The regulatory-compliant medical device plastics recycling platforms market is valued at USD 106 million in 2026 and is projected to reach USD 312.0 million by 2036, reflecting a CAGR of 11.40%. Expansion is supported by growing hospital polymer waste streams, stricter regulatory oversight, and adoption of certified recycling platforms for sensitive medical plastics. Closed-loop compliant systems represent around 49% of platform types, complemented by on-site segregation, off-site certified recycling, and automated compliance monitoring. Cost structures reflect certification requirements, operational complexity, and reporting obligations. Margin concentration favors providers delivering traceable, verified outputs rather than those focused solely on processing volume.
Between 2026 and 2036, application coverage includes rigid medical plastics, flexible disposables, composite device components, and sterile packaging polymers. Value accrues to platform operators offering integrated compliance, process consistency, and documentation capabilities. Key companies such as Veolia, Stericycle, Suez, Clean Harbors, and Republic Services capture stronger margins through multi-application operations and regulatory alignment. Regional adoption varies with hospital density, infrastructure readiness, and enforcement intensity. Providers lacking standardized compliance workflows face narrower returns, whereas those offering end-to-end, certified solutions sustain stable, long-term revenue growth and margin security throughout the forecast period.
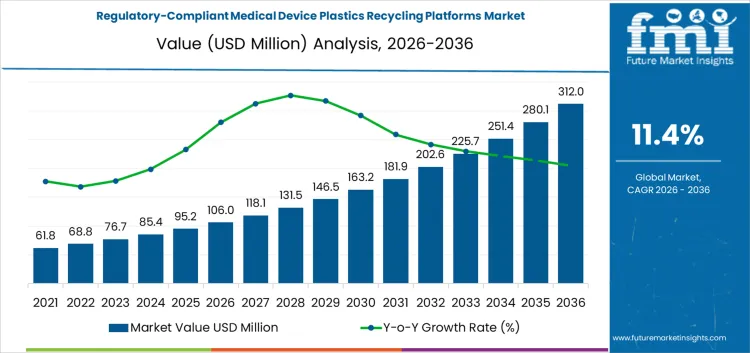
Between 2026 and 2031, the regulatory-compliant medical device plastics recycling platforms market is projected to grow from USD 106 million to USD 178 million, generating an absolute increase of USD 72 million and accounting for approximately 35% of total decade growth. Early-stage growth is driven by hospital adoption of compliant recycling platforms, rising focus on medical waste circularity, and investment in decontamination and polymer recovery technologies. Cost structures are influenced by sterilization, sorting, and process validation requirements. Value capture favors operators providing certified, high-purity recycled plastics suitable for medical applications rather than prioritizing volume throughput.
From 2031 to 2036, the market is expected to expand from USD 178 million to USD 312.0 million, adding USD 134 million and representing roughly 65% of decade growth. Growth accelerates as regulatory enforcement strengthens and recycled plastics are integrated into medical device supply chains. Margins increasingly favor platforms controlling feedstock sourcing, sterilization efficiency, and compliance documentation. Competitive advantage shifts to operators offering traceable, validated output, while late entrants focus on operational efficiency, standardization, and certification to capture incremental market share.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 106 million |
| Forecast Value (2036) | USD 312.0 million |
| Forecast CAGR (2026 to 2036) | 11.40% |
Regulatory-compliant recycling platforms for medical device plastics are increasingly implemented to align polymer recovery with stringent healthcare regulations. Historically, mixed-use plastics from devices such as catheters, tubing, and packaging were discarded due to contamination risk and inconsistent material composition.
Contemporary platforms combine segregation, sterilization, and controlled extrusion to produce regranulates suitable for non-critical applications, while maintaining full regulatory documentation. Hospitals, manufacturers, and recyclers coordinate to ensure traceable handling, process validation, and audit readiness. Demand is driven by operational risk reduction, cost management, and compliance adherence. Value capture prioritizes documented material safety, verified polymer performance, and regulatory auditability, rather than volume throughput, establishing these platforms as critical infrastructure in medical polymer lifecycle management.
