The vaginal yeast infections treatment market begins 2026 at USD 4.4 billion and is projected to reach USD 7.7 billion by 2036 at a 5.70% CAGR. Adoption is driven by prevalence, treatment route, and patient convenience rather than brand visibility. Clinicians select therapy based on efficacy, safety, formulation type, and dosing schedule. Once a protocol is standardized in clinics or retail pharmacies, switching products requires new patient counseling and clinical oversight. Pharmacists monitor stock levels, expiry dates, and regulatory compliance. Patient adherence and comfort influence therapy choice, while procurement ensures steady supply and documentation.
Market growth follows incidence trends, clinical guideline updates, and retail accessibility rather than promotional campaigns. Manufacturers focus on formulation uniformity, stability, and packaging integrity. Release requires quality checks for potency, microbial safety, and labeling accuracy. Distribution is planned around pharmacy chains and regional health centers to prevent stockouts. Training, education, and awareness programs support proper usage and adherence. Profitability depends on predictable reorder patterns, low waste, and consistent demand rather than aggressive pricing or frequent reformulation. Expansion aligns with new product approvals, patient outreach, and broader healthcare coverage.
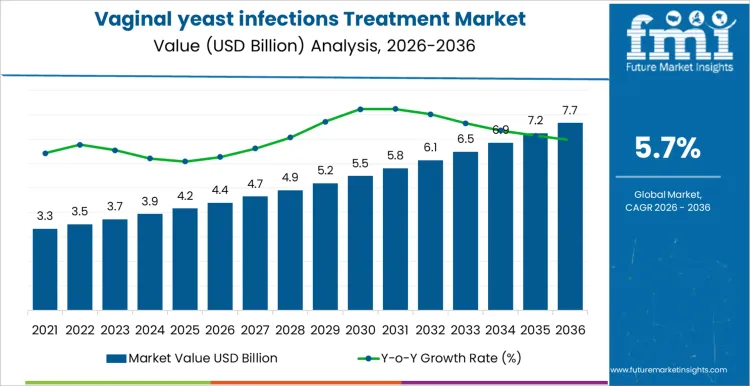
Patient education initiatives and treatment guideline adoption drive growth in the vaginal yeast infections treatment market more than total gynecological clinic visits. In 2026, at about USD 4.4 billion, demand is concentrated in outpatient gynecology centers, women’s health clinics, and telehealth platforms where therapy selection is guided by severity, recurrence risk, and patient preference. Uptake is influenced by product convenience, formulation type, and over-the-counter availability. Expansion occurs as awareness campaigns encourage earlier diagnosis, adherence to treatment regimens improves, and self-care options gain traction. Consumer-led uptake trends track closely with the wider yeast infection treatment segment, especially where self-medication is common. Growth reflects gradual integration of effective therapies into standard care pathways rather than sudden surges in patient numbers.
Distribution strategy and product accessibility shape the later phase of the vaginal yeast infections treatment market. As value approaches roughly USD 7.7 billion by 2036, buyers focus on multi-channel availability, consistent formulation quality, and patient support programs. Manufacturers that invest in direct-to-consumer education, pharmacy partnerships, and scalable production gain competitive advantage. The rise from mid four billion into the low eight billion reflects cumulative adoption across retail, clinical, and digital channels rather than a single intervention. Companies providing reliable supply, strong patient engagement, and evidence-backed treatment options are better positioned to maintain long-term market share.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 4.4 billion |
| Forecast Value (2036) | USD 7.7 billion |
| Forecast CAGR 2026 to 2036 | 5.7% |
Treatments for vaginal yeast infections target Candida overgrowth in the vaginal microbiome to relieve irritation, discharge, and inflammation. Many recurrent-case protocols align closely with vulvovaginal candidiasis treatment pathways used in specialist women’s health clinics. Earlier therapy relied on broad antifungal creams or oral azoles with limited formulation options, which sometimes caused recurrence and variable patient adherence. Modern approaches include single-dose oral antifungals, topical gels, and combination therapies designed for rapid symptom relief and reduced relapse. Healthcare providers evaluate products by efficacy against resistant strains, duration of therapy, local tolerability, and patient convenience. Procurement teams often cross-check formulary decisions with the broader candidiasis therapeutics pipeline when resistance concerns rise. Adoption is concentrated in gynecology clinics, primary care practices, and telehealth platforms where timely diagnosis and accessible treatment improve patient outcomes. Treatment choice also influences adherence, follow-up visits, and overall patient satisfaction.
