Cryo Peptides Market
Cryo Peptides Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Cryo Peptides Market Forecast and Outlook 2026 to 2036
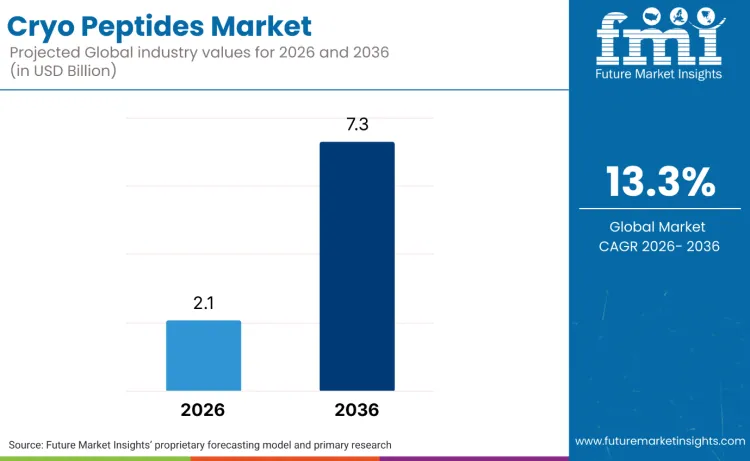
The cryo peptides market is valued at USD 2.1 billion in 2026 and is projected to reach USD 7.3 billion by 2036, expanding at a 13.3% CAGR. Growth quality is being shaped by large dermocosmetic platforms concentrating capital on science-led actives and physician-adjacent distribution. L’Oréal’s agreement to acquire Medik8 is a structural signal that high-efficacy, ingredient-led skincare is being treated as a scalable platform rather than a niche premium label. The company framed Medik8 as a fit with its dermatological beauty strategy, stating that Medik8 has built a strong position in the UK, with accelerating growth in North America, and is well positioned to grow in China.
Protocol-led demand is reinforced by the scale of minimally invasive aesthetics. The American Society of Plastic Surgeons reports neuromodulator injections remained the number one minimally invasive category in 2024, with almost 10 million patients. This procedure volume sustains a large addressable base for topical regimens positioned around wrinkle modulation and post-procedure maintenance.
On the product side, brands are explicitly linking peptide serums to injectable-adjacent expectations. SkinCeuticals positions P-TIOX as a peptide-powered serum inspired by anti-wrinkle neurotoxin injections, reflecting how clinical language is being used to translate procedure logic into retail repeat purchase.
Cryo Peptides Market Key Takeaways
| Metrics | Values |
|---|---|
| Market Value (2026) | USD 2.1 billion |
| Market Value (2036) | USD 7.3 billion |
| CAGR (2026 to 2036) | 13.3% |
Summary of the Cryo Peptides Market
What Is Growth Outlook for Cryo Peptides Market as per Future Market Insights Projection?
Future Market Insights projects the cryo peptides market to grow at a 13.3% CAGR, from USD 2.1 billion in 2026 to USD 7.3 billion by 2036.
FMI Research Approach: Forecasting based on clinic-adjacent anti-ageing protocol adoption, premium serum conversion rates, and peptide-led claim substantiation readiness.
How Do FMI Analysts Perceive the Cryo Peptides Market to Evolve?
FMI sees the market shifting from premium skincare positioning toward protocol-led maintenance, where peptide serums are integrated into injectable and device care routines and sold through channels that can validate outcomes.
FMI Research Approach: Analysis of professional channel expansion, acquisition signals in dermocosmetics, and regimen standardisation across premium aesthetics.
Which Country Holds Largest Share in Global Cryo Peptides Market?
China holds a significant share, supported by a claims governance framework that formalises efficacy evaluation expectations and by ongoing updates to the national cosmetics ingredient inventory, which influences the pace of compliant innovation.
FMI Research Approach: Country-level modelling aligned with regulatory compliance intensity, premium skincare channel scale, and import and domestic brand activation.
How Large Will the Cryo Peptides Market Be by 2036?
By 2036, the cryo peptides market is projected to reach USD 7.3 billion, supported by premium beauty consumer adoption and higher utilisation through dermatology and medical aesthetic centers.
