Chemical Recycling Technologies for Medical-Grade Plastics Market
Chemical Recycling Technologies for Medical-Grade Plastics Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Chemical Recycling Technologies for Medical-Grade Plastics Market Forecast and Outlook 2026 to 2036
The chemical recycling technologies for medical grade plastics market is valued at USD 640 million in 2026 and is projected to reach USD 1,960 million by 2036, reflecting a CAGR of 11.8%. Growth over this period is closely tied to regulatory oversight governing material purity, traceability, and end use approval in healthcare settings. High compliance costs related to validation testing, contamination control, and documentation raise entry thresholds. Certification requirements for medical applications extend qualification timelines and increase capital exposure. Firms with established regulatory pathways progress faster through approval cycles. Smaller technology providers face longer commercialization horizons, affecting adoption rates and limiting early scale deployment.
Between 2026 and 2036, vendor positioning is shaped by the ability to manage certification complexity rather than processing capacity alone. Healthcare buyers prioritize regulatory continuity and audit readiness, reducing tolerance for uncertified outputs. Suppliers offering validated processes secure stronger contract visibility and pricing stability. Compliance driven operating costs remain elevated but restrict competitive entry. Value concentrates among firms integrating regulatory management into technology deployment, allowing customers to mitigate approval risk. As standards converge across regions, differentiation shifts toward reliability of certification maintenance. Profit pools favor vendors aligning process controls with medical compliance frameworks while sustaining predictable operating margins over long contract cycles.
Quick Stats for Chemical Recycling Technologies for Medical Grade Plastics Market
- Chemical Recycling Technologies for Medical Grade Plastics Market Value (2026): USD 640 million
- Chemical Recycling Technologies for Medical Grade Plastics Market Forecast Value (2036): USD 1,960 million
- Chemical Recycling Technologies for Medical Grade Plastics Market Forecast CAGR (2026 to 2036): 11.8%
- Leading Type in Chemical Recycling Technologies for Medical Grade Plastics Market: Sterile grade chemical recycling technologies
- Key Growth Regions in Chemical Recycling Technologies for Medical Grade Plastics Market: Asia Pacific, Europe, North America
- Top Players in Chemical Recycling Technologies for Medical Grade Plastics Market: Eastman, BASF, Wanhua Chemical, GAIL, Teijin, Dow
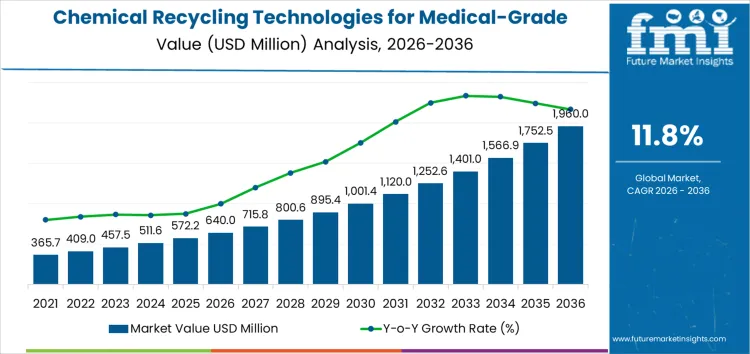
What Is the Growth Forecast for Chemical Recycling Technologies for Medical Grade Plastics Market through 2036?
From 2026 to 2031, the chemical recycling technologies for medical grade plastics market increases from USD 640 million to USD 997 million, reflecting a steady rolling CAGR supported by gradual expansion in regulated recycling capacity. Annual values rise sequentially from USD 414 million to USD 640 million, indicating controlled compounding as adoption progresses. Historical growth was limited by regulatory complexity, validation timelines, and restricted acceptance of recycled content in medical applications. In this phase, rolling growth is shaped by pilot to commercial scale transitions, growing interest from medical device manufacturers, and early integration into closed loop hospital and pharmaceutical waste streams.
