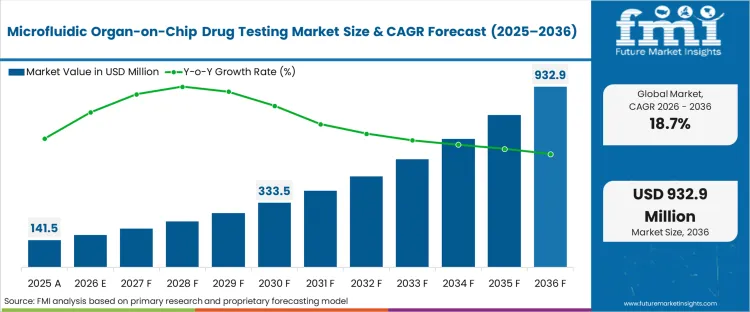
The microfluidic organ-on-chip drug testing market was valued at USD 141.5 million in 2025. Sales are set to reach USD 168.0 million in 2026 at a CAGR of 18.70% during the forecast period. Ongoing investment supports the growth in valuation to USD 932.9 million through 2036 as pharmaceutical organ-on-chip market participants replace high-attrition static cellular assays with dynamic, human-relevant predictive models to salvage late-stage clinical pipelines.
The transition facing biopharmaceutical development teams centers on the deprecation of mandatory animal IND data in favor of qualified in vitro human models. Buyers actively evaluating preclinical toxicology alternatives to animal testing are no longer asking if microfluidic flow improves cell viability; they are deciding which assays can be fully migrated to chip formats to satisfy updated regulatory submission pathways. Delaying this qualification cycle forces developers to rely on animal data that regulators are increasingly scrutinizing for human translational relevance, directly threatening timeline predictability. The broader microphysiological systems sector provides a clear alternative, establishing NAMs in drug development as a baseline requirement for novel biologics.
Before adoption scales from niche investigative toxicology to routine screening, platforms must achieve compatibility with high-throughput organ-on-chip screening infrastructure. The way is the integration of these dynamic flow models into standard 96-well or 384-well automated handling environments. Once hardware form-factors match existing robotic liquid handlers, assay directors can trigger volume transitions without retrofitting entire laboratory ecosystems, accelerating the replacement cycle of conventional lab on chips.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 168.0 million |
| Industry Value (2036) | USD 932.9 million |
| CAGR (2026-2036) | 18.70% |
China organ-on-chip drug testing market leads global expansion, tracking at 20.7% compound, as domestic programs bypass legacy animal testing infrastructure directly for advanced human-model capacity. The United States follows at 19.8%, driven by dedicated regulatory qualification initiatives. The South Korean market is projected to advance at 19.1%, while the Germany organ-on-chip drug testing market expands at 18.9% and the UK registers 18.4%. The Netherlands is likely to grow at 18.2%, and the Japan posts a measured 17.6% trajectory. This geographic divergence highlights a separation between regions building new preclinical capacity from the ground up versus markets managing careful transitional qualification against decades of historical in vivo data.
The market encompasses bioengineered microfluidic devices that culture living human cells within continuously perfused micrometer-sized chambers to replicate the physiological and mechanical functions of intact human organs. Functionally, this defines the boundary between static 3D cellular clusters and dynamic, flow-based physiological models utilized specifically for predicting human responses to pharmacological compounds prior to clinical trials.
Scope includes polymer or glass-based microfluidic chips containing single or multiple organ emulations, the associated pneumatic or mechanical pumping hardware required to maintain flow, and the proprietary software utilized to translate on-chip sensor data into pharmacological endpoints. The market also accounts for specialized consumable kits, validated primary cell lines designed specifically for dynamic perfusion, and contract testing services executed on these platforms. Hardware elements like the high precision microfluidic pump directly tethered to the chip operation are included within system sales.
The market explicitly excludes traditional static 2D cell culture plates, non-perfused transwell inserts, and standalone static organoids that lack a continuous microfluidic flow component. These static methodologies are excluded because they do not simulate the biomechanical forces, such as shear stress or cyclic strain, that define the predictive value of dynamic organ-chip platforms. General-purpose laboratory equipment not engineered specifically for organ-chip operation is also excluded.
