Ascites Fluid Removal Devices Market : Global Industry Analysis 2021 - 2025 and Opportunity Assessment 2026 - 2036
Ascites Fluid Removal Devices Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2021 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Ascites Fluid Removal Devices Market Forecast and Outlook 2026 to 2036
The ascites fluid removal devices market is projected at USD 744.6 million in 2026 and USD 1,592.7 million by 2036, reflecting a 7.9% CAGR. Hospitals and specialty clinics adopt these systems based on patient volume, procedure frequency, and workflow integration rather than device branding. Selection depends on pump reliability, catheter compatibility, and drainage efficiency. Once a protocol is established, hospitals maintain device continuity to limit training requirements and procedural errors. Clinicians prioritize ease of insertion, vacuum control, and safety features. Inventory planning tracks refill sets, tubing, and consumables. Maintenance and service coverage influence procurement decisions. Scheduling must align with operating room or procedure room availability.
Commercial dynamics are driven by procedural throughput and clinical adoption rather than marketing campaigns. Manufacturers focus on pump calibration, tubing integrity, and vacuum stability to ensure consistent performance. Release gates include leak tests, catheter fit checks, and sterility verification. Distribution centers stage devices and consumables near high use hospitals to meet demand without delays. Training programs reduce misuse and adverse events. Supplier scorecards consider field reliability, service response, and product consistency. Profit depends on yield, rework rates, and predictable order patterns rather than promotional discounts or frequent design changes. Equipment expansion follows patient caseload growth and hospital program scaling.
Quick Stats for Ascites Fluid Removal Devices Market
- Ascites Fluid Removal Devices Market Value (2026): USD 744.6 million
- Ascites Fluid Removal Devices Market Forecast Value (2036): USD 1,592.7 million
- Ascites Fluid Removal Devices Market Forecast CAGR (2026-2036): 7.9%
- Leading Type in Ascites Fluid Removal Devices Market: Automated paracentesis systems
- Key Growth Regions in Ascites Fluid Removal Devices Market: Asia Pacific, North America, Europe
- Top Players in Ascites Fluid Removal Devices Market: Sequana Medical, BD, Cardinal Health, AngioDynamics, Cook Medical
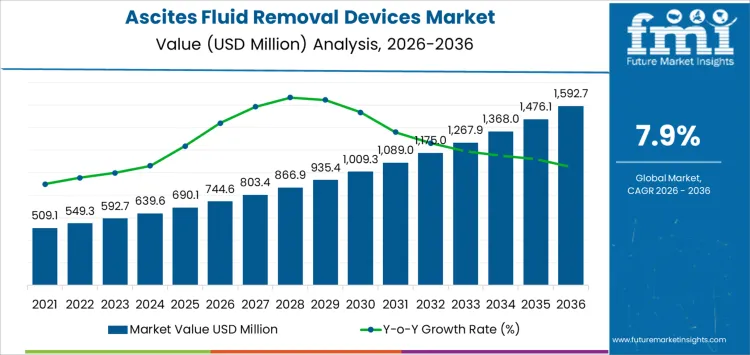
What Is the Growth Forecast for the Ascites Fluid Removal Devices Market through 2036?
Clinical protocol adoption and procedure standardization shape demand for the ascites fluid removal devices market more than general patient population growth. As hospitals expand cirrhosis management programs, demand for specialized fluid removal systems increases alongside traditional therapeutic protocols. In 2026, at about USD 744.6 million, usage is concentrated in hospitals, specialized liver care centers, and outpatient infusion clinics where paracentesis procedures follow defined care pathways. Device selection is locked during procurement and protocol approval, which ties purchasing to treatment program expansions rather than to short term case fluctuations. Utilization grows as more facilities adopt standardized devices for safety, efficiency, and patient comfort, and as repeat interventions increase among chronic care patients. The steady rise reflects broader deployment across clinical networks rather than rapid turnover of existing equipment.
Training requirements and service reliability define the later phase of the ascites fluid removal devices market. As value approaches roughly USD 1,592.7 million by 2036, buyers focus on pump reliability, tubing integrity, and ease of device assembly and cleaning. Clinical teams integrate liver disease diagnostics with procedural planning to identify patients requiring therapeutic paracentesis and ascites fluid removal Manufacturers invest in quality control, ergonomic design, and field support infrastructure as installed bases expand. The move from the mid seven hundred into the high one point eight billion reflects accumulated adoption across hospitals and outpatient centers rather than a single regulatory or guideline change. Suppliers that combine consistent device performance, predictable delivery schedules, and robust technical support tend to secure long term positions in procedural equipment lists.
