The global 3D printed drugs sector is on track to achieve a valuation of USD 1.1 billion by 2036, accelerating from USD 435.8 million in 2026 at a CAGR of 9.8%. As per Future Market Insights, expansion is structurally underpinned by the pharmaceutical industry's need for personalized dosage forms, complex release profiles, and on-demand compounding that cannot be achieved through conventional tablet pressing. The USA Food and Drug Administration approved Spritam (levetiracetam) in 2015 as the first 3D printed drug, and the regulatory pathway established by that approval is now being leveraged by multiple companies for new drug candidates. This regulatory precedent compels pharmaceutical firms to invest in 3D printing platforms that can achieve cGMP compliance at commercial production volumes. Simultaneously, the industry is shifting from single-drug printing to polypill platforms that combine multiple active ingredients in a single personalized dosage form.
In September 2025, Aprecia partnered with Apprentice.io to implement an AI-powered manufacturing platform at its Ohio facility, aiming to scale its proprietary 3DP technology for complex drug delivery solutions using cloud-native digital manufacturing tools. In March 2025, Triastek received IND clearance from the USA FDA for T20G, a 3D printed gastric retention product for cardiovascular conditions utilizing its Melt Extrusion Deposition (MED) technology. FMI opines that Triastek's FDA IND clearance for a 3D printed gastric retention product demonstrates that additive pharmaceutical manufacturing can create drug geometries, such as extended stomach-resident structures, that are physically impossible to produce through conventional tablet compression.
The operational reality for 3D printed drug suppliers is defined by the need to balance the personalization promise of additive manufacturing with the rigorous batch consistency and process validation required by pharmaceutical regulators. In September 2025, Triastek announced the construction of the world's largest commercial 3D printed pharmaceutical facility in Nanjing, capable of producing 300 million tablets annually. As per FMI, the 3D printed drugs market is entering a phase where commercial-scale production validation will be the key barrier to adoption, as the technology must prove it can match conventional manufacturing reliability at volume. FMI is of the opinion that pharmacy-level compounding platforms, such as CurifyLabs Create launched in October 2025, will drive near-term adoption by enabling personalized medications directly from APIs at the point of dispensing.
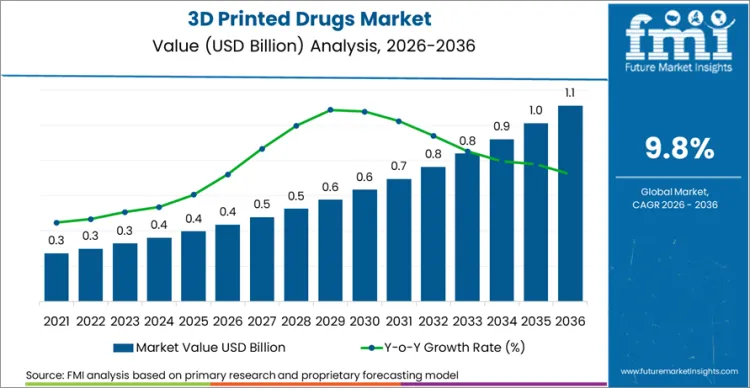
Future Market Insights projects the 3D printed drugs market to expand at a CAGR of 9.8% from 2026 to 2036, increasing from USD 435.8 Million in 2026 to USD 1.1 Billion by 2036.
FMI Research Approach: FMI proprietary forecasting model based on FDA 3D printed drug approval pathway data and pharmaceutical additive manufacturing investment trends.
FMI analysts perceive the market evolving toward commercial-scale validated production facilities capable of hundreds of millions of tablets annually, with pharmacy-level compounding platforms enabling personalized dosing at point of dispensing.
FMI Research Approach: Triastek Nanjing facility (September 2025) and CurifyLabs Create platform (October 2025).
The United States holds a significant share, supported by the FDA's established regulatory pathway for 3D printed drugs following the Spritam approval and Aprecia's commercial manufacturing operations in Ohio.
FMI Research Approach: FMI country-level revenue modeling by FDA drug approval data and 3D pharmaceutical manufacturing capacity.
The global 3D printed drugs market is projected to reach USD 1.1 Billion by 2036.
FMI Research Approach: FMI long-term forecast from pharmaceutical personalization trends and commercial 3D printing production scale-up data.
The 3D printed drugs market includes revenue from pharmaceutical products manufactured using additive technologies, including powder bed fusion, melt extrusion deposition, and inkjet-based printing, and from pharmacy-level 3D compounding platforms.
FMI Research Approach: FMI market taxonomy aligned with FDA additive pharmaceutical manufacturing guidance.