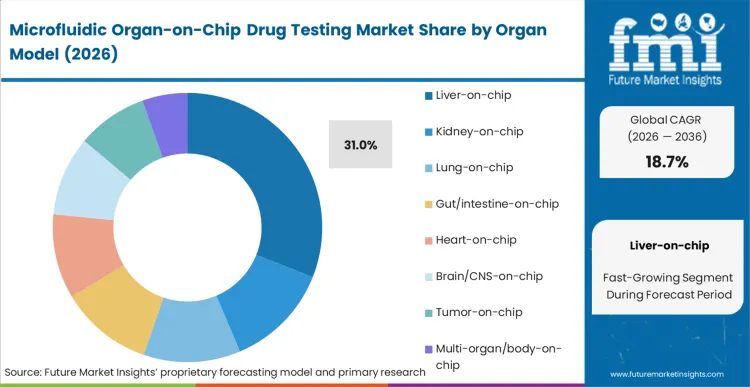
Liver-on-chip holds 31.0% of the market, centred entirely on the financial geometry of drug development within the liver-on-chip drug toxicity testing market: hepatotoxicity is the single most common cause of post-market withdrawal and late-stage clinical failure. According to FMI's estimates, pharmaceutical organisations deploy microfluidics predominantly for liver models because conventional animal and static human models consistently fail to predict human drug-induced liver injury accurately. Tracking drug-induced liver injury organ-on-chip signatures is critical; the dynamic perfusion inside these chips maintains primary hepatocyte functionality and metabolic competence over extended periods, directly resolving the rapid dedifferentiation seen in 2D plates. Kidney-on-chip nephrotoxicity screening market and the lung-on-chip drug testing market also benefit from this continuous flow methodology to isolate organ-specific failure points early. Failure to integrate this specific predictive capability leaves clinical pipelines exposed to catastrophic late-stage failure costs that offset any savings from cheaper legacy screening methods.
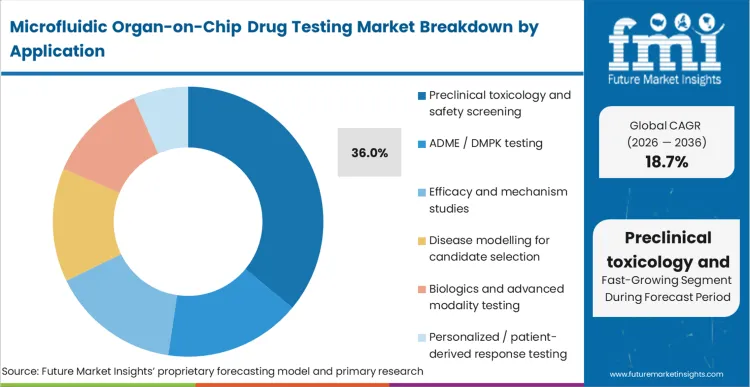
Preclinical toxicology and safety screening, which captures 36.0% share, is garnering the demand for higher throughput and the biological complexity required for true human prediction. Buyers operating within the organ-on-chip toxicology testing market must balance the upfront capital expenditure of establishing toxicology drug screening platforms against the downstream cost of advancing a flawed compound. Additionally, creating a reliable ADME organ-on-chip platform requires rigorous endpoint verification. As per FMI's projection, the immediate cost savings originate not from replacing animal models one-for-one, but from disqualifying toxic hits before they consume extensive in vivo resources. Once integrated, hidden costs emerge in data management and operator training, requiring dedicated personnel who specialize in gut-on-chip drug absorption testing and complex barrier models. However, the total lifecycle comparison heavily favors organ-chips, as identifying a single toxicity failure pre-clinically offsets years of system hardware and consumable investments.
