AAV Packaging Services Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
AAV Packaging Services Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
AAV Packaging Services Market Forecast and Outlook 2026 to 2036
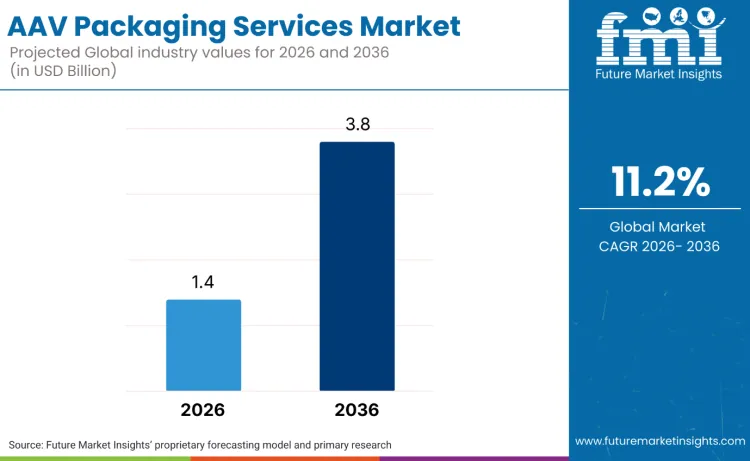
The global AAV packaging services market is projected to reach USD 1.4 billion in 2026 and expand to USD 3.8 billion by 2036, registering a CAGR of 11.2%. As per FMI, this growth depicts the growing adoption of adeno-associated virus vectors in gene therapy development, driven by expanding clinical pipelines across neuromuscular disorders, ophthalmology, and rare genetic conditions. Market expansion is driven by the increasing complexity of AAV manufacturing requirements, with biotechnology companies and pharmaceutical organizations seeking specialized packaging platforms that can navigate regulatory compliance while maintaining vector potency and quality.
The sector demonstrates a clear transition from research-grade packaging toward GMP-compliant manufacturing capabilities as gene therapy programs advance through clinical development phases. Contract development and manufacturing organizations are investing heavily in scalable AAV production platforms that integrate process development, analytical testing, and commercial supply capabilities under unified service frameworks. This consolidation addresses the technical challenges associated with AAV vector production, including capsid optimization, purification protocols, and quality control assays that ensure therapeutic efficacy across diverse genetic targets. Companies, such as Oxford Biomedica are increasingly focusing on GMP capacity to broaden their customer base. In October 2025, the company acquired Resilience’s site in North Carolina, an FDA-approved, commercial-scale viral vector manufacturing facility.
The signing of a late-stage programme, alongside the continued expansion of existing agreements since the start of 2024, demonstrates clients’ confidence in Oxford Biomedica’s ability to deliver process development and GMP manufacturing services. Our Business Development team continues to experience strong demand for process characterisation, validation, and process performance qualification (PPQ) work, showcasing the high quality and reliability of our services. This demand is in line with our forecasts across all key vectors and geographies, validating our growth strategy. We remain confident about our future prospects as the leading global pure-play CDMO in cell and gene therapy and our ability to continue building our client portfolio.”- Dr Sébastien Ribault, Chief Commercial Officer of Oxford Biomedica
Advanced manufacturing technologies, including suspension cell culture systems and downstream purification innovations, are reshaping service delivery models. Providers now offer end-to-end solutions that encompass vector design consultation, process optimization, and regulatory support, enabling gene therapy developers to accelerate development timelines while maintaining manufacturing consistency. The integration of real-time analytics and automated production systems allows for precise control over vector quality attributes, reducing batch-to-batch variability and supporting regulatory submissions across multiple jurisdictions.
Summary of AAV Packaging Services Market
What is the growth outlook on the AAV packaging services market?
The market is projected to grow at an 11.2% CAGR from 2026 to 2036, expanding from USD 1.4 billion to USD 3.8 billion.
How do analysts perceive the AAV packaging services market to evolve?
The market is expected to transition from fragmented research services toward integrated GMP manufacturing platforms that support full development lifecycles from preclinical through commercial production.
Which country holds the largest share in the AAV packaging services market?
The United States accounts for the largest global value share, driven by advanced gene therapy infrastructure and regulatory frameworks.
How large will the AAV packaging services market be by 2036?
The global market is forecast to reach USD 3.8 billion by 2036.