FMI Research Approach: Build-up forecasting using end-user mix, channel conversion, and repeat purchase economics tied to protocol-based regimens.
What Is Cryo Peptides Market Definition in This Study?
In this study, cryo peptides refer to premium skincare formulations where peptide systems are used to target visible ageing, texture, and firmness outcomes, often aligned to professional-grade routine expectations and sold through premium retail, DTC, and clinic-adjacent channels.
FMI Research Approach: Scope anchored to peptide-driven efficacy claims, formulation formats used in premium skincare, and channel behaviours visible in dermocosmetic portfolios.
What Are the Key Trends Strengthening the Cryo Peptides Market Expansion?
FMI identifies a convergence between peptide science and procedure-led demand, where brands adopt injectable-adjacent language and expand professional distribution to capture recurring maintenance spend.
FMI Research Approach: Tracking of portfolio decisions, acquisition signals, and protocol framing by global dermatological beauty groups.
Which Forces Are Accelerating Cryo Peptides Adoption?
The market is accelerating as peptide science becomes easier to convert into repeatable claims and premium channel protocols. Peer-reviewed evidence positions biomimetic peptides as functional cosmetic actives used to influence visible ageing pathways, supporting why signal-peptide messaging is credible at launch and scalable across portfolios. Demand is amplified by the procedural base for wrinkle management. ASPS reports neuromodulator injections remained the leading minimally invasive category in 2024 with 9,883,711 patients, creating a large audience primed for maintenance skincare positioned around wrinkle modulation. Corporate capital allocation confirms category priority, with L’Oréal’s announced acquisition of Medik8 explicitly tied to dermatological beauty expansion and growth ambition in China.
How is the Cryo Peptides Market Segmented?
The cryo peptides market is segmented by product type, peptide type, formulation, application, and end user to reflect how protocol-led skincare is commercialised. Product type captures the outcome promise that anchors purchase, peptide type reflects the mechanism language used in claim architecture, formulation determines achievable concentration and user compliance, application aligns with the highest-frequency routines, and end user separates premium self-directed consumers from clinic-led dispensing models.
What Explains the Leadership of Anti Aging Cryo Peptides in Product Type?
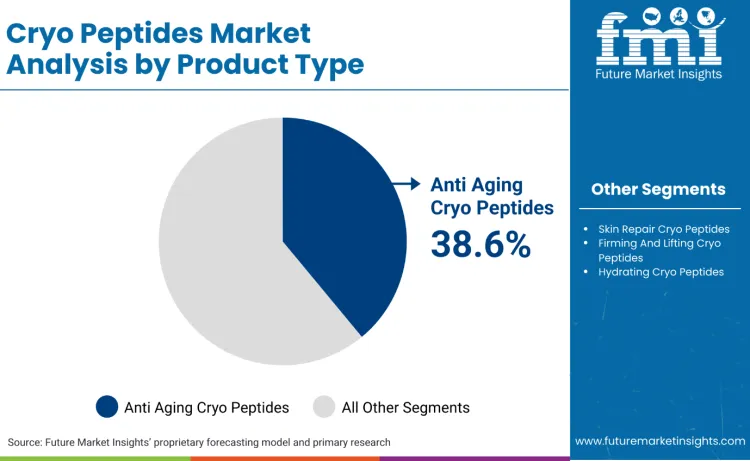
Anti aging cryo peptides lead the product type mix with a 38.6% share because the category monetises maintenance demand created by high, repeatable procedure throughput. Neuromodulator injections remain the dominant minimally invasive procedure in the USA, indicating a large installed base of consumers paying repeatedly for wrinkle-related outcomes. Brands convert this procedural intent into topical regimens by framing peptide serums as adjacent to injection effects rather than as generic moisturisers.
Skin Ceuticals explicitly positions P-TIOX as a peptide-powered serum inspired by anti-wrinkle neurotoxin injections, signalling a deliberate strategy to align topical anti-ageing with the language of clinical intervention. Platform behaviour reinforces this prioritisation. L’Oréal’s planned acquisition of Medik8 is justified through dermatological beauty expansion and international scaling, showing that anti-ageing efficacy stories are being consolidated into groups with professional channel reach.
Why Do Signal Peptides Dominate the Peptide Type Mix?