Future growth depends on evolving regulatory frameworks, hospital sustainability policies, and increasing pressure to minimize hazardous waste streams. Unlike early pilot projects focused on proof-of-concept, modern platforms emphasize reproducible polymer properties, validated decontamination, and controlled additive profiles across multiple sites.
Cost efficiency is influenced by energy usage, sterilization effectiveness, and feedstock preparation, concentrating margins among operators capable of integrating end-to-end compliance. Healthcare providers adopt these systems to reduce liability, document material recovery, and support circularity targets. By 2036, regulatory-compliant medical device plastics recycling platforms are expected to serve as standard solutions for traceable, safe, and operationally efficient management of clinical polymer waste.
The demand for regulatory compliant medical device plastics recycling platforms is segmented by platform type and application focus. Platform types include closed loop compliant recycling systems, on site hospital segregation and processing, off site certified recycling services, and automated compliance tracking and reporting. Application focus covers rigid medical plastics, flexible polymer disposables, composite device plastics, and sterile packaging polymers. Segment selection is influenced by regulatory requirements, traceability standards, and material recovery efficiency. Adoption is driven by the need to maintain compliance with ISO, FDA, and local medical waste guidelines, while ensuring polymer integrity for reuse. Operational feasibility and auditability shape platform preference more than cost considerations.
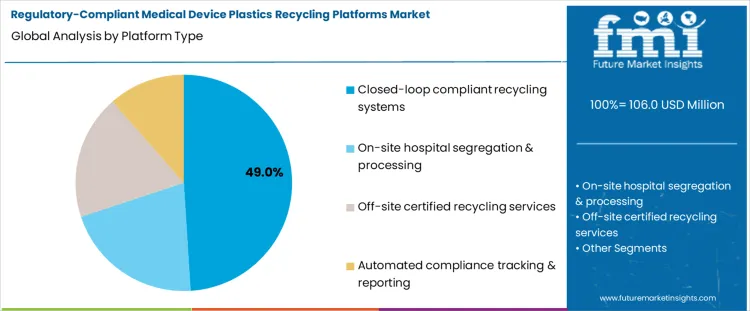
Closed loop compliant recycling systems account for approximately 49% of total platform adoption, making them the leading platform type. This share reflects their ability to maintain controlled material flow from collection through reprocessing, ensuring regulatory traceability and consistent polymer quality. These systems integrate segregation, sterilization, and recycling processes in a controlled environment. Operators favor closed loop platforms because they minimize contamination risk, provide documentation for compliance audits, and support reintegration of recycled material into new medical products.
Demand for closed loop systems is shaped by regulatory scrutiny and operational reliability. Hospitals, manufacturers, and certified recyclers adopt these platforms to ensure traceable recycling of high value medical plastics. The approach reduces risk of cross contamination and provides verifiable evidence of compliance. Systematic monitoring of batch data and material handling ensures output meets safety standards. The segment leads because it delivers controlled, scalable, and compliant recycling pathways for rigid and sensitive medical polymers.
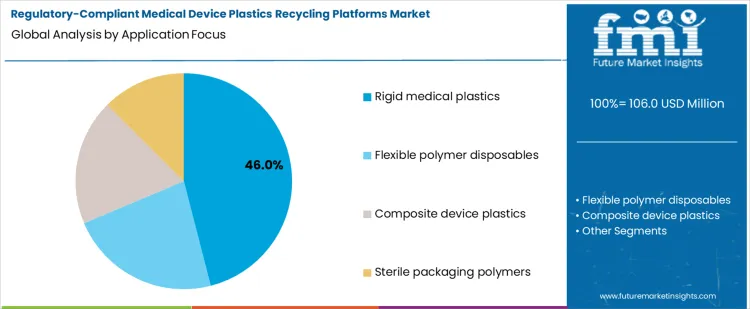
Rigid medical plastics account for approximately 46% of total application focus, making them the largest segment. This reflects high volumes of rigid items such as syringes, housings, and procedural components, which require consistent material properties and strict regulatory compliance. Their geometry and uniformity facilitate collection, sterilization, and controlled recycling through closed loop systems. Recycled output maintains mechanical strength and clarity, supporting reuse in new medical devices.
