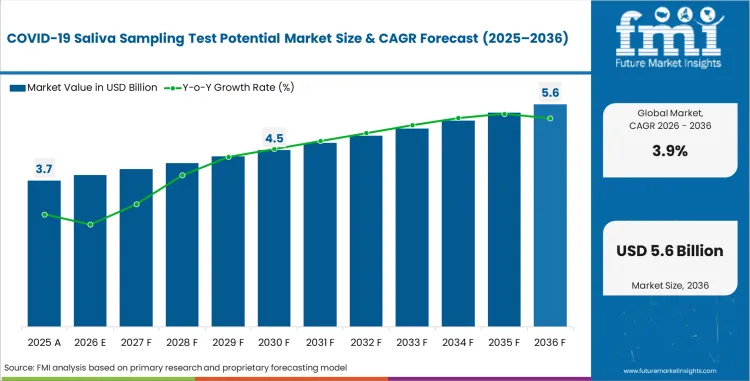
The covid-19 saliva sampling test potential market was valued at USD 3.70 billion in 2025, projected to reach USD 3.84 billion in 2026, and is forecast to expand to USD 5.64 billion by 2036 at a 3.9% CAGR. Saliva-based sampling is gaining traction as a non-invasive alternative to nasopharyngeal swab collection for respiratory pathogen surveillance. Public health agencies and large-venue operators are investing in saliva sampling infrastructure to enable high-throughput, self-administered testing at travel stations, sports arenas, and corporate campuses without requiring trained medical personnel for sample collection.
As per FMI, the saliva sampling test market is evolving from an emergency diagnostic tool into a standing surveillance infrastructure for respiratory pathogen monitoring. The initial pandemic established proof of concept for saliva-based SARS-CoV-2 detection, and public health agencies are now expanding the platform to cover influenza and RSV co-surveillance. FMI analysts observe that the non-invasive collection method and self-administration capability of saliva sampling create a structural cost advantage over nasopharyngeal swab collection, which requires trained healthcare workers and dedicated collection stations. Large-venue operators at travel hubs, sports arenas, and universities are adopting saliva sampling kiosks as a lower-friction entry screening method that maintains throughput without creating bottleneck queues.
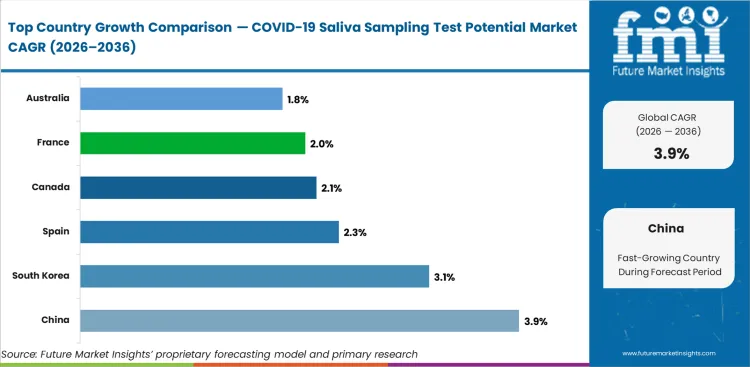
All major markets reflect accelerated deployment parameters. China sets a 3.9% CAGR, South Korea sets a 3.1% CAGR, Spain sets a 2.3% CAGR, France sets a 2.0% CAGR, and Australia sets a 1.8% CAGR. China leads at 3.9% CAGR as its national public health surveillance infrastructure maintains the largest installed base of centralized testing facilities. South Korea at 3.1% reflects early adoption of saliva-based testing platforms across its technology-forward public health system. Spain at 2.3% and France at 2.0% sustain demand through EU-funded respiratory pathogen surveillance networks. Australia at 1.8% maintains testing infrastructure for border health screening applications.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 3.84 billion |
| Industry Value (2036) | USD 5.64 billion |
| CAGR (2026-2036) | 3.9% |
Source: Future Market Insights, 2026
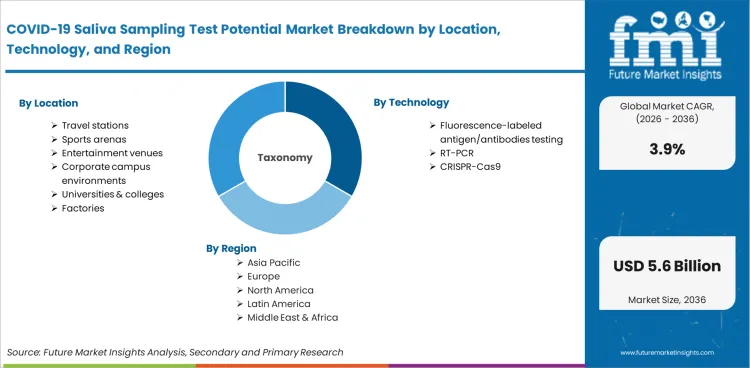
COVID-19 saliva sampling test systems are diagnostic platforms that detect SARS-CoV-2 and related respiratory pathogens using saliva specimens collected through non-invasive self-administration methods. System categories include fluorescence-labeled antigen and antibody assays, RT-PCR testing platforms adapted for saliva matrices, and CRISPR-Cas9 detection technologies. These systems are deployed at travel stations, sports venues, corporate campuses, universities, and factory environments for recurring screening programmes.
