Respiratory Inhaler Devices Market Size, Market Forecast and Outlook By FMI
Summary of the Respiratory Inhaler Devices Market
- Demand And Growth Drivers
- Chronic airway disease treatment keeps device demand active across daily maintenance therapy and rescue therapy.
- Patient handling ease and dose consistency keep inhalers central to routine pulmonary care.
- Treatment access is improving in Asia. Prescription volume is rising in high-population markets.
- Product and Segment View
- Metered dose inhalers lead product demand because prescriber familiarity and rescue use keep this format in heavy circulation.
- Asthma remains the largest disease pool because long treatment duration keeps refill need high across adult and pediatric care.
- Manual inhalers dominate technology demand because connected formats still form a small part of global prescription volume.
- Geography and Competitive Outlook
- India and China stay ahead of the global pace as diagnosis rates improve and inhaler access keeps rising.
- France and Italy post mid-range growth as chronic therapy use stays steady in organized care systems.
- GSK and AstraZeneca hold strong positions through respiratory franchise depth and long-standing prescriber acceptance.
- Analyst Opinion
- , Principal Consultant for Healthcare at FMI, opines, “In respiratory care, device selection depends on intuitive handling and consistent dose delivery. Familiar inhalers therefore remain widely used in asthma and COPD management. Future innovation must improve patient-familiar formats to support adherence and steadier market expansion.”
- Respiratory Inhaler Devices Market Value Analysis
- Recurring therapy use in asthma and chronic obstructive pulmonary disease keeps annual device demand active across prescription-led treatment cycles.
- Value stays concentrated in metered dose inhalers and other familiar formats as established prescribing habits support repeat replacement demand.
- Hospital and retail pharmacy channels retain a major revenue base as chronic respiratory treatment continues through supervised care and refill supply.
- Generic pressure and reimbursement controls keep pricing disciplined across mature markets which limits faster value expansion through the forecast period.
Respiratory Inhaler Devices Market Definition
The respiratory inhaler devices market covers inhalation drug delivery devices used in chronic and acute pulmonary care. Revenue comes from dry powder inhalers and metered dose inhalers. Nebulizers and soft mist inhalers are part of the market as well. The study tracks demand across hospitals and specialty clinics. Retail pharmacy chains and online or mail order pharmacies are covered too.
Respiratory Inhaler Devices Market Inclusions
The report includes global sizing and the forecast from 2026 to 2036. Analysis covers product and drug class. Technology and disease indication are included. Sales channel and country growth are part of the study. Competitive positioning and device use patterns are covered.
Respiratory Inhaler Devices Market Exclusions
General oxygen therapy equipment is outside the scope. Ventilators and CPAP systems are excluded. Standalone pulmonary diagnostics are not included. Oral respiratory drugs and injectable pulmonary therapies are outside the study. Non device respiratory support products are excluded too.
Respiratory Inhaler Devices Market Research Methodology
- Primary Research: Interviews with pulmonologists and respiratory therapists were used to test demand patterns across prescribing and channel settings.
- Desk Research: Public company materials and respiratory care publications were reviewed alongside treatment references and regulatory material.
- Market Sizing and Forecasting: Device demand was mapped to product type and disease pool. Channel use and price mix were then used to reconcile total value.
- Data Validation: Product and disease leadership values came from the source file. Drug class technology sales channel and nebulizer type leadership shares were estimated from the supplied taxonomy to complete the requested six parent segment view.
Why is the Respiratory Inhaler Devices Market Growing?
- Asthma and chronic obstructive pulmonary disease treatment creates repeat inhaler demand across long-term prescription cycles.
- Metered dose inhalers hold a large installed base that supports repeat use across routine respiratory therapy.
- Digital inhalers and low-carbon formats are adding value as suppliers improve adherence support and device performance.
- Hospital distribution and pharmacy refill access keep inhaler use active across chronic care pathways.
Long-term respiratory therapy supports a durable replacement cycle for inhaler devices. Asthma treatment and chronic obstructive pulmonary disease care require repeated dosing over extended periods. That usage pattern influences regular demand across hospitals and pharmacy networks. Ongoing use of metered dose inhalers supports large refill volume across routine airway care. Device familiarity also supports stable prescription use across established respiratory treatment pathways.
Product value is rising as suppliers introduce digital inhalers and lower-emission formats. Healthcare providers are placing greater focus on dose consistency and treatment adherence. That preference propels demand for devices that are easier to use across daily therapy. Newer formats such as smart inhalers and improved dry powder inhalers are gaining attention in selected programs that need better tracking or easier use. Growth also benefits from rising diagnosis levels in India and China which expands access to inhalation therapy.
