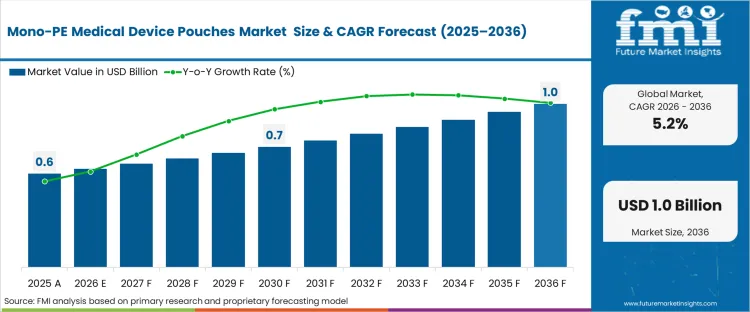
The mono-PE medical device pouches market was valued at USD 0.58 billion in 2025. The sector is expected to reach USD 0.61 billion in 2026 at a CAGR of 5.28% during the forecast period. Sustained investment propels the total opportunity to USD 1.02 billion through 2036 as "Design for Recycling" mandates in healthcare supply chains force a shift away from multi-material laminates toward mono-polymer streams to mitigate the impact of the EU Plastic Levy and escalating national taxes on non-recycled plastic waste.
Procurement directors in the medical device sector are currently navigating a fundamental transition from a protection at any cost mentality to protection within circularity. This decision shift is not merely an environmental preference but a response to hospital-side waste management costs where mixed-material sterile barrier systems are increasingly penalized. The stakes for delay are significant, as non-compliant packaging risks exclusion from large-scale GPO contracts that now weigh sustainability alongside aseptic integrity. The maturation of recyclable packaging solutions has proven that mono-material films can achieve the puncture resistance required for heavy orthopedic implants without relying on nylon or PET tie layers. FMI's analysis suggests that while material costs for advanced mono-PE resins remain stable, the real value is captured through reduced EPR liabilities at the point of end use.
The market enters a self-reinforcing growth phase only once hospital sterile processing departments (SPDs) achieve a critical threshold of waste segregation infrastructure. This gate condition is triggered by the integration of dedicated PE-only collection streams within surgical suites, which justifies the premium paid for mono-material pouches. Once this operational loop is closed, the logistical friction of recycling medical-grade plastics drops significantly, making the next unit of adoption easier than the last.
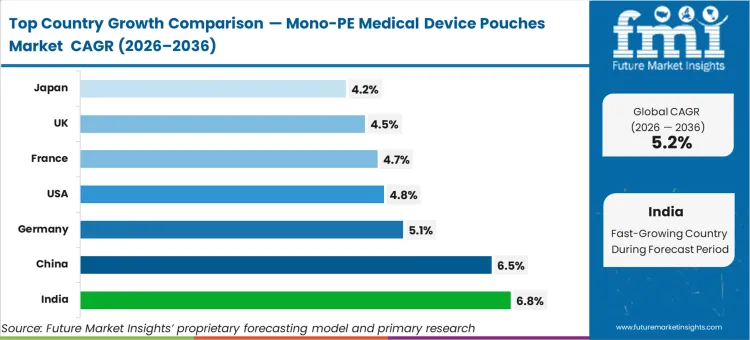
Demand for medical pouches in India is set to grow at a CAGR of 6.8%, driven by domestic manufacturing expansion and export compliance needs. The China market is projected to rise at 6.5% compound annual rate as regional players modernize production lines for high-barrier films. Germany's sector is expected to demonstrate a CAGR of 5.1%, reflecting strict adherence to European packaging waste directives. Sales of mono-PE products in the USA are on track to record a CAGR of 4.8%, while France is likely to post a CAGR of 4.7% through 2036. The UK industry is anticipated to increase by a CAGR of 4.5%, followed by Japan at 4.2% annually. The structural divergence across these rates is governed by the varied pace at which regional healthcare systems are mandating the removal of multi-layer structures from sterile barrier qualifications.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.61 billion |
| Industry Value (2036) | USD 1.02 billion |
| CAGR (2026–2036) | 5.28% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The Mono-PE Medical Device Pouches Market comprises sterile barrier systems constructed entirely from polyethylene-based polymers, including various densities and oriented films. This market is defined by the functional requirement to maintain terminal sterility of medical devices while ensuring the entire pouch structure belongs to a single recycling stream. Unlike traditional multi-material laminates, these pouches eliminate the need for mechanical separation during recycling, focusing on performance parity in seal strength and microbial barrier properties.