Ascites Fluid Removal Devices Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 744.6 million |
| Forecast Value (2036) | USD 1,592.7 million |
| Forecast CAGR (2026-2036) | 7.9% |
How Is the Ascites Fluid Removal Devices Market Supporting Patient Care in Hepatic and Malignant Conditions?
Ascites fluid removal devices are used to drain excess peritoneal fluid in patients with liver disease, cancer, or other causes of fluid accumulation. Facilities utilizing transient elastography devices for non-invasive liver fibrosis assessment also invest in ascites fluid removal systems to manage decompensated cirrhosis. Hospitals managing hepatic encephalopathy and associated ascites complications standardize fluid removal protocols to improve patient outcomes and reduce hospitalization duration. Earlier care used large gauge needles and repeated paracentesis sessions, which increased infection risk and procedural discomfort. Modern devices include catheters with integrated safety features, controlled flow, and one-way valves to improve patient safety and ease of use. Clinicians evaluate these systems by drainage control, patency maintenance, ease of insertion, and compatibility with imaging guidance. Use follows treatment protocols where fluid accumulation is chronic or recurrent. Adoption depends on procedural workflow integration, staff training, and device availability across inpatient and outpatient settings.
Procurement is led by interventional radiology, hepatology, and nursing management because device performance impacts both safety outcomes and operational efficiency. Portal hypertension management programs coordinate ascites drainage with interventional hemodynamic interventions, creating integrated procedural protocols across hepatology centers. Earlier purchasing often relied on standard needles or catheters, which created variable patient experiences and additional monitoring requirements. Current programs emphasize standardized devices that reduce complication rates, simplify training, and ensure regulatory compliance. Manufacturers compete on material durability, flow control reliability, and disposability rather than on cost alone.
Hospitals evaluate total cost through procedure time, post-procedure monitoring, and reintervention rates, not device unit price. As liver fibrosis treatment advances reduce disease progression, recurrent ascites management through specialized fluid removal devices becomes increasingly critical for quality of life. Distributors add value through timely supply, training support, and post-market documentation. Over time, demand tracks prevalence of liver disease, cancer care volume, and outpatient procedure growth, where safe, predictable, and efficient fluid removal is a core component of symptom management and clinical workflow planning.
What Factors Are Shaping the Demand for Ascites Fluid Removal Devices Market in 2026 by Product Type and Application?
Demand in Ascites Fluid Removal Devices Market is primarily influenced by procedural throughput, patient risk management, and regulatory compliance rather than short-term equipment purchases. Hospitals and specialty clinics plan device acquisition based on projected caseload, therapy standardization, and staff availability. Device selection integrates with training programs, sterile workflow requirements, and documentation protocols, which affects adoption speed. Segment structure reflects differences in automation level, disposability, and monitoring capabilities. Buyers evaluate devices based on operational reliability, infection control compliance, and ease of integration into existing clinical workflows. Suppliers that demonstrate consistent performance, streamlined maintenance, and regulatory alignment gain preferential consideration. Growth follows treatment program expansion and standardization initiatives rather than spontaneous demand fluctuations.
How Does Product Type Selection Impact Operational Efficiency and Supplier Requirements in the Ascites Fluid Removal Devices Market?
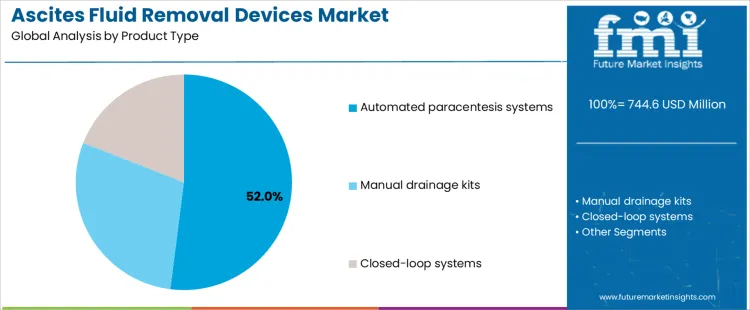
Automated paracentesis systems account for approximately 52% of demand due to their ability to standardize aspiration rates and reduce procedural variability. These systems improve throughput in high-volume liver clinics and require suppliers to provide training, calibration support, and preventive maintenance services. Manual drainage kits are used in smaller or budget-constrained settings, where supplier reliability and ease of distribution influence adoption. Closed-loop systems address advanced treatment protocols that integrate reinfusion or continuous monitoring, creating higher technical support and validation needs. Each product type affects supplier involvement, inventory planning, and after-sales service. Companies offering comprehensive training, predictable maintenance schedules, and modular device options can secure broader adoption across hospital tiers.