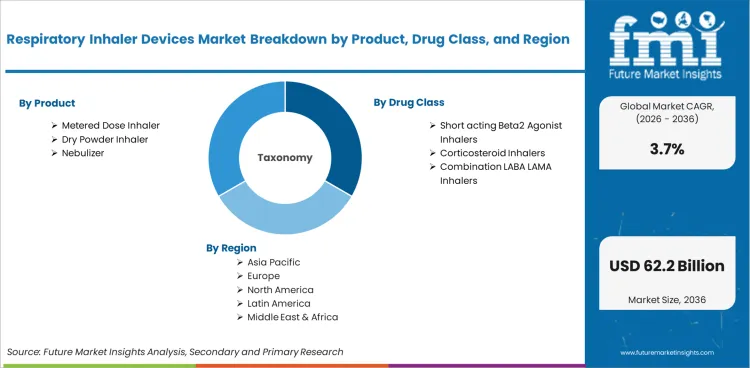
Market Segmentation Analysis
- Metered dose inhalers are expected to account for 71.6% share of product demand in 2026 because rescue therapy use and long prescription familiarity keep this format at the center of routine airway care.
- Short acting beta2 agonist inhalers are projected to represent 24.4% share of drug class demand in 2026 because rescue treatment volume stays large across asthma management.
- Asthma is expected to contribute 36.7% share of disease indication demand in 2026 because long treatment duration keeps inhaler refill frequency high.
- Hospitals are projected to contribute 34.9% share of sales channel demand in 2026 because acute episodes and specialist review still direct initial device choice.
The market is segmented by product, including dry powder inhalers and metered dose inhalers along with nebulizers and soft mist inhalers, with metered dose inhalers leading due to their large prescribed base. It is further segmented by drug class, covering short-acting beta2 agonists and short-acting muscarinic antagonists along with long-acting beta2 agonists and long-acting muscarinic antagonists, while combination inhalers and corticosteroid inhalers support long-term respiratory treatment. By technology and disease indication, the market includes manual inhalers and digital inhalers while serving asthma and chronic obstructive pulmonary disease along with pulmonary arterial hypertension and other severe respiratory conditions. The market is also segmented by sales channel and region, spanning hospitals and specialty clinics along with retail pharmacy chains and online pharmacies across major global markets.
Respiratory Inhaler Devices Market Analysis by Product
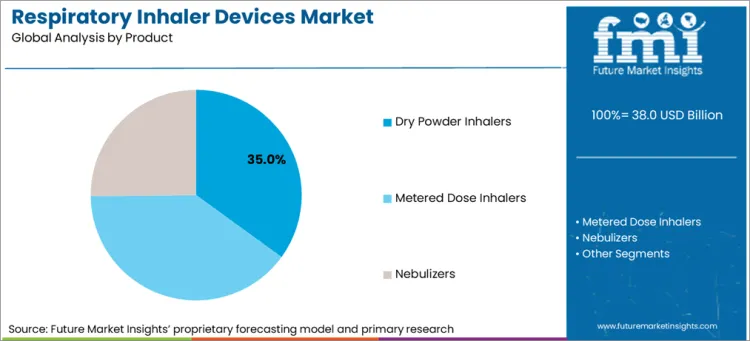
- Metered dose inhalers are anticipated to account for 71.6% share of product type in 2026 as acute relief use and long prescriber familiarity keep this format in the largest prescription pool.
- Routine use across asthma and chronic obstructive pulmonary disease keeps refill volume high for this device type.
Insights into the Soft Mist Inhaler segment
- Soft mist inhalers are likely to expand faster because lower propellant pressure and easier inhalation support adherence in maintenance therapy.
- Climate-linked interest in lower-emission inhaler options is improving attention on this format.
Respiratory Inhaler Devices Market Analysis by Drug Class
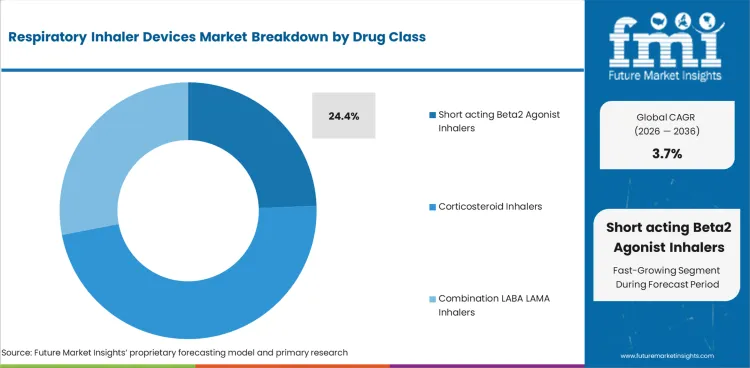
- Short acting Beta2 agonist inhalers are projected to represent 24.4% share of drug class type in 2026 because reliever use stays active across large asthma patient pools.
- Salbutamol-led rescue prescribing keeps this drug class in frequent retail and hospital circulation.
Insights into the Combination ICS LABA Inhalers segment
- Combination ICS LABA inhalers are predicted to rise at a significant pacebecause maintenance regimens are shifting toward dual therapy in patients who need tighter symptom control.
- Long duration use supports repeat refill activity in this segment.
