Digital Colony Counter Market
Digital Colony Counter Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Digital Colony Counter Market Forecast and Outlook 2026 to 2036
The digital colony counter market is set to reach USD 310.0 million in 2026 and is forecasted to reach USD 657.0 million by 2036 at a CAGR of 7.8%. Value formation reflects rising reliance on automated microbial enumeration within regulated laboratory workflows where accuracy, traceability, and throughput directly affect operational continuity. Digital counters replace manual visual counting in environments handling high plate volumes under time constraints. Expenditure concentrates on systems that standardize results across operators, reduce repeat testing, and support documented compliance requirements. Software-enabled image capture and data storage strengthen audit readiness, making capital allocation less discretionary and more procedural within quality-controlled laboratory settings.
Expansion through the forecast period is shaped by validation rigor, workflow integration limits, and cost discipline across laboratory tiers. Adoption remains uneven where low sample volumes do not justify automation investment. Algorithm performance depends on plate quality, lighting consistency, and colony morphology, requiring method verification before routine use. Regulatory expectations for equivalence with established microbiological methods extend qualification timelines. Maintenance of optical components and software updates adds lifecycle cost considerations. Procurement favors platforms compatible with existing plate formats and information systems to avoid workflow disruption. Growth aligns with laboratory capacity expansion, compliance intensity, and replacement of legacy counting practices rather than exploratory research spending.
Quick Stats for Digital Colony Counter Market
- Digital Colony Counter Market Value (2026): USD 310.0 million
- Digital Colony Counter Market Forecast Value (2036): USD 657.0 million
- Digital Colony Counter Market Forecast CAGR 2026 to 2036: 7.8%
- Leading Automation Level by Demand Share: Semi-Automated Image Counters
- Fastest-Growing Countries: India, China, Vietnam, Indonesia, Mexico
- Top Players in Global Demand: Interscience, IUL Instruments, Synbiosis, Schuett-Biotec, Don Whitley Scientific, METER Group, Apogee Instruments, Neutec Group, Labtron Equipment
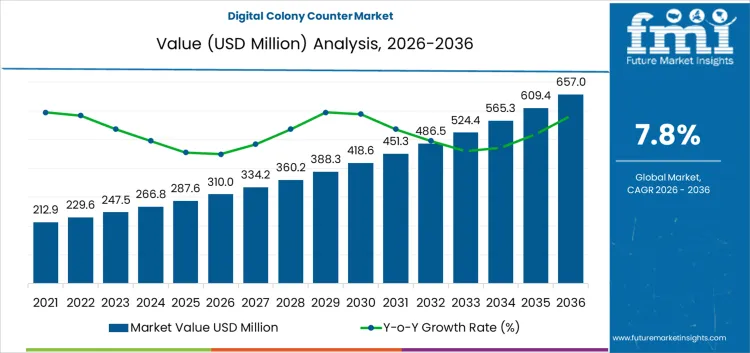
Digital Colony Counter Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 310.0 million |
| Market Forecast Value (2036) | USD 657.0 million |
| Forecast CAGR 2026 to 2036 | 7.8% |
Why is the Demand for Digital Colony Counter Growing?
Demand for digital colony counters is increasing as clinical laboratories, pharmaceutical quality control units, and food safety testing facilities require accurate and efficient enumeration of microbial colonies. Manual counting techniques are time consuming and subject to operator variability, and digital systems provide consistent image capture and algorithm-based quantification that improves reproducibility of results. Microbiology professionals specify digital colony counters that support high throughput workflows and integrate with laboratory information management systems to streamline data reporting and traceability. Regulatory expectations for documented test results in sterility testing, batch release protocols, and environmental monitoring further encourage adoption of automated solutions that generate audit-ready records.
Procurement teams evaluate device resolution, software analytics, user interface clarity, and service support to ensure systems meet laboratory throughput and compliance requirements. Growth in diagnostic testing, product quality programs, and research in microbiome and fermentation sciences reinforces uptake of digital colony counters that reduce turnaround time and human error. Academic and contract research laboratories require tools that support diverse plate formats and lighting conditions while maintaining sensitivity across a range of colony sizes and densities. Advances in imaging sensors and pattern recognition software improve discrimination of overlapping colonies and reduce need for repeat assays. Technical staff benefit from devices that offer customizable protocols and rapid calibration procedures that align with quality control standards. These application demands contribute to sustained growth in digital colony counter deployment across laboratory environments.
