD 195.2 billion |
| Pharmerging Contract Manufacturing Market Forecast Value in (2035 F) | USD 375.5 billion |
| Forecast CAGR (2025 to 2035) | 6.8% |
The Pharmerging Contract Manufacturing market is experiencing significant growth, driven by increasing outsourcing of pharmaceutical production in emerging markets to reduce costs and enhance operational efficiency. Demand is being fueled by the rising prevalence of chronic diseases, expanding access to healthcare, and growing pharmaceutical consumption in these regions. Advancements in manufacturing capabilities, quality assurance processes, and regulatory compliance have enabled contract manufacturers to provide high-quality production services for both branded and generic pharmaceuticals.
The market is further supported by increasing investment in healthcare infrastructure, strategic partnerships, and collaborations between global pharmaceutical companies and regional manufacturers. Rising demand for innovative formulations, personalized medicines, and efficient supply chain solutions is also driving adoption.
As pharmaceutical companies aim to optimize production costs while maintaining product quality, contract manufacturing in pharmerging markets is increasingly preferred The market is expected to continue growing as governments and private enterprises focus on healthcare accessibility, efficient drug production, and compliance with international standards.
The pharmerging contract manufacturing market is segmented by product, therapeutic area, drug class, and geographic regions. By product, pharmerging contract manufacturing market is divided into Branded Generics, Branded Drugs, and Generics. In terms of therapeutic area, pharmerging contract manufacturing market is classified into Oncology, Cardiovascular, Diabetes, Infectious Diseases, and Others. Based on drug class, pharmerging contract manufacturing market is segmented into Small Molecule Drugs, Generic Drugs, Highly-Potent Drugs And Cytotoxic Drugs, Large Molecule Drugs, Monoclonal Antibodies, Vaccines, Interferon, Insulin And Analogs, and Others. Regionally, the pharmerging contract manufacturing industry is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
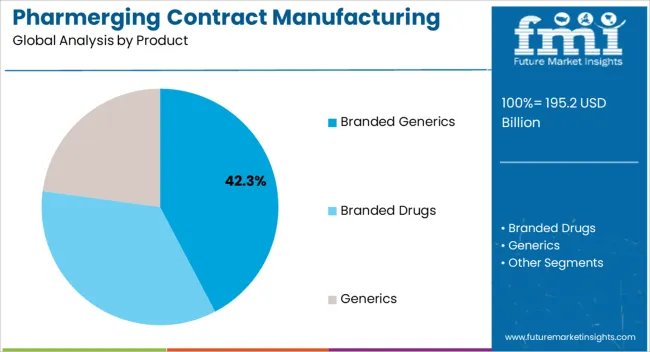
The branded generics product segment is projected to hold 42.3% of the market revenue in 2025, establishing it as the leading product type. Growth is being driven by increasing demand for affordable yet reliable medications in emerging markets. Branded generics provide a cost-effective alternative to innovative drugs while maintaining quality and efficacy, making them highly preferred by healthcare providers and patients.
Contract manufacturers are increasingly focusing on producing branded generics to meet regulatory standards and ensure consistent supply. The ability to scale production quickly in response to market demand further strengthens adoption. Partnerships with global pharmaceutical companies enhance technology transfer, quality assurance, and market reach.
With rising healthcare expenditures and expanding access to medications in pharmerging regions, branded generics are expected to maintain market leadership The segment’s growth is also supported by increasing chronic disease prevalence, government initiatives to improve drug availability, and the need for cost-efficient therapies, positioning it as a core driver of the overall market.
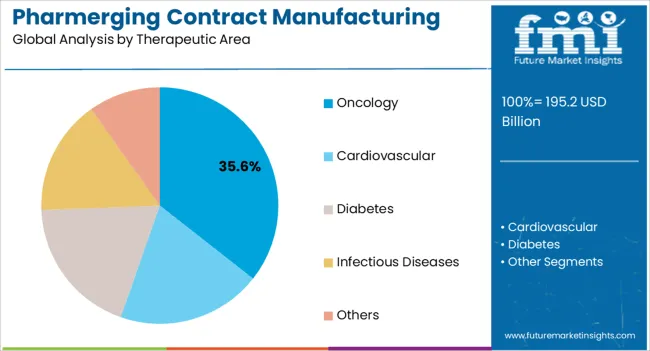
The oncology therapeutic area segment is anticipated to account for 35.6% of the market revenue in 2025, making it the leading therapeutic area. Growth in this segment is being driven by the rising global incidence of cancer and the increasing need for effective treatment options. Contract manufacturing enables the production of high-quality oncology drugs that comply with stringent safety and regulatory requirements.