What is the market definition of AAV packaging services?
It encompasses specialized manufacturing services for adeno-associated virus vector production, including research-grade packaging, GMP clinical manufacturing, analytical testing, and commercial supply operations.
What unique trends are shaping the AAV packaging services market?
Key trends include platform standardization, automated production systems, and integrated service models that combine manufacturing with regulatory consulting and quality assurance.
AAV Packaging Services Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.4 billion |
| Market Forecast Value (2036) | USD 3.8 billion |
| Forecast CAGR (2026-2036) | 11.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
What is Driving the Demand for AAV Packaging Services?
Rising demand for gene therapies is fundamentally reshaping the AAV packaging services market, manifesting as a surge in outsourcing to CMO/CDMOs that can deliver scalable, compliant adeno-associated virus vectors. Integrated solutions that combine upstream production with high-yield vector packaging and analytics are rapidly becoming the new standard for developers seeking reliable supply continuity. Continued prioritization of safety, traceability, and time-to-clinic efficiency is further driving sustained investment in advanced AAV packaging infrastructure.
The shift toward commercialization of approved AAV-based therapies is creating sustained demand for scalable production platforms capable of supporting market supply requirements. Advanced vector engineering approaches, including tissue-specific targeting and enhanced transduction efficiency, require sophisticated manufacturing expertise that extends beyond traditional viral production methods. The increasing complexity of AAV capsid modifications and payload optimization has elevated the technical barriers to in-house manufacturing, driving outsourcing to specialized service providers.
Investment patterns in gene therapy development, particularly venture funding for rare disease applications, are supporting expanded infrastructure development across contract manufacturing organizations. The integration of process analytical technology and quality-by-design principles into AAV manufacturing workflows has increased the value proposition of outsourced services, enabling developers to access state-of-the-art production capabilities without significant capital investment.
How is the AAV Packaging Services Market Segmented?
The AAV packaging services market has been segmented based on service type, therapeutic application, end user, and region. In terms of service type, the market is divided into research-grade AAV packaging, GMP clinical packaging, late-stage commercial supply, analytical and release testing, and process development services. By therapeutic application, the market encompasses ophthalmology, neuromuscular and CNS disorders, metabolic and rare genetic disorders, oncology applications, and cardiovascular indications. By end user, the market includes emerging biotech companies, large pharmaceutical organizations, academic medical centers, government research institutions, and contract research organizations. Regionally, the market spans North America, Europe, East Asia, South Asia, Latin America, and Middle East and Africa.
Why Do GMP Clinical Packaging Services Dominate the AAV Packaging Services Market?
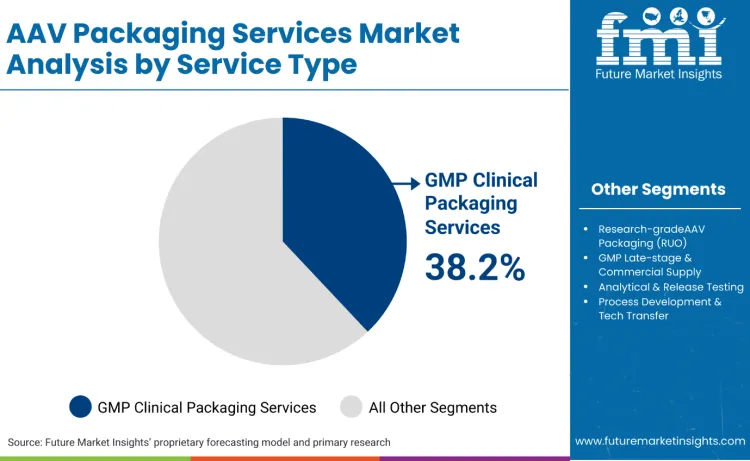
GMP clinical packaging services command the largest market share at 38.2%, driven by the substantial volume of AAV-based gene therapies progressing through Phase I and Phase II clinical trials. This service category addresses the critical manufacturing requirements for clinical-grade vector production, encompassing process validation, batch release testing, and regulatory compliance documentation essential for investigational new drug applications.