Operational efficiency is further affected by device choice. Automated systems reduce labor hours and procedural errors but require integration into monitoring infrastructure and regular quality checks. Manual kits offer lower upfront cost but increase the burden on clinical staff for workflow compliance and training. Ascites fluid removal systems bridge the gap between diagnosis and liver transplantation, managing symptomatic fluid accumulation during the waitlist period.
Closed-loop devices require coordinated calibration, software validation, and consumable supply management. For suppliers, this translates to differentiated service models, tiered support agreements, and predictable consumables demand. Device type selection therefore drives both procurement strategies and supplier engagement models, highlighting the importance of reliability, technical support, and documentation in sales and deployment planning.
Why Does Application Mix Shape Market Opportunity and Revenue Concentration in the Ascites Fluid Removal Devices Market?
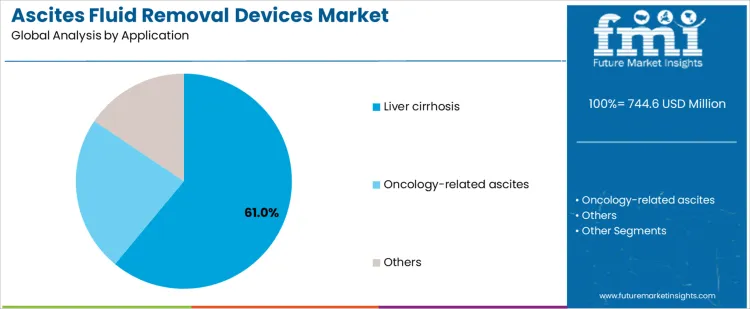
Liver cirrhosis treatment represents approximately 61% of demand due to the frequency and regularity of fluid removal procedures, making this application a high-volume, high-reliability target for manufacturers. Oncology networks implementing standardized malignant ascites management protocols drive adoption of specialized fluid removal devices across cancer treatment centers. Other applications, including secondary ascites causes, create smaller, targeted orders but require flexibility in device configuration. Segment focus enables suppliers to allocate production and service resources efficiently. Companies that align product lines to high-volume applications while providing specialized support for lower-volume segments can maximize revenue and market penetration.
End-use considerations influence purchasing patterns and supplier strategies. High-volume liver programs favor bulk procurement, service contracts, and training bundles to ensure consistent procedural outcomes. Oncology and niche applications prioritize device adaptability, rapid technical support, and documentation support to satisfy clinical and regulatory standards. Suppliers that provide differentiated service offerings and customizable device configurations gain access to multiple revenue streams. Segment shares therefore reflect both procedural volume and clinical complexity, emphasizing how product design, support infrastructure, and regulatory alignment determine commercial opportunity for manufacturers and distributors.
How Is the Ascites Fluid Removal Devices Market Being Driven by Operational Efficiency and Clinical Reliability?
The Ascites Fluid Removal Devices Market is increasingly influenced by hospital priorities for fast, safe, and repeatable fluid drainage procedures. Devices are evaluated not only for sterility but also for ease of setup, handling ergonomics, and compatibility with standard bedside systems. Buyers, including procurement teams, nursing supervisors, and physicians, weigh uptime, disposability, and infection prevention features. For suppliers, this emphasizes the need to offer products that reduce procedural errors, simplify training, and integrate with hospital workflows. Manufacturers that provide reliable devices backed by clear usage protocols can gain preference as long-term vendors.
How Are Device Consistency and Standard Operating Procedures Shaping Supplier Expectations in The Ascites Fluid Removal Devices Market?