This market includes flat, gusseted, and header pouches specifically engineered for medical device sterilization using Ethylene Oxide (EO), Gamma, or E-beam methods. It covers high-performance mono-material packaging films such as MDO-PE and specialized HDPE structures that provide the necessary thermal and mechanical resistance. Inclusion extends to pouches used for surgical instruments, orthopedic implants, and dental tools, provided the primary construction is a mono-polymer polyethylene system.
Exclusions from this market include sterile pouches constructed from mixed-material laminates such as PET/PE, Paper/Film, or Tyvek/Nylon structures where the base polymer is not exclusively polyethylene. Furthermore, standard industrial or food-grade polyethylene pouches that lack the necessary bio-burden controls and medical-grade cleanroom certifications are excluded. Functional components like non-PE heat-seal coatings or paper-based lids that prevent mono-stream recycling are also outside the scope of this analysis.
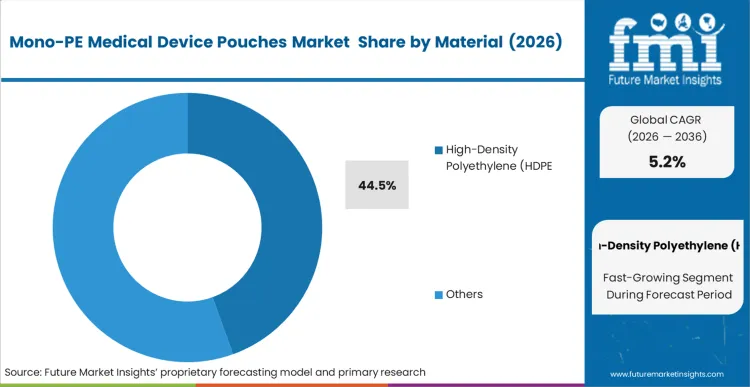
The displacement of multi-material structures is accelerating as incumbents fail to meet the "Design for Recycling" requirements now integrated into hospital procurement scorecards. Material selection is no longer purely a function of barrier properties but an economic hedge against rising waste disposal fees in European and North American markets. According to FMI's estimates, HDPE remains the structural foundation for this transition, as it mimics the mechanical integrity of complex laminates through the use of Biaxially Oriented Polyethylene (BOPE) or MDO-PE, eliminating the recycling penalty of traditional PET/PE structures. Packaging engineers are increasingly specifying oriented PE structures to ensure that large surgical kits do not compromise the pouch during high-vibration transport cycles. The decision to move to medical grade pe resins is often triggered when an OEM initiates a global product refresh, allowing for a single global validation of the sterile barrier. Organizations that cling to legacy laminates face a fragmented regulatory landscape where their packaging is treated as a liability rather than a carrier.
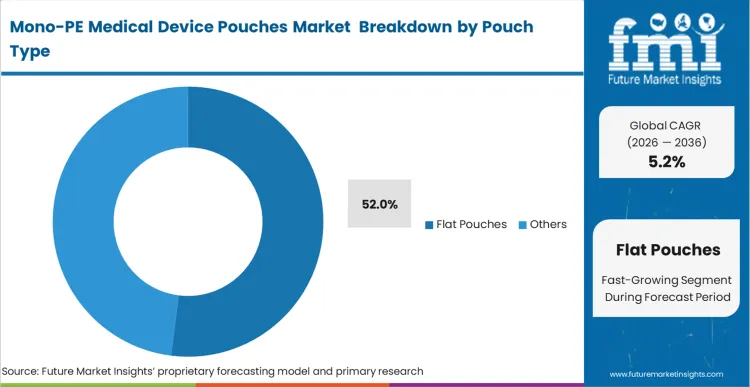
The dominance of flat pouches comes down to a structural logic of high-volume manufacturing: they are the most efficient format for high-speed automated filling lines used for standard surgical disposables. This share figure of 52.0% is not just a legacy preference but a result of the format’s compatibility with established validation protocols in sterile barrier systems. According to FMI's projection, the conversion to pouches market solutions that are 100% recyclable is most advanced in this segment because the flat geometry minimizes the stress on mono-material seals. As manufacturers transition, they are focusing on the heat-seal window, which is narrower for polyethylene than for PET-based laminates. A minor deviation in temperature can lead to seal thinning, a failure mode that is particularly critical for medical devices. The operational consequence of a seal breach is an immediate loss of sterility, necessitating the re-processing or disposal of expensive medical assets.