Market scope includes saliva-based COVID-19 diagnostic test kits, self-collection devices, and associated processing platforms across centralized and decentralized testing models. Regional and country-level market sizes, forecast period 2026 to 2036, and segment breakdowns by venue type, testing technology, and testing model are fully incorporated.
The scope excludes nasopharyngeal swab-based COVID-19 tests, serology and blood-based antibody tests, general-purpose molecular diagnostic platforms not adapted for saliva specimens, and vaccine administration infrastructure.
The Non-Invasive Nature of the COVID-19 Saliva Sampling Test Anticipates its Market Growth
Traditional nasal swabs may be extremely uncomfortable, making many people wary about getting tested. Saliva tests are easy to administer, painless, and capable of being self-collected with ease, thus enabling higher participation rates.
That's where accessibility comes in, be it school-going kids, white-collar workers, or public gatherings in large numbers, testing should be wide-ranging for the purpose of effective monitoring and subsequently conducting outbreak control. Therefore, the improved accessibility of COVID-19 saliva sampling test has significantly contributed to its increase in adoption rate.
Moreover, comfort and ease of saliva testing create a better perception among the public, reducing anxiety related to testing. This factor has led to an increase in demand of testing among populations that are hesitant to the traditional methods of testing. This simplicity in saliva collection makes logistics easier and rapid deployment possible across settings.
As various public health strategies continue to evolve and testing continues to play a crucial role in managing COVID-19, the appeal of non-invasive saliva tests is likely drive their adoption.
With the potential for wide usage in ongoing surveillance and early detection, they are a highly valued tool in fighting the virus and are even more strongly cemented in pandemic management. Therefore, the ease and convenience provided by the saliva sampling test contributes to its future growth prospects.
The Cost Effective Nature of COVID-19 Saliva Sampling Test Propels its Market Adoption
Traditional testing methods through nasal swabs usually require higher expenses because of the resources spent on expertise, equipment, and lab analysis. Saliva tests largely require lower resources, hence lowering both operational expenses and the price per test. This not only makes regular testing more affordable for schools, businesses, and healthcare providers but also starts a regular testing program in these places.
Beyond the greater affordability, lower costs often mean more extensive testing coverage and encourage organizations to adopt regular testing as a way of preventing the spread of the virus. Greater capacity enables rapid detection and isolation, with consequential reductions in rates of transmission and the overall burden on healthcare.