The dominance shows the current maturation stage of the gene therapy sector, where numerous AAV programs are transitioning from preclinical research into human testing phases. Clinical-grade manufacturing demands specialized expertise in vector purification, potency assays, and sterility testing that exceeds research-grade production standards. Companies require partners capable of producing vectors that meet clinical trial material specifications while maintaining scalability for potential commercial manufacturing. Lonza’s official viral vectors platform provides clinical‑ and commercial‑grade GMP manufacturing solutions for adeno‑associated virus (AAV) and other viral vectors, reflecting ongoing investments in regulated manufacturing capacity.
How Do Therapeutic Applications Influence AAV Packaging Service Selection?
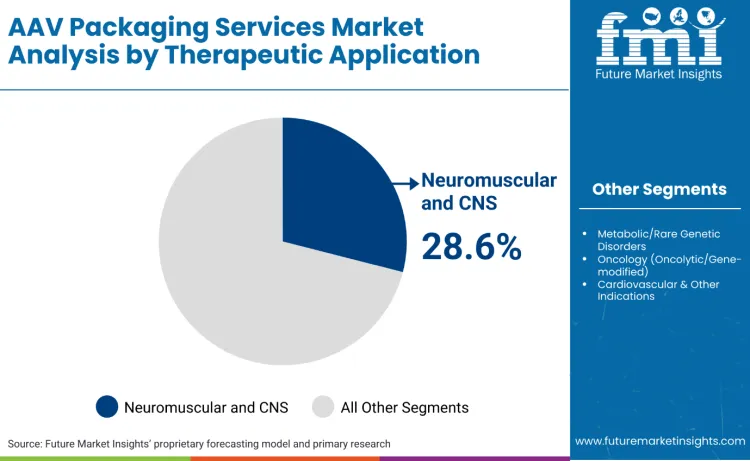
Neuromuscular and CNS applications lead the market with 28.6% share, driven by the exceptional potential of AAV vectors to cross the blood-brain barrier and achieve targeted gene delivery to neural tissues. These applications require specialized vector engineering approaches, including capsid modifications for enhanced CNS tropism and optimized delivery protocols that account for vector distribution patterns across nervous system tissues. Several leading companies in the AAV packaging services market, including Thermo Fisher Scientific, Lonza Group, Catalent, and Oxford Biomedica, are making significant investments in gene therapy infrastructure and viral vector production.
Thermo Fisher Scientific is actively supporting the gene therapy sector by offering end‑to‑end solutions that span research, development, manufacturing, and commercialization of gene‑based therapies. AAV packaging services are crucial for neuromuscular and central nervous system applications due to the complexity of gene therapies targeting these areas. These therapies require precise and scalable viral vector packaging to ensure effective delivery to targeted cells. The need for AAV packaging is driven by the intricate nature of these conditions, where treatment often involves high viral vector doses for efficient gene transfer. Furthermore, maintaining vector stability and minimizing degradation during the packaging process is vital for successful outcomes in clinical trials and patient treatment. The increasing demand for these services reflects the growing focus on gene-based therapies for challenging neurological disorders.
How Is Technological Evolution Reshaping AAV Packaging Services?
The AAV packaging sector is transitioning from traditional adherent cell culture methods toward scalable suspension cell platforms that enable higher vector yields and improved manufacturing economics. Bioreactor technologies specifically designed for AAV production are incorporating real-time monitoring capabilities that track vector formation, cell viability, and contamination risks throughout the production process. Advanced purification technologies, including chromatography systems optimized for AAV capsid separation and concentration, are improving vector recovery rates while reducing impurity levels. The implementation of single-use manufacturing systems is eliminating cross-contamination risks while reducing facility changeover times between different vector constructs.
For instance, Lonza Group AG is actively integrating advanced technologies into AAV packaging services by developing suspension‑based HEK293 platforms that boost AAV production yields and scalability, improving productivity and quality through optimized vector systems and automated processes. Quality control methodologies have advanced to include rapid potency assays and capsid characterization techniques that accelerate batch release timelines. The integration of artificial intelligence into production planning and quality assessment is enabling predictive manufacturing approaches that optimize yield and minimize batch failures. Automated systems for vector harvesting, purification, and formulation are reducing manual intervention while improving process consistency across manufacturing campaigns.
How Is the AAV Packaging Services Market Evolving Globally?