Hospitals and specialty clinics are standardizing paracentesis procedures to maintain uniform patient outcomes and operational efficiency. This drives demand for devices with predictable flow rates, secure connectors, and minimal handling variability. Suppliers are expected to deliver units with consistent quality, clear documentation, and durable packaging to ensure that devices perform reliably across different wards and operators. Companies that can supply pre-configured kits with integrated safety and monitoring components can differentiate themselves. Long-term contracts increasingly favor vendors that can support multi-site standardization with predictable performance.
Where Is Recurring Revenue Opportunity Being Created in The Ascites Fluid Removal Devices Market?
Recurring demand arises from facilities treating patients with chronic or repeated ascites episodes. Each procedure consumes single-use catheters, tubing, and drainage accessories, generating predictable repeat orders. There is also opportunity in providing bundled solutions that combine disposables with training, maintenance support, or inventory management services. Suppliers that align their offering with hospital supply schedules, manage lot traceability, and provide rapid replacement for high-turnover products can secure steady revenue streams. Facilities adopting standardized device kits across multiple units or departments create long-term demand stability for committed suppliers.
Why Do Adoption Barriers and Clinical Risk Management Limit Rapid Expansion in The Ascites Fluid Removal Devices Market?
Even when new device designs offer operational advantages, clinical teams may prefer familiar devices to avoid procedural errors and potential complications. Hospitals also prioritize investments in equipment that has broad utility across departments, which can limit purchase of specialized drainage devices. Supplier switching is further constrained by procurement approval cycles, device validation requirements, and budget allocation processes. Companies entering the market must therefore provide robust clinical support, training materials, and documented performance to reduce perceived risk and gain trust. Adoption is strongest in high-volume or specialty centers where efficiency and safety impact patient care directly.
What is the Demand for Ascites Fluid Removal Devices by Country?
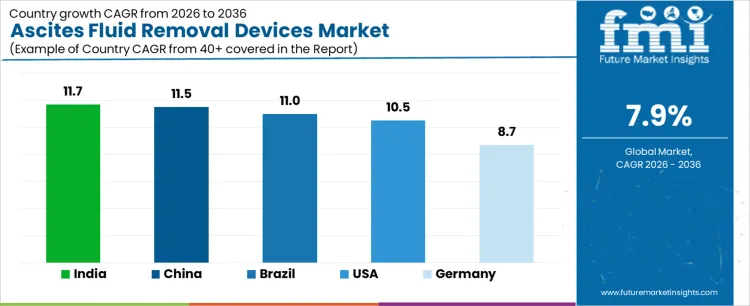
| Country | CAGR |
|---|---|
| USA | 10.5% |
| Brazil | 11.0% |
| China | 11.5% |
| India | 11.7% |
| Germany | 8.7% |
Demand for ascites fluid removal devices is increasing as hospitals and specialty clinics expand treatment options for patients with liver disease, cancer, and congestive conditions requiring therapeutic paracentesis. India leads with an 11.7% CAGR, supported by rising incidence of chronic liver disease, increasing hospital infrastructure, and adoption of minimally invasive fluid management systems. China follows at 11.5%, driven by large patient volumes and growing awareness of device assisted therapy. Brazil records 11.0%, reflecting broader access in private and public healthcare facilities. The USA grows at 10.5%, shaped by outpatient and hospital based procedures. Germany, at 8.7%, reflects stable demand in a mature healthcare system with regulated adoption of specialized devices.
How Are Hospital Procedure Volumes Driving Demand in the Ascites Fluid Removal Devices Market in the United States?
Growing hospital procedure volumes set planning benchmarks around a 10.5% CAGR for ascites fluid removal devices in United States. Capital committees and interventional hepatology teams control most procurement decisions. Adoption focuses on paracentesis, outpatient fluid management, and critical care integration. Device choice emphasizes ease of use, safety features, and rapid drainage capability. Once validated, systems are deployed across multiple hospital sites and specialty clinics. Training programs and physician familiarity influence utilization. Import systems dominate higher specification applications while domestic suppliers serve routine hospital needs. Budget planning aligns with procedural growth and service line expansion. Replacement cycles track usage. Commercial success depends on framework agreements and inclusion in hospital equipment catalogs rather than individual department purchases.
Why Are Private Healthcare Networks Influencing Demand in the Ascites Fluid Removal Devices Market in Brazil?