Between 2031 and 2036, the market expands from USD 997 million to USD 1,960 million, resulting in a materially higher rolling CAGR. Annual values advance from USD 1,110 million to USD 1,960 million, reflecting accelerating compounding over successive years. Earlier growth emphasized feasibility and compliance validation, while future expansion is driven by larger scale facilities, broader regulatory approvals, and stronger alignment with medical grade polymer supply chains. Rising processing volumes, standardized recycling protocols, and increased participation from healthcare packaging and device producers support faster rolling CAGR progression during the latter period.
Chemical Recycling Technologies for Medical Grade Plastics Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 640 million |
| Forecast Value (2036) | USD 1,960 million |
| Forecast CAGR (2026 to 2036) | 11.8% |
What Is Driving Growth in the Chemical Recycling Technologies for Medical-Grade Plastics Market?
Growth in the chemical recycling technologies for medical-grade plastics market stems from a combination of performance demands unique to healthcare applications and increasing expectations for circular material management. Conventional mechanical recycling methods often degrade polymer properties, making recycled outputs unsuitable for high-purity, high-performance medical uses such as sterile packaging, surgical trays, and device components. Medical-grade plastics require stringent chemical and physical consistency, traceability, and absence of contaminants, criteria that mechanical recycling cannot reliably meet.
Chemical recycling technologies such as solvolysis, pyrolysis, and depolymerization break complex medical waste plastics into monomers or intermediate chemicals that can be reprocessed into materials with properties and purity comparable to virgin resins.
The market’s future growth is also shaped by environmental policy initiatives and healthcare sector commitments to reduce waste footprint without jeopardising patient safety. Regulatory frameworks in many regions are increasing pressure to divert plastic waste from incineration and landfill, including controlled medical waste where feasible, and to incorporate recycled content into product supply chains.
Healthcare systems and device manufacturers are setting circularity targets that include reuse of materials where sterilisation and safety criteria can be met. Improvements in feedstock segregation, contamination control, and chemical recycling process design enhance the economic viability of converting mixed or contaminated medical plastics into high-quality output. Investment in chemical recycling infrastructure capable of handling polyolefins, polycarbonates, and fluoropolymers common in medical applications is expanding as collection and pre-processing systems evolve.
What Factors Are Affecting the Demand for Chemical Recycling Technologies for Medical Grade Plastics Market in Terms of Plastic Type and Technology?
The chemical recycling technologies market for medical grade plastics is driven by rising healthcare waste volumes, stricter disposal regulations, and pressure to recover high quality polymers from contaminated streams. Demand centers on solutions capable of handling biohazard exposure while restoring polymer performance. Market leadership reflects safety validation, regulatory acceptance, and scalability rather than experimentation. Adoption patterns are shaped by plastic composition, contamination intensity, and integration with existing medical waste management systems.
What Is Driving Leadership by Plastic Type in the Chemical Recycling Technologies for Medical Grade Plastics Market?
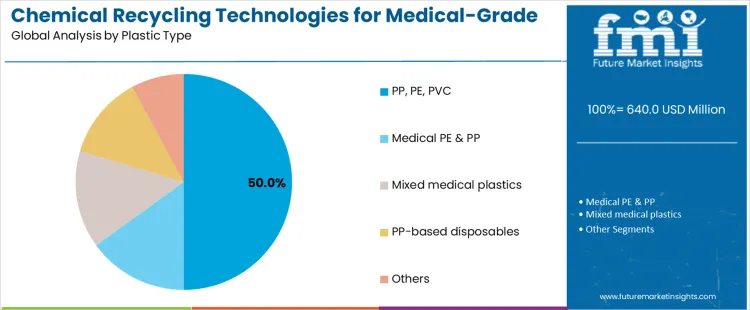
PP, PE, and PVC account for about 50% of demand because these polymers dominate single use medical devices, packaging, and consumables. Syringes, tubing, IV bags, trays, and protective equipment are largely manufactured from these materials, creating a concentrated and recurring waste stream. Their widespread use across hospitals and clinics ensures consistent feedstock availability for recycling technologies. Chemical recycling is favored for these plastics because conventional mechanical recycling struggles with contamination, additive complexity, and performance degradation. The ability to break polymers down to reusable chemical intermediates supports recovery without compromising material integrity. Regulatory frameworks increasingly encourage diversion of these plastics from incineration, reinforcing demand.