This all becomes more attractive in resource-limited settings, where budgetary constrictions can impair testing efforts. Reliably offering a low-resource alternative, saliva tests can support communities in sustaining public health efforts without necessarily straining the financial resources of that community.
Overall, affordable testing through saliva sampling puts these tests in a very attractive position and enhances their contribution to pandemic management by promoting wide market adoption.
Integration of COVID-19 Saliva Sampling Test into Public Health Strategies Surges its Market Growth
The integration of COVID-19 saliva sampling tests into public health strategies can greatly increases their market growth by positioning these tests as an essential tool in the management of the pandemic.
Saliva tests enable broad screening, which makes them fundamental tools in the monitoring of community transmission and for the early detection of outbreaks. This capacity aligns with public health objectives to enhance surveillance, particularly in high-risk settings such as schools, workplaces, and events.
Saliva tests normally are part of comprehensive response plans because public health authorities are now aware of the need for accessible testing. Their non-invasive and easy-to-administer nature allows them to be deployed on a large scale, hence making testing more feasible and less disrupting.
Saliva tests are well-placed for regular monitoring at higher frequency without being onerous on healthcare systems. Funding and supporting public health initiatives increasingly focuses on these methods of testing due to cost-effectiveness, which further propel their growing adoption.
This factors places saliva testing in a strategic position within the framework of public health, where improvements in community health outcomes are correspondingly entrenching the market position for these tests and securing their applicability in future health strategies.
Variation in Results Owing to Sensitivity and Specificity of Saliva tests hinders its Market Growth
Sensitivity often refers to the ability of test to correctly identify the virus. The inconsistent sensitivity and specificity demonstrated by saliva test have raised many concerns about their reliability in comparison with the established nasal swab tests.
Poor consistency in results might drive up the rate of false negatives, mislabeling infectious people as free of the virus. This can pose certain public health risks because undetected transmission could set off more outbreaks.
On the other hand, false positives sometimes could even lead to unjustified isolation or medical intervention. Such variations in test accuracy erodes confidence in health professionals and patients who might opt for more conventional forms of testing that are reliable.
These challenges, in the end, preclude large-scale adoption and acceptance of saliva tests, invariably holding them back from achieving more robust market growth in a diagnostic sector where high degrees of accuracy will be expected. The cautious approach may affect the integration of saliva testing into routine COVID-19 screening practices.
Tier 1 companies comprise market leaders with a market revenue of above USD 100 million capturing significant market share of 63.6% in global market. These market leaders are characterized by high production capacity, wide product portfolio, and range of services in the market.
These market leaders are differentiating themselves from the market based on extensive expertise in manufacturing and broad geographical outreach reach in the market. These factors have aided them in serving a wide range of consumer base. Prominent companies within tier 1 include ARUP Laboratories, Vatic Health and Mogene, Abbott
Tier 2 companies include mid-size players with revenue of USD 50 to 100 million having presence in specific regions with their high influence on the local market. These players hold around 27.6% share of the market as they are characterized by a strong presence overseas and strong market knowledge.
These market players have good technology and ensure regulatory compliance but may be limited to their availability in certain markets. Prominent companies in tier 2 include Psomagen Inc, DxTerity Diagnostics Inc, and Chembio Diagnostics.
Tier 3 includes the majority of companies that have limited geographical outreach or are operating at the local presence and serving niche markets having revenue below USD 50 million. These companies are focused towards fulfilling the demand from local market and are therefore classified as the tier 3 share segment.
They are comparatively small-scale players and are recognized as an unorganized market, which doesn’t have extensive infrastructure structure or higher production capabilities.
The section below covers the industry analysis for the COVID-19 saliva sampling test potential cover market for different countries. Market demand analysis on key countries in several regions of the globe, including North America, Asia Pacific, Europe, and others, is provided. The USA is anticipated to remain at the forefront in North America, and is projected to grow at a CAGR of 1.2% through 2036.