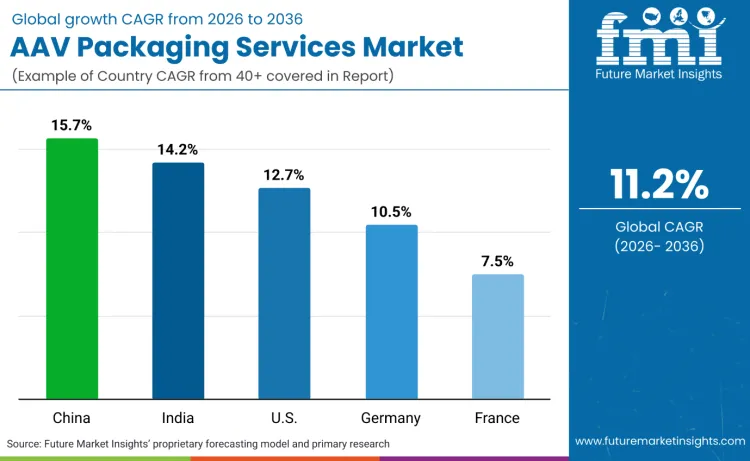
Global demand for AAV packaging services is accelerating as gene therapy development expands across diverse therapeutic areas and geographic regions. Growth demonstrates increasing adoption of outsourced manufacturing models, with biotechnology companies prioritizing access to specialized expertise over internal capability development. Service selection emphasizes manufacturing reliability, regulatory compliance, and technical support capabilities across pharmaceutical institutions and research organizations. China records 15.7% CAGR, India records 14.2% CAGR, USA records 12.7% CAGR, Germany records 10.5% CAGR, and France records 7.5% CAGR. Adoption patterns reflect regional gene therapy development maturity and regulatory infrastructure supporting clinical advancement.
| Country | CAGR (2026-2036) |
|---|---|
| China | 15.7% |
| India | 14.2% |
| USA | 12.7% |
| Germany | 10.5% |
| France | 7.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
China: How is Expanding Gene Therapy Infrastructure Driving Service Adoption?
China's AAV packaging services market is experiencing rapid expansion driven by government initiatives supporting gene therapy development and the establishment of specialized manufacturing facilities across major biotechnology hubs. Domestic pharmaceutical companies are advancing AAV-based clinical programs targeting rare genetic disorders and cancer applications, creating sustained demand for manufacturing services that meet international quality standards. The rise in demand for cancer gene therapy and precision genomics is fostering the market growth.
The Chinese AAV packaging services market is set to grow at 15.7% CAGR during the study period. The regulatory environment supporting gene therapy clinical trials has matured significantly, with streamlined approval pathways encouraging domestic and international companies to conduct clinical studies within China. Investment in contract manufacturing infrastructure is expanding local production capabilities while reducing dependence on overseas suppliers for clinical trial materials.
India: Is Research Excellence Elevating Manufacturing Standards?
India's market growth shows expanding biotechnology research capabilities and the development of manufacturing infrastructure supporting gene therapy applications. Academic medical centers are increasing collaboration with contract manufacturing organizations to advance translational research programs targeting genetic disorders prevalent in South Asian populations. The government is also taking active steps in advancing gene therapy. The Government of India has launched initiatives to strengthen biotechnology and gene therapy innovation under programs like the Biopharma SHAKTI.
The emergence of specialized AAV manufacturing facilities is positioning India as a regional hub for gene therapy production, supported by skilled scientific workforce and competitive manufacturing costs. Regulatory frameworks are evolving to support gene therapy development while maintaining quality standards aligned with international guidelines. The Indian AAV packaging services market is set to grow at 14.2% CAGR during the study period.
USA: How is the Innovation Leadership Strategy Advancing Therapeutic Development?
The USA market maintains leadership through advanced manufacturing technologies and regulatory expertise that supports rapid clinical program advancement. The FDA's established gene therapy guidance documents provide clear development pathways that enable efficient manufacturing planning and regulatory submissions. The USA AAV packaging services market is set to grow at 12.7% CAGR during the study period.
Investment in next-generation manufacturing platforms is concentrated in major biotechnology clusters, where contract organizations are developing automated production systems capable of supporting both clinical and commercial supply requirements. The integration of advanced analytics and process control technologies is establishing new benchmarks for manufacturing efficiency and product quality. Several key players in the AAV packaging services market, such as Thermo Fisher Scientific Inc. have made substantial investments in gene therapy manufacturing capabilities, strengthening their support for the rapidly growing field of cell and gene therapies. In 2025, Thermo Fisher Scientific Inc. opened its state‑of‑the‑art Advanced Therapies Collaboration Center in Carlsbad, California.
