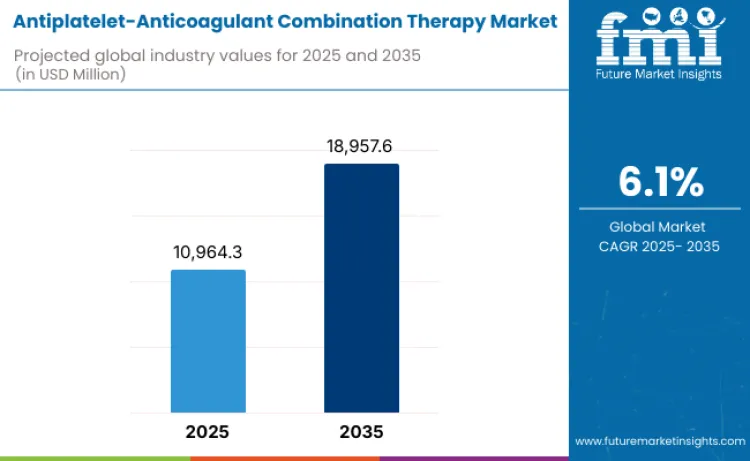
Antiplatelet-anticoagulant combination therapy market is projected to reach USD 18,957.6 million by 2035, recording an absolute increase of USD 7,993.3 million over the forecast period. This market is valued at USD 10,964.3 million in 2025 and is set to rise at a CAGR of 6.1% during the assessment period.
Overall size is expected to expand by nearly 1.7x times, supported by rising prevalence of acute coronary syndromes, increasing incidence of atherothrombotic events, and growing therapeutic adoption for high-risk cardiovascular patients requiring intensified thromboprophylaxis. Concerns related to bleeding risk, variable patient response, and cautious reimbursement policies in several regions may restrain near-term expansion.
Antiplatelet-Anticoagulant Combination Therapy Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 10,964.3 million |
| Market Forecast Value (2035) | USD 18,957.6 million |
| Forecast CAGR (2025 to 2035) | 6.1% |
Therapeutic advancements in factor Xa inhibitors, P2Y12 antagonists, and direct thrombin inhibitors are reshaping the combination therapy landscape. Modern regimens incorporate targeted agents with predictable pharmacokinetics, reduced drug interactions, and improved safety profiles, enabling optimized dosing strategies across diverse patient populations. Integration of combination therapy into post-PCI management, atrial fibrillation care frameworks, and peripheral artery disease treatment pathways supports broad clinical applicability.
Government cardiovascular disease-reduction initiatives, public health screening programs, and investment in advanced cardiac care infrastructure contribute to acceleration. Hospitals and specialty cardiac networks are adopting standardized antithrombotic protocols to address rising hospitalization rates associated with myocardial infarction, stroke, and thromboembolic complications.
Between 2025 and 2030, antiplatelet-anticoagulant combination therapy market is projected to expand from USD 10,964.3 million to USD 14,417.2 million, resulting in a value increase of USD 3,452.9 million, which represents 43.2% of total forecast growth for the decade. This phase will be shaped by increasing treatment demand among patients with acute coronary syndromes, rising use of intensified thromboprophylaxis in high-risk cardiovascular populations, and broader clinical adoption supported by evolving guideline recommendations.
From 2030 to 2035, growth is forecast to expand from USD 14,417.2 million to USD 18,957.6 million, adding another USD 4,540.4 million, which constitutes 56.8% of overall ten-year expansion. This period is expected to be characterized by expanded integration of precision-based antithrombotic regimens, increasing availability of next-generation factors Xa and P2Y12 inhibitor combinations, and strengthened real-world evidence supporting long-term use in complex patient subsets.
Antiplatelet-anticoagulant combination therapy market grows by enabling cardiovascular clinicians to address residual thrombotic risk that persists despite single-agent therapy, supporting improved protection against myocardial infarction, stroke, and systemic embolic events in high-risk patient groups. Cardiologists face mounting pressure to reduce recurrent ischemic outcomes across acute coronary syndrome patients, post-PCI populations, and individuals with overlapping atherosclerotic and thromboembolic conditions, with combination regimens demonstrating superior event-reduction benefits compared to monotherapy options in multiple large-scale clinical studies.