How Are the Segments Classified in the Digital Colony Counter Market?
Demand for digital colony counters is shaped by microbiological testing accuracy requirements, laboratory throughput targets, and compliance with standardized counting protocols. Adoption supports reduction of manual counting variability and improvement of documentation reliability. Usage spans quality control, diagnostics, and research environments requiring repeatable microbial enumeration. System selection prioritizes image resolution, counting consistency, and compatibility with common culture formats. Segment classification reflects differentiation by automation capability, laboratory end use, and plate format compatibility. Structure highlights how workflow intensity, regulatory oversight, and sample presentation influence specification priorities and deployment patterns across microbiology laboratories.
Which Automation Level Represents the Largest Share of Demand?
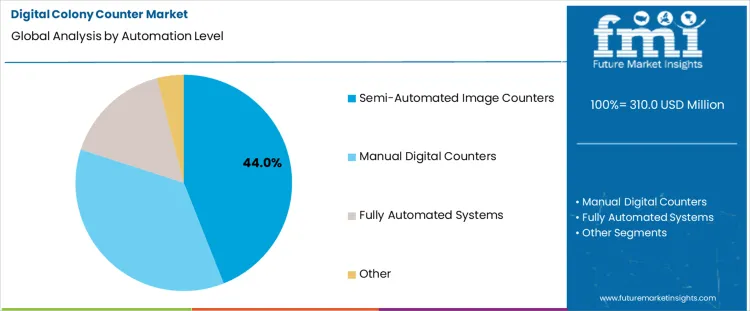
Semi-automated image counters hold 44.0%, representing the largest share among automation levels due to balance between accuracy and operator control. These systems combine digital imaging with user validation to improve counting consistency. Laboratories retain oversight while reducing fatigue and subjective error associated with manual counting. Integration supports moderate sample throughput without complexity of full automation. Manual digital counters remain relevant for low-volume testing environments. Fully automated systems address high-throughput laboratories with higher capital requirements. Automation segmentation reflects preference for hybrid solutions delivering efficiency gains while maintaining procedural flexibility.
Key Points
- Semi-automated systems balance accuracy with operator oversight.
- Digital imaging reduces variability in colony enumeration.
- Other automation levels address low-volume or high-throughput needs.
Which End Use Accounts for the Highest Share of Demand?
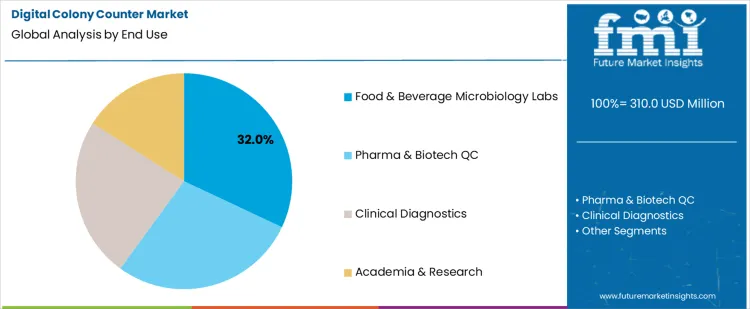
Food and beverage microbiology labs hold 32.0%, representing the largest share among end-use settings due to routine safety testing requirements. These laboratories perform frequent microbial counts to verify hygiene and regulatory compliance. High sample volumes increase need for consistent and traceable results. Digital counters support standardized reporting and audit readiness. Pharmaceutical and biotech quality control labs emphasize validation and precision. Clinical diagnostics apply counting for infection monitoring. Academic research uses systems for experimental analysis. End-use segmentation reflects concentration of demand where routine testing frequency and compliance obligations remain highest.
Key Points
- Food safety testing requires frequent microbial enumeration.
- Digital counters support compliance documentation needs.
- Other sectors apply systems for quality control and diagnostics.
Which Plate Format Support Category Drives the Largest Share of Demand?