Medical grade PE and PP form a strong secondary segment due to their prevalence in disposables requiring sterility and mechanical reliability. Mixed medical plastics also contribute volume but introduce variability that increases processing complexity. PP based disposables are attractive where standardized product design simplifies sorting. Other plastics remain marginal due to lower volumes or challenging compositions. Plastic type leadership therefore reflects waste stream dominance, material uniformity, and feasibility of decontamination rather than diversity of polymer applications within healthcare settings.
What Is Driving Leadership by Technology in the Chemical Recycling Technologies for Medical Grade Plastics Market?
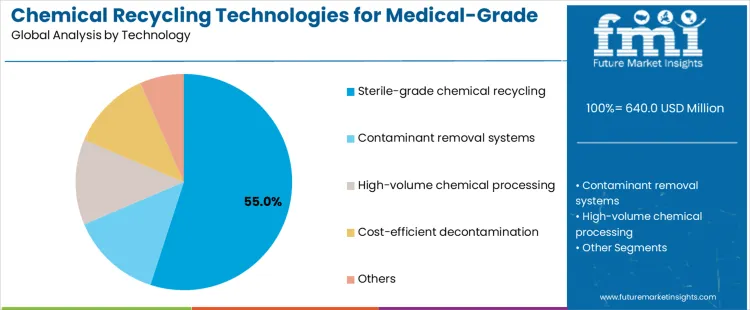
Sterile grade chemical recycling technologies account for about 55% of demand because medical applications require exceptionally high safety and purity standards. These systems combine depolymerization with validated sterilization and contaminant destruction, enabling recovered materials to meet regulatory requirements for reuse. Hospitals and waste processors favor technologies that demonstrably neutralize biological and chemical hazards while producing consistent output. Sterile grade systems align with compliance expectations for handling infectious waste and support closed loop ambitions in regulated healthcare supply chains.
Contaminant removal systems and high volume chemical processing technologies support broader implementation but remain secondary. Decontamination focused systems emphasize pathogen and residue elimination but may not fully restore polymer quality. High volume processing targets scale efficiency but requires controlled feedstock streams. Cost efficient decontamination solutions appeal where reuse standards are lower, such as non-clinical products. Other technologies remain in development or pilot stages. Technology leadership therefore reflects regulatory confidence, safety assurance, and compatibility with medical waste oversight rather than throughput alone.
How Are Chemical Recycling Technologies for Medical-Grade Plastics Being Applied Across Healthcare and Pharmaceutical Sectors?
Use appears in hospitals, laboratories, and pharmaceutical manufacturing where high-purity polymer waste such as polypropylene, polyethylene, and PVC must be recovered safely. Operators adopt chemical recycling to depolymerize medical-grade plastics from syringes, IV sets, and packaging into monomers or oligomers. Recycled polymers are reintegrated into medical devices, protective equipment, and packaging while meeting regulatory standards. These applications reflect operational and compliance priorities rather than aesthetic purposes, with adoption driven by waste reduction, circularity, and safe handling of sensitive materials.
What Operational and Material Conditions Support Adoption of Chemical Recycling for Medical-Grade Plastics?
Selection aligns with facilities that can maintain contamination-free feedstock, precise reaction conditions, and rigorous quality control. Technologies such as hydrolysis, glycolysis, or enzymatic depolymerization are optimized for temperature, catalysts, and residence time to recover high-purity polymers. Operators monitor feedstock composition, sterilization requirements, and downstream purification to meet ISO and FDA standards. Integration with extrusion or polymerization lines ensures recycled materials retain mechanical, chemical, and barrier properties. These conditions emerge from operational efficiency, product integrity, and regulatory compliance priorities in structured recycling workflows.
Which Practical Considerations Limit Wider Use of Chemical Recycling for Medical-Grade Plastics?