Government-led cardiovascular disease control programs, expansion of tertiary cardiac care infrastructure, and increasing deployment of guideline-directed therapy pathways drive broader clinical adoption of combination regimens across hospitals and specialty cardiac centers. Clinical societies, including ESC, ACC, and AHA, continue to emphasize risk-stratified use of dual-pathway inhibition, reinforcing clinician confidence and accelerating therapy integration across diverse care settings. Rising global adoption of minimally invasive coronary interventions and expanding use of PCI in low- and middle-income regions contribute to sustained therapeutic demand.
Antiplatelet-anticoagulant combination therapy market is segmented by regimen type, antiplatelet backbone, indication, end user, and region. By regimen type, it is divided into DOAC plus antiplatelet (dual pathway inhibition), VKA plus antiplatelet, parenteral anticoagulant plus antiplatelet, and triple therapy short-course. Based on antiplatelet backbone, it is categorized into aspirin plus P2Y12, aspirin only plus OAC, P2Y12 only plus OAC, and other adjunct combinations.
By indication, it is segmented into stable CAD or chronic coronary syndrome, ACS or post-stent PCI, peripheral artery disease, atrial fibrillation with PCI, and VTE with coexisting ASCVD. End users include hospitals, cardiology clinics, retail or e-pharmacies, and integrated health systems. Regionally, it is divided into Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
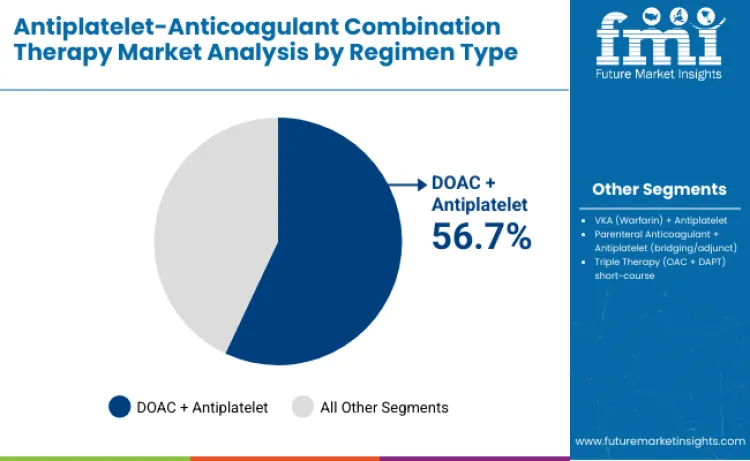
DOAC plus antiplatelet segment represents the dominant force, capturing 56.7% of total share in 2025. This segment encompasses dual-pathway inhibition regimens combining factor Xa or thrombin inhibition with targeted platelet suppression, supported by strong clinical evidence demonstrating reduced major cardiovascular events with improved safety profiles. Its dominant position is driven by predictable pharmacokinetics, simplified dosing, fewer interactions, and strong real-world adoption across post-PCI and chronic coronary syndrome populations where ischemic protection must be balanced with bleeding risk.
VKA plus antiplatelet segment maintains a 19.3% share, primarily serving regions where VKA remains standard due to cost or long-term treatment familiarity. Parenteral anticoagulant plus antiplatelet combinations account for 9.6%, used primarily for bridging or acute in-hospital management. Triple therapy short-course contributes 14.4%, reserved for selective high-risk patients requiring temporary intensified thromboprophylaxis.
