Demand for peripherally inserted central catheters in the UK is projected to be valued at USD 119.2 million in 2026 and is expected to reach USD 157.7 million by 2036, expanding at a 2.8% CAGR. This trajectory is defined by clinical decisions within NHS trusts and private healthcare facilities seeking reliable, cost-effective vascular access for medium to long-term therapies. The adoption curve is shaped by daily patient care protocols, infection control standards, and the strategic planning of infusion services across acute and community settings.
PICC lines are deployed in environments where catheter dwell time, complication rates, and patient comfort directly influence clinical outcomes and departmental efficiency. Procurement teams judge performance through reductions in catheter-related bloodstream infections, improved first-insertion success rates, material biocompatibility, and the device’s ability to withstand specific therapeutic regimens. Clinical leads prioritize products that enhance insertion safety, minimize maintenance interventions, and support a broad range of infusion protocols.
For clinical procurement specialists and vascular access teams, the evaluation extends beyond the catheter itself. Decisions are intrinsically linked to insertion kit compatibility, securement and dressing solutions, clinician training requirements, and the manufacturer’s support for clinical guidelines.
The ability to standardize products across hospital networks, train staff efficiently on insertion techniques, and access reliable clinical evidence supports adoption across large teaching hospitals and smaller community units alike.
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 119.2 million |
| Industry Forecast Value (2036) | USD 157.7 million |
| Forecast CAGR 2026 to 2036 | 2.8% |
Demand in the UK grows as healthcare providers intensify focus on patient safety, therapy delivery efficiency, and the shift of care into community settings. Hospitals face pressure to reduce repeated peripheral cannulations, lower the risk of phlebitis, and provide stable venous access for complex drug regimens. A reliable PICC line supports timely administration of therapies, protects peripheral vasculature, and can facilitate earlier patient discharge with ongoing infusion needs.
Clinical practice evolution and demographic trends influence procurement cycles. An ageing population and increasing prevalence of conditions requiring long-term intravenous therapy underpin steady demand. In this environment, vascular access teams focus on devices that minimize insertion complications, offer proven material safety, and align with UK guidelines on vascular access device management.
Therapy-specific requirements also guide product selection. Oncology units administering vesicant chemotherapy agents prioritize catheters with confirmed compatibility and tip location accuracy. Teams managing patients requiring parenteral nutrition evaluate lumen design and infection risk profiles.
Many procurement teams also cross-reference PICC selections against options for implantable port systems used in long-term oncology care when determining the most appropriate vascular access strategy for a patient’s full treatment pathway.
Procurement decisions are shaped by safety and cost-containment objectives. Emphasis on reducing healthcare-associated infections directs buyers toward products with antimicrobial technologies and securement systems designed to minimize microbial migration.
This pushes clinical buyers to value designs supported by strong clinical outcome data, comprehensive insertion training programs, and a total cost-of-care rationale that accounts for potential complication-related expenses.
The segmentation of the PICC landscape in the UK reflects how clinical teams match device characteristics to specific therapeutic needs, patient populations, and care settings.
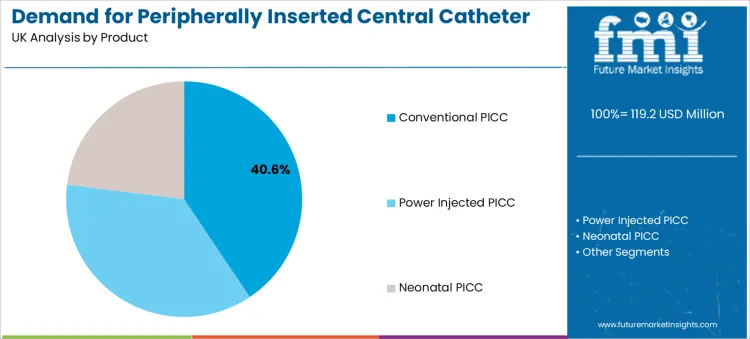
Conventional PICC lines hold a 40.6% share as the leading product type. This reflects their established role as a versatile, cost-effective solution for a wide array of intravenous therapies requiring secure central access. For hospital procurement, these devices represent a reliable standard of care with which clinical staff have extensive familiarity, supporting consistent insertion and maintenance protocols across diverse departments.
Their sustained use is often seen in settings where power-injection capability is not a routine requirement for the patient population. Evaluation of these devices frequently connects to broader procurement strategies for vascular access devices that improve patient comfort.
Single lumen PICCs account for a 52.0% share, indicating that a majority of clinical requirements are for single-therapy infusion or intermittent medication delivery. This configuration is often selected for its lower infection risk profile relative to multi-lumen devices, simpler maintenance, and smaller catheter diameter, which can be beneficial for patient vasculature.