Demand for rigid medical plastics is driven by operational scale, regulatory mandates, and quality assurance needs. Hospitals and manufacturers prioritize recycling streams where material integrity can be reliably preserved. Rigid polymers simplify processing and documentation, enabling integration into audit compliant systems. The segment leads because rigid medical plastics combine high material demand, predictable processing behavior, and regulatory importance, making them the primary target for investment in compliant recycling platforms.
Regulatory-compliant medical device plastics recycling platforms focus on recovering polymers from single-use devices while maintaining adherence to stringent health authority standards. Adoption is strongest where hospitals, clinics, and device manufacturers face mandatory disposal or recycled content requirements. Systems emphasize validated sterilization, polymer integrity, and traceable documentation to meet local compliance frameworks. Growth reflects regulatory alignment and risk mitigation rather than overall volume expansion. Operators providing repeatable, certified recycling processes gain preference from healthcare networks. Investment is concentrated in regions with strict enforcement of medical waste segregation, audit protocols, and certification requirements, ensuring recycled polymers meet defined quality and safety thresholds.
Adoption is driven by country-specific medical waste and device regulations requiring secure handling and validated decontamination of single-use plastics. Hospitals and manufacturers prioritize suppliers that can deliver traceable, certified recycling outputs. Regulatory frameworks often mandate process validation for sterilization, contaminant removal, and polymer property retention. Platforms that demonstrate adherence to local health authority guidelines reduce audit risk and enhance approval timelines. Funding programs or institutional incentives in certain regions further encourage adoption. The driver is regulatory certainty and risk reduction, enabling healthcare facilities to meet compliance obligations while promoting circularity in medical device plastics.
Adoption is constrained by high capital costs for sterilization and polymer processing lines, stringent infection control protocols, and variability in feedstock composition. Technical expertise is required to maintain polymer performance and compliance documentation. Smaller hospitals or recyclers may lack scale or resources to justify investment. Logistical challenges in segregating, transporting, and storing contaminated device plastics increase operational complexity. Inconsistent enforcement or unclear approval criteria across regions slows uptake. These barriers limit adoption to technically capable, well-funded operators able to provide audit-ready, reproducible processes for medical device plastic recycling in compliance with local regulations.
Trends emphasize integrated process design combining sterilization, polymer recovery, and quality monitoring to meet regulatory standards. Partnerships between hospitals, device manufacturers, and recyclers secure feedstock and facilitate compliance documentation. Pilot and demonstration facilities validate reproducibility and safety before industrial deployment. Digital traceability systems support batch-level certification and audit readiness. Platforms increasingly adopt modular, scalable units to adapt to diverse facility sizes and waste streams. Focus is on reproducibility, compliance, and stakeholder integration rather than throughput or cost. Success is measured by the ability to consistently recycle medical device plastics while meeting local regulatory and institutional requirements.
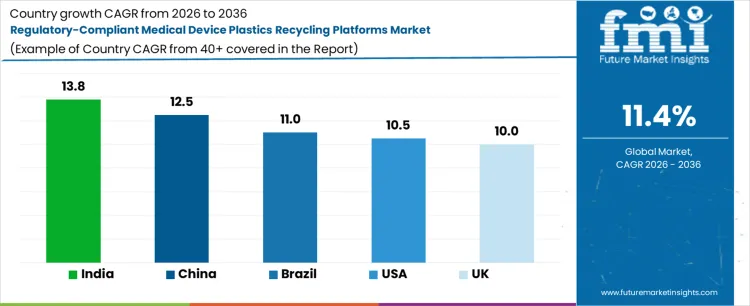
| Country | CAGR (%) |
|---|---|
| USA | 10.5 |
| UK | 10.0 |
| China | 12.5 |
| India | 13.8 |
| Brazil | 11.0 |
Demand for regulatory-compliant medical device plastics recycling platforms is growing rapidly as healthcare providers and manufacturers prioritize high-purity recycling and compliance with stringent medical standards. India leads with a 13.8% CAGR, driven by expanding hospital networks, high usage of single-use plastics, and adoption of certified recycling platforms for polycarbonate, polypropylene, and other medical-grade polymers. China follows at 12.5%, supported by large-scale medical device production, regulatory alignment, and integrated recycling solutions to recover valuable polymers. Brazil records 11.0% growth as hospitals and clinics implement compliance-driven recycling systems. The USA grows at 10.5%, reflecting investments in FDA-aligned platforms and hospital partnerships for safe polymer recovery. The UK shows 10.0% CAGR, shaped by adoption of certified recycling protocols and government-supported medical waste management initiatives.