Trends include construction of commercial-scale 3D pharmaceutical factories, FDA IND clearances for complex-geometry drug candidates, and pharmacy-level compounding platforms for personalized medications.
FMI Research Approach: Triastek Nanjing 300M tablet facility (September 2025), T20G IND clearance (March 2025), and Aprecia-Apprentice.io AI manufacturing (September 2025).
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 435.8 Million |
| Industry Value (2036) | USD 1.1 Billion |
| CAGR (2026 to 2036) | 9.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
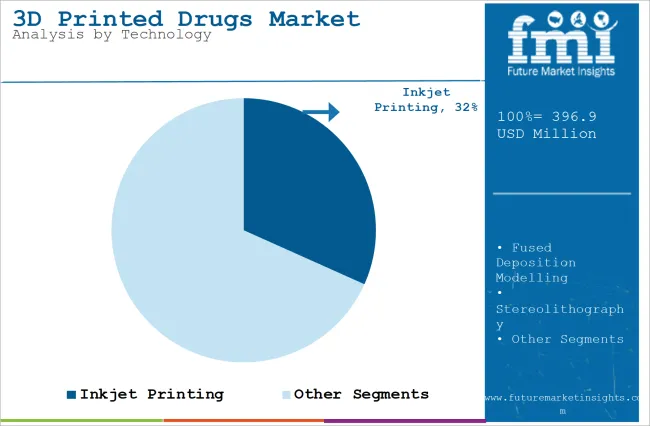
The section contains information about the leading segments in the industry. Based on technology, the inkjet printing segment is expected to account for 31.7% of the global share in 2026.
| Technology | Inkjet Printing |
|---|---|
| Value Share (2026) | 31.7% |
The inkjet printing segment is projected to be a dominating segment in terms of revenue, accounting for almost 31.7% of the market share in 2026.
The market is expected to be dominated by the Inkjet printing segment due to its versatility, precision, and efficiency in drug production. Inkjet printing enables the precise deposition of drug formulations, which allows for the creation of complex drug structures and personalized dosages. This technology is highly effective in producing multi-layered drug tablets and controlled-release formulations, which are crucial for personalized medicine.
Other significant advantages of the inkjet printer include faster speeds than other types of 3D printing techniques, which have enabled it for high-throughput drug manufacturing. The ability of this printer to print small batch orders on-demand minimizes cost waste and satisfies the demand of specific dosages or formulations of the drugs for unique patients. The relatively low equipment cost and ease of integration into existing pharmaceutical manufacturing processes add to the attractions of inkjet printing. The advantages make inkjet printing the dominant technology in the evolving 3D printed drugs market.
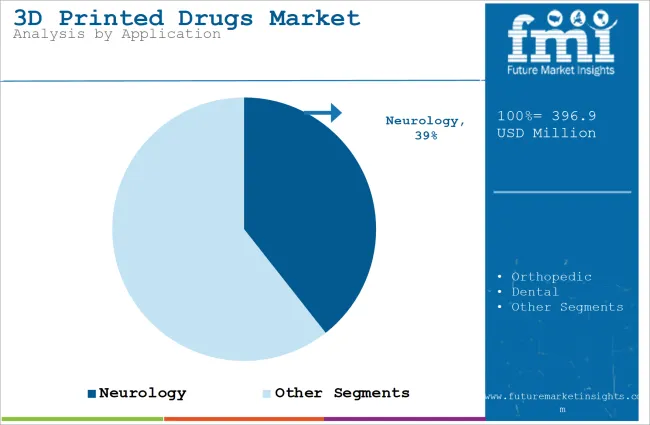
| Application | Neurology |
|---|---|
| Value Share (2026) | 39.4% |
The neurology segment will dominate the industry in terms of revenue, accounting for almost 39.4% of the market share in 2026. This is due to the high prevalence of knee injuries and degenerative conditions such as osteoarthritis, which commonly affect the knee joint.
The neurology segment is likely to be the prominent one in market owing to the rapidly increasing need for personalized treatment procedures for neurological disorders like Alzheimer's disease, Parkinson's disease, and epilepsy. Customization of drug formulation through 3D printing aids in formulating drugs for dosing precisely suited to a specific patient, more so in terms of conditions involving controlled-release mechanisms.
Neurological disorders often demand lengthy, complicated treatments, and 3D printing technology can be used for the development of customized drug delivery systems that ensure that the treatment will be consistent and effective. The technologies such as Inkjet printing enable the production of multi-layered tablets or multi-drug combinations in order to increase the targeting and absorption of drugs in the brain.