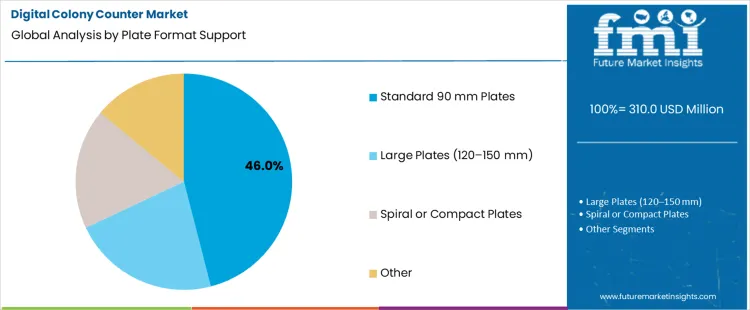
Standard 90 mm plates hold 46.0%, representing the largest share among plate format support categories due to widespread laboratory use. This plate size aligns with common culture protocols across food, pharmaceutical, and clinical laboratories. Equipment compatibility with standard plates simplifies integration into existing workflows. Imaging calibration and counting algorithms are optimized for this format. Larger plates support specialized applications with lower usage frequency. Spiral and compact plates address space-efficient or automated plating methods. Plate format segmentation reflects dominance of standard culture practices shaping equipment specification priorities.
Key Points
- Standard plates dominate routine microbiological testing.
- Broad compatibility simplifies laboratory workflow integration.
- Other formats support specialized or space-efficient protocols.
What are the Key Dynamics in the Digital Colony Counter Market?
Demand for digital colony counters reflects laboratory need for accurate, repeatable quantification of microbial colonies under regulated testing conditions. Adoption concentrates within clinical diagnostics, pharmaceutical quality control, food safety testing, environmental monitoring, and academic research laboratories. Global scope aligns with standardization of microbiological analysis and reduction of manual counting variability. Usage centers on image-based systems combining optical hardware and analytical software for plate-based colony enumeration.
How do laboratory standardization and accuracy requirements shape adoption?
Microbiological testing relies on precise colony counts to support contamination assessment, sterility testing, and regulatory reporting. Demand increases as laboratories seek to reduce operator-dependent variability inherent in manual counting. Digital systems enable consistent detection across varying colony sizes, colors, and densities. Pharmaceutical and food testing laboratories adopt automated counting to support compliance with validated test protocols and audit readiness. Image archiving supports result traceability and repeat analysis. High-throughput environments value faster processing of multiple plates without compromising accuracy. Integration with laboratory information management systems improves data integrity. Adoption reflects operational need for reproducible results under quality-controlled laboratory workflows.
How does validation burden, cost sensitivity, and technical limits influence market scalability?
Digital colony counters require validation against established microbiological methods, increasing implementation effort. Demand sensitivity rises where equipment cost exceeds manual alternatives in low-volume laboratories. Performance varies with plate quality, lighting conditions, and colony morphology complexity. Software algorithms require tuning to avoid miscounting merged or irregular colonies. Maintenance of optical components affects long-term accuracy. Regulatory environments require documented method equivalence, extending adoption timelines. Training needs persist despite automation. Supply chain dependence on imaging sensors and software updates constrains scalable deployment across budget-constrained laboratory networks.
How Is Demand for Digital Colony Counters Evolving Globally?
Demand for digital colony counters is expanding globally due to laboratory automation needs, accuracy requirements, and higher microbiological testing volumes. Adoption aligns with pharmaceutical quality control, food safety testing, water analysis, and academic research workflows requiring repeatable enumeration and audit trails. Transition from manual counting reduces operator variability and improves throughput under regulatory scrutiny. Growth rates in India at 9.6%, China at 8.9%, Vietnam at 8.2%, Indonesia at 7.9%, and Mexico at 7.1% indicate sustained expansion driven by laboratory capacity additions, compliance intensity, and lifecycle replacement of manual and semi-automated systems.
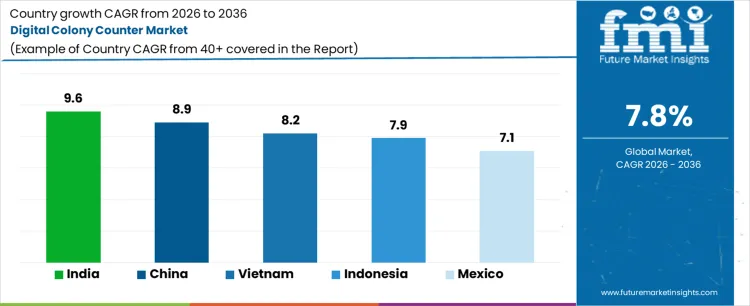
| Country | CAGR (%) |
|---|---|
| India | 9.6% |
| China | 8.9% |
| Vietnam | 8.2% |
| Indonesia | 7.9% |
| Mexico | 7.1% |
What Is Driving Strong Growth of Digital Colony Counter Demand in India?