This segment fits a high volume of patients receiving extended antibiotic courses, hydration therapy, or specific chemotherapy regimens not requiring concurrent infusion. The choice of lumen number is a fundamental part of clinical decision-making for intravenous drug delivery systems.
Chemotherapy represents a 34.7% share as the leading application. The mandatory need for secure, reliable venous access for often prolonged and frequent cycles of vesicant or irritant drugs makes PICCs a cornerstone of oncology care. The primary clinical driver is patient safety, ensuring cytotoxic agents are delivered centrally to minimize tissue damage and extravasation risk.
Oncology teams prioritize catheter designs with strong evidence for chemical compatibility and mechanical stability. This application area is closely linked to technologies for safe drug delivery in oncology settings.
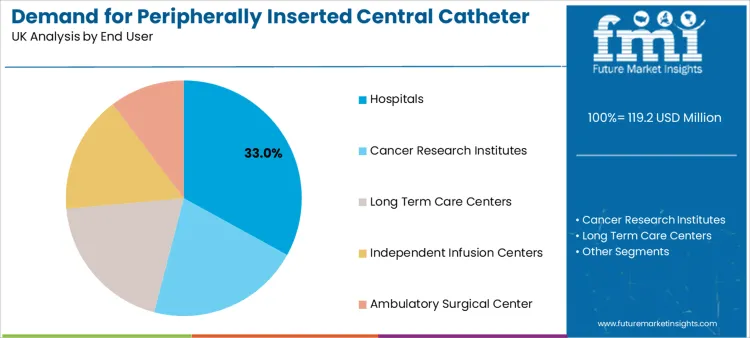
Hospitals represent a 33.0% share as the leading end-user segment. Acute care settings are where the majority of PICC insertions occur, driven by inpatient initiation of long-term therapies, management of complex infections, and perioperative nutritional support.
High-volume purchasing, stringent tendering processes, and a requirement for products that integrate seamlessly with existing insertion protocols and ward-based care characterize hospital procurement. Investment in hospital PICCs is frequently evaluated alongside needs for complementary infusion pump technology to ensure a fully compatible therapy delivery ecosystem.
Patient safety and service efficiency are primary drivers. Clinical governance leads demand devices with low thrombosis and infection rates. Procurement teams seek products that simplify inventory and standardize care pathways. Finance departments evaluate total treatment cost, favoring devices that reduce complication-related expenditures.
Vascular access nurses prioritize products that enhance insertion success and patient comfort. Regulatory compliance remains a constant dynamic, with adherence to Medicines and Healthcare products Regulatory Agency (MHRA) regulations underpinning procurement safety standards.
Budgetary constraints within NHS trusts create a significant barrier, often favoring familiar, cost-led products over premium-priced alternatives with advanced features. Clinical inertia and the need for comprehensive re-training on new insertion techniques for different device designs can delay uptake.
The complexity of demonstrating clear cost-benefit superiority in real-world settings can hinder adoption. Procurement is heavily influenced by the need for robust clinical evidence and health economic analysis before switching established product portfolios.
Differentiation through clinical evidence and service is a major opportunity. Manufacturers that provide comprehensive outcome data, hands-on simulation training for inserters, and dedicated clinical support specialists build loyal adoption.
Another opportunity exists in developing integrated solutions that bundle the catheter with ultrasound-guided insertion systems and securement/dressing products to improve overall procedure success and durability. Growth in community-based care presents an opportunity for designs facilitating easier patient self-care or community nurse management.
Competition from alternative vascular access devices such as midline catheters for shorter-term needs or totally implantable ports for very long-term therapy, can segment demand. Significant downward pressure on healthcare procurement budgets can freeze or slow device upgrades.
Any high-profile safety concerns or product recalls related to catheter materials or design can severely damage trust in a specific brand or technology, shifting demand rapidly. This reinforces the critical importance of post-market surveillance and transparent incident reporting.
Regional adoption varies based on the concentration of specialist clinical services, oncology center locations, demographic health needs, and the maturity of community-based IV therapy programs.
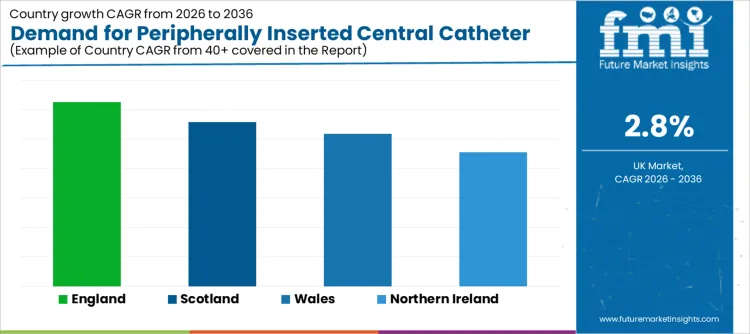
| Region | CAGR 2026 to 2036 |
|---|---|
| England | 3.1% |
| Scotland | 2.8% |
| Wales | 2.6% |
| Northern Ireland | 2.3% |
England grows at 3.1%, supported by its high density of major oncology centers, large teaching hospitals with active vascular access teams, and well-established ambulatory care pathways. Larger procurement frameworks and greater access to clinical specialist training foster confidence in adopting and standardizing devices. The scale of patient volume encourages investment in products that improve efficiency and reduce variation across regional networks.