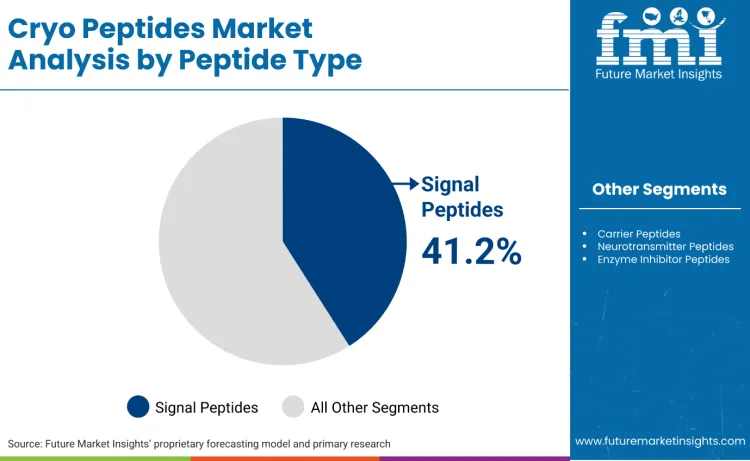
Signal peptides hold a 41.2% share because they convert best into defensible, repeatable claims across regulated markets while staying within cosmetic boundaries. Biomimetic peptide literature frames peptides as cosmetic actives that can be designed to interact with skin biology and visible ageing pathways, which supports consistent messaging around firmness, texture, and wrinkle appearance within cosmetic regulatory boundaries. This matters in markets tightening claims governance.
China’s NMPA issued a national specification for evaluating cosmetic efficacy claims, raising the compliance value of mechanisms that can be supported through structured substantiation rather than vague sensory narratives. Signal peptides offer a scalable mechanism language that can be packaged into dossiers, clinical-style testing, and claims files. Portfolio strategy also supports dominance. Dermatological beauty groups are consolidating efficacy-led brands and scaling them internationally, as seen in L’Oréal’s move for Medik8, which it links to science-led skincare and growth ambitions across regions including China.
What Makes Serums the Primary Formulation Format?
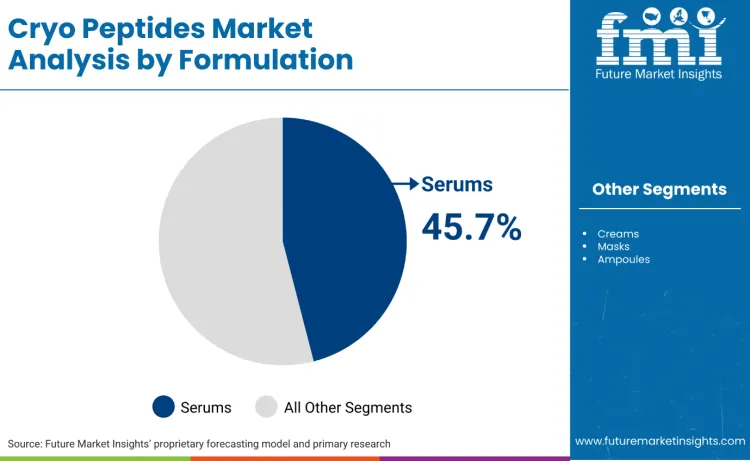
Serums lead the formulation mix with a 45.7% share because they provide the highest commercial efficiency for premium actives, the cleanest protocol compliance, and the fastest cross-channel scaling. In practice, the category’s leading narratives are being built around serum-first regimens that promise measurable improvement and are easy to layer with procedures and devices. SkinCeuticals’ positioning of P-TIOX as a peptide-powered serum linked to injectable-adjacent expectations illustrates how brands use serum format to signal potency and routine integration.
The format also aligns with the market’s dominant use case. Facial care holds the highest application share because daily face routines drive frequency, and serums fit step-based routines where concentration and sensory finish can be tuned without changing the cleanser or moisturiser backbone. Procedure dynamics reinforce this logic. With neuromodulator injections and other minimally invasive treatments sustaining high volumes, maintenance routines need a lightweight, repeatable format that consumers will use consistently between visits.
What Are the Key Trends and Restraints in the Cryo Peptides Market?