Digital colony counter demand in India is growing at a CAGR of 9.6%, supported by expansion of pharmaceutical manufacturing, diagnostics, and food testing laboratories. Regulatory compliance requires accurate microbial enumeration for batch release and surveillance. Contract research and testing organizations scale throughput, favoring automated counting to reduce analyst time. Public health laboratories increase microbiology testing volumes for water and clinical screening. Academic research funding sustains adoption across universities. Demand growth reflects capacity expansion and compliance-driven accuracy needs rather than discretionary laboratory upgrades.
- Pharmaceutical quality control expansion
- Higher testing volumes in diagnostics and food safety
- CRO laboratory throughput optimization
- Academic and public health laboratory adoption
Why Is China Sustaining Elevated Demand for Digital Colony Counters?
China is expanding at a CAGR of 8.9%, driven by large-scale pharmaceutical production and food safety oversight. Manufacturing quality systems emphasize traceable, repeatable microbial counts. Centralized laboratories adopt digital imaging to standardize results across sites. Growth in biotechnology research increases plate-based assays requiring rapid enumeration. Domestic instrument manufacturing supports availability and service coverage. Demand growth reflects testing standardization and industrial scale rather than short-term research spending cycles.
- Pharmaceutical manufacturing quality standardization
- Food safety surveillance intensity
- Biotechnology research assay growth
- Strong domestic instrument supply and service
How Is Vietnam Influencing Growth of Digital Colony Counter Adoption?
Digital colony counter demand in Vietnam is growing at a CAGR of 8.2%, shaped by expansion of food processing, export compliance testing, and pharmaceutical packaging operations. Laboratories serving seafood, agriculture, and beverage exporters require validated microbial counts. Automation reduces training burden and improves consistency in growing lab networks. University laboratories increase microbiology programs supporting skilled workforce development. Demand growth reflects export compliance requirements and laboratory network expansion rather than replacement of mature installed bases.
- Export food testing compliance requirements
- Expansion of pharmaceutical packaging quality labs
- Consistency needs across growing lab networks
- Academic microbiology program growth
What Factors Are Supporting Digital Colony Counter Demand in Indonesia?
Indonesia is expanding at a CAGR of 7.9%, influenced by food safety monitoring and water quality testing. Tropical conditions increase microbial testing frequency across processing facilities. Public laboratories adopt digital tools to improve accuracy and reporting speed. Private testing services scale operations for industrial clients. Budget sensitivity favors compact, reliable systems with low maintenance needs. Demand growth reflects testing volume increases and operational efficiency requirements rather than advanced research intensity.
- Increased microbial testing under tropical conditions
- Public laboratory modernization initiatives
- Growth of private testing service providers
- Preference for cost-efficient, reliable instruments
Why Is Mexico Experiencing Steady Expansion in Digital Colony Counter Demand?
Digital colony counter demand in Mexico is growing at a CAGR of 7.1%, supported by export-oriented food manufacturing and pharmaceutical quality control. Laboratories serving North American supply chains require standardized microbial enumeration. Automation supports compliance documentation and audit readiness. University research labs contribute incremental demand through teaching and applied research. Stable industrial testing volumes sustain predictable procurement cycles. Growth reflects export compliance and quality assurance needs rather than rapid laboratory sector expansion.
- Export food and pharma compliance testing
- Standardization for North American supply chains
- Audit-ready laboratory documentation needs
- Stable demand from academic research laboratories
What is the competitive landscape of demand for the digital colony counter market globally?
Demand for digital colony counters is driven by microbiology testing in clinical diagnostics, food safety, pharmaceutical quality control, environmental monitoring, and academic research. Buyers evaluate counting accuracy, ease of use, imaging resolution, automation capability, compatibility with Petri dish formats, and analytical software integration. Procurement teams prioritize suppliers with validated performance data, global service support, regulatory compliance for laboratory equipment, and strong user interface design. Trend in the global market reflects increased adoption of automated imaging, AI-assisted colony recognition, and integration with laboratory information management systems to reduce manual error and improve throughput.