Respiratory Inhaler Devices Market Analysis by Technology
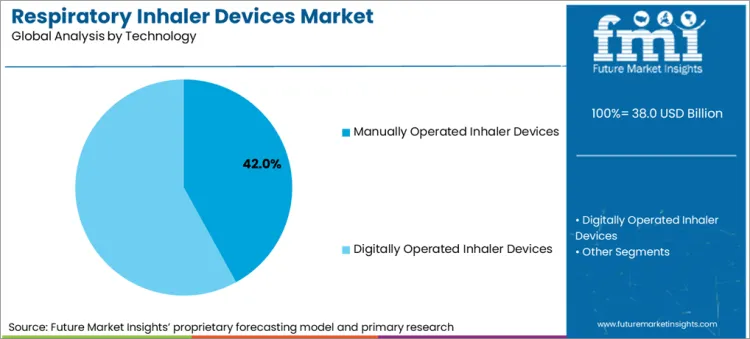
- Manual inhalers are expected to account for 91.0% share of technology demand in 2026 because prescription volume stays with standard non-connected devices across most care systems.
- Digital formats are drawing interest. Installed prescription behavior still favors simpler hardware with lower upfront cost.
Insights into the Digital Inhalers segment
- Digital inhalers are projected to expand at a prominent expansion rate because adherence tracking and remote monitoring support better control in long-duration therapy.
- Specialty programs use this format first in settings that value follow-up data.
Respiratory Inhaler Devices Market Analysis by Disease Indication
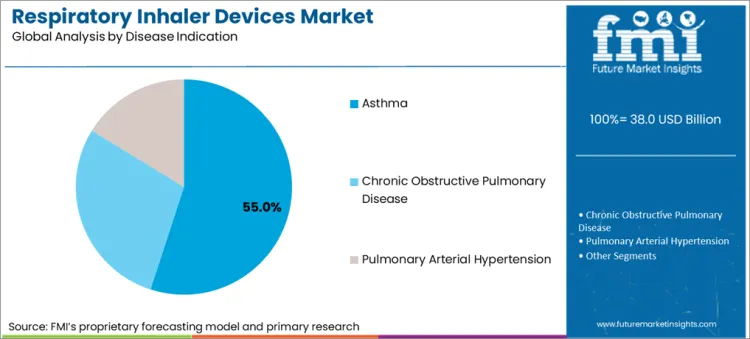
- Asthma is expected to hold 36.7% share of disease indication demand in 2026 because early diagnosis and long-term inhaled treatment keep refill activity high across adult and pediatric care.
- Ongoing need for maintenance and rescue therapy keeps device use more regular in asthma than in smaller indication pools.
Insights into the Chronic Obstructive Pulmonary Disease segment
- Chronic obstructive pulmonary disease therapeutics is likely to record steady expansion because aging patient pools and long-duration bronchodilator use influences demand growth for inhalers.
- Maintenance therapy intensity supports repeat device replacement in this segment.
Respiratory Inhaler Devices Market Analysis by Sales Channel
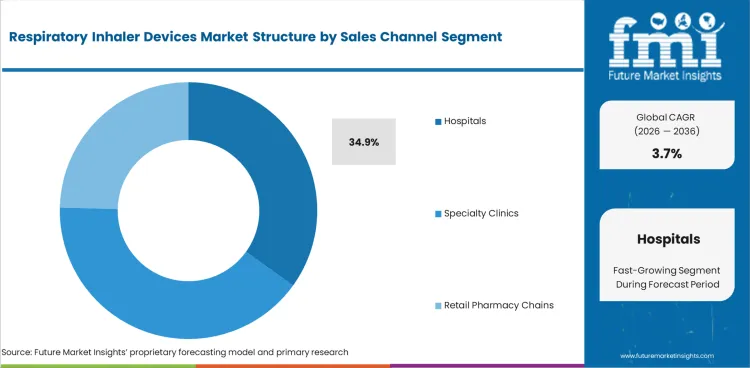
- Hospitals are projected to account for 34.9% share of sales channel demand in 2026 because acute respiratory episodes and specialist reviews often set the first device choice.
- Institutional care still shapes early device training and therapy change decisions for many patients.
Insights into the Online Pharmacies and Mail Order Pharmacies segment
- Online pharmacies and mail order pharmacies are expected to expand faster because repeat inhaler refills are well suited to home delivery and chronic therapy schedules.
- Digital refill convenience support stronger continuity in stable maintenance treatment.
Respiratory Inhaler Devices Market Analysis by Nebulizer Type
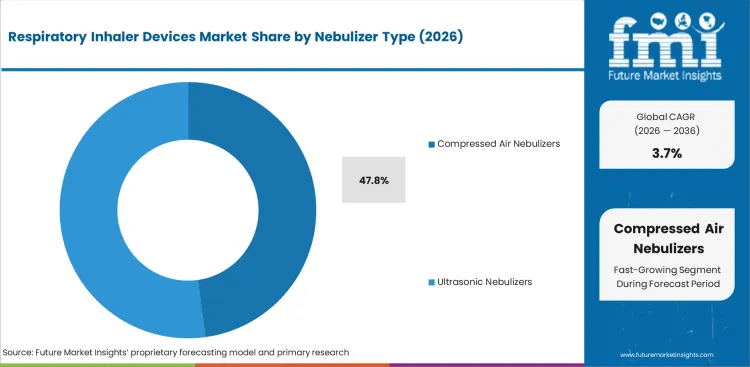
- Compressed air nebulizers are expected to represent an estimated 47.8% share of nebulizer type in 2026 because installed base depth and lower unit cost keep this format in routine clinical use.