.webp)
| Countries | Value CAGR (2026 to 2036) |
|---|---|
| Canada | 2.1% |
| Spain | 2.3% |
| France | 2.0% |
| South Korea | 3.1% |
| China | 3.9% |
| Australia | 1.8% |
Ease of administration and lesser discomfort provided by COVID-19 saliva sampling test as compared to the conventional nasal swabbing offer greater adoption rates among the public. As, they are particularly useful in schools, at workplaces, and in events where testing regularly is very important for maintaining safety.
Their quick turnaround time for results enhances responsiveness to outbreaks, offering more support to public health campaigns. These tests have been endorsed by various health authorities and organizations, further nurturing their adoption among the general public. In addition, ongoing technological improvements with major focus on enhancing accuracy and reliability of the tests further surges their market growth.
Therefore, the increased demand, innovations, and the need for easy test methods when dealing with COVID-19, the market is bound to increase which demonstrates positive cycle of growth in the country. Moreover, advancing technology, growing investment in research and development for improving saliva testing technologies anticipates the growth of the market in the country.
Saliva tests are self-collecting tests that avoid the invasion of traditional nasal swabs. Therefore, ease of use lessens most of the apprehension or hesitation about testing, which lead to increased adoption in populations where people have phobias for medical procedures, and needle.
The comfort factor trigger regular testing to take place in schools, workplaces, and at events within the community where consistent monitoring for safety is necessary. It follows public health guidelines that are insisted on focusing the way for curbing the spread of COVID-19.
Furthermore, the endorsement of health authorities instills greater confidence in saliva tests, making them the tests of preference. Convenience and comfort are what people want, and thus it creates a market of tests for saliva sampling. This enhances their role in pandemic management and ensures effective public health strategies which have significantly contributed to its market growth, throughout the country.
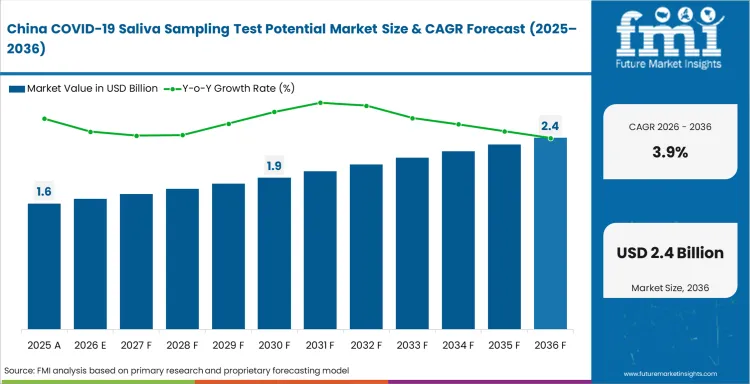
While the country has articulated a focus on rapid and effective case-finding to control outbreaks, accessible testing options have become sought after at an unprecedented level. Saliva tests, being non-invasive and easy to administer, it has witnessed a significant adoption in schools, and workplaces of the country.
This demand is further driven by government policies that outline regular testing as one of the public health strategies. Controlling outbreaks and making environments safe for day-to-day activities ensure scalable solutions in testing.
Moreover, as saliva tests has rapid results it aid in initiation of planning for rapid response activities. With increased awareness among the people for testing, there is a tendency to go for convenience, thereby reinforcing market growth.
In such a dynamic environment, continuous investments in research and development are foreseen to further bring in innovations that improve test accuracy and reliability. These factors aid saliva sampling tests to hold an edge in the Chinese market.
The section contains information about the leading segments in the industry. By technology, the fluorescence-labeled antigen/antibodies testing segment is estimated to account for significant share of the market share and is anticipated to witness significant growth during the forecast period.