Expansion of private hospital chains and specialty hepatology units assumes a 11% CAGR for ascites fluid removal devices in Brazil. Procurement authority sits with hospital network committees and procedural leads. Device selection emphasizes safety, workflow integration, and patient throughput. Import systems dominate advanced paracentesis and automated drainage platforms. Local distributors manage installation, training, and field support. Once approved, rollout spans multiple affiliated centers. Adoption follows outpatient and inpatient procedure growth. Replacement cycles depend on device wear and catheter lifespan. Spending aligns with hospital expansion and new treatment line openings. Commercial visibility depends on contracts with large networks and reference site inclusion rather than dispersed sales to small clinics or standalone practices.
What Institutional Programs Drive Scale in the Ascites Fluid Removal Devices Market in China?
National hospital network expansion and chronic liver care programs support a 11.5% CAGR for ascites fluid removal devices in China. Major tertiary hospitals and specialized liver centers control procurement. Adoption targets paracentesis, fluid management in cirrhosis, and oncology support. Domestic manufacturers supply standard devices while international suppliers retain positions in higher specification systems. Tender cycles and multi-year planning govern timing. Clinical training, safety features, and procedure integration influence selection. Once approved, deployment replicates across multiple hospital sites. Replacement demand follows procedure volume rather than machine aging alone. Capital allocation aligns with hospital construction and service expansion schedules. Commercial success depends on early access to large hospital networks and framework agreements rather than sales to independent clinics or small private facilities.
Why Are Hepatology Service Expansions Driving Growth in the Ascites Fluid Removal Devices Market in India?
Expansion of hepatology programs and outpatient liver clinics drives a 11.7% CAGR for ascites fluid removal devices in India. Large private hospital chains and specialty centers lead procurement. Device choice emphasizes safety, procedure efficiency, and disposable consumable compatibility. Import devices dominate high acuity applications while domestic suppliers cover standard clinical setups. Procurement follows central committee approvals. Once validated, devices are deployed across multiple affiliated facilities. Training programs and procedural standardization affect adoption. Replacement cycles track catheter lifespan and usage. Budget release aligns with hospital openings and service expansion. Commercial success depends on inclusion in hospital network protocols and reference site adoption rather than sales to individual clinics or ad hoc departments. Regulatory clearance influences early adoption timelines.
How Are Guidelines and Reimbursement Structures Shaping Demand in the Ascites Fluid Removal Devices Market in Germany?
Clinical guidelines and reimbursement frameworks assume a 8.7% CAGR for ascites fluid removal devices in Germany. University hospitals, hepatology centers, and outpatient clinics control most procurement. Evaluation prioritizes safety features, drainage accuracy, and workflow integration. Procurement follows structured health technology assessment processes. Import suppliers dominate premium platforms while local distributors provide standard systems. Once approved, devices deploy across multiple sites. Training and certification affect utilization. Spending follows procedure volume growth and service expansion schedules. Replacement cycles are based on catheter wear. Commercial success depends on inclusion in guideline driven procurement frameworks and reference adoption across hospital networks rather than isolated departmental purchases. Audit and compliance requirements influence approval timing.
How Do Manufacturers Compete for Clinical Adoption in the Ascites Fluid Removal Devices Market?
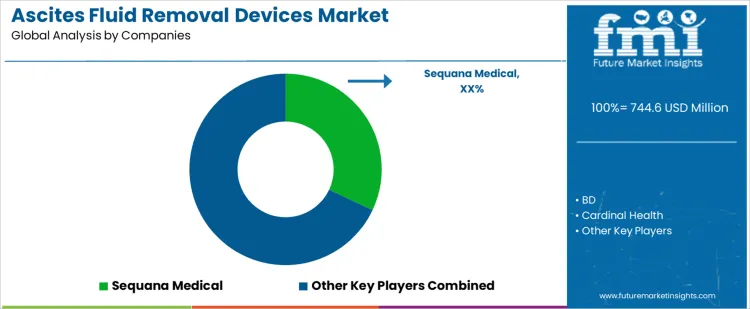
Ascites fluid removal device selection occurs within procedure workflow planning and hospital protocol design rather than at purchasing alone. Physicians define catheter type, drainage volume, dwell time, and bedside handling requirements before naming devices. Sequana Medical, BD, Cardinal Health, AngioDynamics, Cook Medical compete during those evaluation stages with different product philosophies. Once a device enters protocol documents, training programs, documentation, and service logistics fix the choice. Sequana Medical positions its automated and low flow systems for chronic fluid management programs. BD supplies single use catheters and drainage kits integrated with hospital inventory systems. Cardinal Health supports multi-site standardization and procedural consistency. AngioDynamics focuses on minimally invasive access and patient comfort. Cook Medical offers broad product ranges across fluid removal and intervention tools.