The decision to adopt mono-PE for surgical instruments is driven by the sheer volume of waste generated by high-frequency surgical procedures in acute care settings. This application segment is the primary target for sustainability initiatives because it offers the highest potential for measurable recycling impact. FMI analysts opine that surgical device manufacturers are being forced to decide between maintaining legacy packaging or restructuring their entire validation file to meet the procurement standards of "green hospitals." The transition is often led by the surgical device packaging sector, where the move toward mono-material streams provides a clear competitive advantage in tender processes. Delaying this transition complicates the regulatory pathway in regions like the EU, regions like the EU, where the Packaging and Packaging Waste Regulation (PPWR) increasingly mandates specific recyclability targets for healthcare primary packaging.
The consequence of hospital systems failing to adopt mono-material waste streams is a direct increase in variable operating costs associated with clinical waste disposal. Hospitals are no longer passive recipients of packaging; they are active influencers of the supply chain, demanding healthcare packaging that aligns with their internal carbon-neutral goals. FMI's view is that the adoption sequence starts with large academic medical centers that have the infrastructure for dedicated plastic recycling. These facilities are forcing a change in the procurement cycle, where the "recyclability" of a pouch is weighed as heavily as its unit price. Diagnostic labs and smaller surgical centers are expected to follow as the secondary ecosystem for medical-grade plastic recycling matures, making it easier to manage mono-polymer waste.
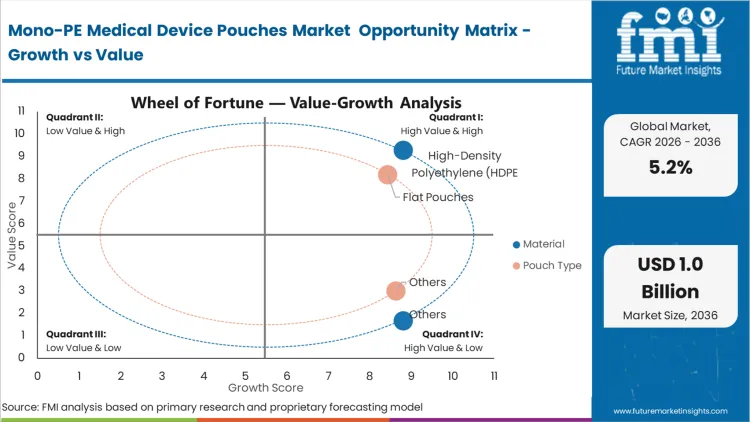
The primary driver for this market is the structural shift in how healthcare systems quantify the "total cost" of a medical device, including its end-of-life disposal. Regulatory frameworks such as the EU Packaging and Packaging Waste Regulation (PPWR) are forcing medical device manufacturers to move away from multi-material laminates that are traditionally incinerated as medical waste. This forcing condition compels procurement directors at global device firms to initiate large-scale sterilization pouches market conversion projects. The commercial stakes are high: failing to provide a recyclable packaging format can lead to the loss of preferred supplier status in highly regulated European and North American hospital networks.
The primary restraint in the market is the structural "qualification inertia" that defines medical device manufacturing. Unlike food packaging, a material change in a sterile pouch requires a full re-validation of the sterile barrier system according to ISO 11607 standards, which can take 12 to 18 months. This friction is not just about the cost of the film, but the organizational resources required for stability testing, shipping trials, and regulatory filings. Furthermore, the existing installed base of heat-sealing equipment in many plants is optimized for PET/PE laminates, which have a much wider sealing window than mono-material polyethylene. This technical gap creates a barrier for smaller manufacturers who lack the capital to upgrade to precision-controlled sealing lines.