- Hospitals and home care programs still rely on this design for dependable aerosol delivery across age groups.
Insights into the Mesh Nebulizers segment
- Mesh nebulizers are expected to expand rapidly due to portable size and quiet use support home-based therapy and pediatric care.
- Interest in compact handheld mesh nebulizer formats is improving in self-care settings.
Respiratory Inhaler Devices Market Drivers Restraints and Opportunities
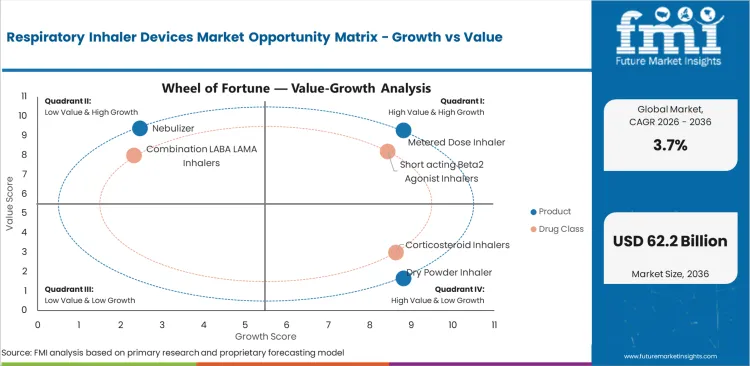
- Recurring asthma and chronic obstructive pulmonary disease therapy keeps inhaler demand active across hospitals and pharmacy refill channels.
- Generic competition and reimbursement control limit pricing headroom across standard inhaler lines in mature prescription markets.
- Digital inhalers and patient-friendly formats are opening higher-value scope across adherence-focused respiratory treatment.
Respiratory inhaler device demand is expected to remain steady, supported by long‑duration chronic therapy and the continued need for reliable, easy‑to‑use devices that sustain regular refill volumes. Supplier selection is increasingly shaped by dose consistency and patient handling simplicity, which reinforce adherence in routine care. Although value growth in mature prescription markets faces pressure from generic competition and reimbursement controls, product innovation is creating steady upside. Digital support features and lower‑emission inhaler formats are improving treatment outcomes and positioning suppliers for incremental upgrades rather than volume‑led expansion.
Chronic Respiratory Therapy Continuity
Asthma and chronic obstructive pulmonary disease treatment keep inhaler demand active across long-duration care pathways. Respiratory care teams rely on inhaled maintenance therapy for routine disease control and rescue treatment. Product use stays active in outpatient settings and institutional settings because inhalation therapy stays central to targeted airway drug delivery. Rising pulmonary disease burden adds more prescription volume to this base.
Generic Price Pressure and Technique Variability
Standard inhaler lines face steady pricing pressure across mature therapy markets. Generic competition limits premium retention in established device classes and reimbursement discipline keeps value growth measured. Incorrect inhaler use creates another constraint because handling errors reduce treatment outcomes and weaken the benefit of prescribed therapy. Mature markets such as the United States and Germany show this pattern more clearly because replacement demand is stronger than fresh device conversion.
Patient-Friendly and Digital Inhaler Expansion
Patient-friendly inhalers and digitally supportive formats create the significant expansion outlook in this market. Respiratory care providers are placing greater focus on device usability and therapy adherence support across chronic airway disease management. Low-carbon inhaler development adds another value layer as suppliers move selected products toward updated formats. Product differentiation is shifting toward easier handling and stronger treatment continuity rather than simple volume growth.
Regional Analysis
.webp)
| Country |
CAGR |
| India |
7.2% |
| China |
7.0% |
| South Korea |
6.6% |
| France |
4.5% |
| Italy |
3.7% |
| Japan |
3.2% |
| United States |
1.7% |
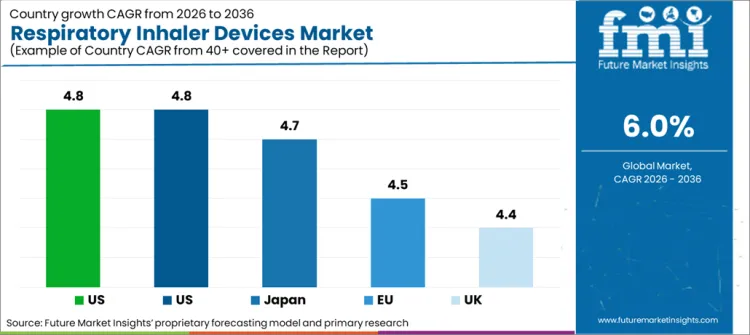
Source. Future Market Insights analysis based on the supplied market model and covered country narratives.
Analysis of Respiratory Inhaler Devices Market by Key Countries
- India is projected to post the fastest pace at 7.2% CAGR by 2036 as inhaler access keeps rising across asthma and chronic obstructive pulmonary disease treatment.
- China is expected to follow at 7.0% CAGR through 2036 as diagnosis rates and retail channel reach keep improving.