Germany: Can Advanced Bioprocessing Capabilities Strengthen Competitive Position?
Germany's market growth is supported by established pharmaceutical manufacturing expertise and expanding gene therapy development infrastructure. Contract manufacturing organizations are leveraging advanced bioprocessing capabilities to support European clinical programs while meeting stringent regulatory requirements across multiple jurisdictions.
The German AAV packaging services market is set to grow at 10.5% CAGR during the study period. The focus on manufacturing innovation includes development of platform technologies that can accommodate diverse vector constructs while maintaining consistent production standards. Investment in automated manufacturing systems is enhancing production capacity while reducing operational costs across major manufacturing facilities.
France: How is the Transformation in Gene Therapy Taking the Healthcare Industry By Storm?
The French AAV packaging services market is growing because the overall gene therapy sector in France is expanding rapidly with strong revenue growth and rising demand for viral vector‑based treatments, especially those using AAV vectors. Increasing investments in research and development of gene therapies and more clinical programs require reliable and scalable AAV production capacity locally. France’s strong biotechnology infrastructure and supportive regulatory environment attract both domestic and international developers to outsource packaging services to specialized providers within the country, which further boosts the market for AAV packaging services.
As one of the leading biotech hubs in Europe, France offers robust government support for biotechnology innovation, including incentives for gene therapy companies. The country's collaboration with academic institutions and its well-established clinical trial networks contribute to the acceleration of gene therapies, driving up the need for AAV packaging services.
Companies are also securing heavy funds from the government to develop novel gene therapies. For instance, in 2024, the government of France allocated USD 5.7 million to Vivet Therapeutics (“Vivet”), a clinical stage biotech company to advance the development of a gene therapy for the treatment of cerebrotendinous xanthomatosis (CTX), a rare neurodegenerative disease. The French AAV packaging services market is set to grow at 7.5% CAGR during the study period.
Who are the Major Players Active in the AAV Packaging Services Market?
The competitive landscape is witnessing stiff competition among established contract development and manufacturing organizations competing on manufacturing capacity, technical expertise, and regulatory compliance capabilities. Major players are investing heavily in expanding their production capacities and enhancing their technological capabilities to support clinical and commercial-scale gene therapy manufacturing. These companies work closely with biotech firms to provide flexible and scalable solutions for viral vector production, ensuring the successful development and delivery of gene therapies. As the AAV packaging services market continues to grow, key players like.
Thermo Fisher Scientific, Lonza, and Catalent are competing by offering comprehensive solutions that address the entire gene therapy development and manufacturing process. These companies are investing heavily in expanding their viral vector production capacities, incorporating cutting-edge technologies to streamline gene therapy development. Additionally, the industry is increasingly embracing artificial intelligence and automation to enhance efficiency and precision in packaging and manufacturing. This shift allows smaller biotech firms to focus on research and development while outsourcing the complex manufacturing processes to these larger service providers.
The competitive landscape is also witnessing regional differences in strategy. While North American players like Thermo Fisher and Catalent lead the market with robust infrastructure and proximity to top-tier research institutions, European companies like Oxford Biomedica and AGC Biologics are focusing on strengthening their regulatory compliance and capacity for large-scale production. Asia-Pacific players, particularly in China and South Korea, are rapidly expanding their viral vector capabilities, leveraging cost advantages and increasing investment in high-throughput systems. This global expansion is fostering a competitive environment where innovation in vector production and packaging services remains crucial for maintaining market leadership.
Key Industry Developments:
- In October 2024, Lonza acquired Genentech large-scale biologics manufacturing site in Vacaville, California (US) from Roche for USD 1.2 billion.
- In January 2024, Oxford Biomedica plc acquired ABL Europe to boost its position as a world-leading cell and gene therapy CDMO.
Key Players in the AAV Packaging Services Market
- Thermo Fisher Scientific Inc.
- Lonza Group AG
- Catalent, Inc.
- Viralgen Vector Core S.L.
- WuXi Advanced Therapies Inc.
- Oxford Biomedica plc
- FUJIFILM Diosynth Biotechnologies (UK) Limited
- Andelyn Biosciences, Inc.