Based on the regional analysis, the Mono-PE Medical Device Pouches market is segmented into North America, Europe, Asia Pacific, Latin America, Middle East, and Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 6.8% |
| China | 6.5% |
| Germany | 5.1% |
| USA | 4.8% |
| France | 4.7% |
| UK | 4.5% |
| Japan | 4.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Infrastructure-led growth defines the Asia Pacific region, particularly in hubs where new medical device plants are being built with integrated sustainable packaging capabilities from the start. Unlike older facilities in Western markets that must undergo expensive retrofitting, Asian manufacturers are leapfrogging legacy laminates to meet the stringent "Design for Recycling" requirements of their export customers in Europe. According to FMI's assessment, the rapid decentralization of medical device manufacturing is creating a surge in demand for high-barrier mono-PE films that can survive long-haul sea freight. Procurement directors in China and India are increasingly prioritizing hdpe packaging solutions that offer both mechanical protection and compliance with global circularity standards.
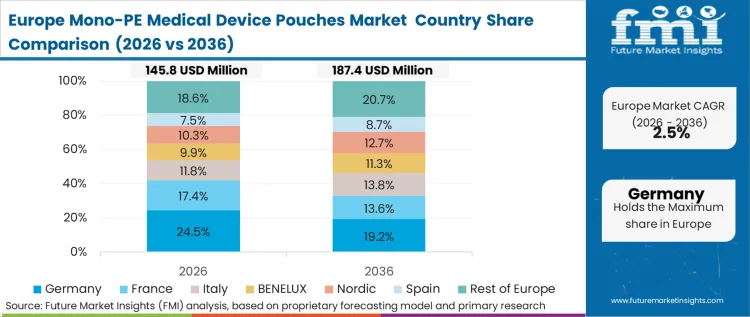
Policy-led dynamics dominate the European landscape, where the updated Packaging and Packaging Waste Regulation (PPWR) creates a direct economic penalty for non-recyclable sterile barrier systems. This regulatory environment makes the transition to mono-PE a commercial necessity rather than an environmental choice for medical device firms operating in the region. Based on FMI's assessment, the "incineration tax" applied to mixed-material medical waste is a significant driver for medical pouches market conversion in high-volume hospitals. Clinical waste managers are actively seeking to divert sterile packaging from the red-bag waste stream into the plastic recycling loop to meet national sustainability quotas.
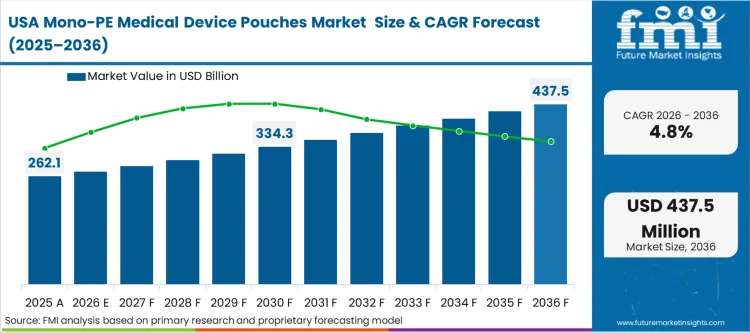
Buyer behavior-led shifts are the primary catalyst in North America, where the consolidation of healthcare systems into massive Group Purchasing Organizations (GPOs) is centralizing the demand for sustainable solutions. GPOs are increasingly adding "environmental impact" as a weighted criteria in their multi-year sourcing reviews for sterile disposables. In FMI's view, the North American aseptic packaging market is responding to this pressure by investing in resin technologies that bridge the performance gap between mono-PE and legacy laminates.
FMI's report includes detailed analysis of Canada, Brazil, Mexico, and other emerging markets. The structural pattern across these additional regions shows that mono-material adoption tracks closely with the export-readiness of local device manufacturers.
The structural concentration of the Mono-PE Medical Device Pouches Market is high because the expertise required to produce medical-grade oriented films is limited to a small group of tier-1 converters. Buyers distinguish qualified vendors not on unit price, but on their ability to provide a complete validation package that includes stability data for mono-material seals. Leading companies like Amcor plc, Mondi Group, and Berry Global Inc. have established dominance by investing in proprietary MDO-PE lines that produce films with the mechanical and thermal properties necessary for sterile barrier systems.