- South Korea is likely to record 6.6% CAGR during the forecast period as organized respiratory care supports faster uptake of inhaled therapy.
- United States demand is expected to rise at 1.7% CAGR through 2036 because replacement demand and generic competition keep value growth measured.
Country growth differs with diagnosis scale and prescription access. Asia leads because treatment access is improving across large patient pools. Mature Western markets expand more slowly because replacement demand dominates new unit growth.
Demand outlook for Respiratory Inhaler Devices in India
Hospital expansion is lifting inhaler use in India across chronic respiratory care. The market in India is projected to register a 7.2% CAGR through 2036 because asthma and chronic obstructive pulmonary disease treatment demand is rising and inhaler access is widening across hospital and retail channels. Chronic maintenance therapy is taking a larger share of routine respiratory treatment across the country.
- Hospital channels are widening access to inhaler-based treatment across major care settings in India.
- Retail distribution is supporting refill activity for long-duration respiratory therapy across the country.
- Asthma and chronic obstructive pulmonary disease treatment keep prescription demand active in routine care.
Future outlook for Respiratory Inhaler Devices in China
Urban treatment reach is strengthening inhaler use in China across expanding respiratory care pathways. Sector adoption in China is likely to post a 7.0% CAGR through 2036 because chronic respiratory disease burden is rising and therapy access is improving through stronger diagnosis and retail channel penetration. Prescription growth is moving beyond top hospital networks into a wider treatment base.
- Diagnosis expansion is increasing the treated pool for inhaler-based respiratory therapy in China.
- Retail channel penetration is improving product access across a wider set of care locations.
- Chronic respiratory disease burden is sustaining stronger prescription demand through the forecast period.
Opportunity analysis of Respiratory Inhaler Devices in South Korea
Treatment access is improving in South Korea across a concentrated respiratory care system. The industry in South Korea is expected to grow at a 6.6% CAGR through 2036 because respiratory care demand is increasing and inhaler therapy use is rising across organized channels. Channel concentration keeps supplier selection tight across established treatment networks.
- Organized treatment channels support wider use of inhaler therapies in South Korea.
- Rising respiratory care demand is expanding prescription activity across chronic pulmonary treatment.
- Channel concentration keeps competitive access selective for inhaler suppliers in this market.
In-depth analysis of Respiratory Inhaler Devices in France
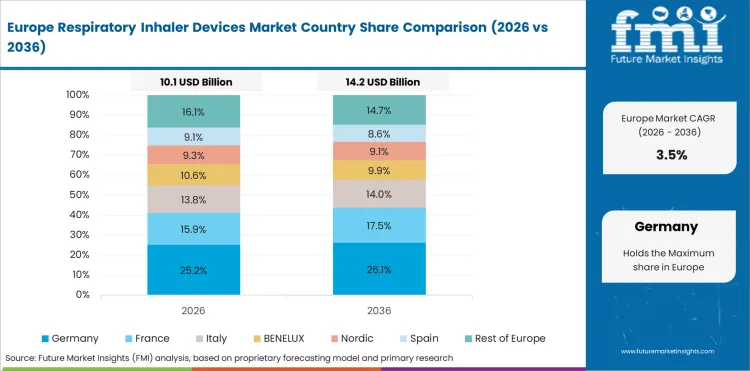
Specialist prescribing supports steady inhaler demand in France across structured pulmonary care. Sector in France is forecast to advance at a 4.5% CAGR through 2036 because respiratory therapy demand stays active through specialist prescribing and retail access in managed care settings. Ongoing therapy use keeps refill volume stable across chronic treatment pathways.
- Specialist prescribing supports continued use of inhaler-based therapy in France.
- Retail access keeps respiratory medicines available across recurring treatment cycles.
- Managed care settings support steady device turnover across chronic pulmonary care.
Sales analysis of Respiratory Inhaler Devices in Italy
Outpatient treatment is promoting the inhaler use in Italy across asthma and chronic pulmonary care. Demand in Italy is expected to record a 3.7% CAGR through 2036 because asthma and chronic obstructive pulmonary disease treatment demand stays active and inhalation therapy use continues across outpatient settings. Prescription continuity supports measured expansion rather than a sharp volume step-up.
- Outpatient care keeps inhaler prescriptions active across routine respiratory treatment in Italy.
- Asthma therapy and chronic pulmonary treatment support steady device demand across the country.
- Inhalation-based treatment continues to hold a stable place in physician-led care.
Future outlook for Respiratory Inhaler Devices in Japan
Clinical routines drives the inhaler demand stable in Japan across mature respiratory care settings. Japan is projected to expand at a 3.2% CAGR through 2036 because chronic pulmonary treatment demand stays steady and inhaler-based therapy remains widely used in structured clinical environments. Market maturity keeps growth moderate even though prescription continuity stays firm.
- Structured clinical environments support steady inhaler use across chronic respiratory treatment in Japan.
- Prescription continuity keeps device demand active across established pulmonary care pathways.
- Mature treatment patterns keep expansion moderate through the study period.