- AGC Biologics A/S
- Batavia Biosciences B.V.
Market Definition
The AAV packaging services market refers to specialized manufacturing and development services supporting adeno-associated virus vector production for gene therapy applications. It includes research-grade packaging for preclinical studies, GMP-compliant manufacturing for clinical trials, analytical testing services, and commercial supply operations. The market encompasses service providers that offer vector production capabilities across diverse therapeutic applications including ophthalmology, neuromuscular disorders, and rare genetic conditions. The market includes both contract development and manufacturing organizations that provide integrated services spanning process development through commercial supply, as well as specialized providers focusing on specific aspects of AAV manufacturing. Market sizing reflects service revenues generated through outsourced AAV production rather than internal manufacturing activities conducted by pharmaceutical companies.
Market Inclusion
Included in the AAV packaging services market are manufacturing services categorized by development stage (research-grade, clinical, commercial), therapeutic application (neuromuscular, ophthalmology, metabolic disorders), and service scope (process development, analytical testing, regulatory support). The scope encompasses vector production platforms designed for therapeutic applications, with segmentation based on manufacturing complexity, regulatory compliance requirements, and production scale. Geographically, the market includes major regions supporting gene therapy development: North America, Europe, East Asia, South Asia, Latin America, and Middle East and Africa, with detailed analysis for countries demonstrating significant market activity including United States, China, Germany, and India.
Market Exclusion
Excluded from the market scope are non-AAV viral vector services, including lentiviral and retroviral manufacturing platforms. Research reagent production for academic applications outside therapeutic development pathways is not included. Manufacturing services for gene therapy applications using non-viral delivery systems are excluded from market analysis. The market excludes internal manufacturing activities conducted by pharmaceutical companies for proprietary programs, focusing specifically on outsourced service revenues. Equipment sales and technology licensing arrangements are not included in market sizing calculations.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Service Type | Research-grade AAV Packaging (RUO); GMP Clinical AAV Packaging (Phase I/II); GMP Late-stage & Commercial Supply (Phase III/PPQ/Commercial); Analytical & Release Testing (Potency, VG, Empty/Full, Sterility); Process Development & Tech Transfer (Packaging-related) |
| Therapeutic Application | Ophthalmology (Retinal/Ocular); Neuromuscular & CNS; Metabolic/Rare Genetic Disorders; Oncology (Oncolytic/Gene-modified); Cardiovascular & Other Indications |
| End User | Emerging Biotech & CGT Start-ups; Large Biopharma/Pharma Partners; Academic Medical Centers & Translational Labs; Government/Public-sector Research; CROs/Specialty Contract Labs (Channel) |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | China, India, USA, Germany, France, and 40+ countries |
| Key Companies Profiled | Thermo Fisher Scientific; Lonza; Catalent; Viralgen; WuXi Advanced Therapies; Others |
| Additional Attributes | Revenue analysis by service type, therapeutic application, and end user; performance in manufacturing applications and production capabilities across pharmaceutical institutions, research facilities, and biotechnology applications; manufacturing efficiency enhancement, quality optimization, and platform benefits under clinical applications; impact on development outcomes, production precision, and platform differentiation during procurement processes; compatibility with regulatory protocols and development preferences; procurement dynamics driven by GMP positioning, manufacturing innovation programs, and long-term development partnerships. |
AAV Packaging Services Market by Segment
Service Type:
- Research-grade AAV Packaging (RUO)
- GMP Clinical AAV Packaging (Phase I/II)
- GMP Late-stage & Commercial Supply
- Analytical & Release Testing
- Process Development & Tech Transfer
Therapeutic Application:
- Ophthalmology (Retinal/Ocular)
- Neuromuscular & CNS
- Metabolic/Rare Genetic Disorders
- Oncology (Oncolytic/Gene-modified)
- Cardiovascular & Other Indications
End User:
- Emerging Biotech & CGT Start-ups
- Large Biopharma/Pharma Partners
- Academic Medical Centers & Translational Labs
- Government/Public-sector Research
- CROs/Specialty Contract Labs
Region:
- East Asia
- China
- Japan
- South Korea
- Taiwan
- Rest of East Asia
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- BENELUX
- Nordic
- Rest of Europe
- North America
- USA
- Canada
- Mexico
- South Asia
- India
- Australia & New Zealand
- Southeast Asia
- Rest of South Asia
- Latin America
- Brazil
- Chile
- Colombia
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- USA Food and Drug Administration. (2024). Considerations for the development of chimeric antigen receptor (CAR) T cell products: Guidance for industry. USA Department of Health and Human Services.