High capital expenditure and energy requirements can limit adoption to large-scale or specialized facilities. Feedstock segregation, contamination, and sterilization must be strictly controlled to maintain polymer purity. Regulatory approvals for recycled medical plastics differ across regions and applications. Skilled operators, process monitoring, and continuous quality validation are necessary. These factors lead to selective deployment where material recovery, compliance, and operational feasibility justify incremental cost, infrastructure investment, and technical expertise in sensitive healthcare recycling workflows.
What is the Demand for Chemical Recycling Technologies for Medical Grade Plastics by Country?
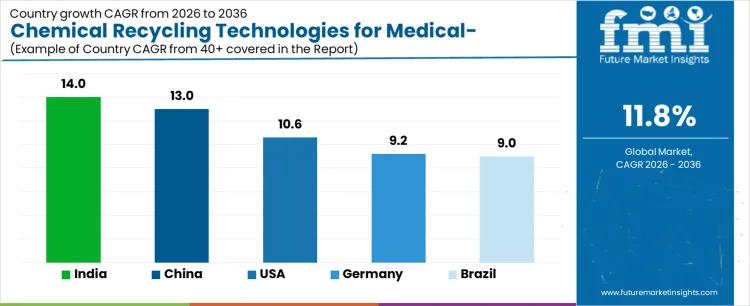
| Country | CAGR (%) |
|---|---|
| USA | 10.6% |
| Germany | 9.2% |
| China | 13.0% |
| India | 14.0% |
| Brazil | 9.0% |
The demand for chemical recycling technologies for medical grade plastics varies across countries, driven by healthcare waste management, sustainability initiatives, and adoption of advanced recycling methods. India leads with a 14.0% CAGR, supported by growing healthcare infrastructure, increasing medical plastic consumption, and rising emphasis on recycling and circular economy practices. China follows at 13.0%, driven by large scale medical device manufacturing, regulatory compliance, and adoption of chemical recycling for high quality recycled plastics. The USA grows at 10.6%, reflecting adoption in hospitals, pharmaceutical production, and medical packaging. Germany records 9.2%, shaped by mature healthcare systems and stringent environmental regulations. Brazil posts 9.0%, supported by developing medical infrastructure and growing recycling initiatives.
How Are Chemical Recycling Technologies Expanding Medical-Grade Plastics Recovery in the United States?
In the United States, the Chemical Recycling Technologies for Medical-Grade Plastics Market is growing at a CAGR of 10.6% through 2036, driven by increasing demand for recycling of post-consumer and post-industrial medical plastics, including syringes, vials, and packaging. Hospitals, medical device manufacturers, and specialized recycling facilities are adopting chemical depolymerization and purification processes to recover high-purity monomers and meet FDA compliance. Compared to Germany, where regulatory oversight is the main driver, the U.S. market balances environmental sustainability with operational efficiency. Domestic suppliers are providing high-performance, modular systems capable of handling recurring medical-grade plastic streams.
- Hospitals, medical device manufacturers, and recycling facilities drive adoption
- Chemical recycling recovers high-purity monomers for medical use
- Domestic suppliers provide modular, high-performance depolymerization systems
- Recurring medical plastic feedstock sustains predictable adoption
How Are Regulatory Compliance and Recycling Standards Driving Growth in Germany?
In Germany, revenue is growing at a CAGR of 9.2% through 2036, supported by strict EU and national regulations on medical waste disposal and recycling. Hospitals, pharmaceutical facilities, and medical device manufacturers are adopting chemical recycling technologies to recover medical-grade plastics, reduce hazardous waste, and maintain regulatory compliance. Demand is concentrated in urban medical hubs, pharmaceutical manufacturing clusters, and specialized recycling facilities. Compared to the USA, adoption is more compliance-driven than operational efficiency-focused. Domestic suppliers provide high-performance recycling technologies designed for traceability, monomer purity, and integration with existing processing infrastructure. Recurring medical plastic streams and regulatory audits sustain steady adoption.
- Urban medical hubs, pharmaceutical clusters, and recycling facilities drive adoption
- Technologies recover medical-grade plastics and ensure compliance
- Domestic suppliers provide high-performance, traceable recycling solutions
- Recurring medical plastic streams sustain steady adoption
How Is Industrial Scale and Healthcare Expansion Driving Adoption in China?