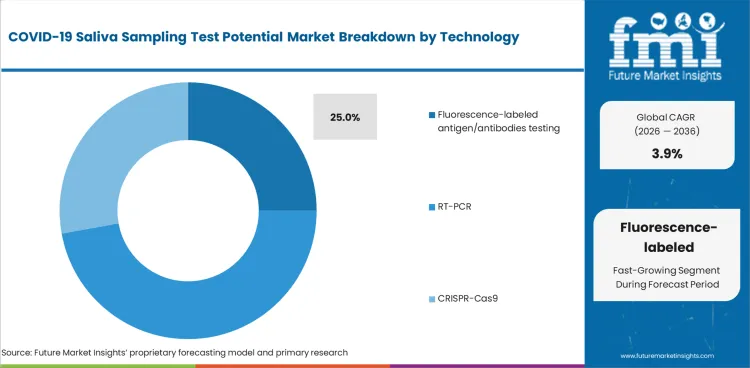
| Technology | Fluorescence-labeled antigen/antibodies testing |
|---|---|
| Value Share (2036) | 25.0% |
Fluorescence-labeled antigen/antibody testing holds the largest share of 25.0% in the potential market of COVID-19 saliva testing due to its higher sensitivity and specificity. This technique make use of fluorescent tracers in detecting antigens or antibodies of viruses present in the saliva for the precise identification of the current or past infection.
Speedy returns are important in outbreak management; hence, this testing modality has a high value in ensuring responses to public health.
In addition, fluorescence-based assays can easily be transitioned to high-throughput formats. The technology has continuously benefited from improvement of assay design and fluorescence labeling techniques for better performance and reliability.
In addition, the growing focus on non-invasive test methods extends a good match with the ease and comfort of saliva collection, hence improving compliance among users. Collectively, these factors contribute fluorescence-labeled antigen/antibody testing to hold largest share in the evolving COVID-19 saliva sampling test market.
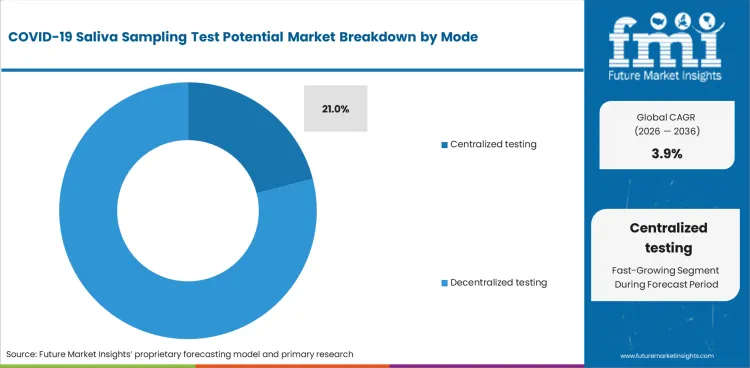
| Mode | Centralized Testing |
|---|---|
| Value Share (2036) | 21.0% |
Centralized testing has the largest share of 21.0% in the potential market of COVID-19 saliva sampling tests, owing to efficiency and reliability concerning large-scale management of testing operations.
The centralized facilities develop processes that ensure samples collection, process, and analyze in a systematic manner. This factor is particularly advantageous in a public health crisis where results have rapid turnaround times for timely interventions.
Moreover, advanced laboratory technologies and expertise, possible in centralized testing, become quite limited at decentralized sites. This improves the accuracy and reliability of the test results so as to enhance public trust in the testing process. It also allows for better quality control and standardization across different testing sites.
Besides, large centralized facilities can process numerous samples, which are ideal for mass testing initiatives that are required across the highly populous areas. Centralized testing allows resources to be distributed more effectively, ensuring personnel and equipment are used to maximum efficiency. All these factors together allows centralized testing to dominate the COVID-19 saliva sampling test market.