Specialized manufacturing capabilities, including complex formulations and advanced delivery systems, support the production of oncology therapeutics. The ability to scale production rapidly to meet growing demand ensures timely availability of critical cancer treatments.
Investment in research collaborations and partnerships between contract manufacturers and pharmaceutical companies further enhances capabilities in oncology drug production As awareness of cancer treatment options increases and access to therapies improves in emerging markets, the oncology segment is expected to remain a key growth driver, supported by innovation in drug formulations, cost-effective manufacturing, and expanding healthcare infrastructure.
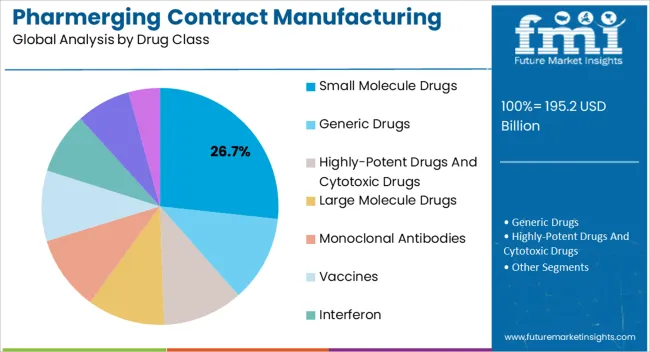
The small molecule drugs segment is projected to hold 26.7% of the market revenue in 2025, establishing it as the leading drug class. Growth in this segment is being driven by the high demand for orally administered and cost-effective therapeutic options across various disease areas. Small molecule drugs are easier to manufacture at scale compared with biologics, making them highly suitable for contract manufacturing in pharmerging markets.
Their stability, established regulatory pathways, and proven efficacy support widespread adoption. Contract manufacturers are leveraging advanced production techniques and quality control processes to ensure consistency and compliance.
The ability to provide scalable, reliable, and cost-efficient production aligns with the needs of pharmaceutical companies seeking to optimize supply chains As emerging markets continue to expand access to essential medicines, small molecule drugs are expected to maintain their leading position, supported by demand for affordable, high-quality therapies and the growth of chronic and lifestyle disease treatment.
Pharmerging markets indicate the most promising emerging markets in the pharmaceutical domain. Most of these markets are currently exhibiting growth rates moderate to high indicating sustainable demand prospects. The global pharmaceutical market is projected to expand at CAGR of 4–7% between 2025 and 2026 to reach US$1.5 trillion by 2026. This indicates revenues from companies at ex-manufacturer price levels excluding rebates and discounts.
Industry experts note that the USA and pharmerging markets are expected to account for more than 60% of sales and 80% of sales growth over the next five years. Contract manufacturing of pharmaceuticals is witnessing steady acceleration in pharmerging markets given high demand for medicines in these countries.
For example, consumer medicines form a substantially large portion of pharmerging markets and manufacturers want to source the production of these medicines in the target market themselves. Given the growing complexity in supply-chain of highly potent drugs (both small molecules and biologics); there is a growing trend of outsourcing early-stage analytical, formulation, and commercial development of the medicines.
This shift offers CDMOs more opportunities to partner and offer services within the drug product lifecycle. Specifically experts notes that outsourcing the bulk manufacturing of drugs is gradually gaining traction in countries in Asia-Pacific.