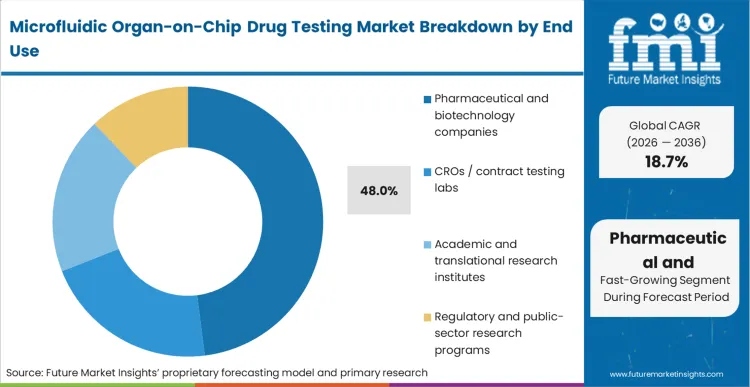
Pharmaceutical and biotechnology companies, represents 48.0% of the market. The key consideration has shifted from evaluating microfluidic models to determining whether to internalize the infrastructure or outsource testing. Tier-1 companies act as the first-movers in the organ-on-chip preclinical testing market, absorbing the heavy internal qualification burden to build proprietary, predictive drug discovery informatics libraries. FMI predicts that these organizations adopt biotech organ-chip testing platforms to gain a decisive speed advantage in candidate selection. Mid-cap biotech firms follow, often utilizing the validated protocols established by the tier-1 leaders. Translational medicine organ-chip platforms at academic institutes, while critical for initial biological validation, adopt commercial systems later in the cycle to standardize outputs for licensing discussions. Consequently, toxicology labs organ-on-chip adoption and CRO adoption of organ-on-chip workflows remain robust, accelerating the transition from bespoke lab-built chips to commercial platforms.
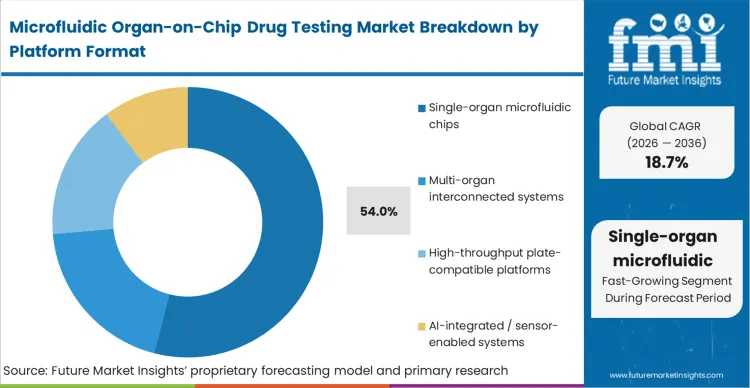
Single-organ microfluidic chips displace complex multi-organ concepts to hold 54.0% share, because multi-organ systems fail to meet the immediate throughput requirements of early-stage screening. Assay directors select single-organ formats because they map directly onto existing organ-specific toxicity protocols, requiring less complex high throughput screening validation. Based on FMI's assessment, the initial purchase decision is validated during internal qualification when the single-organ data successfully aligns with historical monotypic baselines. The simplicity of controlling single-tissue variables drives renewal and protocol expansion. Conversely, attempting to leapfrog directly into the multi-organ chip drug development market introduces unmanageable biological variables, delaying operational qualification and forcing researchers to revert to static models to meet strict pipeline deadlines.
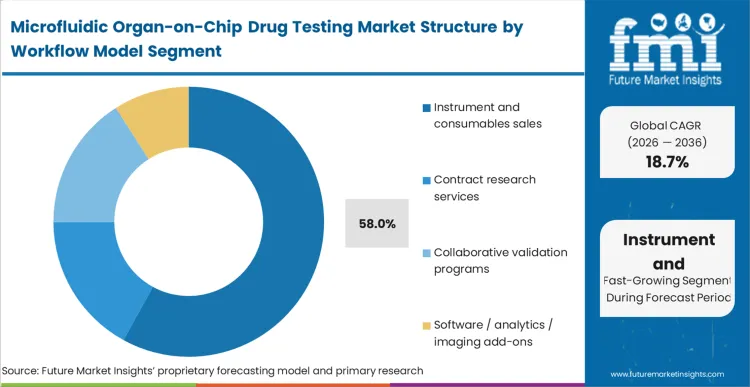
Instrument and consumables sales are heading toward a dominant 58.0% position as the market transitions from an outsourced service model to decentralised internal capacity. Initially, complex lab chip devices required the specialized expertise of the platform developers, limiting pharma engagement to organ-on-chip contract testing services. In FMI's view, the stabilization of polymer manufacturing and standardized consumable kits allows vendors to move their revenue model toward recurring hardware installations. Supply constraints center heavily on the sourcing of primary human cells qualified for dynamic flow, forcing teams who buy organ-on-chip system for drug screening to plan procurement cycles carefully. Moving toward 2036, the supply landscape will centralize around vendors who can bundle the hardware, the predictive software, and the validated biological consumables into a single closed ecosystem, pushing organ-on-chip CRO services to handle highly specialized overflow.