- European Medicines Agency. (2023). Guideline on quality, non-clinical and clinical requirements for gene therapy medicinal products. European Medicines Agency.
- National Institutes of Health. (2023). Gene therapy research supported by the NIH: Program overview and funding priorities. National Institutes of Health.
- World Health Organization. (2024). WHO technical report series: Recommendations for the quality control of gene therapy products. World Health Organization.
Frequently Asked Questions
How big is the AAV packaging services market in 2026?
The global AAV packaging services market is estimated to be valued at USD 1.4 billion in 2026.
What will be the size of the AAV packaging services market in 2036?
The market size for AAV packaging services is projected to reach USD 3.8 billion by 2036.
How much will the AAV packaging services market grow between 2026 and 2036?
The AAV packaging services market is expected to grow at an 11.2% CAGR between 2026 and 2036.
What are the key service types in the AAV packaging services market?
The key service types include research-grade AAV packaging, GMP clinical packaging, late-stage commercial supply, and analytical testing services.
Which end user will contribute a significant share in the AAV packaging services market in 2026?
Emerging biotech and CGT start-ups are set to command a 54.3% share in the AAV packaging services market in 2026.
Table of Content
- AAV Packaging Services Market Size and Share Forecast Outlook (2026 to 2036)
- AAV Packaging Services Market Forecast and Outlook (2026-2036)
- AAV Packaging Services Market Key Takeaways
- Key Shifts in the AAV Packaging Services Market from 2026 to 2036
- AAV Packaging Services Market Segmentation Analysis
- By Service Type
- Research-grade AAV Packaging (RUO)
- GMP Clinical AAV Packaging (Phase I/II)
- GMP Late-stage & Commercial Supply (Phase III/PPQ/Commercial)
- Analytical & Release Testing (Potency, VG, Empty/Full, Sterility)
- Process Development & Tech Transfer (Packaging-related)
- By Therapeutic Application
- Ophthalmology (Retinal/Ocular)
- Neuromuscular & CNS
- Metabolic/Rare Genetic Disorders
- Oncology (Oncolytic/Gene-modified)
- Cardiovascular & Other Indications
- By End User
- Emerging Biotech & CGT Start-ups
- Large Biopharma/Pharma Partners
- Academic Medical Centers & Translational Labs
- Government/Public-sector Research
- CROs/Specialty Contract Labs (Channel)
- By Region
- By Service Type
- Market Dynamics and Growth Analysis
- Growth Drivers
- Market Restraints
- Strategic Trends
- AAV Packaging Services Market Global Analysis
- Asia Pacific
- North America
- Europe
- Latin America
- Middle East & Africa
- Competitive Landscape Analysis
- Key Players in the AAV Packaging Services Market
- Market Definition, Inclusion, and Exclusion Criteria
- Scope of the AAV Packaging Services Market Report
- AAV Packaging Services Market by Segment
- Bibliography
- Frequently Asked Questions (FAQs)
List of Tables
- Global AAV Packaging Services Market Size (USD Billion), 2026-2036
- AAV Packaging Services Market Value and CAGR Summary
- AAV Packaging Services Market Share by Service Type, 2026
- AAV Packaging Services Market Share by Therapeutic Application, 2026
- AAV Packaging Services Market Share by End User, 2026
- AAV Packaging Services Market CAGR by Country, 2026-2036
- AAV Packaging Services Market Regional Growth Rates
- Competitive Market Share of Leading Players
- AAV Packaging Services Market Segment-wise Revenue Contribution
- AAV Packaging Services Market Opportunity Analysis
List of Figures
- Global AAV Packaging Services Market Size Trend (2026-2036)
- AAV Packaging Services Market Growth Rate Overview
- AAV Packaging Services Market Share by Service Type (%)
- AAV Packaging Services Market Share by Therapeutic Application (%)
- AAV Packaging Services Market Share by End User (%)
- Regional Distribution of AAV Packaging Services Market
- Competitive Landscape of AAV Packaging Services Market
- Key Technological Trends Impacting AAV Packaging Services Market