In China, the market is expanding at a CAGR of 13% through 2036, fueled by rapid growth of hospitals, medical device manufacturing, and plastic-intensive healthcare facilities. Chemical recycling technologies are deployed to recover monomers from medical-grade plastics, reduce disposal costs, and produce compliant recycled material. Demand is concentrated in urban medical centers, large pharmaceutical clusters, and specialized recycling plants. Compared to Brazil, adoption focuses on high-volume, urban medical and industrial hubs. Domestic suppliers are scaling modular, high-capacity chemical recycling systems compatible with recurring medical plastic streams. Government incentives and industrial expansion sustain rapid adoption nationwide.
- Urban medical centers, pharmaceutical clusters, and recycling plants drive adoption
- Technologies recover monomers and reduce disposal costs
- Domestic suppliers scale modular, high-capacity recycling systems
- Recurring medical plastic streams sustain rapid adoption
How Is Emerging Healthcare Infrastructure Supporting Growth in India?
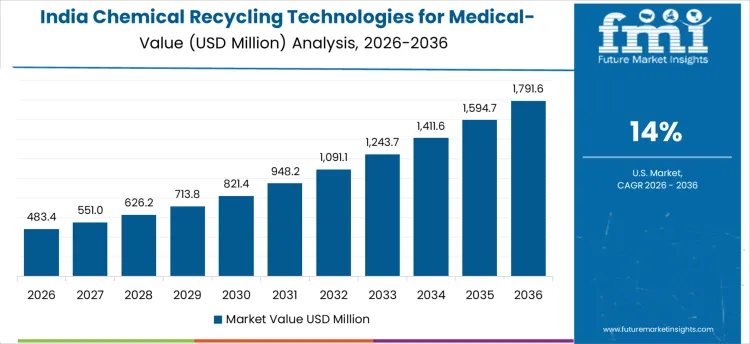
In India, revenue is growing at a CAGR of 14% through 2036, driven by rapid expansion of hospitals, diagnostic centers, and medical device manufacturing units. Facilities are adopting chemical recycling technologies to recover medical-grade plastics, reduce environmental impact, and comply with emerging regulations. Demand is concentrated in urban medical hubs, pharmaceutical manufacturing units, and healthcare-associated recycling facilities. Domestic suppliers provide high-performance, cost-effective systems compatible with diverse plastic streams. Compared to China, adoption is influenced by emerging healthcare zones and government incentives promoting recycling. Recurring plastic waste streams and infrastructure expansion are sustaining robust adoption nationwide.
- Urban medical hubs, pharmaceutical units, and healthcare recycling facilities drive adoption
- Technologies recover medical-grade plastics and reduce environmental impact
- Domestic suppliers provide cost-effective, high-performance systems
- Recurring plastic waste streams sustain robust adoption
How Are Chemical Recycling Hubs Supporting Adoption in Brazil?
In Brazil, the market is growing at a CAGR of 9% through 2036, supported by established chemical recycling hubs and adoption of medical-grade plastics recovery in hospitals and pharmaceutical facilities. Facilities are integrating chemical recycling technologies to recover high-purity monomers, reduce medical waste, and comply with environmental regulations. Demand is concentrated in industrial zones, urban medical centers, and specialized recycling plants. Domestic suppliers provide modular, high-performance recycling systems compatible with recurring medical plastic streams. Compared to India and China, adoption is focused on established hubs rather than rapid industrial or healthcare expansion. Recurring waste streams sustain steady adoption nationwide.
- Industrial zones, urban medical centers, and recycling plants drive adoption
- Technologies recover high-purity monomers and reduce medical waste
- Domestic suppliers provide modular, high-performance systems
- Recurring medical plastic streams sustain steady adoption
Which Strategic Approaches Define Competition in the Chemical Recycling Technologies for Medical Grade Plastics Market through Validation, Risk Control, and Supply Integrity?