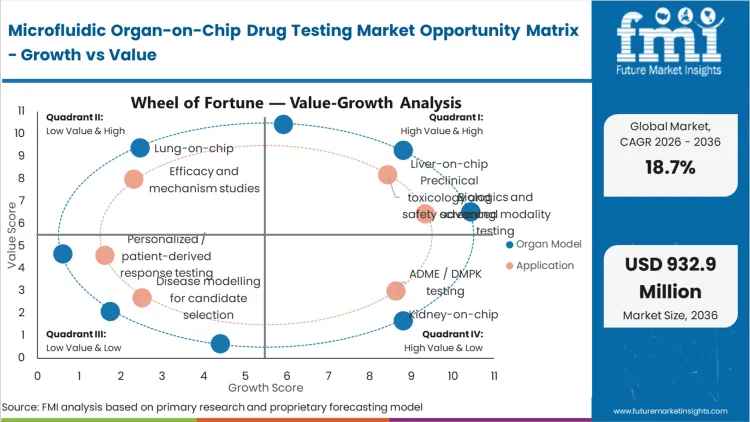
The regulatory use of organ-on-chip in drug development compelling pharmaceutical toxicology leads to qualify human-relevant microfluidic models serves as the primary driver. This forces organizations to rapidly validate alternative methodologies to maintain pipeline velocity, as relying solely on traditional in vivo models for complex modalities increasingly yields non-predictive clinical outcomes. Evaluating the organ-on-chip ROI compared with conventional assays reveals that identifying a failure early completely justifies the integration costs. Evaluating organ-on-chip vs animal testing, organizations find that failure to integrate these dynamic platforms leaves developers exposed to protracted regulatory scrutiny and competitive disadvantages in securing trial approvals.
The single biggest buyer friction is the high-throughput organ-chip platform price compounded by the rigorous requirement to standardize microfluidic biological endpoints against decades of legacy historical in vivo data. This qualification gap slows adoption because toxicologists cannot simply accept novel in vitro readouts without a direct translation to established risk-assessment algorithms utilizing traditional toxicology screening systems. While bioinformatics platforms are emerging to bridge these datasets, their utility remains limited by the proprietary nature of pharma data, meaning each organization must laboriously validate the system internally before trusting it for go/no-go pipeline decisions.
.webp)
Regional segmentation places the Microfluidic Organ-on-Chip Drug Testing market across North America, Europe, and Asia Pacific, covering over 40 countries.
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 20.70% |
| United States | 19.80% |
| South Korea | 19.10% |
| Germany | 18.90% |
| United Kingdom | 18.40% |
| Netherlands | 18.20% |
| Japan | 17.60% |
.webp)
The regulatory environment in North America, explicitly driven by the FDA's Modernization Act 2.0 and NIH/NCATS funding, shapes buyer behaviour by providing clear qualification pathways for new approach methodologies. This policy-led framework reduces the perceived risk for pharmaceutical organizations looking to replace traditional preclinical medical device testing services and drug screening protocols with microfluidic alternatives. According to FMI's estimates, the focus is not merely on acquiring hardware, but on participating in public-private consortiums that establish accepted standards of evidence.
FMI's report includes extensive analysis of the Canadian landscape. Organizations across North American hubs demonstrate a unified approach to data standard harmonization, actively pooling pre-competitive validation results to accelerate broad regulatory acceptance.