Key advantages supporting DOAC plus antiplatelet dominance include:
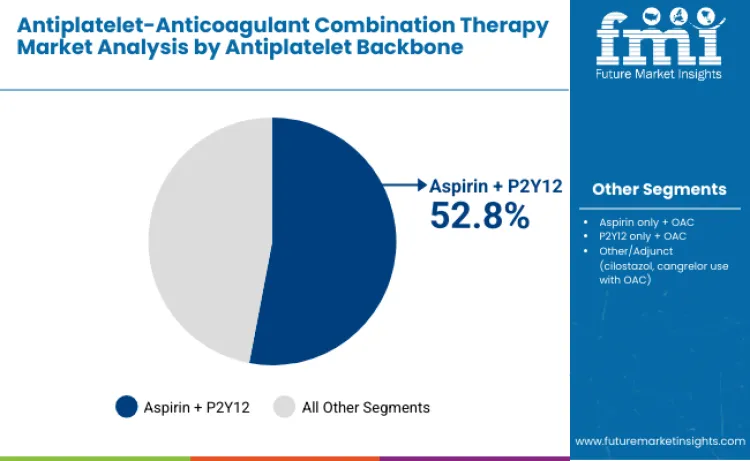
Aspirin plus P2Y12 segment dominates with 52.8% share in 2025, reflecting its status as the cornerstone antiplatelet backbone in dual-pathway inhibition regimens. This segment benefits from robust clinical validation, established PCI protocols, and strong integration into global ACS and stable CAD treatment guidelines. Combination's ability to deliver comprehensive platelet inhibition while supporting compatibility with DOAC therapy strengthens its leadership across acute and chronic cardiovascular management pathways.
Aspirin only plus OAC holds 18.7%, used in patients with elevated bleeding risk or intolerance to dual antiplatelet therapy. P2Y12 only plus OAC accounts for 21.6%, increasingly adopted in individuals requiring aspirin-sparing strategies. Other adjunct combinations represent 6.9%, supporting specialized clinical needs such as cilostazol or cangrelor use in select populations.
Key market dynamics supporting aspirin + P2Y12 dominance include:
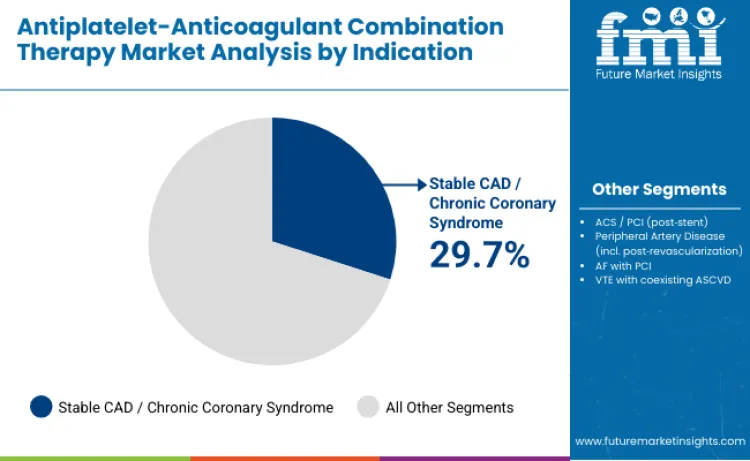
Stable CAD or chronic coronary syndrome represents the largest segment in 2025, accounting for 29.7% of total share. This dominance is driven by growing global burden of chronic atherosclerotic disease, rising management of long-term residual risk, and expanded use of dual-pathway inhibition in chronic coronary populations based on emerging evidence demonstrating reduction in long-term cardiovascular events. Strong uptake within primary and secondary prevention frameworks reinforces this segment's leadership.
ACS or post-stent PCI follows closely with 27.6%, driven by expanding global PCI volumes and structured post-procedure antithrombotic protocols. Peripheral artery disease contributes 18.9%, strengthened by increasing recognition of limb and cardiovascular event risk in PAD populations. Atrial fibrillation with PCI accounts for 17.4%, while VTE with coexisting ASCVD represents 6.4%.