Incumbents maintain a significant advantage through their established relationships with medical device OEMs and their deep understanding of ISO 11607 compliance. A challenger must build more than just a production line; they must develop a technical support infrastructure capable of helping buyers navigate the 18-month requalification cycle. The primary variable buyers use to distinguish vendors is the "sealing window" consistency of their films, as mono-PE requires much tighter process control than traditional PET/PE laminates. One FMI hyperlink within a mechanism sentence flexible packaging solutions indicates that winners in this space are those who provide the material and the sealing expertise as a bundled service.
Large buyers like global surgical device brands are increasingly resisting vendor lock-in by demanding that mono-PE structures be cross-compatible with multiple equipment manufacturers. The structural tension between these buyer preferences and the incentive for vendors to create proprietary, high-margin film structures will define the competitive landscape toward 2036. The market is expected to remain concentrated as the capital requirements for medical-grade cleanroom extrusion and high-precision testing facilities continue to rise, favoring players with global scale.
| Metric | Value |
|---|---|
| Quantitative Units | USD 0.61 billion to USD 1.02 billion, at a CAGR of 5.28% |
| Market Definition | Sterile barrier systems constructed from 100% polyethylene resins, designed to maintain device sterility while allowing for circular economy recycling in a single polymer stream. |
| Material Segmentation | High-Density Polyethylene (HDPE), Low-Density Polyethylene (LDPE), Linear Low-Density Polyethylene (LLDPE) |
| Pouch Type Segmentation | Flat Pouches, Gusseted Pouches, Header Pouches |
| Application Segmentation | Surgical Instruments, Dental Tools, Orthopedic Implants, Cardiovascular Devices |
| End Use Segmentation | Hospitals, Ambulatory Surgical Centers, Diagnostic Labs |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East, Africa |
| Countries Covered | India, China, Germany, USA, France, UK, Japan, and 40 plus countries |
| Key Companies Profiled | Amcor plc, Mondi Group, Berry Global Inc., Huhtamaki Oyj, Constantia Flexibles, ProAmpac, Sealed Air Corporation, Winpak Ltd., Oliver Healthcare Packaging, SteriPack Group |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews were conducted with sterile barrier engineers and procurement directors. Baseline sizing utilized unit consumption data from surgical settings, validated against resin trade volumes and OEM sustainability roadmaps. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Mono-PE Medical Device Pouches Market in 2026?
The market is estimated to reach USD 0.61 billion in 2026. This figure signals a fundamental shift in the healthcare packaging sector as manufacturers move away from incineration-prone mixed materials toward circular mono-polymer streams.
What will it be valued at by 2036?
The industry is projected to cross USD 1.02 billion by 2036. This valuation reflects the complete integration of recyclable sterile barrier systems into global healthcare supply chains, particularly as EPR penalties for multi-material waste become standardized.
What CAGR is projected for the Mono-PE Medical Device Pouches Market?
The market is expected to grow at a CAGR of 5.28% between 2026 and 2036. This rate is anchored to the 18-month qualification cycles of medical device OEMs, which prevents immediate surges but ensures a steady, structural transition toward sustainable formats.
Which Material segment leads the market?
High-Density Polyethylene (HDPE) is expected to hold 44.5% share in 2026. This leadership is due to HDPE's inherent stiffness and puncture resistance, which allow it to replace multi-layer laminates in surgical tool applications without compromising the integrity of the sterile barrier.
Which Pouch Type segment leads the market?
Flat Pouches are projected to grab 52.0% share in 2026. This segment leads because the flat geometry is the most compatible with high-speed automated filling lines and established sterile barrier validation protocols for high-volume surgical disposables.
Which Application segment leads the market?
Surgical Instruments lead the market due to the high turnover of sterilized kits in acute care settings. This segment is the primary target for hospital sustainability initiatives because it generates the highest volume of plastic waste that can be diverted into PE recycling streams.
What drives rapid growth in the Mono-PE Medical Device Pouches Market?
Growth is primarily driven by "Design for Recycling" mandates within hospital GPO contracts, which prioritize mono-material packaging to reduce clinical waste costs. This structural pressure forces device manufacturers to requalify their sterile barriers to maintain access to major healthcare networks.
What is the primary restraint for the Mono-PE Medical Device Pouches Market?