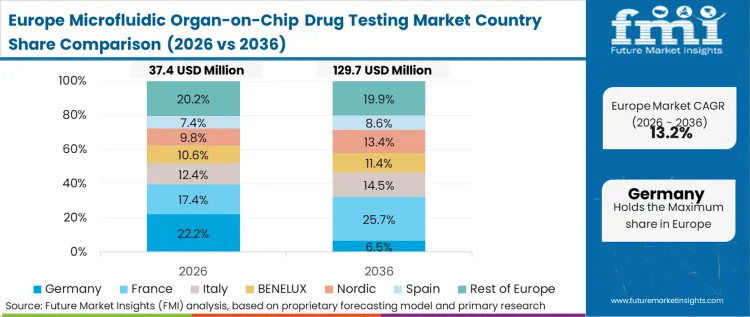
Cost structures and capital availability define the European approach, where rigorous scientific capacity is balanced against stringent, often slower, internal pharmaceutical validation cycles. Economics-led decisions force procurement directors to demand robust return-on-investment models before scaling 3d cell culture and microfluidic infrastructure. FMI predicts that European buyers heavily scrutinize the total cost of ownership, including the hidden costs of specialized operator training and complex bio-consumables sourcing, resulting in a more measured but deeply entrenched adoption curve.
FMI's report includes coverage of France, Italy, and the broader EU consortiums. The pattern across these nations is the reliance on centralized, EU-funded standardization roadmaps to underwrite the financial risk of platform acquisition for mid-sized players.
The physical and digital infrastructure scale-up across Asia Pacific allows the region to bypass the limitations of legacy screening environments. This infrastructure-led expansion means buyers are not retrofitting old laboratories; they are building advanced biochips and microfluidic capacities directly into new, massive-scale biotech parks. As per FMI's projection, this clean-slate approach enables a faster transition to high-throughput, plate-compatible platforms, skipping the early single-chip manual iteration phases that characterized Western adoption.
FMI's report includes analysis of India and Australia. These markets demonstrate a distinct pattern of adopting proven commercial single-organ platforms for targeted academic research before attempting to scale into full industrial pharmaceutical deployment.
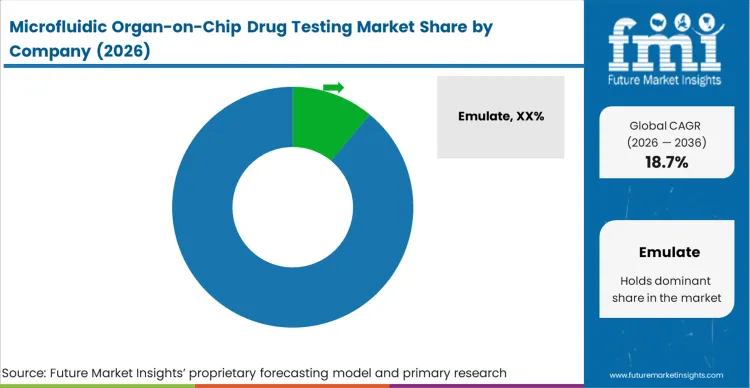
The competitive structure of this market is heavily consolidated around a few dominant platform ecosystems because the capital required to validate biological predictability creates an enormous barrier to entry. Evaluating which companies lead microfluidic organ-on-chip drug testing, Emulate, MIMETAS, and CN Bio lead because buyers utilize the volume of published peer-reviewed validation data as the primary variable to distinguish qualified organ-on-chip suppliers for pharma from experimental alternatives. Procurement directors searching for the best organ-on-chip platform for toxicology do not simply buy polymer chips; they purchase the established confidence that regulatory bodies have already accepted data generated by these specific platforms. Emulate maintains its 15.8% share leadership by marketing a dedicated ecosystem that bridges hardware, automated cell biology systems, and proprietary qualification software.
Incumbents possess deep libraries of historically qualified physiological endpoints that challengers and other organ-on-chip platform vendors must replicate. TissUse and AlveoliX maintain their advantage because their platforms are embedded deeply within multi-year pharmaceutical development programs, making the switching costs for assay directors prohibitively high. An evaluator attempting to compare Emulate vs MIMETAS vs CN Bio for organ-chip workflows immediately recognises that challengers must build a comprehensive library of human-translational validation data to compete, not merely a functionally superior pump. The integration of high-throughput compatibility, such as Emulate’s 96-emulation AVA workstation, demonstrates how incumbents secure their base against organ-chip platform alternatives by aligning their complex biology with the standardised robotic workflows pharma already utilises.