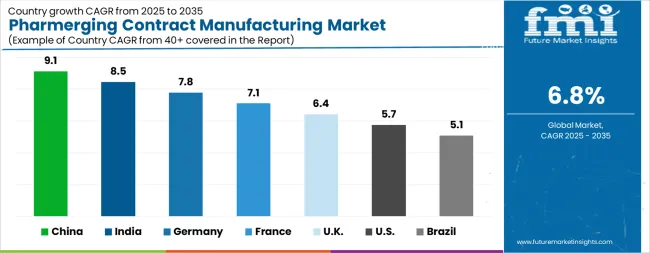
| Country | CAGR |
|---|---|
| China | 9.1% |
| India | 8.5% |
| Germany | 7.8% |
| France | 7.1% |
| UK | 6.4% |
| USA | 5.7% |
| Brazil | 5.1% |
The Pharmerging Contract Manufacturing Market is expected to register a CAGR of 6.8% during the forecast period, exhibiting varied country level momentum. China leads with the highest CAGR of 9.1%, followed by India at 8.5%. Developed markets such as Germany, France, and the UK continue to expand steadily, while the USA is likely to grow at consistent rates. Brazil posts the lowest CAGR at 5.1%, yet still underscores a broadly positive trajectory for the global Pharmerging Contract Manufacturing Market. In 2024, Germany held a dominant revenue in the Western Europe market and is expected to grow with a CAGR of 7.8%. The USA Pharmerging Contract Manufacturing Market is estimated to be valued at USD 68.6 billion in 2025 and is anticipated to reach a valuation of USD 119.9 billion by 2035. Sales are projected to rise at a CAGR of 5.7% over the forecast period between 2025 and 2035. While Japan and South Korea markets are estimated to be valued at USD 9.2 billion and USD 4.9 billion respectively in 2025.
| Item | Value |
|---|---|
| Quantitative Units | USD 195.2 Billion |
| Product | Branded Generics, Branded Drugs, and Generics |
| Therapeutic Area | Oncology, Cardiovascular, Diabetes, Infectious Diseases, and Others |
| Drug Class | Small Molecule Drugs, Generic Drugs, Highly-Potent Drugs And Cytotoxic Drugs, Large Molecule Drugs, Monoclonal Antibodies, Vaccines, Interferon, Insulin And Analogs, and Others |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, Middle East & Africa |
| Country Covered | United States, Canada, Germany, France, United Kingdom, China, Japan, India, Brazil, South Africa |
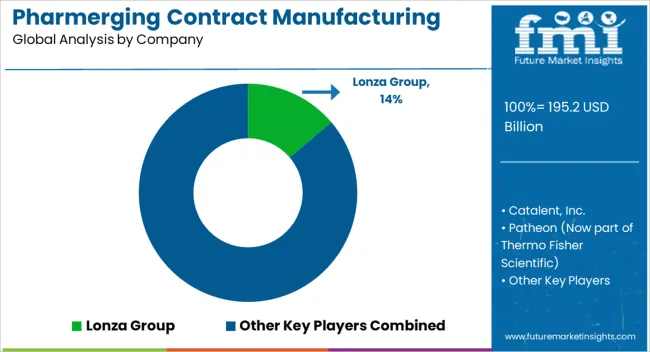
| Key Companies Profiled | Lonza Group, Catalent, Inc., Patheon (Now part of Thermo Fisher Scientific), Recipharm AB, Boehringer Ingelheim, Dr. Reddy's Laboratories, Jubilant Life Sciences, Fareva, Vetter Pharma, Evonik Industries, WuXi AppTec, Pfizer CentreOne, and Almac Group |
How big is the pharmerging contract manufacturing market in 2025?
The global pharmerging contract manufacturing market is estimated to be valued at USD 195.2 billion in 2025.
What will be the size of pharmerging contract manufacturing market in 2035?
The market size for the pharmerging contract manufacturing market is projected to reach USD 375.5 billion by 2035.
How much will be the pharmerging contract manufacturing market growth between 2025 and 2035?
The pharmerging contract manufacturing market is expected to grow at a 6.8% CAGR between 2025 and 2035.
What are the key product types in the pharmerging contract manufacturing market?
The key product types in pharmerging contract manufacturing market are branded generics, branded drugs and generics.
Which therapeutic area segment to contribute significant share in the pharmerging contract manufacturing market in 2025?
In terms of therapeutic area, oncology segment to command 35.6% share in the pharmerging contract manufacturing market in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.