Moreover, the ability to print drugs with a specific release profile minimizes the side effects and optimizes therapeutic outcomes. As the incidence of neurological disorders increases across the globe, the demand for 3D-printed drugs in this segment is expected to increase rapidly, thereby driving its market dominance.
Increasing FDA approvals and regulatory support is driving the market growth
FDA approvals have driven considerable growth within the market by providing assurance that regulatory standards for safety and effectiveness are satisfied. As oversight organizations like the FDA continue extending regulatory support to this emerging field, the industry has been granted improved legitimacy and assurance.
Most notably, the approval of Spritam, the first 3D-printed pharmaceutical, demonstrated to manufacturers and investors that customized medicines produced through these techniques could satisfy requirements for quality while revolutionizing delivery.
Additionally, approvals have amplified adoption by signaling to all stakeholders that 3D-printed formulations can benefit patients through precisely tailored dosing while clearing mandated hurdles, revealing the technology's potential to individualize treatment.
In evolving 3D-printing technologies, FDA and regulatory bodies have been changing their frameworks accordingly, allowing new ways to accommodate such innovations. With approvals for more 3D-printed drugs, trust in this technology would rise and pave the way for pharma companies to enter and explore 3D printing for their future drug development, thereby enhancing the market potential.
Expansion of bioprinting application is driving the industry growth
Growing 3D printing technologies for the creation of biological tissues, drug delivery systems, and even the organs themselves, bioprinting provides drug development with an even more tailored level of accuracy. The possibility of creating complex structures with bioprinting allows pharmaceutical companies to design and manufacture personalized drug delivery systems for the unique needs of individual patients, thereby increasing the efficacy of treatment.
Further, advanced formulations such as multiple-drug combinations, controlled release systems, and tissue engineered drug delivery devices could be provided using bioprinting technology. Such applications can be critical for treating serious disorders such as cancer, neurology diseases, and chronic ailments requiring the highest possible degree of precise targeted delivery of drugs to be effective.
As research continues in bioprinting, the technology itself is becoming much more accessible and versatile, creating new potential avenues for pharmaceutical use. This, in turn, increases demand for 3D-printed drugs and unlocks new opportunities to provide more efficient, personalized treatments, further boosting the market expansion.
Advancements in 3D printing technology creates further growth opportunity for 3D printed drugs
Advancements in 3D printing technology open significant opportunities for the market of 3D printed drugs. Higher precision allows for more intricate and complex drug formulations to be produced with exact dosages and release profiles tailored to individual patients' needs. Such accuracy enhances therapeutic outcomes, especially in personalized medicine.
The speed of 3D printing has also improved, shortening the development and manufacturing time of new drug formulations. This is also enabling faster production cycles to meet the market needs of the rapidly increasing demand for customized treatments from pharmaceutical companies.
Moreover, new biocompatible polymers, APIs, and innovative excipients also expand the range of printable drugs. These enable more complex drug delivery systems such as controlled-release or multi-drug combinations.
As these improvements in technology are made, it can make 3D printing a more viable and scalable process for the production of drugs, opening many doors to further innovation in the pharmaceutical industry.
High initial investment costs of 3D printed drugs may restrict market growth
High initial investment costs are a significant restraint for the 3D printed drugs. The upfront costs of acquiring the 3D printing equipment, specialized materials, and setting up the necessary infrastructure can be costly, especially to smaller pharmaceutical companies or startups.
Unlike traditional methods of drug manufacturing that have established processes, 3D printing requires much capital for advanced printing machines, software, and research into bio-compatible materials that are suitable. This can become a barrier to entry for financially constrained companies.
For example, 3D printers, which can print pharmaceutical-grade products, have prices ranging from tens of thousands of dollars to hundreds of thousands of dollars. Also, the materials used in 3D printing, such as bio-inks and APIs, are costlier than their counterparts used in the drug industry traditionally. This tends to raise production costs and would also mean that small companies are locked out of 3D printing technologies.
As a result, the high initial investment needed to establish 3D printing technology limits its extensive adoption, particularly in regions or by companies that do not have the financial backup to invest in such advanced technologies.
Traditionally, pharmaceutical manufacturing used batch processing and mass production techniques. Drugs were manufactured in large quantities using conventional methods like compression, granulation, and coating. Such processes involved standardized formulations that were not adaptable to custom dosages or tailor-made treatments for patients.