Manufacturers differentiate through formulation stability, dose optimization, and spectrum of antifungal activity rather than price alone. Portfolio planning typically references the broader anti-fungal therapy class mix to anticipate demand shifts across azoles and non-azoles. Earlier reliance on generics and limited product lines reduced differentiation and restricted brand positioning. Current programs emphasize novel delivery systems, patient-friendly packaging, and education on recurrence prevention. Clinics and pharmacies assess total operational value through reduced return visits, improved patient compliance, and minimized side effects, rather than per-unit cost. Suppliers add value through consistent supply, packaging innovation, and technical guidance for healthcare staff. Over time, growth is driven by increasing awareness, rising outpatient consultation, and telemedicine adoption, where rapid, reliable, and easy-to-administer therapies enhance patient experience and clinical workflow efficiency.
Clinical prevalence, treatment efficacy, and patient convenience drive the Vaginal Yeast Infections Treatment Market in 2026 rather than short-term purchasing fluctuations. Healthcare providers and pharmacies prioritize therapies that ensure rapid symptom relief, minimize recurrence, and align with treatment guidelines. Segment adoption varies based on mechanism of action, formulation, and route of administration. Benchmarking against the vaginal antifungals landscape helps teams justify channel strategy across OTC and prescription formats. Suppliers providing consistent supply chains, patient guidance, and regulatory documentation can secure long-term adoption. Market growth is concentrated in prescription azole antifungals and OTC polyene antifungals, creating predictable demand for manufacturers that combine therapeutic effectiveness, patient support, and reliable product availability across clinical and retail channels.
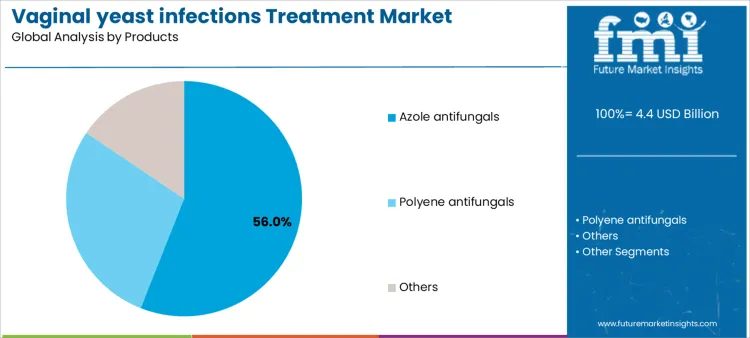
Azole antifungals represent approximately 56% of demand due to their broad-spectrum activity and established use in prescription treatment. Suppliers must ensure batch consistency, regulatory compliance, and education for healthcare professionals on dosing and duration. Polyene antifungals are frequently used in OTC treatments, requiring reliable packaging, patient guidance, and supply logistics to maintain proper usage. Other therapeutic classes cater to specialized indications, demanding technical support and clinical guidance. Product type determines supplier engagement in training, inventory planning, and technical assistance, influencing adoption rates across prescription and self-care settings.
Therapeutic characteristics and administration routes further shape workflow and supplier strategy. Azole antifungals require adherence monitoring, clinician guidance, and pharmacy supply coordination, creating recurring supplier touchpoints. Polyene antifungals emphasize user-friendly packaging, patient instructions, and predictable retail supply, reducing procedural complexity but increasing demand for educational support. Specialty therapies require targeted guidance and integration into clinical protocols. Hospitals, clinics, and pharmacies standardizing on specific product types rely on suppliers for consistent quality, training, and availability. Providers offering comprehensive support, guidance, and inventory reliability can strengthen adoption and retention.