In the United States, the regulatory-compliant medical device plastics recycling platforms market is expanding at a CAGR of 10.5%, driven by stringent FDA and EPA guidance for the management of single-use device waste. Recyclers and medical device manufacturers are deploying integrated platforms that combine segregation, cleaning, sterilization, and quality verification to produce safe, compliant feedstock. Demand is concentrated among large hospitals, contract recyclers, and certified platform operators. Investments prioritize process validation, traceability, and adherence to regulatory requirements rather than rapid capacity expansion. Growth reflects strategic adoption of compliant platforms to meet sustainability targets while ensuring patient safety.
The United Kingdom market for regulatory-compliant medical device plastics recycling platforms is growing at a CAGR of 10.0%, supported by healthcare regulations promoting safe material recovery and circular economy principles. Recycling operators are implementing validated cleaning, sterilization, and verification protocols to produce reusable or downcycled plastics. Demand is concentrated among hospitals and certified platform providers serving the healthcare sector. Investments focus on process standardization, quality assurance, and traceable operations rather than large-scale capacity additions. Growth reflects structured integration of compliant platforms into hospital waste management systems.
China is witnessing strong growth in the regulatory-compliant medical device plastics recycling platforms market at a CAGR of 12.5%, fueled by hospitals and recyclers meeting international compliance expectations. Integrated platforms are used to process single-use medical devices, producing high-quality feedstock suitable for reuse or industrial applications. Demand is concentrated in urban hospital clusters and large healthcare networks. Investments emphasize traceability, process validation, and operational reliability. Growth is shaped by global buyer requirements and environmental regulations rather than domestic mandates alone.
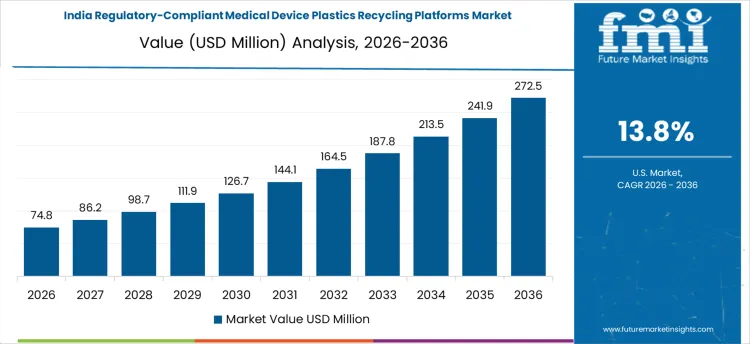
India shows the fastest expansion in regulatory-compliant medical device plastics recycling platforms, growing at a CAGR of 13.8%, driven by increasing hospital capacity and rising usage of single-use medical devices. Recyclers are deploying integrated platforms that combine segregation, sterilization, and verification processes to recover high-quality plastics. Demand is concentrated among organized recyclers and hospital networks engaged in compliance-driven sustainability initiatives. Investments focus on process reliability, contamination control, and certification. Growth reflects both increased device volumes and the need to supply certified recycled feedstock for domestic and international healthcare applications.
Brazil is recording steady growth in the regulatory-compliant medical device plastics recycling platforms market at a CAGR of 11.0%, supported by improved hospital waste management infrastructure and environmental regulation enforcement. Recycling operators are deploying platforms for segregation, cleaning, and sterilization of single-use medical devices to produce safe, compliant feedstock. Demand is concentrated in urban hospitals and healthcare networks with recycling programs. Investments emphasize process standardization, contamination reduction, and traceable documentation rather than rapid expansion. Growth reflects gradual integration of compliant platforms into established hospital waste management systems.