Key factors supporting stable CAD segment dominance include:
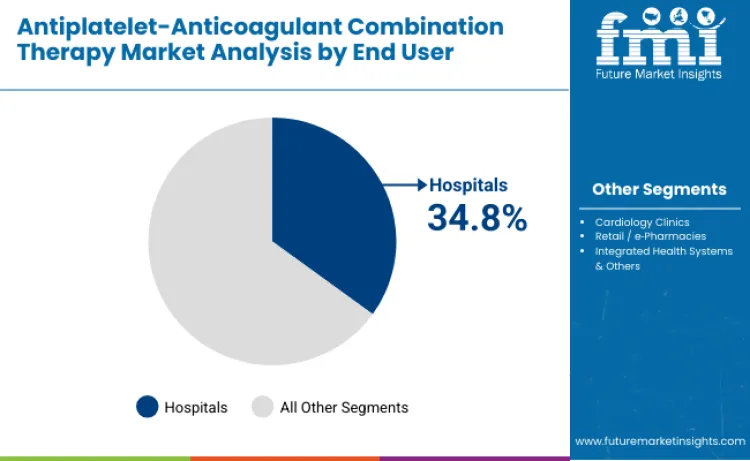
Hospitals dominate with 34.8% share in 2025, reflecting their central role in acute cardiovascular management, PCI procedures, and initiation of antithrombotic therapy. Hospitals serve as primary hubs for ACS stabilization, interventional cardiology procedures, and in-hospital optimization of dual-pathway regimens, resulting in high therapeutic utilization across catheterization labs, cardiac care units, and inpatient monitoring settings.
Cardiology clinics account for 27.9%, driven by structured follow-up, risk reassessment, and long-term therapy adjustments. Retail and e-pharmacies contribute 31.6%, supported by growing chronic therapy continuation and increased patient access to refills outside hospital systems. Integrated health systems and other centers represent 5.7%, providing coordinated long-term management for complex cardiovascular patients.
Key market dynamics supporting hospital leadership include:
The market expands due to rising cardiovascular disease prevalence, increased atrial fibrillation diagnoses, and broader use of dual-pathway inhibition strategies. Growing global CVD burden among aging and metabolic-risk populations drives demand for intensified regimens that provide stronger ischemic protection. Clinical guidelines increasingly endorse DOAC-antiplatelet combinations, especially after PCI, where residual thrombotic risk persists despite monotherapy. Rapid growth in interventional cardiology procedures, including PCI and revascularization, strengthens the need for optimized peri-procedural and post-discharge antithrombotic therapies that reduce long-term cardiovascular events and improve patient outcomes across hospital and outpatient settings.
What Are the Major Restraints Limiting Adoption of Combination Antithrombotic Therapy?
Key restraints include heightened bleeding risk, which necessitates careful patient selection and limits broad prescribing confidence. Combination therapy often requires structured monitoring, making adoption difficult in resource-constrained healthcare systems. Cost differences between DOAC-based combinations and VKA-anchored regimens restrict access, especially where reimbursement is inconsistent. Regulatory caution after bleeding events or in multi-comorbidity patients slows initiation. Long-term adherence challenges, limited use of risk-assessment tools, and regional disparities in payer coverage further contribute to uptake variability, reducing uniform global adoption despite strong clinical evidence supporting therapeutic effectiveness.
What Emerging Trends Are Shaping the Future of the Antiplatelet-Anticoagulant Combination Therapy Market?
Emerging trends emphasize precision-based antithrombotic strategies using risk-stratification algorithms to tailor therapy duration and intensity. Real-world evidence, digital adherence monitoring, and remote patient-management tools enhance safety tracking and long-term outcome optimization. New DOAC formulations and reduced-dose combination regimens are being evaluated in expanded clinical trials to balance efficacy and bleeding risk. Outpatient cardiology networks increasingly integrate structured therapy pathways. Market evolution may shift as AI-enabled bleeding-risk prediction models, novel vascular-protective agents, and next-generation antithrombotics compete for inclusion in secondary-prevention frameworks across high-risk cardiovascular populations.