The most important trend is protocol convergence between aesthetics and premium skincare. Brands are using injectable-adjacent framing to translate clinical outcome expectations into daily topical use, making peptide serums a maintenance layer rather than an optional upgrade. SkinCeuticals’ positioning of a peptide serum inspired by neurotoxin injections is a clear indicator of how messaging is being redesigned to capture post-procedure intent. This trend is supported by the continued dominance of injectables in the procedure mix, which sustains recurring touchpoints where clinicians, medical spas, and premium retailers can recommend topical regimens that extend perceived results.
The main restraint is rising compliance load in the largest premium markets, which increases time-to-market and documentation cost for peptide-led claims. In the USA, MoCRA introduced new requirements across facility registration, product listing, and serious adverse event reporting, forcing brands to formalise systems and supplier traceability before scaling. In China, NMPA’s efficacy claim evaluation specification raises expectations for substantiation discipline, tightening the cost-benefit of aggressive performance claims without robust testing and documentation. These controls do not remove demand, but they constrain smaller brands that lack regulatory infrastructure and slow international rollouts when dossiers and process readiness are not designed for multi-market governance from day one.
Cryo Peptides Market Analysis by Key Countries
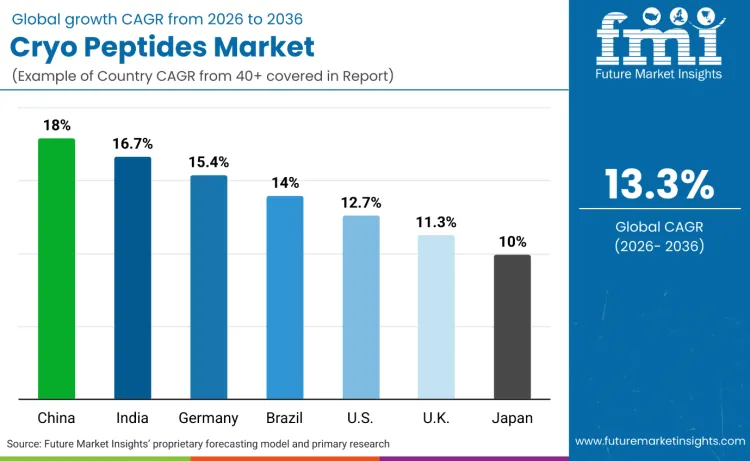
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 18.0% |
| India | 16.7% |
| Germany | 15.4% |
| Brazil | 14.0% |
| USA | 12.7% |
| UK | 11.3% |
| Japan | 10.0% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
Why Is China a High-Growth Market for Cryo Peptides Through 2036?
China is projected to expand at an 18.09% CAGR across 2026 to 2036, anchored in regulatory formalisation that rewards documented efficacy and accelerates compliant portfolio scaling. NMPA’s specification for evaluating cosmetic efficacy claims raises the commercial value of peptide systems that can be supported through structured substantiation rather than broad sensory promises. The regulatory environment also evolves through updates to the national cosmetics ingredient inventory, influencing how quickly brands can refresh compliant formulations while staying aligned with permissible ingredient frameworks. On the supply side, global dermocosmetic groups are explicitly prioritising China as a growth engine for science-led skincare. L’Oréal’s planned acquisition of Medik8 is framed with growth ambition in China, signalling that international platforms expect protocol-led, efficacy-focused skincare to scale through Chinese premium channels. This combination of claims governance and platform investment concentrates launches into peptide-heavy serums, sustaining above-average CAGR.
What Sustains India’s Expansion in Cryo Peptides Across 2026 to 2036?
India is forecast to grow at a 16.75% CAGR from 2026 to 2036, driven by a tightening regulatory base and the rapid formalisation of premium skincare distribution, which favours brands able to comply and educate. The Cosmetics Rules, 2020 establish enforcement pathways including non-compliance provisions, pushing premium importers and domestic brands toward stronger documentation and labelling discipline as they scale. This benefits peptide-led products that can be positioned with clearer performance architecture and ingredient rationale. Cross-market platform behaviour also matters because multinational dermocosmetic groups are expanding in India as dermocosmetics demand rises. L’Oréal describes dermatological beauty momentum and explicitly notes strong growth in India within its broader strategy narrative, reinforcing that global portfolios see India as a priority geography for clinically positioned skincare. As premium beauty consumers increase spend in online and urban retail ecosystems, peptide serums become a repeatable routine purchase tied to visible-ageing outcomes, lifting long-run adoption.