Demand outlook for Respiratory Inhaler Devices in the United States
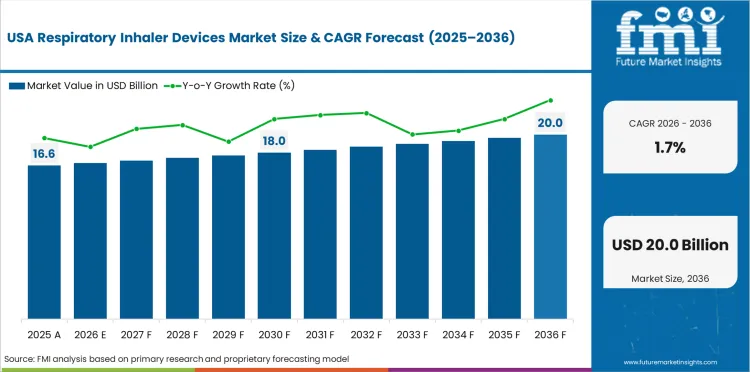
Prescription depth keeps respiratory inhaler use active in the United States across chronic pulmonary treatment. The United States industry is expected to grow at a 1.7% CAGR through 2036 because chronic respiratory drug demand stays steady and inhaler use remains common in asthma and chronic obstructive pulmonary disease care despite market maturity and strong competition. Replacement-led volume keeps the installed base active across established prescription channels.
- High prescription volume supports continued inhaler replacement across routine respiratory treatment.
- Asthma and chronic obstructive pulmonary disease care keep device demand active in mature settings.
- Strong competition limits faster value expansion in this established market.
Competitive Landscape and Strategic Positioning
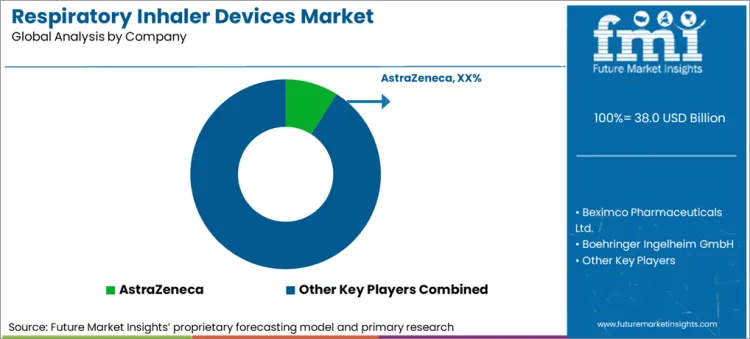
- Competition is moderately concentrated around respiratory drug companies with established inhaler franchises.
- Leading firms compete on device familiarity and dose consistency. Channel reach and therapy portfolio depth shape their position.
- Smaller specialists gain ground in nebulizer systems and home care use. They compete through portability and device handling benefits.
- Entry stays difficult because respiratory therapy approval and physician familiarity create a high switching barrier.
Established respiratory franchises hold an advantage because prescribers and care teams already know the device formats and drug combinations in their portfolios. GSK and AstraZeneca benefit from deep inhaler lines and long-standing respiratory use. Boehringer Ingelheim adds strength through chronic obstructive pulmonary disease treatment depth. Ongoing activity in triple-therapy inhalers and bronchodilator therapy supports that position.
Generic and value-oriented suppliers compete through cost access and refill continuity. Teva Respiratory and Viatris stay important in mature channels that need familiar formats at tighter price points. Cipla and Lupin support expansion in cost-sensitive markets through large respiratory portfolios. Demand still favors suppliers that keep supply continuity intact across hospital and retail channels.
Device- specialists compete in nebulizer and home care use. OMRON and PARI support this part of the market through portable systems and established aerosol delivery knowledge. Interest in nebulizers and connected drug delivery devices keeps this part of the market active in pediatric care and home therapy.
Entry remains difficult because inhaler switching needs clinical confidence and stable patient training. New suppliers must show reliable dose delivery and secure physician trust before larger prescription flows move. Climate pressure adds a second hurdle because propellant transition work raises development effort for pressurized systems. Recent investment in lower-emission inhalers and new nebulized combinations shows that product change still demands time and capital.
Key Companies in the Respiratory Inhaler Devices Market
Company positions reflect respiratory franchise depth and device know-how across prescription inhalers and nebulizer systems.
- Global Leaders: GSK plc and AstraZeneca lead through deep respiratory portfolios and strong prescriber familiarity. Boehringer Ingelheim adds chronic obstructive pulmonary disease strength. These firms benefit from global channel reach and long-term therapy presence across asthma and chronic obstructive pulmonary disease care.
- Value and Generic Suppliers: Teva Respiratory and Viatris keep a solid role in mature prescription channels through familiar inhaler lines and price access. Cipla and Lupin add strength in cost-sensitive markets through large respiratory portfolios and expanding distribution support across hospital and retail settings.
- Device and Nebulizer Specialists: OMRON Corporation and PARI GmbH stand out in nebulizer systems through portable device strength and home care use. CHIESI Farmaceutici adds inhaler depth through pressurized inhaler development and ongoing propellant transition work in regulated respiratory programs.