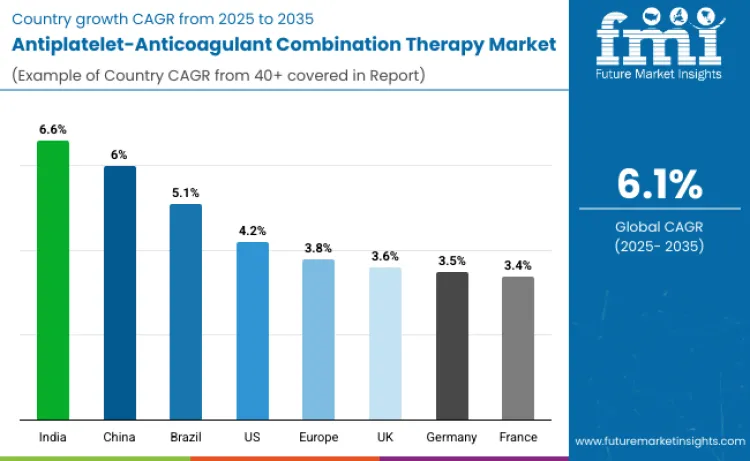
| Country/Region | CAGR (2025-2035) |
|---|---|
| India | 6.6% |
| China | 6.0% |
| Brazil | 5.1% |
| USA | 4.2% |
| Europe | 3.8% |
| UK | 3.6% |
| Germany | 3.5% |
| France | 3.4% |
The antiplatelet-anticoagulant combination therapy landscape is expanding globally, led by India (6.6%) due to rising cardiovascular disease burden, growing PCI volumes, and rapid adoption of DOAC-based dual-pathway inhibition. China (6.0%) follows with national cardiovascular programs and modernized cardiology departments. Brazil (5.1%) records strong momentum through private-sector expansion. The USA (4.2%) advances steadily with mature interventional networks, while Europe (3.8%), including Germany (3.5%), France (3.4%), and the UK (3.6%), maintains structured adoption through guideline-driven care. Asia anchors high-growth potential, while North America and Europe sustain evidence-based premium therapy uptake.
The report covers an in-depth analysis of 40+ countries top-performing countries are highlighted below.
China shows the strongest growth potential in antiplatelet-anticoagulant combination therapy with a CAGR of 6.0% through 2035, driven by expansion of nationwide cardiovascular programs, large-scale screening, and significant investment in tertiary cardiac-care infrastructure. Major hubs such as Beijing, Shanghai, Guangzhou, and Chengdu are integrating dual-pathway inhibition into standardized PCI and stroke-care algorithms. Rising access to DOAC-based regimens, wider guideline-aligned therapy adoption, and growth in outpatient cardiology networks support uptake, reinforced by Healthy China policies promoting prevention and evidence-based treatment continuity.
Key market factors:
In India’s metropolitan medical hubs and tertiary cardiac centers, adoption of antiplatelet-anticoagulant combination therapy is accelerating across interventional cardiology units, post-PCI programs, and chronic CVD management pathways. Growth is reinforced by Ayushman Bharat-driven screening expansion, rising cardiac patient volumes, and rapid PCI capacity development. With a CAGR of 6.6% through 2035, uptake strengthens as DOAC-based regimens and standardized dual-pathway inhibition protocols gain wider clinical integration.
Key market factors:
Germany’s advanced cardiovascular ecosystem shows strong adoption of antiplatelet-anticoagulant combination therapy, supported by robust evidence demonstrating reduced recurrent ischemic events in post-PCI patients using structured dual-pathway inhibition. Major centers in Berlin, Munich, Hamburg, and Frankfurt integrate these regimens into evidence-based care aligned with ESC guidelines and high procedural standards. German clinicians prioritize treatment precision, predictable pharmacology, and strong safety profiles, driving preference for premium DOAC-anchored combinations. With a CAGR of 3.5% through 2035, growth is sustained by focus on high-risk segments and consistent guideline-driven implementation.
Key development areas:
Brazil leads Latin American adoption of antiplatelet-anticoagulant combination therapy, driven by rising cardiovascular disease prevalence, increasing PCI procedure volumes, and expanding access to advanced antithrombotic options in major metropolitan regions. With a CAGR of 5.1% through 2035, momentum is supported by higher ACS hospitalization rates, broader DOAC availability in private insurance networks, and strengthened guideline-aligned dual-pathway inhibition across tertiary centers in São Paulo, Rio de Janeiro, Brasília, and Porto Alegre. Cardiologists increasingly apply combination regimens to reduce recurrent ischemic events, supported by improving diagnostics, interventional capacity, and expanded therapy distribution channels.