Scotland expands at 2.8%, shaped by its integrated national health service structure and the need to deliver complex care across geographically dispersed populations. Procurement decisions often emphasize product reliability, strong clinical evidence, and designs that are suitable for use in both central hospitals and more remote community settings, supporting patient management closer to home.
Wales grows at 2.6%, driven by health board-level procurement strategies that aim to standardize care. There is a focus on selecting devices that balance clinical performance with cost-effectiveness, supporting predictable budgeting across acute and community services. Adoption is linked to the development of unified clinical guidelines for vascular access.
Northern Ireland rises at 2.3%, reflecting a careful, evidence-based approach within its integrated health and social care system. Procurement teams frequently prioritize products with a strong proven track record, clear benefits for patient safety, and reliable supplier support for training and clinical queries, ensuring sustainable implementation within existing care structures.
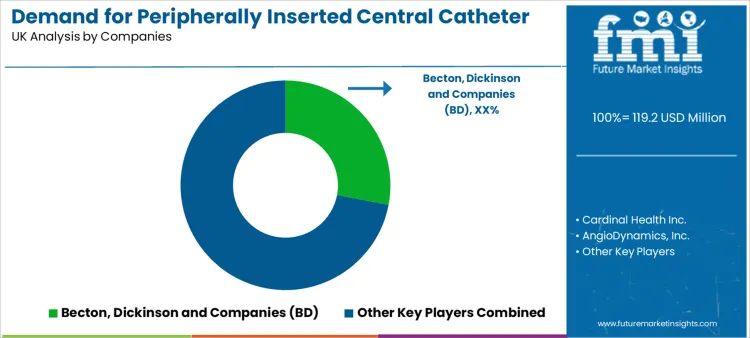
Clinical evidence, product reliability, strength of clinical education programs, and the depth of technical support shape competition. Consumers evaluate suppliers on practical factors such as catheter material safety data, insertion success rates supported by real-world evidence, compatibility with a wide range of therapeutics, and the vendor’s ability to support continuous clinical training.
Becton, Dickinson and Company (BD) is associated with a comprehensive vascular access portfolio and strong emphasis on clinical education and procedural safety. Cardinal Health Inc. competes through a broad range of medical device offerings, including PICCs designed for clinical efficiency and patient comfort. AngioDynamics, Inc. is positioned with specialized vascular access and interventional products, often featuring advanced material technologies.
Teleflex Incorporated provides a wide array of PICCs, including power-injectable models, supported by procedural kits and educational resources. Vygon Group contributes to the landscape with a focus on specialized catheters, including neonatal PICCs, catering to specific patient population needs.
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| Product | Conventional PICC; Power Injected PICC; Neonatal PICC |
| Lumen | Single Lumen; Double Lumen; Multiple Lumen |
| Application | Chemotherapy; Long-term IV Infusion; Parenteral Nutrition; Blood Transfusion; Difficult Intravenous Access |
| End User | Hospitals; Cancer Research Institutes; Long Term Care Centers; Independent Infusion Centers; Ambulatory Surgical Center |
| Regions Covered | England; Scotland; Wales; Northern Ireland |
| Key Companies Profiled | Becton, Dickinson and Company (BD); Cardinal Health Inc.; AngioDynamics, Inc.; Teleflex Incorporated; Vygon Group |
How big is the demand for peripherally inserted central catheter in uk in 2026?
The demand for peripherally inserted central catheter in uk is estimated to be valued at USD 119.2 million in 2026.
What will be the size of peripherally inserted central catheter in uk in 2036?
The market size for the peripherally inserted central catheter in uk is projected to reach USD 157.7 million by 2036.
How much will be the demand for peripherally inserted central catheter in uk growth between 2026 and 2036?
The demand for peripherally inserted central catheter in uk is expected to grow at a 2.8% CAGR between 2026 and 2036.
What are the key product types in the peripherally inserted central catheter in uk?
The key product types in peripherally inserted central catheter in uk are conventional picc, power injected picc and neonatal picc.
Which lumen segment is expected to contribute significant share in the peripherally inserted central catheter in uk in 2026?
In terms of lumen, single lumen segment is expected to command 52.0% share in the peripherally inserted central catheter in uk in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.