Why Does Germany Show Strong CAGR in Cryo Peptides?
Germany is projected to expand at a 15.41% CAGR during 2026 to 2036, reflecting the scale of the national beauty and home care market and the strength of structured retail and pharmacy ecosystems that support premium, science-led positioning. The German cosmetic industry association IKW reports record consumer spending on beauty and home care products, highlighting the country’s capacity to absorb higher-value skincare categories. Germany also has a dense pharmacy infrastructure and established consumer trust in health-adjacent purchase environments, creating favourable conditions for peptide serums positioned around efficacy and tolerance rather than novelty. Compliance discipline is reinforced by EU cosmetics governance, which standardises safety and claims expectations across the market. These factors raise conversion in premium segments that value substantiation and professional recommendation, supporting strong CAGR even without the extreme procedure-led scaling seen in the USA.
What Drives Brazil’s Cryo Peptides Growth Through 2036?
Brazil is forecast to grow at a 14.07% CAGR from 2026 to 2036, linked to the scale and visibility of aesthetic procedures and a regulatory framework that structures how cosmetics are notified and controlled. The ISAPS global survey reports large national procedure markets and tracks Brazil as a dedicated location for procedure statistics, reinforcing the size of the clinic and medical aesthetic center ecosystem that feeds protocol-led skincare demand. On the regulatory side, ANVISA sets requirements for cosmetics and fragrances, including registration and notification pathways and post-market controls, shaping how brands structure compliant premium launches. This combination supports a channel mix where medical aesthetic centers and premium consumers drive repeat usage of peptide serums aligned to firmness and anti-ageing outcomes, with clinics acting as both demand generators and trust infrastructure.
Why Does the United States Remain a Core Market for Cryo Peptides?
The United States is projected to expand at a 12.73% CAGR across 2026 to 2036 because procedure volumes create a continuous maintenance market and regulation is forcing higher operational maturity that advantages scaled players. ASPS reports neuromodulator injections reached 9,883,711 patients in 2024, maintaining injectables as the largest minimally invasive category and sustaining routine demand for adjunct topical regimens. At the same time, MoCRA requirements around facility registration, product listing, and serious adverse event reporting increase compliance overhead, which favours brands embedded in large platforms and professional channels. These conditions align with physician-dispensed and premium retail strategies, where peptide serums can be sold as part of ongoing protocol routines rather than one-time prestige purchases, sustaining steady double-digit growth even as governance tightens.
What Explains the United Kingdom’s Sustained Growth Trajectory?
The United Kingdom is forecast to grow at an 11.39% CAGR from 2026 to 2036, supported by a post-Brexit compliance system that formalises market access and rewards brands with strong regulatory and documentation capabilities, while premium consumer demand concentrates in higher-value skincare categories. The UK requires cosmetic product notifications through the Submit Cosmetic Product Notification service, creating a defined compliance step for brands selling into the market. This procedural governance favours companies with disciplined product information files and stable supply chains, which aligns with peptide-led products that depend on clear claims architecture. On the corporate side, the UK’s importance as a base for science-led premium skincare is visible in platform acquisition logic, with L’Oréal explicitly highlighting Medik8’s strong position in the UK as part of the rationale for acquisition and scaling. The result is a market where premium serums remain a primary growth engine.
Why Is Japan’s Growth More Measured Than Other High-Premium Markets?
Japan is projected to expand at a 10.05% CAGR during 2026 to 2036, moderated by regulatory categorisation and ingredient governance that shapes how performance claims can be made and how products are positioned between cosmetics and quasi-drugs. Japan’s Standards for Cosmetic Products define prohibited ingredients and compliance expectations, constraining certain actives and tightening formulation choices. Government guidance also clarifies that products positioned closer to functional outcomes may fall under quasi-drug frameworks, shaping launch strategy and slowing the pace of aggressive performance claims in mainstream cosmetic channels. These controls encourage conservative claim architecture and raise the value of trusted distribution and brand equity. The market still grows as premium consumers adopt peptide serums for facial care, but governance and classification effects keep CAGR below China and India.
What Is Shaping Competition in the Cryo Peptides Market?