Large pharmaceutical buyers actively resist ecosystem lock-in by engaging multiple vendors for different organ models, preventing any single provider from dictating enterprise-wide preclinical architecture. The tension moving toward 2036 involves pharma’s preference for open, interoperable cell line development services and standard plate formats against the dominant vendors' incentive to maintain closed, proprietary hardware-software loops. As regulatory standards solidify, the market will likely consolidate further around platforms that seamlessly export standardized biological metadata, while fragmented component suppliers who cannot offer full predictive workflows will be relegated to the academic research tier.
| Metric | Value |
|---|---|
| Quantitative Units | USD 168.0 million to USD 932.9 million, at a CAGR of 18.70% |
| Market Definition | The market encompasses bioengineered microfluidic devices that culture living human cells within continuously perfused micrometer-sized chambers to replicate the physiological and mechanical functions of intact human organs. |
| Organ Model Segmentation | Liver-on-chip, Kidney-on-chip, Lung-on-chip, Gut/intestine-on-chip, Heart-on-chip, Brain/CNS-on-chip, Tumor-on-chip, Multi-organ/body-on-chip |
| Application Segmentation | Preclinical toxicology and safety screening, ADME / DMPK testing, Efficacy and mechanism studies, Disease modelling for candidate selection, Biologics and advanced modality testing, Personalized / patient-derived response testing |
| End User Segmentation | Pharmaceutical and biotechnology companies, CROs / contract testing labs, Academic and translational research institutes, Regulatory and public-sector research programs |
| Platform Format Segmentation | Single-organ microfluidic chips, Multi-organ interconnected systems, High-throughput plate-compatible platforms, AI-integrated / sensor-enabled systems |
| Regions Covered | North America, Europe, Asia Pacific |
| Countries Covered | United States, China, Germany, United Kingdom, Netherlands, Japan, South Korea, and 40 plus countries |
| Key Companies Profiled | Emulate, MIMETAS, CN Bio, TissUse, AlveoliX, InSphero, Quris-AI |
| Forecast Period | 2026 to 2036 |
| Approach | Primary research engaged preclinical assay directors, toxicologists, and translational science leads at top-tier pharmaceutical organizations. Desk research aggregated regulatory guidance on new approach methodologies and public-private validation consortium data. Forecasts were cross-validated against vendor revenue disclosures and specific contract research capacity metrics in independent testing laboratories. |
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
What is the forecast for organ-on-chip drug testing through 2036?
The valuation is projected to reach USD 932.9 million. This expansion signals the transformation from niche investigative pilot studies into mandatory, scaled deployment for high-risk biologics screening across all major pharmaceutical pipelines.
What is the organ chip drug testing market size in 2026?
The market is valued at USD 168.0 million. This figure reflects the baseline capital already committed by tier-1 pharmaceutical companies toward early hardware integration and initial biological validation protocols.
What CAGR is projected?
The market tracks at an 18.70% CAGR. This aggressive rate is maintained by the continuous replacement cycle of traditional static in vitro assays, accelerated by regulatory updates that actively accept non-animal predictive data.
Which Organ Model segment leads?
Liver-on-chip commands a 31.0% share. Assay directors prioritize hepatic models because drug-induced liver injury remains the primary financial driver of late-stage clinical trial failures, making early detection critical.
Which Application segment leads?
Preclinical toxicology and safety screening dominates with 36.0% share. Buyers utilize chips here first because eliminating a single toxic compound before it enters costly in vivo testing immediately justifies the platform's capital expenditure.
Which End User segment leads?
Pharmaceutical and biotechnology companies hold 48.0% of the market. These organizations possess the capital depth required to internalize the complex infrastructure and build proprietary, predictive data libraries for their own candidate selection.
How does organ-on-chip improve drug testing?
Continuous microfluidic perfusion simulates actual human biomechanical forces like shear stress and cyclic strain that static plates cannot replicate. This dynamic environment maintains cell viability and metabolic competence over weeks, allowing toxicologists to detect chronic, long-term human safety signals early in the pipeline.
What are the challenges of organ-on-chip adoption?
The rigorous requirement to standardize microfluidic biological endpoints against decades of legacy historical data poses the heaviest friction. Toxicologists cannot fully adopt the platform until they internally validate the new 3D microfluidic readouts against accepted in vivo baseline risk calculations.