Personalized medicines have specific dosing levels and the designed release profile that can be printed using 3D printing. Compared to traditional ways of drug manufacture, 3D printing creates a drug upon demand, lessens waste material, and can enhance efficiency through it. On-demand production offers 3D printing the chance to produce very complex drug forms like multi-drug combinations or controlled-release systems difficult to achieve in other conventional manufacturing methods. Finally, 3D printing may provide smaller manufacturing runs, increasing its flexibility with respect to individual treatments and small scale manufacture.
The major drivers in the growth of the market are cost-effectiveness and production efficiency. In contrast to the traditional method of large-scale production with substantial waste generation, 3D printing permits on-demand drug production with less material wastage and operational expenses in terms of inventory. Printing in exact dosages and customized formulations minimizes excess raw materials and packaging. Thirdly, 3D printing shortens production times, thereby allowing for a quicker turnaround with the fast time-to-market new formulation drugs.
This efficiency is very important for personalized medicine, in which small, customized batches are needed. Due to streamlined production, 3D printing is cheaper and drives their adoption among pharmaceutical companies that need higher quality while hoping to reduce costs.
Tier 1 companies are the industry leaders with 59.5% of the global industry. These companies stand out for having a large product portfolio and a high production capacity. These industry leaders also stand out for having a wide geographic reach, a strong customer base, and substantial experience in manufacturing and having enough financial resources which enables them to enhance their research and development efforts and expand into new industries.
The companies within tier 1 have a good reputation and high brand value. These companies frequently get involved in strategies such as acquisition and product launches. Prominent companies within tier 1 include Aprecia Pharmaceuticals LLC, Cycle Pharmaceuticals, Teva Pharmaceuticals and FabRx Ltd.
Tier 2 companies are relatively smaller as compared with tier 1 players. The tier 2 companies hold a market share of 26.6% worldwide. These firms may not have cutting-edge technology or a broad global reach, but they do ensure regulatory compliance and have good technology.
The players are more competitive when it comes to pricing and target niche markets. Key Companies under this category include Hewlett Packard Caribe, Osmotica Pharmaceuticals, Extend Biosciences, Bioduro Affinity Therapeutics and others
The market analysis for 3D printed drugs in various nations is covered in the section below. An analysis of important nations in North America, Latin America, Western Europe, Eastern Europe, South Asia & Pacific, and Middle East & Africa of the world has been mentioned below.
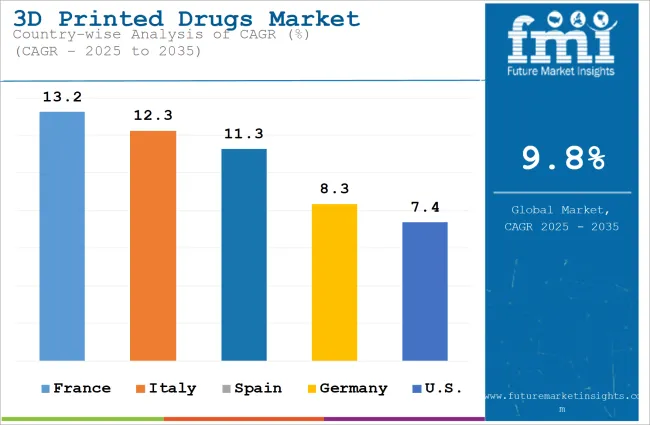
It is projected that the United States will maintain its leading position in North America through 2036, holding a value share of 88.6%. By 2036, China is expected to experience a CAGR of 12.7% in the Asia-Pacific region.
| Countries | Value CAGR (2026 to 2036) |
|---|---|
| USA | 11.4% |
| Germany | 10.7% |
| UK | 10.1% |
| France | 9.5% |
| China | 12.7% |
| South Korea | 10.9% |
| India | 10.0% |
Rising focus on precision medicine is a major factor for growth of this market in Germany
Germany’s 3D printed drugs market is poised to exhibit a CAGR of 10.7% between 2026 and 2036. The Germany holds highest market share in European market.
The strong push for precision medicine in Germany fuels the growth of the 3D printed drugs industry. 3D printing provides a significant platform for this aspect as it makes it possible to have customized drug formulations that are formulated to meet individual needs.
This means that pharmaceutical companies can make personalized doses, complex drug combinations, and targeted delivery systems, which would be extremely challenging or even impossible to achieve in a traditional manufacturing setup. This custom level enhances the efficacy of treatments, reduces side effects, and improves patient outcomes.
Consequently, if Germany continues to stand out in precision medicine research and innovation, it's only necessary for the demand for 3D-printed medicines to rise with it. It is through such capabilities that deliver highly personal patient-specific treatments that healthcare can advance and more adopt 3D printing technologies within the pharmaceutical industries.
Advanced research and development is a growth factor in the United States
United States is anticipated to show a CAGR of 11.4% between 2026 and 2036.
