Cleanroom traffic management places the dynamic pass box market at USD 500 million in 2026, with installed programs carrying demand to USD 1,130.5 million by 2036 at an 8.5% CAGR. Facilities adopt these units to control material transfer between classified zones without disrupting pressure cascades. Validation teams define airflow class, interlock logic, and decontamination method before any layout drawing closes. Once a room qualifies, equipment models remain fixed inside protocols and training files. Operators judge units by purge recovery time, filter access, and alarm behavior. Purchasing checks documentation depth and service reach. Installation planning coordinates wall thickness, utilities, and commissioning tests.
Business in this category follows validation schedules, retrofit cycles, and expansion of controlled manufacturing areas. Engineering groups specify stainless grades, glazing, and gasket systems to match cleaning regimes. Assembly work focuses on leak integrity, door alignment, and fan stability. Factory release depends on airflow mapping and interlock verification. Integrators influence selection through room packages rather than standalone units. Service teams handle filter change programs and periodic requalification support. Spare parts list emphasizes fans, sensors, and control boards. Warranty exposure guides conservative design margins. Profit formation follows build quality, documentation discipline, and predictable field service workload rather than catalog breadth or price promotions.
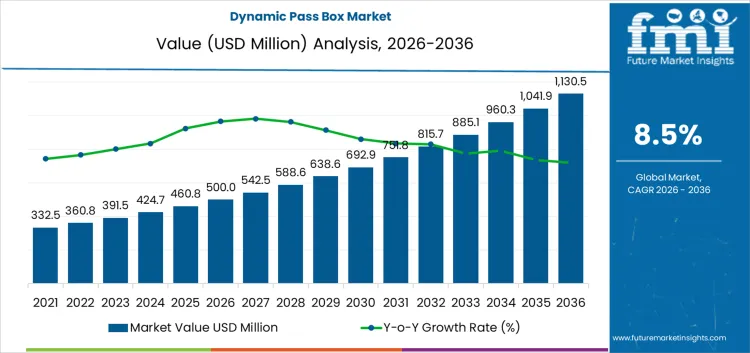
Facility layout discipline and contamination control protocols set the pace for the dynamic pass box market more than overall cleanroom construction volume. In 2026, at about USD 500 million, demand is concentrated in pharmaceutical manufacturing, biotechnology plants, and medical device facilities where material transfer rules are defined in validation documents. Equipment selection is locked during plant design or major retrofit programs, which ties purchasing to capital project timing rather than to routine maintenance budgets. Adoption rises as production lines become more segmented and as audit requirements place greater weight on controlled transfer steps. The upward path reflects wider use across qualified rooms and process stages rather than rapid replacement of existing hardware.
Compliance burden and service support shape the later phase of the dynamic pass box market. As value approaches roughly USD 1,130.5 million by 2036, buyers focus on airflow stability, interlock reliability, and ease of cleaning validation. Product ranges expand toward larger formats and higher throughput configurations. Manufacturing effort increases as documentation, factory testing, and site acceptance procedures become more demanding. The move from the five hundred million range into the low one point one billions reflects accumulated additions across many production sites rather than a single investment cycle. Suppliers that combine installation competence, predictable performance, and responsive service networks tend to secure repeat positions in regulated facilities.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 500 million |
| Forecast Value (2036) | USD 1,130.5 million |
| Forecast CAGR 2026 to 2036 | 8.5% |
Dynamic pass boxes are used to transfer materials between rooms of different cleanliness levels while maintaining pressure balance and contamination control. They include built in air handling, filtration, and interlocking doors to prevent cross flow during loading and unloading. Earlier cleanroom layouts depended on simple pass through cabinets, which could not control particles or pressure disturbance during frequent transfers. Dynamic designs address this by creating a short, controlled purge cycle inside the chamber. Pharmaceutical plants, biotech facilities, and electronics fabs specify these units by air change rate, filter grade, leakage limits, and door cycle reliability. Placement is tied to process flow, not corridor convenience. Engineers also consider cleanability, surface finish, and alarm integration because these units become part of routine material movement and audit inspection paths across daily operations.
Specification and purchasing decisions are led by quality, validation, and facility engineering teams rather than by general equipment buyers. Earlier installations often varied by room, which increased spare parts range and validation effort. Current projects aim to standardize chamber sizes, control logic, and filter formats across entire sites. Manufacturers compete on airflow stability, control system reliability, and documentation packages instead of cabinet thickness alone. Users evaluate total cost through downtime risk, filter replacement intervals, and cleaning labor, not through purchase price. Integrators add value through installation, pressure balancing, and site acceptance testing. Over time, demand follows growth in regulated production capacity and tighter segregation of process steps, where predictable material transfer becomes a routine control point rather than a special handling exception within audited facilities and daily batch operations.
Specification in Dynamic Pass Box Market is governed by contamination control strategy, room pressure zoning, and inspection protocols rather than by furniture or equipment replacement cycles. Pass boxes are defined during cleanroom layout approval and become part of the validated material flow concept. Once transfer paths, door logic, and filtration requirements are accepted, equipment choice is fixed within the facility design. This ties demand to new cleanroom builds, capacity expansions, and major requalification programs. Segment structure reflects differences in airflow management, control logic, and monitoring requirements. Buyers prioritize validation documentation, reliability of interlocks, and service access because transfer failures affect batch integrity and compliance status.

Standard dynamic pass boxes represent about 45% of demand in the Dynamic Pass Box Market because many facilities require controlled transfer with moderate airflow and straightforward operating logic. These units integrate into material flow paths without adding complex control systems. Pass boxes equipped with HEPA or ULPA filtration serve areas where particulate control during transfer is critical, which expands qualification testing and filter integrity monitoring. Interlocking system variants enforce procedural discipline by preventing simultaneous door opening, increasing control over pressure cascade integrity. Auto sensing door models introduce automation to reduce operator error, though they require additional sensor validation and maintenance protocols. Each type establishes a different level of documentation, testing frequency, and service planning.
From an operational risk perspective, type selection determines inspection burden and deviation handling. Basic units rely on procedural controls and routine cleaning, which simplifies maintenance but increases dependence on training. Filtered and interlocked systems impose periodic performance checks, airflow verification, and control logic testing. Auto sensing configurations add software and sensor calibration into the validation matrix. Once a facility is qualified with a specific configuration, changes are avoided because revalidation affects room classification and material flow approvals. Demand by type therefore follows cleanroom design standards and corporate quality policies rather than short term equipment purchasing decisions.

Pharmaceutical and biotech cleanrooms account for about 48% of demand in the Dynamic Pass Box Market because these facilities move high volumes of materials between classified areas under strict contamination control rules. This environment requires frequent transfers with documented control at each interface. Hospital and healthcare clean zones apply pass boxes to manage supplies between controlled and semi controlled spaces, where reliability and ease of cleaning are emphasized. Microelectronics and semiconductor cleanrooms require extremely low particle levels, which increases specification strictness and favors advanced filtration and interlocking. Research laboratories operate smaller systems with varied workflows, leading to more customized and lower volume installations.
Application mix also shapes procurement and qualification processes. Pharmaceutical manufacturers purchase through formal capital projects with extensive user requirement specifications and validation plans. Hospitals follow facility upgrade programs and infection control policies. Semiconductor plants integrate pass boxes into broader cleanroom automation and monitoring systems, extending approval cycles. Research organizations acquire equipment through project based funding with flexible specifications. The resulting demand structure concentrates volume in regulated life science facilities while technical rigor intensifies in microelectronics environments. Segment shares therefore move with cleanroom capacity expansion, regulatory inspection cycles, and laboratory infrastructure investment rather than with short term equipment replacement trends.
The Dynamic Pass Box Market is shaped by how pharmaceutical, biotechnology, and cleanroom operators control material transfer between rooms of different cleanliness classes. Pass boxes sit at critical interfaces where poor process discipline can compromise entire production areas. Buyers focus on air flow behavior, interlocking reliability, filtration performance, and ease of cleaning. Selection decisions involve quality assurance, validation, and facility engineering teams rather than only procurement. This positions dynamic pass boxes as part of contamination control architecture rather than as simple cabinets, making documentation quality, validation support, and integration with room layouts central to purchasing decisions.
Many facilities aim to standardize room layouts and material transfer practices across production lines and sites. This is changing expectations in the Dynamic Pass Box Market toward equipment that fits repeatable cleanroom modules and established operating procedures. Buyers prefer consistent door sizes, control interfaces, and service routines to reduce training and maintenance complexity. This also encourages selection of pass boxes that integrate smoothly with existing wall systems and monitoring practices. As a result, suppliers are evaluated on product consistency, documentation clarity, and long-term availability rather than on custom features for single installations.
Long-term demand is being built in facilities that expand production capacity in phases or add new process rooms over time. Each new cleanroom, packaging suite, or preparation area requires controlled transfer points, which creates repeat need for dynamic pass boxes. The Dynamic Pass Box Market also benefits from upgrades and revalidation projects, where older or simpler transfer cabinets are replaced with higher performance units. There is opportunity for suppliers that become part of standard facility design templates used by engineering contractors and pharmaceutical groups, securing repeat orders across multi-year build and expansion programs.
Each cleanroom project comes with its own layout constraints, user requirements, and validation plans. This makes it difficult to use exactly the same pass box design everywhere. The Dynamic Pass Box Market also faces long qualification cycles because equipment must be tested, documented, and approved as part of regulated production environments. Buyers are cautious about introducing new designs without proven references. In addition, budget control in facility projects often prioritizes core process equipment over transfer hardware. These factors slow wider standardization and keep purchasing decisions tied to project timelines and validation schedules.
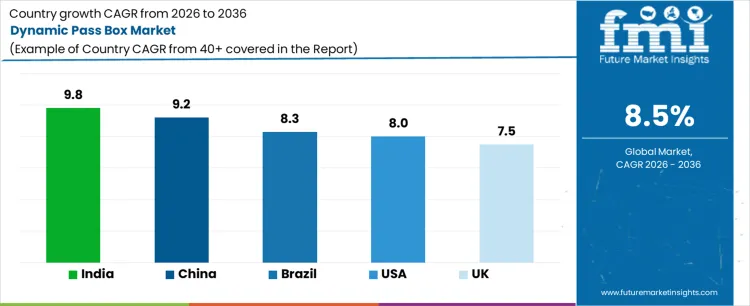
| Country | CAGR |
|---|---|
| USA | 8.0% |
| UK | 7.5% |
| China | 9.2% |
| India | 9.8% |
| Brazil | 8.3% |
Demand for dynamic pass boxes is rising as pharmaceutical, biotech, and cleanroom based manufacturing facilities tighten contamination control and material transfer protocols. India leads with a 9.8% CAGR, supported by expansion of drug manufacturing, vaccine production, and new cleanroom capacity in contract manufacturing facilities. China follows at 9.2%, driven by growth in biologics, medical device assembly, and regulated laboratory infrastructure. Brazil records 8.3%, reflecting gradual upgrades in hospital pharmacies and pharmaceutical plants. The USA grows at 8.0%, shaped by compliance driven replacement of older transfer systems and investment in high containment facilities. The UK, at 7.5%, reflects steady demand from mature but regulation intensive life sciences and healthcare manufacturing environments.
Cleanroom construction programs place the dynamic pass box market in the United States on an 8.0% CAGR path. Pharmaceutical sites, biotech plants, and hospital compounding units’ account for most installations. Engineering teams specify interlocked doors, airflow control, pressure cascades, and surface finishes. Procurement follows turnkey cleanroom projects rather than isolated equipment orders. Factory acceptance tests, smoke studies, and on site commissioning reports control release status. Maintenance planning covers filter integrity, seal condition, interlock timing, and control panel uptime. Replacement demand appears after audit findings related to leakage or door wear. Distributors coordinate regional service coverage and spare parts availability. Quality groups archive records for inspections and periodic requalification. Installation scheduling aligns with project milestones. Commercial success depends on specification inclusion and dependable service response across multi-site operators nationwide.
Framework purchasing structures keep the dynamic pass box market in the United Kingdom aligned with a 7.5% CAGR. Hospital pharmacies, aseptic suites, and biologics facilities drive most demand. Integrators manage audited equipment lists and service obligations. Design reviews emphasize pressure regimes, door interlocks, airflow patterns, and cleanability during operation. Site acceptance relies on smoke visualization, recovery time checks, and recorded particle counts. Maintenance contractors schedule filter changes, hinge inspection, interlock testing, and controller calibration. Replacement follows audit findings, impact damage, or persistent pressure imbalance. Distributors hold spares near major facility clusters. Documentation remains part of quality files supporting inspections and revalidation cycles. Installation work follows capital programs rather than local department requests. Commercial access depends on framework inclusion and reliable response times across managed estates.
Centralized construction programs place the dynamic pass box market in China on a 9.2% growth track. Pharmaceutical plants, device factories, and vaccine facilities deploy units across high throughput cleanrooms. Design institutes publish standard layouts covering pressure cascades, interlocks, finishes, and monitoring points. Procurement aggregates volumes through program packages and schedules deliveries with construction timelines. Domestic manufacturing supports capacity, spare parts supply, and coordinated commissioning. Factory tests verify airflow balance, recovery time, enclosure integrity, and controller functions. Quality teams record inspections and corrective actions inside project systems. Replacement follows audit findings related to leakage or unstable pressure. Regional warehouses stage spares and support synchronized handovers. Installation teams follow uniform procedures across provinces. Commercial success depends on inclusion within national programs and consistent execution across contractor networks.
Export driven pharmaceutical investment fixes the dynamic pass box market in India at a 9.8% CAGR. Biosimilar plants, formulation sites, and hospital compounding units generate steady installation volume. Project consultants define door interlocks, airflow recovery targets, finishes, and cleaning access. Procurement aligns purchases with turnkey cleanroom builds. Local fabricators integrate enclosures while control components arrive through qualified suppliers. Commissioning teams perform smoke tests, recovery measurements, and interlock verification. Maintenance focuses on filter replacement, gasket condition, hinge alignment, and controller reliability. Replacement demand rises after audit observations or mechanical damage. Distributors provide spares and installation support across clusters. Documentation supports inspections and customer audits. Scheduling follows construction milestones rather than departmental requests. Commercial continuity depends on approval lists and dependable service delivery across regulated manufacturing sites.
Regulatory driven facility upgrades keep the dynamic pass box market in Brazil near an 8.3% CAGR. Hospital pharmacies, contract manufacturers, and research laboratories represent the main installation base. Engineering firms specify pressure regimes, interlocks, finishes, and cleaning access during design. Procurement follows capital projects rather than ad hoc equipment requests. Import logistics influence lead times while local partners handle installation and commissioning. Acceptance relies on smoke visualization, recovery checks, and recorded particle counts. Maintenance plans cover filter replacement, seal condition, hinge wear, and controller diagnostics. Replacement follows audit findings, repeated alarms, or pressure stability issues. Distributors stock spares near major cities. Documentation remains part of quality records. Commercial results depend on approved lists and dependable field response within regulated facilities.

Dynamic pass box selection follows facility logistics design rather than equipment brand comparison. Planners model material routes, queue times, wipe down steps, and operator handling points before defining transfer equipment. AirClean Systems, GKE Corporation, Terra Universal, Cleanrooms International, Aeromech compete inside those layout decisions. AirClean Systems supplies catalog units suited to laboratory retrofits and modular walls. Terra Universal builds high specification systems for semiconductor and advanced life science environments. GKE Corporation focuses on pharmaceutical installations that require integrated monitoring and documented airflow performance. Cleanrooms International links pass boxes with full room build contracts and wall systems. Aeromech serves industrial clean spaces where durability and access for maintenance matter more than finish detail. Once layouts receive approval, airflow tests, procedures, and training materials fix the equipment choice for extended operating periods.
Procurement behavior reflects validation effort, maintenance workload, and construction sequencing rather than unit price comparisons. Engineering teams review recovery time, interlock behavior, surface finish, filter access, and cleaning ergonomics during technical evaluation. Project managers favor suppliers that deliver frames and doors aligned with wall schedules and trade coordination. Maintenance groups prefer designs with predictable gasket replacement and simple filter changes. AirClean Systems benefits from repeat orders in regulated laboratory networks. Terra Universal holds positions where documentation depth and performance testing govern acceptance. GKE Corporation relies on pharmaceutical references and regional service coverage. Cleanrooms International secures work through integrated build scope. Aeromech competes in facilities that prioritize mechanical robustness. Market outcomes differ by contamination class, build strategy, and owner operating discipline.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Type | Standard dynamic pass boxes, Dynamic pass boxes with HEPA/ULPA filtration, Dynamic pass boxes with interlocking systems, Dynamic pass boxes with auto-sensing doors |
| Application | Pharmaceutical & biotech cleanrooms, Hospital and healthcare clean zones, Microelectronics & semiconductor cleanrooms, Research laboratories |
| Region | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | AirClean Systems, GKE Corporation, Terra Universal, Cleanrooms International, Aeromech |
| Additional Attributes | Dollar by sales by type and application; demand driven by cleanroom expansion and validation programs; purchasing governed by airflow class, interlock logic, and documentation; selection shaped by installation integration, filter access, recovery time, and long-term service support rather than unit price. |
How big is the dynamic pass box market in 2026?
The global dynamic pass box market is estimated to be valued at USD 500.0 million in 2026.
What will be the size of dynamic pass box market in 2036?
The market size for the dynamic pass box market is projected to reach USD 1,130.5 million by 2036.
How much will be the dynamic pass box market growth between 2026 and 2036?
The dynamic pass box market is expected to grow at a 8.5% CAGR between 2026 and 2036.
What are the key product types in the dynamic pass box market?
The key product types in dynamic pass box market are standard dynamic pass boxes , dynamic pass boxes with hepa/ulpa filtration, dynamic pass boxes with interlocking systems and dynamic pass boxes with auto‑sensing doors.
Which application segment to contribute significant share in the dynamic pass box market in 2026?
In terms of application, pharmaceutical & biotech cleanrooms segment to command 48.0% share in the dynamic pass box market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.