Which country grows fastest?
China advances at a leading 20.70% compound rate. Unlike Western markets managing careful transitional qualification against legacy protocols, Chinese facilities are building advanced microfluidic capacities directly into new, massive-scale biotech infrastructure.
Is organ-on-chip replacing animal testing?
Yes, but progressively rather than instantly. Regulatory shifts like the FDA Modernization Act 2.0 remove the mandatory animal requirement for specific IND submissions, transitioning microfluidic models from supplementary tools to qualified primary data sources for novel biologics where animal models fail to predict human immune responses.
Why is organ-on-chip used in preclinical research?
High late-stage clinical attrition rates drive the transition. Pharmaceutical development teams use these platforms because relying solely on traditional in vivo data yields non-predictive outcomes for complex new modalities, costing organizations billions in late-stage failures.
Which organ-on-chip models are used for toxicology?
Liver-on-chip and Kidney-on-chip formats are prioritized because hepatic and renal toxicities are the primary drivers of late-stage clinical trial halts. Procurement directors allocate capital toward these specific models first to secure the highest immediate return on risk avoidance.
Organoids vs organ-on-chip drug testing: how do they compare?
While both utilize 3D cell structures, organoids are static models that lack functional vascularization and fluid dynamics. Adding the microfluidic flow of organ-chips provides the biomechanical stress necessary to sustain mature tissue functions, bridging the gap between a cluster of cells and a functional physiological system.
Organ-on-chip vs 3D cell culture: what is the difference?
Traditional 3D cell cultures sit in static wells, accumulating their own waste and exhausting nutrients rapidly. Organ-chips incorporate continuous microfluidic perfusion, mimicking blood flow to provide fresh nutrients and clear waste, enabling the long-term, repeat-dose toxicity studies impossible in static formats.
Organ-on-chip vs animal testing: what drives the transition?
Species-specific biological differences mean animals often process complex human biologics, such as bispecific antibodies, differently than patients do. Organ-chips utilizing primary human cells provide a human-relevant alternative that bypasses these species translation errors.
What is organ-on-chip drug testing and how does it define the market boundary?
It involves culturing living human cells within continuously perfused micrometer-sized chambers to replicate physiological functions. Functionally, this defines the commercial boundary between basic static cellular clusters and the dynamic physiological platforms explicitly validated for pharmacological screening.
Summarize organ-on-chip market drivers and barriers?
The primary driver is the regulatory acceptance of non-animal data coupled with pharma's urgent need to reduce late-stage clinical attrition. The dominant barrier remains the high integration cost and the labor-intensive requirement to standardize new microfluidic readouts against historical in vivo toxicity baselines.
Explain the commercial outlook for organ-on-chip drug testing?
The commercial trajectory turns from exploratory academic use toward mandatory, scaled deployment for high-risk screening. As high-throughput, plate-compatible hardware standardizes, laboratory managers are internalizing the infrastructure, moving vendor revenue heavily toward recurring consumable and software models.
Organ-on-chip CRO for toxicology studies: why outsource?
Tier-1 pharmaceutical companies rapidly exhaust their internal microfluidic screening capacity as they scale operations. They utilize specialized CROs to capture this overflow through dedicated validation protocols that perfectly align with the strict, proprietary standards set internally.
Organ-on-chip ROI compared with conventional assays: how is it measured?
Financial controllers recognize the return on investment entirely in the avoidance of late-stage failure costs. Disqualifying a single hepatotoxic compound pre-clinically offsets years of system hardware, specialized operator training, and expensive consumable investments.
How fast is organ-on-chip replacing animal models in pharma?
The pace is dictated by qualification timelines rather than simple purchasing capability. Integration tracks the regulatory calendar, accelerating significantly as public-private consortiums pool pre-competitive validation data, allowing pharma to confidently replace animal IND submissions within the next decade.
Microfluidic organ-on-chip market size: what validates the baseline?
The baseline valuation is anchored to verified hardware installation bases and annualized consumable run-rates across major global pharmaceutical research hubs, cross-validated against the specific contract research capacity metrics maintained by independent testing laboratories.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.