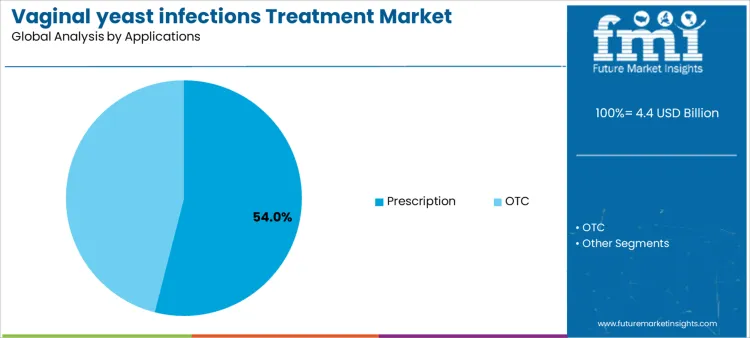
Prescription applications account for approximately 54% of demand because moderate-to-severe infections require clinician-supervised therapy, generating predictable, high-volume adoption. OTC use, while smaller in volume, provides patients with self-managed relief, creating stable demand across retail channels. Application-specific requirements guide supplier inventory management, patient education initiatives, and service programs. Aligning product supply and support services with both prescription and OTC applications allows suppliers to maximize market reach, procedural efficiency, and revenue predictability while addressing the full spectrum of patient needs.
Healthcare delivery channels and patient behaviors further shape supplier strategy. High-volume prescription use emphasizes training for clinicians, consistent drug supply, and adherence monitoring programs to ensure therapeutic success. OTC applications require clear labeling, accessible instructions, and reliable retail availability, supporting patient self-management. Specialty or alternative therapies demand precise guidance, supply reliability, and technical support. Segment distribution is therefore determined by treatment setting, patient preference, and clinical complexity rather than short-term purchasing trends. Suppliers providing guidance, consistent supply, and post-sale support can secure broader adoption and long-term engagement.
The Vaginal Yeast Infections Treatment Market is shaped by hospitals, gynecology clinics, and outpatient care centers managing recurrent and acute infections. Treatments are evaluated based on therapeutic effectiveness, safety profile, formulation convenience, and adherence potential. Selection decisions involve gynecologists, pharmacists, and procurement teams. Suppliers that provide therapies with consistent efficacy, clear dosing instructions, and minimal side effects are preferred. This positions treatments as essential for women’s health management rather than optional interventions, emphasizing reliable outcomes, patient adherence, and predictable clinical results.
How Are Standardized Care Guidelines Influencing Supplier Expectations in the Vaginal Yeast Infections Treatment Market?
Healthcare providers increasingly follow evidence-based treatment protocols to ensure consistent patient outcomes and reduce recurrence. This drives demand in the Vaginal Yeast Infections Treatment Market toward therapies that integrate with existing clinical guidelines, provide predictable relief, and minimize adverse effects. Buyers prefer products backed by clinical studies, clear administration protocols, and patient support materials. Suppliers are evaluated on product consistency, regulatory compliance, and technical support rather than minor formulation differences. Treatments aligned with standardized guidelines are more likely to be adopted across multiple clinics and hospital networks.
Where is Recurring Demand Being Built in the Vaginal Yeast Infections Treatment Market?
Recurring demand comes from hospitals, outpatient clinics, and gynecology centers treating patients with recurrent or chronic infections. Each treatment cycle generates predictable repeat orders. The Vaginal Yeast Infections Treatment Market also benefits from multi-site healthcare systems standardizing on approved therapy options. Suppliers that provide dosing support, patient education, and reliable supply chains secure long-term engagement. Embedding treatments into standard care pathways ensures ongoing demand tied to patient volume and treatment frequency.
Why do Cost and Safety Considerations Limit Broader Adoption in the Vaginal Yeast Infections Treatment Market?