The primary restraint is the structural qualification inertia inherent in the healthcare industry. A change in pouch material requires a full ISO 11607 validation, a process that demands significant organizational resources and time, slowing the adoption rate despite clear sustainability benefits.
Which country grows fastest in the Mono-PE Medical Device Pouches Market?
India is the fastest-growing market with a 6.8% CAGR. This rapid pace, compared to China’s 6.5%, is due to the greenfield nature of India’s new medical device manufacturing zones, which are adopting mono-PE lines immediately to ensure export compliance for regulated Western markets.
How does ISO 11607-1 compliance affect the transition to Mono-PE?
Compliance with ISO 11607-1 requires that the sterile barrier system maintains integrity through the entire distribution and sterilization cycle. Mono-PE films must be engineered with specific orientation to match the mechanical properties of legacy laminates while navigating a much narrower heat-sealing window during production.
What is the role of MDO-PE in the medical pouches market?
Machine Direction Orientation (MDO) technology allows polyethylene to achieve clarity and stiffness comparable to polyester. This enables the creation of all-PE pouches that allow for visual inspection of the device, a critical requirement for clinical safety and aseptic presentation in the OR.
How are hospital procurement directors influencing packaging decisions?
Procurement directors are increasingly weighing "end-of-life cost" alongside unit price, favoring packaging that can be diverted from expensive medical waste streams. This behavior forces vendors to justify their material choices through the lens of hospital-side waste management efficiency.
Is Mono-PE compatible with steam sterilization?
Mono-PE requires specialized resin blends and oriented structures to withstand the high temperatures of steam sterilization without excessive shrinkage or seal creep. While traditionally used for EO or Gamma, Standard Mono-PE is generally optimized for EO and Gamma; however, specialized high-heat HDPE blends and cross-linked resins are currently being qualified for 121°C gravity steam cycles.
What are the consequences for OEMs who delay the move to mono-material pouches?
OEMs who delay face the risk of exclusion from "green tenders" in European and North American hospital systems. As GPOs move toward carbon-neutral supply chains, non-recyclable sterile barriers are being categorized as high-liability items that complicate a hospital's internal sustainability reporting.
Does the conversion to Mono-PE require new sealing equipment?
While not always requiring entirely new machines, the transition often necessitates high-precision temperature control upgrades. Polyethylene has a much narrower thermal sealing window than legacy laminates, making process stability critical to avoiding sterile barrier failure.
What is the expected growth rate for the German market?
The German market is projected to grow at 5.1% CAGR. This rate reflects the high maturity of Germany's recycling infrastructure and the early adoption of extended producer responsibility mandates that penalize mixed-material medical packaging.
How does the US market differ from Europe in its adoption of Mono-PE?
In the USA, adoption is driven primarily by GPO procurement preferences and corporate sustainability goals, whereas in Europe, growth is heavily mandated by national and regional packaging waste directives that include financial penalties for non-recyclable waste.
Can Mono-PE pouches provide adequate puncture resistance for orthopedic implants?
Yes, high-density MDO-PE films are specifically designed to provide the mechanical toughness required for heavy orthopedic implants. These films are often used in multi-layer PE structures that provide the necessary gauge while remaining in a single-polymer recycling stream.
What is the focus of competitive players in this market?
Competitive players are focusing on providing a "bundled validation" service, helping OEMs manage the complex testing required to prove that mono-PE seals remain sterile through the entire product shelf life. This technical support is a key differentiator in a high-stakes regulated market.
Are there exclusions to the mono-material trend?
Exclusions include multi-material pouches using paper or Tyvek lids that are not compatible with PE-stream recycling. While these remain common, the market shift is toward finding mono-polymer lids or all-PE header pouches to achieve a fully recyclable sterile barrier.
What is the structural trajectory of the market toward 2036?
The market is heading toward a state where mono-PE becomes the default standard for high-volume surgical disposables. By 2036, the structural tension between environmental goals and validation costs will be resolved through standardized testing protocols that lower the barrier to mono-material adoption.
How does FMI validate the data for the Mono-PE medical pouches report?
FMI validates sizing and growth data by triangulating resin production volumes from medical-grade suppliers with the annual surgical procedure rates in key regional markets. This approach is further cross-checked with the disclosed packaging reduction targets of global healthcare giants.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.