Competition in chemical recycling for medical grade plastics is determined first by risk tolerance within regulated healthcare supply chains. Medical device and pharmaceutical packaging users require traceability, contaminant control, and reproducibility that exceed conventional recycling thresholds. Eastman and BASF compete by positioning chemical recycling as an extension of established polymer and specialty chemicals operations, where process control, documentation, and regulatory engagement are already institutionalized.
Their strategies emphasize closed mass balance systems, controlled feedstock sourcing, and alignment with existing medical material approvals. Teijin applies a different lens, prioritizing material purity and molecular consistency to support applications where performance deviation carries clinical risk. Across these players, competitive strength is measured by audit readiness, impurity management, and the ability to support long qualification cycles demanded by medical customers.
A contrasting competitive dynamic emerges among regionally anchored and pilot driven participants. Wanhua Chemical approaches the market through integration with domestic polymer value chains, focusing on technical feasibility and gradual engagement with regulated end uses.
GAIL participates primarily through pilot scale initiatives, where chemical recycling is evaluated as a future compliance and waste management pathway rather than a near term commercial solution. Dow positions its involvement around technology assessment and partnership based exploration, maintaining optionality rather than committing to dedicated medical grade recycling infrastructure. In this market, competitive advantage does not arise from processing volume or speed. It is built through credibility with regulators, controlled scaling, and the ability to demonstrate consistent material behavior across repeated validation cycles.
Key Players in the Chemical Recycling Technologies for Medical Grade Plastics Market
- Eastman
- BASF
- Wanhua Chemical
- GAIL (pilot projects)
- Teijin
- Dow
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Type | Pyrolysis and Depolymerization, Solvent Based Recovery, Hybrid Recycling Systems, Compact Chemical Recyclers, Other Technologies |
| Application | Medical Devices and Disposables, Hospital Waste Recycling, Medical Manufacturing Scrap, Disposable Medical Supplies, High Purity Medical Polymer Recovery |
| End User | Hospitals and Healthcare Facilities, Medical Device Manufacturers, Pharmaceutical Manufacturers, Healthcare Waste Management Operators |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | USA, Germany, China, India, Japan, South Korea, Brazil, United Kingdom, France, Italy, Canada, Mexico, ASEAN countries, GCC countries, and other global regions |
| Key Companies Profiled | Eastman, BASF, Wanhua Chemical, GAIL, Teijin, Dow |
| Additional Attributes | Dollar sales by technology type and plastic type categories, regional adoption trends across Asia Pacific, Europe, and North America, competitive landscape across global and regional chemical recycling providers, regulatory validation and certification requirements for medical grade polymer reuse, integration with hospital waste management systems and medical polymer supply chains |
Chemical Recycling Technologies for Medical Grade Plastics Market Segmentation
By End-Use:
- Medical devices & disposables
- Hospital waste recycling
- Medical manufacturing scrap
- Disposable medical supplies
- Healthcare waste
- High-purity medical polymers
By Technology Type:
- Pyrolysis & depolymerization
- Solvent-based recovery
- Hybrid recycling systems
- Compact chemical recyclers
- Others
By Plastic Type:
- PP, PE, PVC
- Medical PE & PP
- Mixed medical plastics
- PP-based disposables
- Others
By Technology:
- Sterile-grade chemical recycling
- Contaminant removal systems
- High-volume chemical processing
- Cost-efficient decontamination
- Others
By Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- European Commission. (2020). Circular economy action plan: For a cleaner and more competitive Europe. Publications Office of the European Union.
- USA Food and Drug Administration. (2023). Use of recycled plastics in medical devices: Guidance for industry. Center for Devices and Radiological Health.
Frequently Asked Questions
How big is the chemical recycling technologies for medical-grade plastics market in 2026?
The global chemical recycling technologies for medical-grade plastics market is estimated to be valued at USD 640.0 million in 2026.
What will be the size of chemical recycling technologies for medical-grade plastics market in 2036?
The market size for the chemical recycling technologies for medical-grade plastics market is projected to reach USD 1,960.0 million by 2036.
