The global endoluminal suturing device market is forecasted to reach USD 85.2 million in 2026 and expand to USD 158.7 million by 2036, advancing at a CAGR of 6.4%. FMI opines that this trajectory reflects a structural shift in gastrointestinal and bariatric interventions, where clinicians are increasingly prioritizing incisionless, tissue-preserving procedures over open or laparoscopic surgery. Demand is being shaped by rising procedure volumes in obesity management, GERD treatment, and defect closure following advanced endoscopic resections.
Two converging forces are expanding market adoption. First, the global rise in obesity and reflux-related disorders is accelerating demand for minimally invasive endoscopic therapies that reduce recovery time and procedural risk. Second, hospitals are under pressure to lower surgical costs and shorten inpatient stays, favoring endoluminal solutions that can be performed in outpatient or ambulatory settings. This dual pressure is positioning endoluminal suturing as a viable alternative to conventional surgical suturing in selected indications.
The value proposition of these devices is evolving from simple tissue approximation toward durable anatomical modification. Next-generation platforms are improving suture strength, precision, and maneuverability within confined luminal spaces. Clinical preference is shifting toward systems that enable full-thickness suturing with consistent tension control, supporting applications such as endoscopic sleeve gastroplasty and post-procedural defect management. Ongoing innovation is focused on ergonomics, reduced learning curves, and compatibility with flexible endoscopy workflows.
Commercial scalability depends on clinical validation and reimbursement alignment. In the US, growing procedural acceptance and favorable reimbursement pathways for therapeutic endoscopy are supporting adoption. European markets remain more conservative, with slower uptake driven by stringent evidence requirements and training availability. As endoscopic therapy continues to replace select surgical interventions, endoluminal suturing devices are expected to gain a stronger foothold within advanced GI treatment algorithms.
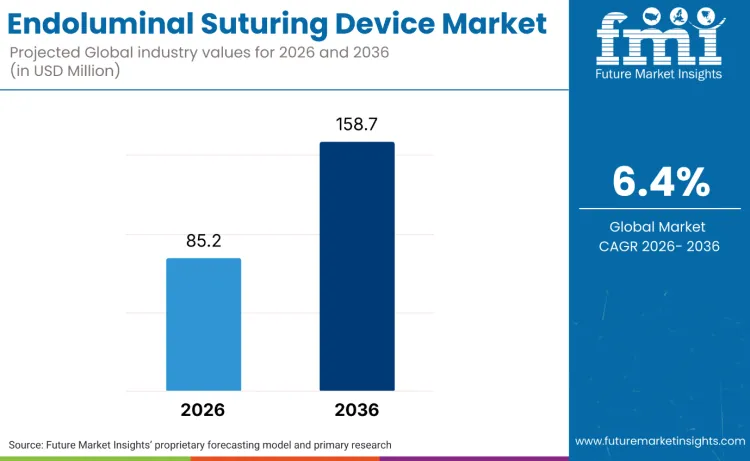
| Metric | Value |
|---|---|
| Market Value (2026) | USD 85.2 million |
| Market Forecast Value (2036) | USD 158.7 million |
| Forecast CAGR (2026 to 2036) | 6.4% |
Endoluminal suturing devices are regulated as medical devices and must meet defined certification and approval requirements before clinical use. In the United States, these systems are generally classified as Class II medical devices. Manufacturers must obtain FDA clearance through the 510(k) pathway by demonstrating substantial equivalence to an already marketed device. Compliance with quality system regulations covering design controls, manufacturing processes, labeling, risk management, and post market surveillance is mandatory. Devices must also follow unique device identification and adverse event reporting requirements.
In the European Union, certification is obtained through CE marking under the Medical Device Regulation. These devices are typically categorized as Class IIa or IIb, depending on intended use and duration of contact. Manufacturers must maintain ISO 13485 compliant quality management systems, prepare detailed technical documentation, conduct clinical evaluations, and implement post market surveillance plans. Approval involves review by a notified body before commercial distribution across EU member states.
Endoluminal suturing devices play a critical role in delivering tissue approximation, surgical precision, and patient safety across gastrointestinal procedures, bariatric surgery, and GERD treatment applications. Adoption is influenced by procedural requirements, cost considerations, device compatibility, and clinical performance criteria. Segmentation by application type, end-use facility, and technology type highlights how medical professionals select specific suturing solutions to meet surgical standards, cost targets, and patient outcomes across diverse clinical procedures.