Competitive Benchmarking: Respiratory Inhaler Devices Market
| Company |
Respiratory Franchise Depth |
Device Breadth |
Channel Access |
Geographic Footprint |
| GSK plc |
High |
High |
High |
Global |
| AstraZeneca |
High |
Moderate |
High |
Global |
| Boehringer Ingelheim International GmbH |
High |
Moderate |
High |
Global |
| Teva Respiratory LLC |
Moderate |
Moderate |
High |
Global |
| Cipla LTD |
Moderate |
Moderate |
Moderate |
Global |
| OMRON Corporation |
Moderate |
High |
Moderate |
Global |
| PARI GmbH |
Moderate |
High |
Moderate |
Regional |
Source. Future Market Insights analysis based on the supplied market model and covered country narratives.
Key Developments in the Respiratory Inhaler Devices Market
- In October 2025, AeroRx Therapeutics announced a USD 21 million Series A financing to support late-stage work on a nebulized LABA LAMA therapy for chronic obstructive pulmonary disease.
- In December 2025, CHIESI UK and Ireland submitted beclometasone with a next-generation propellant to the MHRA as part of its lower-emission pressurized inhaler program.
- In January 2026, Kinaset Therapeutics announced the closing of a USD 103 million Series B financing to advance an inhaled therapeutic candidate for respiratory disease.
Key Players in the Respiratory Inhaler Devices Market
Major Global Players
- GSK plc
- AstraZeneca
- Novartis AG
- Teva Respiratory LLC
- Boehringer Ingelheim International GmbH
- Cipla LTD
- Lupin Ltd
Device And Therapy Specialists
- Viatris Inc.
- CHIESI Farmaceutici S.p.A.
- OMRON Corporation
- Koninklijke Philips N.V.
- PARI GmbH
Report Scope and Coverage
| Item |
Value |
| Quantitative Units |
USD 40.2 billion in 2025 to USD 60.0 billion in 2036 at a 3.7% CAGR |
| Market Definition |
Inhalation drug delivery devices used across chronic and acute pulmonary care |
| Product Segmentation |
Dry Powder Inhaler and Metered Dose Inhaler and Nebulizer and Soft Mist Inhaler |
| Nebulizer Type |
Compressed Air Nebulizers and Ultrasonic Nebulizers and Mesh Nebulizers |
| Drug Class Segmentation |
SABAs and SAMA and LABAs and LAMA and ICS and Combination LABA LAMA and Combination ICS LABA |
| Technology Segmentation |
Manual Inhalers and Digital Inhalers |
| Disease Indication |
Asthma and Chronic Obstructive Pulmonary Disease and Pulmonary Arterial Hypertension and Acute Respiratory Distress and Pulmonary Fibrosis |
| Sales Channel |
Hospitals and Specialty Clinics and Retail Pharmacy Chains and Online Pharmacies or Mail Order Pharmacies |
| Regions Covered |
North America and Latin America and East Asia and South Asia and Pacific and Western Europe and Eastern Europe and Middle East and Africa |
| Countries Covered |
United States and Canada and United Kingdom and Germany and France and China and Japan and South Korea and India and 40 plus countries |
| Key Companies Profiled |
GSK plc and AstraZeneca and Novartis AG and Teva Respiratory LLC and Boehringer Ingelheim International GmbH and Cipla LTD and Lupin Ltd and Viatris Inc. and CHIESI Farmaceutici S.p.A. and OMRON Corporation and PARI GmbH |
| Forecast Period |
2026 to 2036 |
| Historical Reference Year |
2025 |
| Approach |
Hybrid top down and bottom-up modelling aligned to device volume and disease pool and channel mix |
Source. Future Market Insights analysis based on the supplied market model and covered country narratives.
Respiratory Inhaler Devices Market breakdown by Product, Nebulizer Type, Drug Class, Technology, Disease Indication, Sales Channel and Region
Respiratory Inhaler Devices Market Segmented by Product
- Dry Powder Inhaler
- Metered Dose Inhaler
- Nebulizer
- Soft Mist Inhaler
Respiratory Inhaler Devices Market segmented by Nebulizer Type
- Compressed Air Nebulizers
- Ultrasonic Nebulizers
- Mesh Nebulizers
Respiratory Inhaler Devices Market Segmented by Drug Class
- Short acting Beta2 Agonist Inhalers
- Short Acting Muscarinic Antagonist
- Long acting Beta2 Agonist Inhalers
- Long Acting Muscarinic Antagonist
- Corticosteroid Inhalers
- Combination LABA LAMA Inhalers
- Combination ICS LABA Inhalers
Respiratory Inhaler Devices Market Segmented by Technology
- Manual Inhalers
- Digital Inhalers
Respiratory Inhaler Devices Market Segmented by Disease Indication
- Asthma
- Chronic Obstructive Pulmonary Disease
- Pulmonary Arterial Hypertension
- Acute Respiratory Distress
- Pulmonary Fibrosis
Respiratory Inhaler Devices Market Segmented by Sales Channel
- Hospitals
- Specialty Clinics
- Retail Pharmacy Chains
- Online Pharmacies and Mail Order Pharmacies
Respiratory Inhaler Devices Market by Region
- North America
- Latin America
- Brazil
- Rest of Latin America
- East Asia
- South Asia and Pacific
- India
- ASEAN Countries
- Australia and New Zealand
- Rest of South Asia
- Western Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- Eastern Europe
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- Middle East and Africa
- Saudi Arabia
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East and Africa
Research Sources and Bibliography
- GSK plc. (2025, October 22). GSK announces positive pivotal phase III data for next-generation low-carbon version of Ventolin (salbutamol) metered dose inhaler.