How much will be the chemical recycling technologies for medical-grade plastics market growth between 2026 and 2036?
The chemical recycling technologies for medical-grade plastics market is expected to grow at a 11.8% CAGR between 2026 and 2036.
What are the key product types in the chemical recycling technologies for medical-grade plastics market?
The key product types in chemical recycling technologies for medical-grade plastics market are medical dEVices & disposables, hospital waste recycling, medical manufacturing scrap, disposable medical supplies, healthcare waste and high-purity medical polymers.
Which technology type segment to contribute significant share in the chemical recycling technologies for medical-grade plastics market in 2026?
In terms of technology type, pyrolysis & depolymerization segment to command 45.0% share in the chemical recycling technologies for medical-grade plastics market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-use , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-use , 2026 to 2036
- Medical devices & disposables
- Hospital waste recycling
- Medical manufacturing scrap
- Disposable medical supplies
- Healthcare waste
- High-purity medical polymers
- Medical devices & disposables
- Y to o to Y Growth Trend Analysis By End-use , 2021 to 2025
- Absolute $ Opportunity Analysis By End-use , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology Type, 2026 to 2036
- Pyrolysis & depolymerization
- Solvent-based recovery
- Hybrid recycling systems
- Compact chemical recyclers
- Others
- Pyrolysis & depolymerization
- Y to o to Y Growth Trend Analysis By Technology Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Plastic Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Plastic Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Plastic Type, 2026 to 2036
- PP, PE, PVC
- Medical PE & PP
- Mixed medical plastics
- PP-based disposables
- Others
- PP, PE, PVC
- Y to o to Y Growth Trend Analysis By Plastic Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Plastic Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Sterile-grade chemical recycling
- Contaminant removal systems
- High-volume chemical processing
- Cost-efficient decontamination
- Others
- Sterile-grade chemical recycling
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By End-use
- By Technology Type
- By Plastic Type
- By Technology
- Competition Analysis
- Competition Deep Dive
- Eastman
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- BASF
- Wanhua Chemical
- GAIL (pilot projects)
- Teijin
- Dow
- Eastman
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Technology Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Plastic Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Technology Type, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Plastic Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Technology Type, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Plastic Type, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Technology Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Plastic Type, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Technology Type, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Plastic Type, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Technology Type, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Plastic Type, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Technology Type, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Plastic Type, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Technology Type, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Plastic Type, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by End-use
- Figure 6: Global Market Value Share and BPS Analysis by Technology Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Technology Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Technology Type
- Figure 9: Global Market Value Share and BPS Analysis by Plastic Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Plastic Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Plastic Type
- Figure 12: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Technology
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by End-use
- Figure 29: North America Market Value Share and BPS Analysis by Technology Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Technology Type, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Technology Type
- Figure 32: North America Market Value Share and BPS Analysis by Plastic Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Plastic Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Plastic Type
- Figure 35: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Technology
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End-use
- Figure 42: Latin America Market Value Share and BPS Analysis by Technology Type, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Technology Type, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Technology Type
- Figure 45: Latin America Market Value Share and BPS Analysis by Plastic Type, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Plastic Type, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Plastic Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Technology
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by End-use
- Figure 55: Western Europe Market Value Share and BPS Analysis by Technology Type, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Technology Type, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Technology Type
- Figure 58: Western Europe Market Value Share and BPS Analysis by Plastic Type, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Plastic Type, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Plastic Type
- Figure 61: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Technology
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by End-use
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Technology Type, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Technology Type, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Technology Type
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Plastic Type, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Plastic Type, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Plastic Type
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by End-use
- Figure 81: East Asia Market Value Share and BPS Analysis by Technology Type, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Technology Type, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Technology Type
- Figure 84: East Asia Market Value Share and BPS Analysis by Plastic Type, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Plastic Type, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Plastic Type
- Figure 87: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Technology
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by End-use
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Technology Type, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology Type, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Technology Type
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Plastic Type, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Plastic Type, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Plastic Type
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by End-use
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Technology Type, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Technology Type, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Technology Type
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Plastic Type, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Plastic Type, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Plastic Type
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