Competitive outcomes reflect reliability, procedure safety, and support coverage rather than unit price alone. Clinicians evaluate ease of insertion, flow control, risk of occlusion, and material biocompatibility during selection. Nursing teams assess assembly simplicity, device labeling, and waste disposal. Hospitals favor suppliers with consistent supply, staff training support, and clear regulatory documentation. Sequana Medical benefits from clinical studies and program integration in chronic management. BD secures positions through device availability and hospital relationships. Cardinal Health gains share via multi-site logistics and bundled procurement. AngioDynamics competes where procedural ergonomics and access design drive adoption. Cook Medical retains accounts with flexible product portfolios. Market positions vary by hospital policy, patient volume, and treatment protocol adoption across regions.
Key Players in the Ascites Fluid Removal Devices Market
- Sequana Medical
- BD
- Cardinal Health
- AngioDynamics
- Cook Medical
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Million |
| Product Type | Automated paracentesis systems; Manual drainage kits; Closed-loop systems |
| Application | Liver cirrhosis; Oncology-related ascites; Others |
| End User | Hospitals; Home care; Clinics |
| Region | Asia Pacific; Europe; North America; Latin America; Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, Rest of regions |
| Key Companies Profiled | Sequana Medical; BD; Cardinal Health; AngioDynamics; Cook Medical |
| Additional Attributes | Dollar by sales by product and application, procedure volume driven demand, disposable versus system mix, consumables attachment rate, training and protocol standardization impact, service and maintenance intensity, multi-site hospital adoption patterns, inventory turnover and refill cycle behavior, regulatory and documentation requirements |
Ascites Fluid Removal Devices Market Segmentation
Product Type:
- Automated paracentesis systems
- Manual drainage kits
- Closed-loop systems
Application:
- Liver cirrhosis
- Oncology-related ascites
- Others
End User:
- Hospitals
- Home care
- Clinics
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- Aponte, E. M. (2023). Paracentesis. StatPearls. National Library of Medicine.
- Harvey, J. J. (2023). Diagnostic and therapeutic abdominal paracentesis: Overview and safety considerations. Medical Journal of Australia, 219(3), 120–125.
- Bartal, C., Ginès, P., Wong, F., et al. (2025). Proposed clinical practice guidance for large-volume paracentesis in ascites management. Journal of Clinical Medicine, 14(2), 412.
- Lan, T., Xu, L., Yu, Y., & Chen, Y. (2024). Recent developments in the management of ascites: Pathogenesis and treatment. United European Gastroenterology Journal, 12(1), 3–15.
Frequently Asked Questions
How big is the ascites fluid removal devices market in 2026?
The global ascites fluid removal devices market is estimated to be valued at USD 744.6 million in 2026.
What will be the size of ascites fluid removal devices market in 2036?
The market size for the ascites fluid removal devices market is projected to reach USD 1,592.7 million by 2036.
How much will be the ascites fluid removal devices market growth between 2026 and 2036?
The ascites fluid removal devices market is expected to grow at a 7.9% CAGR between 2026 and 2036.
What are the key product types in the ascites fluid removal devices market?
The key product types in ascites fluid removal devices market are automated paracentesis systems, manual drainage kits and closed-loop systems.
Which application segment to contribute significant share in the ascites fluid removal devices market in 2026?
In terms of application, liver cirrhosis segment to command 61.0% share in the ascites fluid removal devices market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Automated paracentesis systems
- Manual drainage kits
- Closed-loop systems
- Automated paracentesis systems
- Y-o-Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Liver cirrhosis
- Oncology-related ascites
- Others
- Liver cirrhosis
- Y-o-Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Home care
- Clinics
- Hospitals
- Y-o-Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Sequana Medical
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- BD
- Cardinal Health
- AngioDynamics
- Cook Medical
- Others
- Sequana Medical
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