Leading market segments:
The United States shows mature adoption of antiplatelet-anticoagulant combination therapy, supported by advanced interventional cardiology networks, high procedural volumes, and broad integration of dual-pathway inhibition across leading cardiac centers. Major hospital systems in California, New York, Texas, Massachusetts, and Pennsylvania routinely apply evidence-based regimens for high-risk coronary and AF-PCI patients. With a CAGR of 4.2% through 2035, growth is driven by strong adherence to ACC/AHA guidelines, high diagnostic precision, and wide DOAC availability across insurance channels. USA pharmaceutical and research institutions further align innovation with optimized treatment pathways.
Key market characteristics:
In London, Manchester, Birmingham, and Edinburgh healthcare clusters, tertiary cardiac centers and interventional cardiology units are increasingly implementing antiplatelet-anticoagulant combination therapy to improve secondary-prevention outcomes in high-risk cardiovascular patients. Clinical results show reduced recurrent ischemic events and stronger long-term management of AF-PCI and chronic coronary populations through adherence to NICE-aligned dual-pathway inhibition protocols. With a CAGR of 3.6% through 2035, growth is supported by expanding AF detection efforts, structured cardiovascular strategies, and integrated care investments. Clinicians across NHS and private networks continue adopting evidence-based combination regimens to meet rising cardiovascular demands.
Market development factors:
Europe’s antiplatelet-anticoagulant combination therapy market is projected to expand from USD 3,113.9 million in 2025 to USD 5,194.4 million by 2035, registering a CAGR of 5.2% as cardiovascular networks increasingly integrate structured dual-pathway inhibition into secondary-prevention pathways. Germany remains the region’s anchor, holding 27.4% share in 2025 and moderating slightly to 27.1% by 2035, supported by advanced cardiology infrastructure, strong ESC guideline adherence, and high procedural volumes concentrated in major cardiac hubs such as Berlin, Munich, and Hamburg.
France follows with growth from 21.6% to 21.9%, driven by structured national cardiovascular strategies, broad DOAC availability, and consistent therapy integration across Paris, Lyon, and Marseille hospital systems. The United Kingdom rises from 19.1% to 19.4% through expanding AF detection initiatives and strengthened NHS integrated-care models. Italy maintains a stable 13.0% share through established coronary care units and rising PCI activity in Milan, Rome, and Turin. Spain advances from 9.4% to 9.6% amid cardiac-care modernization programs, while the Netherlands sustains 5.3% through strong guideline compliance. Rest of Europe increases from 4.2% to 4.6%, supported by broader dual-pathway uptake across Nordic and Central & Eastern European health systems.
Japan’s antiplatelet-anticoagulant combination therapy landscape is mature and highly quality-focused, marked by sophisticated integration of dual-pathway inhibition within advanced cardiovascular care workflows across major hospital systems. Strong focus on clinical precision, dosing accuracy, and evidence-based risk control drives demand for premium combination regimens that meet strict patient-safety and therapeutic performance standards.
Close alignment between cardiovascular research institutions and national guideline bodies supports treatment pathways prioritizing predictable pharmacokinetics and controlled bleeding risk. Cardiology centers in Tokyo, Osaka, and other metropolitan hubs demonstrate advanced adoption, achieving consistent ischemic-event reduction and safety outcomes aligned with Japanese Circulation Society protocols.
The South Korean antiplatelet-anticoagulant combination therapy market is expanding through increasing participation from international pharmaceutical companies offering advanced clinical support programs and therapy-optimization services for ASCVD, AF-PCI, and complex coronary patients. Precision-focused cardiovascular management is strengthening adoption as Korean cardiologists seek combination regimens aligned with domestic guidelines and integrated into structured hospital pathways across Seoul, Busan, Daegu, and Incheon.