Treatment cost, side effects, and patient tolerance can limit adoption in smaller clinics or low-volume facilities. The market also faces adoption constraints from regulatory approvals, formulary inclusion, and clinician familiarity with existing products. Physicians may be cautious about switching therapies mid-treatment due to potential patient discomfort or adherence issues. Suppliers entering the market must provide clinical guidance, educational materials, and consistent product availability. Adoption is concentrated in high-volume gynecology centers and specialized women’s health clinics where efficacy, adherence, and patient outcomes are prioritized.
| Country | CAGR |
|---|---|
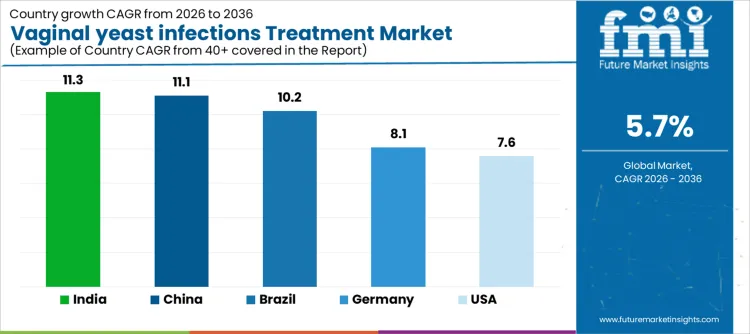
| USA | 7.6% |
| Brazil | 10.2% |
| China | 11.1% |
| India | 11.3% |
| Germany | 8.1% |
Demand for vaginal yeast infections treatment is rising as hospitals, clinics, and retail pharmacies expand access to antifungal therapies and patient awareness programs. India leads with an 11.3% CAGR, supported by increasing women’s health initiatives, higher diagnosis rates, and adoption of over the counter and prescription treatments. China follows at 11.1%, driven by growing healthcare access, rising awareness of gynecological health, and higher treatment adoption. Brazil records 10.2%, reflecting broader availability of antifungal medications in public and private healthcare systems. The USA grows at 7.6%, shaped by steady demand in established clinical and pharmacy channels. Germany, at 8.1%, reflects stable uptake in a mature healthcare market with standardized treatment protocols.
United States demand for vaginal yeast infection treatments is growing along a 7.6% CAGR as hospital gynecology programs expand outpatient care, preventive screening, and patient education initiatives. Procurement decisions are managed by pharmacy committees, gynecology directors, and clinical program boards. Adoption emphasizes rapid-acting antifungal therapies, topical and oral formulations, and standardized treatment protocols. Once selected, therapies are integrated across multiple hospitals and outpatient clinics.
Physician training, workflow integration, and patient monitoring influence adoption. Import formulations dominate premium treatment regimens, while domestic suppliers provide standard antifungal therapies. Capital allocation follows program expansion and patient volume growth. Replacement cycles track therapy duration and adherence. Commercial success depends on inclusion in hospital formularies, clinic treatment protocols, and multi-year supply agreements.
Brazil is experiencing a 10.2% CAGR in vaginal yeast infection treatments due to expansion of private hospital gynecology units, outpatient care programs, and women’s wellness clinics. Procurement authority rests with hospital network pharmacy committees, gynecology department directors, and clinical oversight boards. Adoption focuses on topical, oral, and combination antifungal therapies with high efficacy and rapid symptom relief. Import products dominate premium regimens, while domestic suppliers cover standard therapies.
Once approved, rollout spans multiple affiliated hospitals and outpatient centers. Physician and nursing training, workflow alignment, and patient education influence adoption. Capital allocation aligns with hospital network expansion and new patient programs. Replacement cycles follow treatment protocols. Commercial success depends on inclusion in network formularies, reference site adoption, and long-term supply agreements rather than single clinic or department purchases.