Competition is consolidating around dermocosmetic platforms with the compliance infrastructure to scale peptide-led claims across regulated markets. SkinCeuticals is the leading brand in this study’s scope, supported by L’Oréal’s Dermatological Beauty portfolio scale and its stated strategy to expand science-led skincare through targeted acquisitions.
North America is defined by professional dispensing power and procedure adjacency, where MoCRA compliance and high injectable volumes reward brands that can document safety, manage listings, and convert clinic protocols into repeat serum usage. Europe remains more fragmented, with strong premium physician-dispensed brands and UK-origin science-led players now being pulled into global platforms via acquisition. Asia diverges by governance structure. In China, efficacy claim evaluation and ingredient inventory updates reward companies that can build substantiation files at scale. In Japan, cosmetics standards and quasi-drug categorisation constrain claim aggressiveness, limiting direct transfer of global leadership into local dominance.
Recent Developments:
- In June 2025, L’Oréal signed an agreement to acquire Medik8, positioning the deal as an expansion move for Dermatological Beauty and science-led skincare.
- In 2024 to 2025, SkinCeuticals advanced peptide-serum positioning tied to injectable-adjacent expectations through P-TIOX protocol language.
- In 2024 to 2026, FDA expanded MoCRA implementation resources and guidance for cosmetics registration and listing, raising compliance requirements for brands operating in the USA.
Key Players in the Cryo Peptides Market
- SkinCeuticals
- SkinMedica
- Dr. Barbara Sturm
- Allies of Skin
- Revision Skincare
- Medik8
- PCA Skin
- iS Clinical
- Alastin Skincare
- ZO Skin Health
Market Definition
Cryo peptides are premium skincare products in which peptide systems form the core functional active platform for visible-ageing, firmness, texture, and wrinkle appearance outcomes, typically delivered in serum, cream, mask, or ampoule formats. The category is defined by protocol language and claim architecture that aligns topical use with professional aesthetic expectations. Products are commercialised through premium beauty retail, online direct-to-consumer, dermatology clinics, and medical aesthetic centers. The market is assessed in value terms and focuses on skincare use cases rather than therapeutic drug treatment. Scope emphasises biomimetic and signal-peptide systems positioned for facial anti-ageing maintenance and repeat purchase behaviour.
Market Inclusion
This study includes peptide-led premium skincare formulations marketed for anti-ageing, firming, lifting, hydration, and skin repair outcomes when peptides are presented as the primary active technology. Included formats cover serums, creams, masks, and ampoules used for facial care, eye care, neck and décolletage care, and professional maintenance routines. Included channels span online direct-to-consumer, premium beauty retail, dermatology clinics, and medical aesthetic centers. The scope includes products positioned with substantiated efficacy and safety documentation pathways in regulated markets, including the USA under MoCRA and China under NMPA efficacy claim governance.
Market Exclusion
This study excludes injectable peptides, prescription therapeutics, medical devices, and in-clinic procedures where the primary intervention is not a topical skincare product. It excludes products where peptides are incidental minor ingredients without a peptide-led claim architecture. It excludes general moisturisers, cleansers, sunscreens, and fragrance-led prestige skincare unless peptides are the core functional platform. It excludes aesthetic injectables and neuromodulators as revenue categories, while recognising their role in shaping adjacent topical maintenance demand. It excludes food, supplements, and ingestible collagen or peptide products.