Regional distributors gain momentum through partnerships with global manufacturers providing localized education, Korean-language clinical resources, and reimbursement guidance. Collaboration between multinational firms and national cardiovascular associations supports adoption models that blend global clinical evidence with local patient-management requirements.
The antiplatelet-anticoagulant combination therapy landscape remains concentrated, led by a select group of established companies with significant global influence. Leadership is anchored by Bayer and Johnson & Johnson, supported by a well-established dual-pathway inhibition franchise built around rivaroxaban combined with acetylsalicylic acid and reinforced by strong clinical evidence from major cardiovascular trials. Alongside them, the Bristol Myers Squibb-Pfizer alliance and AstraZeneca maintain substantial competitive strength through large cardiovascular research programs, broad geographic presence, and deep integration into acute coronary syndrome, chronic coronary disease, and atrial-fibrillation-with-PCI treatment pathways.
Challenger companies such as Sanofi, Daiichi Sankyo with Eli Lilly, and BoehringerIngelheim compete through differentiated mechanisms of action, strong peri-procedural data, and targeted physician-training initiatives, giving them a solid position in high-risk and interventional care segments. Product specialists including Novartis, Teva, Viatris, Sandoz, Dr. Reddy’s, Sun Pharma, Aurobindo, and Cipla expand therapy access through generics, cost efficiency, and broad distribution networks.
Key Players in the Antiplatelet-Anticoagulant Combination Therapy Market
| Items | Values |
|---|---|
| Quantitative Units | USD 10,964.3 million |
| Regimen Type | DOAC + Antiplatelet (Dual Pathway Inhibition), VKA (Warfarin) + Antiplatelet, Parenteral Anticoagulant + Antiplatelet (bridging/adjunct), Triple Therapy (OAC + DAPT) short-course |
| Antiplatelet Backbone | Aspirin + P2Y12 (clopidogrel / prasugrel / ticagrelor), Aspirin only + OAC, P2Y12 only + OAC, Other/Adjunct (cilostazol, cangrelor use with OAC) |
| Indication | Stable CAD/Chronic Coronary Syndrome, ACS/PCI (post-stent), Peripheral Artery Disease (post-revascularization), AF with PCI, VTE with coexisting ASCVD |
| End User/Channel | Hospitals (inpatient & cath -lab discharge), Cardiology Clinics, Retail/e-Pharmacies, Integrated Health Systems & Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Country Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40+ countries |
| Key Companies Profiled | Bayer / Johnson & Johnson (rivaroxaban + ASA use), BMS / Pfizer (apixaban combos), AstraZeneca (ticagrelor backbones), Sanofi (clopidogrel; enoxaparin bridging), Daiichi Sankyo / Eli Lilly (prasugrel), Boehringer Ingelheim (dabigatran combos), Novartis (incl. regional antiplatelet rights), Teva / Viatris / Sandoz (generics, multi ‑ region), Dr. Reddy's / Sun / Aurobindo / Cipla (generics) Others |
| Additional Attributes | Dollar sales by regimen intensity and antiplatelet backbone, alongside regional adoption across major geographies, reflect competition among cardiovascular therapy manufacturers. Evolving evidence standards, pathway integration, dual-pathway inhibition advances, and optimized antithrombotic formulations are driving safer bleeding-risk control and improved long-term vascular protection within specialized cardiac workflows. |
How big is the antiplatelet-anticoagulant combination therapy market in 2025?
The global antiplatelet-anticoagulant combination therapy market is valued at USD 10,964.3 million in 2025.
What will be the size of the antiplatelet-anticoagulant combination therapy market in 2035?
The market is projected to reach USD 18,957.6 million by 2035.
How fast will the antiplatelet-anticoagulant combination therapy market grow between 2025 and 2035?
The market will grow at a CAGR of 6.1% from 2025 to 2035.
Which regimen type leads the antiplatelet-anticoagulant combination therapy market in 2025?
DOAC plus antiplatelet therapy leads the market with a 56.7% share in 2025.
Who are the major players in the antiplatelet-anticoagulant combination therapy market?
Key players include Bayer/Johnson & Johnson, BMS/Pfizer, AstraZeneca, and Sanofi.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.