China is experiencing an 11.1% CAGR in vaginal yeast infection treatments due to expansion of hospital gynecology programs, outpatient women’s health centers, and public wellness initiatives. Major hospitals and clinical networks control most procurement. Adoption emphasizes rapid-acting antifungal therapies, patient compliance, and integration with outpatient care workflows. Domestic suppliers provide standard treatments, while international brands retain positions in premium formulations. Procurement follows multi-year hospital planning and centralized tenders. Once validated, therapies are implemented across multiple hospitals and outpatient clinics. Physician training, patient education, and treatment adherence monitoring influence utilization. Replacement cycles follow therapy duration rather than product expiration. Capital allocation aligns with hospital expansion and new clinic launches. Commercial success depends on inclusion in provincial and national hospital formularies and network treatment protocols.
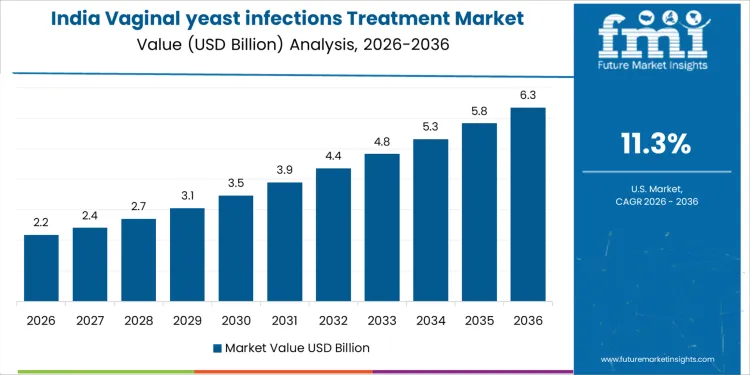
India is witnessing an 11.3% CAGR in vaginal yeast infection treatments as hospital gynecology programs, outpatient women’s health clinics, and wellness initiatives expand. Procurement authority rests with hospital pharmacy boards, gynecology committees, and program directors. Therapy selection emphasizes rapid-acting antifungal agents, treatment adherence, and patient safety. Import products dominate high specification treatments while domestic suppliers cover standard regimens. Once approved, therapies are deployed across multiple affiliated hospitals and clinics. Training, workflow standardization, and patient education influence utilization. Replacement cycles track therapy duration and adherence. Capital allocation aligns with hospital program expansion and patient volume growth. Commercial success depends on network formulary inclusion, reference site adoption, and long-term supply contracts rather than isolated hospital or clinic purchases.
How Are Guidelines and Reimbursement Policies Shaping the Vaginal Yeast Infections Treatment Market in Germany?
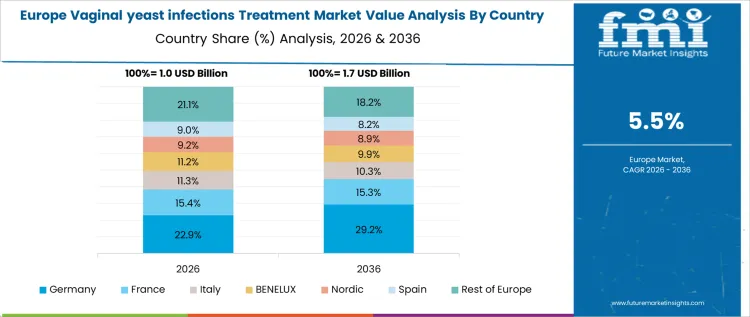
Germany is experiencing an 8.1% CAGR in vaginal yeast infection treatments due to structured clinical guidelines, reimbursement frameworks, and standardized hospital gynecology protocols. University hospitals and regional wellness centers control most procurement. Adoption emphasizes rapid-acting antifungal therapies, patient adherence, and workflow integration. Device and therapy evaluation prioritizes safety, efficacy, and treatment reproducibility. Procurement follows health technology assessments and hospital capital planning schedules. Import suppliers dominate premium regimens while domestic suppliers provide standard therapies. Once approved, therapies are deployed across multiple departments and hospitals. Training, documentation, and patient monitoring influence utilization. Replacement cycles follow treatment duration. Capital allocation aligns with hospital program expansion. Commercial success depends on framework inclusion, reference site adoption, and integrated supplier support rather than isolated clinic purchases.