Interscience holds leading positioning through extensive digital colony counter systems and imaging solutions widely used in food, pharmaceutical, and research laboratories supported by global distribution. IUL Instruments supports demand with high-resolution imaging counters and intuitive software for accurate enumeration across diverse microbiology applications. Synbiosis participates with colony counters and automated imaging platforms designed for high throughput and flexible analysis. Don Whitley Scientific contributes digital counting systems and incubation-integrated solutions for controlled microbial assessment. Schuett-Biotec supplies compact and modular digital colony counters serving quality control and academic laboratories. Competitive differentiation depends on image processing accuracy, software sophistication, throughput capability, ease of integration, and ability to meet diverse laboratory requirements.
Key Players in the Digital Colony Counter Market
- Interscience
- IUL Instruments
- Synbiosis
- Schuett-Biotec
- Don Whitley Scientific
- METER Group
- Apogee Instruments
- Neutec Group
- Labtron Equipment
- Regional Manufacturers
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Automation Level | Semi-Automated Image Counters; Manual Digital Counters; Fully Automated Systems; Other |
| End Use | Food & Beverage Microbiology Labs; Pharma & Biotech QC; Clinical Diagnostics; Academia & Research |
| Plate Format Support | Standard 90 mm Plates; Large Plates (120-150 mm); Spiral or Compact Plates; Other |
| Sales Channel | Direct Manufacturer Sales; Laboratory Distributors; Bundled with Microbiology Workflows; Other |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Vietnam, Indonesia, Mexico, and 40+ countries |
| Key Companies Profiled | Interscience; IUL Instruments; Synbiosis; Schuett-Biotec; Don Whitley Scientific; METER Group; Apogee Instruments; Neutec Group; Labtron Equipment; Regional Manufacturers |
| Additional Attributes | Dollar sales by automation level and end use; adoption trends for fully automated image analysis supporting higher throughput and reduced operator variability; counting accuracy, reproducibility, and detection sensitivity performance metrics; compatibility with chromogenic media and diverse colony morphologies; software capabilities for data storage, audit trails, and regulatory documentation; workflow integration with incubators and spiral platers; calibration, maintenance, and total cost of ownership considerations; compliance with laboratory quality standards and validation requirements influencing digital colony counter selection. |
Digital Colony Counter Market by Segment
Automation Level:
- Semi-Automated Image Counters
- Manual Digital Counters
- Fully Automated Systems
- Other
End Use:
- Food & Beverage Microbiology Labs
- Pharma & Biotech QC
- Clinical Diagnostics
- Academia & Research
Plate Format Support:
- Standard 90 mm Plates
- Large Plates (120-150 mm)
- Spiral or Compact Plates
- Other
Sales Channel:
- Direct Manufacturer Sales
- Laboratory Distributors
- Bundled with Microbiology Workflows
- Other
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- International Organization for Standardization. (2023). ISO 7218: Microbiology of the food chain-General requirements and guidance for microbiological examinations, including colony counting methods. International Organization for Standardization.
- International Organization for Standardization. (2023). ISO 11133: Microbiology of food, animal feed, and water-Preparation, production, storage, and performance testing of culture media used for colony enumeration. International Organization for Standardization.
- United States Food and Drug Administration. (2024). Bacteriological Analytical Manual (BAM): Enumeration of microorganisms and use of automated and digital colony counting systems. USA Food and Drug Administration.
- European Pharmacopoeia Commission. (2023). European Pharmacopoeia, Chapter 2.6.12 and 2.6.13: Microbiological examination and colony counting requirements for pharmaceutical quality control. Council of Europe.
- United States Pharmacopeial Convention. (2024). USP <61>, <62>, and <1223>: Microbiological examination, validation of alternative microbiological methods, and automated colony counting guidance. United States Pharmacopeial Convention.
- World Health Organization. (2023). Quality assurance of microbiological testing laboratories: Data integrity, automated analysis, and result traceability. World Health Organization.
Frequently Asked Questions
How big is the digital colony counter market in 2026?
The global digital colony counter market is estimated to be valued at USD 310.0 million in 2026.
What will be the size of digital colony counter market in 2036?
The market size for the digital colony counter market is projected to reach USD 657.0 million by 2036.
How much will be the digital colony counter market growth between 2026 and 2036?
The digital colony counter market is expected to grow at a 7.8% CAGR between 2026 and 2036.
What are the key product types in the digital colony counter market?
The key product types in digital colony counter market are semi-automated image counters, manual digital counters, fully automated systems and other.
Which end use segment to contribute significant share in the digital colony counter market in 2026?