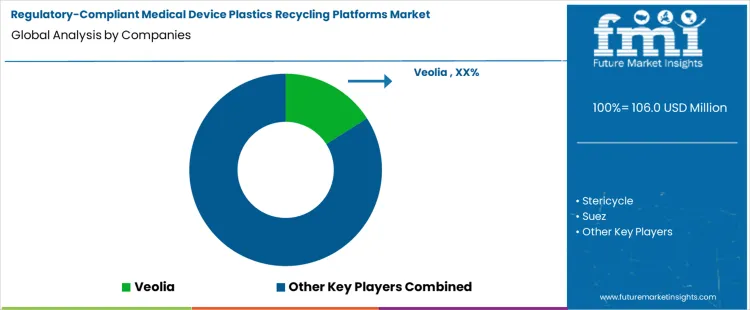
Competition in regulatory-compliant medical device plastics recycling platforms is defined by companies’ ability to manage regulated waste streams, maintain chain of custody, and deliver recycled polymers that meet safety and compliance requirements. Veolia competes through integrated environmental services that link healthcare waste collection with advanced recycling operations, offering customers a unified platform that handles segregation, decontamination, and material recovery. Stericycle focuses on regulated medical waste collection and compliance expertise, developing recycling pathways in coordination with treatment facilities while ensuring strict adherence to clinical waste standards. Suez leverages its logistics network and processing infrastructure to serve multi-site healthcare providers, emphasizing data transparency and audit readiness that support regulatory record-keeping.
Other participants differentiate by service breadth, technology integration, and geography. CleanHarbors integrates decontamination, waste handling, and recycling partnerships to offer a full-service platform that aligns with US regulatory frameworks for clinical waste and plastics recovery. Republic Services brings scale in waste collection and materials processing, often bundling recycling solutions with broader waste management contracts for health systems. Competitors vary in how they balance on-site collection services with off-site recycling capabilities, and in the depth of data reporting they provide to support compliance audits. Firms that offer robust traceability, customized segregation protocols, and documentation systems to meet healthcare regulatory standards tend to secure long-term contracts with medical facilities seeking reliable, compliant recycling solutions.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Platform Type | Closed loop compliant recycling systems, On site hospital segregation & processing, Off site certified recycling services, Automated compliance tracking & reporting |
| Application Focus | Rigid medical plastics, Flexible polymer disposables, Composite device plastics, Sterile packaging polymers |
| Region | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union |
| Key Companies Profiled | Veolia, Stericycle, Suez, Clean Harbors, Republic Services |
| Additional Attributes | Dollar sales by platform type and application, regulatory compliance and certification metrics, audit-ready traceability, process validation for sterilization and polymer integrity, operational integration with hospital workflows, scalability of platforms, risk mitigation, regulatory alignment (ISO, FDA), multi-site deployment capabilities, hospital network coverage |
How big is the regulatory-compliant medical device plastics recycling platforms market in 2026?
The global regulatory-compliant medical device plastics recycling platforms market is estimated to be valued at USD 106.0 million in 2026.
What will be the size of regulatory-compliant medical device plastics recycling platforms market in 2036?
The market size for the regulatory-compliant medical device plastics recycling platforms market is projected to reach USD 312.0 million by 2036.
How much will be the regulatory-compliant medical device plastics recycling platforms market growth between 2026 and 2036?
The regulatory-compliant medical device plastics recycling platforms market is expected to grow at a 11.4% CAGR between 2026 and 2036.
What are the key platform types in the regulatory-compliant medical device plastics recycling platforms market?
The key platform types in regulatory-compliant medical device plastics recycling platforms market are closed‑loop compliant recycling systems, on‑site hospital segregation & processing, off‑site certified recycling services and automated compliance tracking & reporting.
Which application focus segment will contribute significant share in the regulatory-compliant medical device plastics recycling platforms market in 2026?
In terms of application focus, rigid medical plastics segment to command 46.0% share in the regulatory-compliant medical device plastics recycling platforms market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.