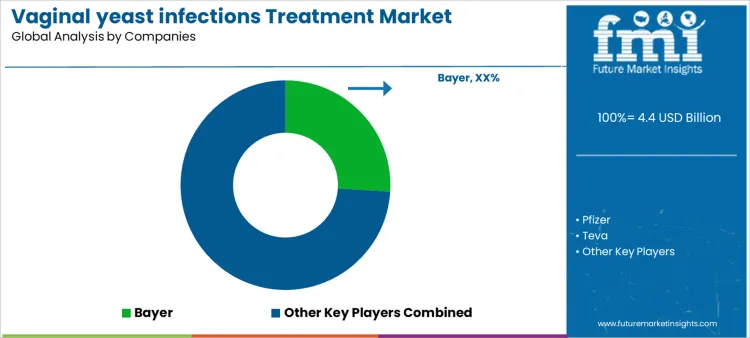
Treatment selection for vaginal yeast infections follows clinical guidelines, symptom severity, and patient adherence considerations rather than simple catalog comparison. Clinicians define formulation type, dosing duration, drug interactions, and pregnancy safety before approving therapies. Bayer, Pfizer, Teva, GSK, Sanofi compete during protocol evaluation, guideline alignment, and formulary decision stages. Once a therapy enters treatment pathways, pharmacy stocking, patient counseling, and prescription monitoring anchor adoption. Bayer positions products through OTC and prescription channels with established clinical data. Pfizer emphasizes short course therapies and clinician familiarity. Teva leverages generic options for affordability and formulary inclusion. GSK targets branded formulations with patient support programs. Sanofi integrates treatment with broader women’s health offerings.
Competitive outcomes reflect efficacy, safety, patient adherence, and availability rather than price alone. Physicians evaluate symptom resolution rates, recurrence prevention, tolerability, and ease of administration during selection. Pharmacists review dispensing requirements, shelf life, and labeling. Bayer benefits from established brand trust and broad channel coverage. Pfizer secures placements where rapid symptom relief is prioritized. Teva gains share through cost efficiency and accessibility. GSK competes via patient education and support integration. Sanofi holds positions in programs linking antifungal treatment with wider women’s health initiatives. Market positions vary by region, prescribing patterns, insurance coverage, and patient preference for formulation type.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Billion |
| Product Type | Azole antifungals; Polyene antifungals; Others |
| Application | Prescription; OTC |
| End User / Sales Channel | Retail pharmacies; Drug stores; E-commerce |
| Region | Asia Pacific; Europe; North America; Latin America; Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, Rest of regions |
| Key Companies Profiled | Bayer; Pfizer; Teva; GSK; Sanofi |
| Additional Attributes | Dollar by sales by product and application, prescription versus OTC mix, azole versus polyene usage split, retail versus online channel share, recurrence driven demand patterns, shelf life and inventory turnover, regulatory and labeling impact, patient adherence and treatment completion trends, pharmacy led education role |
How big is the vaginal yeast infections treatment market in 2026?
The global vaginal yeast infections treatment market is estimated to be valued at USD 4.4 billion in 2026.
What will be the size of vaginal yeast infections treatment market in 2036?
The market size for the vaginal yeast infections treatment market is projected to reach USD 7.7 billion by 2036.
How much will be the vaginal yeast infections treatment market growth between 2026 and 2036?
The vaginal yeast infections treatment market is expected to grow at a 5.7% CAGR between 2026 and 2036.
What are the key product types in the vaginal yeast infections treatment market?
The key product types in vaginal yeast infections treatment market are azole antifungals, polyene antifungals and others.
Which applications segment to contribute significant share in the vaginal yeast infections treatment market in 2026?
In terms of applications, prescription segment to command 54.0% share in the vaginal yeast infections treatment market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.