In terms of end use, food & beverage microbiology labs segment to command 32.0% share in the digital colony counter market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Automation Level
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Automation Level , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Automation Level , 2026 to 2036
- Semi-Automated Image Counters
- Manual Digital Counters
- Fully Automated Systems
- Other
- Semi-Automated Image Counters
- Y to o to Y Growth Trend Analysis By Automation Level , 2021 to 2025
- Absolute $ Opportunity Analysis By Automation Level , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Food & Beverage Microbiology Labs
- Pharma & Biotech QC
- Clinical Diagnostics
- Academia & Research
- Food & Beverage Microbiology Labs
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Plate Format Support
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Plate Format Support, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Plate Format Support, 2026 to 2036
- Standard 90 mm Plates
- Large Plates (120-150 mm)
- Spiral or Compact Plates
- Other
- Standard 90 mm Plates
- Y to o to Y Growth Trend Analysis By Plate Format Support, 2021 to 2025
- Absolute $ Opportunity Analysis By Plate Format Support, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sales Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sales Channel, 2026 to 2036
- Direct Manufacturer Sales
- Laboratory Distributors
- Bundled with Microbiology Workflows
- Other
- Direct Manufacturer Sales
- Y to o to Y Growth Trend Analysis By Sales Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Automation Level
- By End Use
- By Plate Format Support
- By Sales Channel
- Competition Analysis
- Competition Deep Dive
- Interscience
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- IUL Instruments
- Synbiosis
- Schuett-Biotec
- Don Whitley Scientific
- METER Group
- Apogee Instruments
- Neutec Group
- Labtron Equipment
- Regional Manufacturers
- Interscience
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Plate Format Support, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Plate Format Support, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Plate Format Support, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Plate Format Support, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Plate Format Support, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Plate Format Support, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Plate Format Support, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Plate Format Support, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Automation Level , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Automation Level
- Figure 6: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value Share and BPS Analysis by Plate Format Support, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Plate Format Support, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Plate Format Support
- Figure 12: Global Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Sales Channel, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Sales Channel
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Automation Level , 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Automation Level
- Figure 29: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End Use
- Figure 32: North America Market Value Share and BPS Analysis by Plate Format Support, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Plate Format Support, 2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Plate Format Support
- Figure 35: North America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Sales Channel, 2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Sales Channel
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Automation Level , 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by Automation Level
- Figure 42: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 44: Latin America Market Attractiveness Analysis by End Use
- Figure 45: Latin America Market Value Share and BPS Analysis by Plate Format Support, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Plate Format Support, 2026 to 2036
- Figure 47: Latin America Market Attractiveness Analysis by Plate Format Support
- Figure 48: Latin America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Sales Channel, 2026 to 2036
- Figure 50: Latin America Market Attractiveness Analysis by Sales Channel
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Automation Level , 2026 to 2036
- Figure 54: Western Europe Market Attractiveness Analysis by Automation Level
- Figure 55: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 57: Western Europe Market Attractiveness Analysis by End Use
- Figure 58: Western Europe Market Value Share and BPS Analysis by Plate Format Support, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Plate Format Support, 2026 to 2036
- Figure 60: Western Europe Market Attractiveness Analysis by Plate Format Support
- Figure 61: Western Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026 to 2036
- Figure 63: Western Europe Market Attractiveness Analysis by Sales Channel
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Automation Level , 2026 to 2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Automation Level
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Plate Format Support, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Plate Format Support, 2026 to 2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Plate Format Support
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026 to 2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Sales Channel
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Automation Level , 2026 to 2036
- Figure 80: East Asia Market Attractiveness Analysis by Automation Level
- Figure 81: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 83: East Asia Market Attractiveness Analysis by End Use
- Figure 84: East Asia Market Value Share and BPS Analysis by Plate Format Support, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Plate Format Support, 2026 to 2036
- Figure 86: East Asia Market Attractiveness Analysis by Plate Format Support
- Figure 87: East Asia Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Sales Channel, 2026 to 2036
- Figure 89: East Asia Market Attractiveness Analysis by Sales Channel
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Automation Level , 2026 to 2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Automation Level
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Plate Format Support, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Plate Format Support, 2026 to 2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Plate Format Support
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Sales Channel, 2026 to 2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Sales Channel
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Automation Level , 2026 to 2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Automation Level
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Plate Format Support, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Plate Format Support, 2026 to 2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Plate Format Support
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Sales Channel, 2026 to 2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Sales Channel
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis