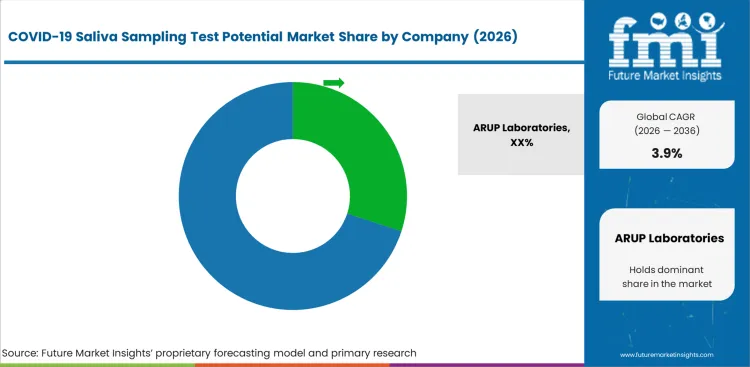
Key players operating in the COVID-19 saliva sampling test potential cover market are investing in development of advanced and innovative products that are more reliable and efficient. Also, many of the key players are emphasizing on making strategic collaborations and acquisition for expansion of their geographical presence and strengthening their market share.
Recent Industry Developments in COVID-19 Saliva Sampling Test Potential Market
| Metric | Value |
|---|---|
| Quantitative Units | USD 3.84 billion to USD 5.64 billion, at a CAGR of 3.9% |
| Market Definition | COVID-19 saliva sampling test systems encompass non-invasive diagnostic platforms using saliva specimens for SARS-CoV-2 detection across high-traffic venues and surveillance testing facilities. |
| Venue Type Segmentation | Travel Stations, Sports Arenas, Entertainment Venues, Corporate Campus Environments, Universities & Colleges, Factories |
| Testing Technology Segmentation | Fluorescence-labeled Antigen/Antibodies Testing, RT-PCR, CRISPR-Cas9 |
| Testing Model Segmentation | Centralized Testing, Decentralized Testing |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, South Korea, Spain, France, Australia, and 40 plus countries |
| Key Companies Profiled | ARUP Laboratories, Vatic Health, Mogene, Psomagen Inc, DxTerity Diagnostics Inc, Chembio Diagnostics, Abbott |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with installed testing facility capacity and recurring test volumes, cross-validated against public health programme budgets and diagnostic manufacturer revenue disclosures. |
In terms of Location, the industry is divided into travel stations, sports arenas, entertainment venues, corporate campus environments, universities & colleges and factories
In terms of technology, the industry is divided into fluorescence-labeled antigen/antibodies testing, RT-PCR, CRISPR-Cas9
In terms of mode, the industry is segregated into centralized testing and decentralized testing
Key countries of North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and Middle East and Africa (MEA), have been covered in the report.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
How large is the demand for COVID-19 saliva sampling tests in the global market in 2026?
Demand for COVID-19 saliva sampling tests in the global market is estimated to be valued at USD 3.84 billion in 2026.
What will be the market size of COVID-19 saliva sampling tests by 2036?
Market size for COVID-19 saliva sampling tests is projected to reach USD 5.64 billion by 2036.
What is the expected demand growth between 2026 and 2036?
Demand for COVID-19 saliva sampling tests is expected to grow at a CAGR of 3.9% between 2026 and 2036.
Which venue type is a primary deployment channel?
Travel stations are a primary deployment channel as border health screening and international transit requirements maintain consistent demand for high-throughput, non-invasive testing infrastructure.
What testing technologies are covered?
The report covers fluorescence-labeled antigen/antibody testing, RT-PCR, and CRISPR-Cas9 detection technologies adapted for saliva sample matrices.
What is driving demand in China?
China maintains the largest installed base of centralized testing facilities within its national public health surveillance infrastructure, sustaining demand at 3.9% CAGR through 2036.
What is included in the scope of this report?
The market covers saliva-based COVID-19 test kits, self-collection devices, and processing platforms across centralized and decentralized models, with breakdowns by venue type, testing technology, and testing model.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.