The USA is home to many top-notch companies and research institutions that spearhead the race for innovations in 3D printing technologies. They are pouring much money into researching how 3D printing could change drug formulation and delivery systems, especially with personalized medicine. This will involve the formulation of customized drugs with specific dosages, release profiles, and combinations of active ingredients.
With continuous R&D activities, new advancements in 3D printing materials, techniques, and precision can lead to increasingly efficient, economical, and individualized treatments for patients. Based in the USA, two companies, namely Aprecia Pharmaceuticals and Organovo, spearhead these emerging innovations, offering personalized medications as well as difficult-to-produce drug delivery systems that were unavailable using conventional production methods. Continuous investment in R&D ensures that the United States remains a hub in the development of 3D-printed drugs, further propelling this innovative market.
Increasing collaborations between Chinese companies and global pharmaceutical players is driving market in China
China is anticipated to show a CAGR of 12.7% between 2026 and 2036.
Partnerships with international pharmaceutical companies help the market to grow in China. With the major strengthening of the pharmaceutical industry in China, partnerships with the international players are speeding up the pace of adaptation to 3D printing technologies, and these collaborations have allowed Chinese companies to gain access to superior expertise, innovative technologies, and latest research in 3D printing, which are necessary to create personalized drug formulations and complex drug delivery systems.
Through these partnerships, Chinese firms will be able to benefit from international best practices in drug development, production efficiency, and compliance with regulations. As a result of partnership with multi-national pharmaceutical firms, state-of-the-art 3D printing techniques are imported into China.
Such innovation has created opportunities for developing customized treatments for chronic and complex diseases. Also, these collaborations ensure easier and faster market access of 3D-printed drugs. As such, collaborations are significantly driving the growth and commercialization of 3D-printed drug solutions across the market in China.
Key players in the market are adopting various growth strategies to strengthen their market position. These include strategic partnerships and collaborations with pharmaceutical companies and research institutions to leverage expertise and accelerate innovation. Companies are also investing in research and development (R&D) to improve 3D printing technologies, enhance precision, and develop customized drug formulations.
Additionally, market players are expanding their product portfolios by introducing new personalized drug delivery systems and controlled-release formulations. Moreover, some companies are focusing on geographical expansion to tap into emerging markets with growing healthcare needs.
Recent Developments
The 3D printed drugs market represents revenue from pharmaceutical products manufactured using additive manufacturing technologies and from 3D pharmaceutical printing platforms used in commercial production and pharmacy compounding.
Inclusions cover commercially manufactured 3D printed tablets, pharmacy-level 3D compounding equipment and consumables, and contract 3D pharmaceutical manufacturing services.
Exclusions include 3D printed medical devices (classified separately), bioprinted tissues, and general pharmaceutical manufacturing equipment not using additive processes.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 435.8 Million |
| Product Type | 3D Printed Tablets, Pharmacy Compounding Platforms, Contract Manufacturing Services |
| Application | Personalized Dosing, Complex Release Profiles, Polypill Formulations |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Key Companies Profiled | Aprecia, Triastek, CurifyLabs, FabRx, Merck KGaA |
In terms of technology, the industry is divided into inkjet printing, fused deposition modelling, stereolithography, zipdose technology, powder bed fusion and other technology.
In terms of application, the industry is segregated into neurology, orthopedic, dental, and other applications
In terms of end user, the industry is divided into hospitals & clinics, research laboratories and others
Key countries of North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe and Middle East and Africa (MEA) have been covered in the report.
What is the current global market size for 3D Printed Drugs?
The global market is valued at USD 435.8 Million in 2026, driven by FDA regulatory pathway maturation for additive drug manufacturing and the construction of commercial-scale 3D pharmaceutical production facilities.
What is the projected Compound Annual Growth Rate (CAGR) for the market over the next 10 years?
The market is projected to grow at a CAGR of 9.8% from 2026 to 2036.
Which regions are experiencing the fastest expansion?
North America leads with Aprecia's commercial manufacturing and FDA IND clearances, followed by China where Triastek is building the world's largest 3D printed pharmaceutical facility in Nanjing.
What are the primary market drivers?
FDA IND clearances for complex-geometry drug candidates and the construction of commercial-scale production facilities capable of 300 million tablets annually are the primary growth catalysts.
Who are the leading suppliers in the industry?
Aprecia, Triastek, CurifyLabs, FabRx, and Merck KGaA are key players, differentiating through proprietary 3D printing technologies, FDA-cleared production platforms, and pharmacy-level personalized compounding systems.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.