Scope of Report Table
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 2.1 billion |
| Forecast Value (2036) | USD 7.3 billion |
| CAGR (2026 to 2036) | 13.3% |
| Product Type Segments | Anti Aging Cryo Peptides; Skin Repair Cryo Peptides; Firming and Lifting Cryo Peptides; Hydrating Cryo Peptides |
| Peptide Type Segments | Signal Peptides; Carrier Peptides; Neurotransmitter Peptides; Enzyme Inhibitor Peptides |
| Formulation Segments | Serums; Creams; Masks; Ampoules |
| Application Segments | Facial Care; Eye Care; Neck and Décolletage Care; Professional Treatments |
| End User Segments | Premium Beauty Consumers; Dermatology Clinics; Medical Aesthetic Centers |
| Countries Covered | China; India; Germany; Brazil; USA; UK; Japan |
| Key Companies Profiled | SkinCeuticals; SkinMedica; Dr. Barbara Sturm; Allies of Skin; Revision Skincare; Medik8; PCA Skin; iS Clinical; Alastin Skincare; ZO Skin Health |
| Additional Attributes | Dollar sales by product type, peptide type, formulation, application, and end user; channel economics split between premium retail, online DTC, dermatology clinics, and medical aesthetic centers; compliance readiness under MoCRA registration and adverse event requirements in the USA and NMPA efficacy claim governance in China; claim substantiation and product information file discipline under EU and UK notification frameworks; peptide sourcing, stability, and quality documentation requirements aligned to premium efficacy claims and post-market controls; role of procedure adjacency and protocol language in converting injectable demand into topical repeat purchase. |
Cryo Peptides Market by Key Segments
By Product Type:
Anti Aging Cryo Peptides; Skin Repair Cryo Peptides; Firming and Lifting Cryo Peptides; Hydrating Cryo Peptides
By Peptide Type:
Signal Peptides; Carrier Peptides; Neurotransmitter Peptides; Enzyme Inhibitor Peptides
By Formulation:
Serums; Creams; Masks; Ampoules
By Application:
Facial Care; Eye Care; Neck and Décolletage Care; Professional Treatments
By End User:
Premium Beauty Consumers; Dermatology Clinics; Medical Aesthetic Centers
Bibliography (APA Style)
- American Society of Plastic Surgeons. (2024). 2024 plastic surgery statistics report.
- L’Oréal. (2024). Annual report 2024 essentials.
- L’Oréal. (2025). L’Oréal signs agreement to acquire a majority stake in Medik8 [Press release].
- Ministry of Health, Labour and Welfare. (2020). Standards for cosmetic products (Japan).
- National Medical Products Administration. (2021). Specification for evaluation of cosmetic efficacy claims (announcement and requirements) (Notice No. 50, 2021).
- SkinCeuticals. (n.d.). P-TIOX advanced peptide serum product information.
- USA Food and Drug Administration. (2026). Modernization of Cosmetics Regulation Act of 2022 (MoCRA).
Frequently Asked Questions
What is the projected size of the cryo peptides market in 2036?
The market is projected to reach USD 7.3 billion by 2036.
Which segment leads the product type in the cryo peptides market in 2026?
Anti aging cryo peptides lead the product type mix with a 38.6% share.
Which peptide type dominates the cryo peptides market structure in 2026?
Signal peptides lead the peptide type mix with a 41.2% share.
Which formulation format holds the largest share in the cryo peptides market?
Serums lead the formulation mix with a 45.7% share, reflecting protocol-led repeat usage.
What is the most binding constraint for brands scaling globally?
Regulatory compliance load across major markets, including MoCRA operational requirements in the USA and efficacy-claim governance in China, raises documentation and time-to-market demands for peptide-led launches.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Product Type, 2026 to 2036
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Peptide Type
- Y to o to Y Growth Trend Analysis By Peptide Type
- Absolute $ Opportunity Analysis By Peptide Type
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Formulation
- Y to o to Y Growth Trend Analysis By Formulation
- Absolute $ Opportunity Analysis By Formulation
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Y to o to Y Growth Trend Analysis By Application
- Absolute $ Opportunity Analysis By Application
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Y to o to Y Growth Trend Analysis By End User
- Absolute $ Opportunity Analysis By End User
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- Key Countries Market Analysis
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Peptide Type
- By Formulation
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Cryo Peptides Market Key Takeaways
- Global Cryo Peptides Market Size, 2021 to 2036
- Cryo Peptides Market Size by Product Type
- Cryo Peptides Market Size by Peptide Type
- Cryo Peptides Market Size by Formulation
- Cryo Peptides Market Size by Application
- Cryo Peptides Market Size by End User
- Cryo Peptides Market Size by Region
- Cryo Peptides Market CAGR by Country
- Scope of Report
List of Figures
- Cryo Peptides Market Growth Trajectory, 2026 to 2036
- Protocol-led Demand Conversion Framework
- Product Type Share Analysis
- Peptide Type Share Distribution
- Formulation Mix Analysis
- Regional Market Attractiveness Analysis
- Competitive Landscape Overview
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