- Halpin, D. M. G., Masekela, R., Vogelmeier, C. F., Ozoh, O. B., Cruz, A. A., Reddel, H. K., Yorgancıoğlu, A., Agustí, A., & the Boards of Directors of the Global Initiative for Chronic Obstructive Lung Disease (GOLD) and the Global Initiative for Asthma (GINA). (2026). Addressing the global challenges of COPD and asthma: A shared vision from the Global Initiative for Chronic Obstructive Pulmonary Disease (GOLD) and the Global Initiative for Asthma (GINA). European Respiratory Journal, 67(2), 2502244.
- Global Initiative for Asthma. (2023). Asthma management and prevention for adults, adolescents, and children 6–11 years: A pocket guide for health professionals.
- World Health Organization. (2024, May 6). Asthma.
- Global Initiative for Asthma. (2025). World Asthma Day 2025.
- Kinaset Therapeutics. (2026, January 10). Kinaset Therapeutics announces $103 million oversubscribed Series B financing to advance novel, inhaled therapeutic candidate for respiratory diseases.
- Chiesi Farmaceutici S.p.A. (2025, December 18). MHRA submission marks new step in Chiesi’s carbon minimal inhaler journey.
- Smith, D. (2025, November 17). 1nhaler secures £1.5 million in funding to advance paper inhaler platform. 1nhaler.
- AeroRx Therapeutics, Inc. (2025, October 7). AeroRx Therapeutics announces $21 million Series A financing to advance inhaled AERO-007, the first nebulized LABA/LAMA for COPD, into late-stage clinical development.
The bibliography is provided for reader reference.
This Report Answers
- What size is the respiratory inhaler devices market in 2026 and how large can it reach by 2036?
- How fast is respiratory inhaler device demand expected to expand from 2026 to 2036?
- Which product segment leads respiratory inhaler device demand in 2026 and what supports that lead?
- Which disease indication leads inhaler device use in 2026 and why does it keep the largest share?
- Why do chronic therapy continuity and handling ease keep familiar inhaler formats in strong circulation?
- Which countries are expected to post the fastest growth through 2036 for respiratory inhaler devices?
- Which companies hold the clearest current positions across prescription inhalers and nebulizer systems?
Frequently Asked Questions
How large is the global respiratory inhaler devices market in 2026?
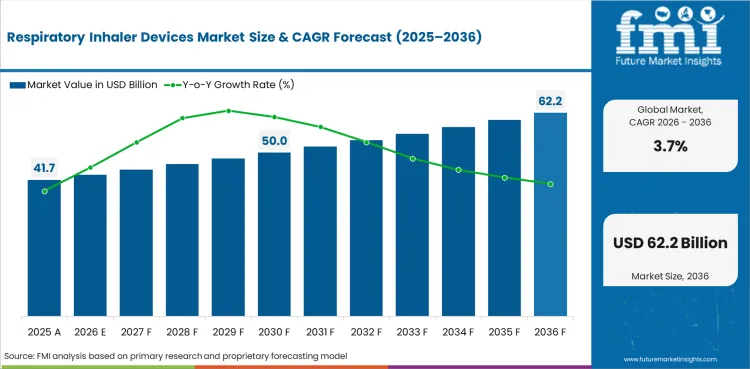
In 2026, the global respiratory inhaler devices market is expected to reach USD 41.7 billion across prescription inhaler and nebulizer demand worldwide.
How big can the respiratory inhaler devices market become by 2036?
By 2036, the respiratory inhaler devices market is projected to reach USD 60.0 billion as chronic therapy demand keeps refill activity firm.
What growth rate is expected for the respiratory inhaler devices market through the forecast period?
The respiratory inhaler devices market is projected to expand at a 3.7% CAGR from 2026 to 2036 during the forecast period.
Which product segment leads respiratory inhaler devices demand in 2026?
Metered dose inhalers are expected to account for 71.6% share of product demand in 2026 because rescue use and prescriber familiarity stay strong.
Which disease indication leads respiratory inhaler device use in 2026?
Asthma is expected to represent 36.7% share of disease indication demand in 2026 as long-duration treatment keeps inhaler refill need high.
Which country is expected to post the fastest growth for respiratory inhaler devices?
India is expected to record the fastest rise at 7.2% CAGR through 2036 because treatment access and diagnosis rates keep improving.
What does the respiratory inhaler devices market include?
The market includes inhalation drug delivery devices used across chronic and acute pulmonary care through dry powder and metered dose inhalers and nebulizers.
How was the respiratory inhaler devices forecast developed?
The forecast was developed through therapy demand mapping and route analysis then validated against channel spread and current company activity by region.








.webp)