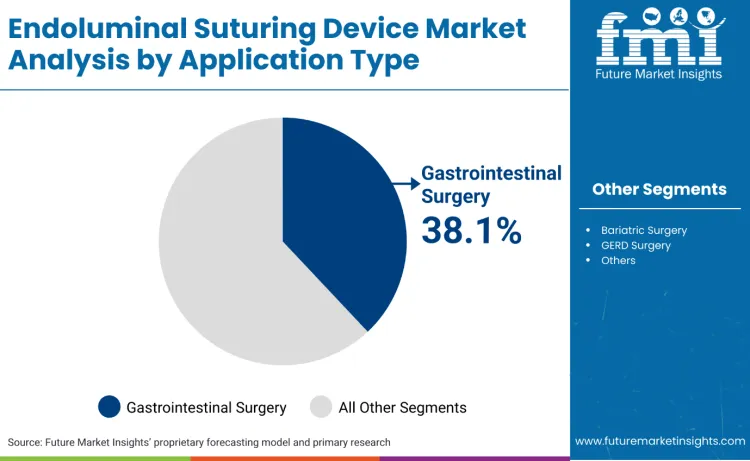
Gastrointestinal surgery accounts for 38.1%, driven by extensive use in bowel resections, perforation repairs, and endoscopic procedures. Bariatric surgery holds 29.4%, supporting weight loss procedures, gastric bypass operations, and revision surgeries. GERD surgery represents 21.7%, favored for fundoplication procedures, reflux treatment, and esophageal interventions. Other applications contribute 10.8%, used in specialized procedures and emerging clinical applications.
Key Points:
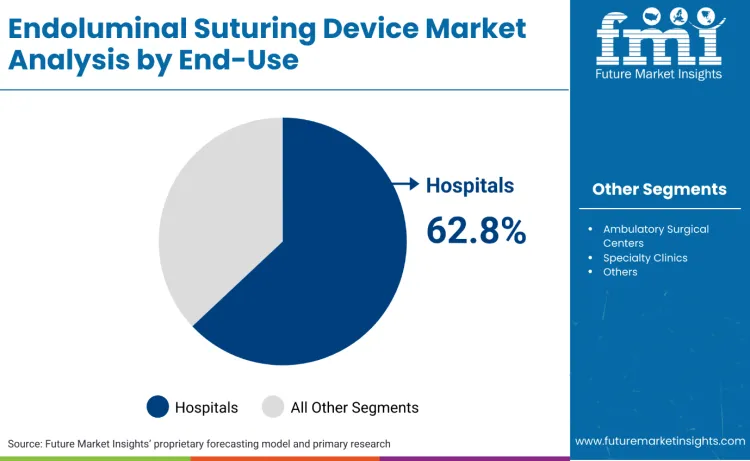
Hospitals represent 62.8%, reflecting priority on comprehensive surgical capabilities and advanced procedure volumes. Ambulatory surgical centers account for 28.3%, suitable for outpatient procedures requiring efficient device utilization. Specialty clinics hold 6.4%, used for focused procedures and targeted patient populations. Other facilities contribute 2.5%.
Key Points:
Based on Future Market Insights’ analysis, pre to 2026, the drug-drug interaction (DDI) testing and management market was largely reactive, focused on retrospective analysis within clinical settings. Healthcare systems relied on static, rule-based software embedded in electronic health records that often missed complex interactions and generated high alert fatigue. The lack of dynamic predictive disease analytics limited real-world utility and clinician trust.
In 2026, regulatory bodies and standards organizations began mandating AI-augmented DDI assessment in clinical decision support systems. This regulatory evolution removed ambiguity around acceptable algorithmic methods and required transparent validation of predictive models. As a result, DDI solutions shifted from basic flags to clinically validated, risk-stratified insights that meaningfully influence prescribing behavior and patient safety.
Consumer and clinician engagement has shifted dramatically. Traditional DDI alerts competed on breadth of coverage, generating high false positives that desensitized users. From 2026 to 2036, real-time, context-aware DDI platforms integrate with digital health records and patient-facing apps, delivering personalized risk scores and actionable guidance. These systems use patient genetics, comorbidities, and longitudinal medication data to tailor recommendations, transforming DDI outputs into trusted clinical touchpoints rather than generic warnings.
The manufacturing and development paradigm is equally significant. Previously, static rule libraries dominated product development, with long update cycles and limited responsiveness to emerging evidence. In 2026 to 2036, modular AI-driven pipelines enable continuous learning and real-world evidence integration. Lightweight model updates and agile deployment accelerate innovation and ensure solutions stay current with new drugs and interaction pathways.
Global demand for endoluminal suturing devices is increasing as healthcare facilities expand advanced 3D surgical models while addressing patient safety, procedural efficiency, and minimally invasive surgery application requirements. Growth reflects rising use of advanced device designs, robotic integration systems, and precision-optimized specifications across gastrointestinal departments, bariatric centers, and surgical facilities. Device selection focuses on suturing precision, ergonomic efficiency, and clinical performance under various surgical scenarios. India records 7.8% CAGR, USA records 6.2% CAGR, Germany records 5.9% CAGR, Japan records 5.4% CAGR, and UK records 5.1% CAGR. Adoption remains driven by clinical requirements and surgical efficiency rather than volume expansion alone.
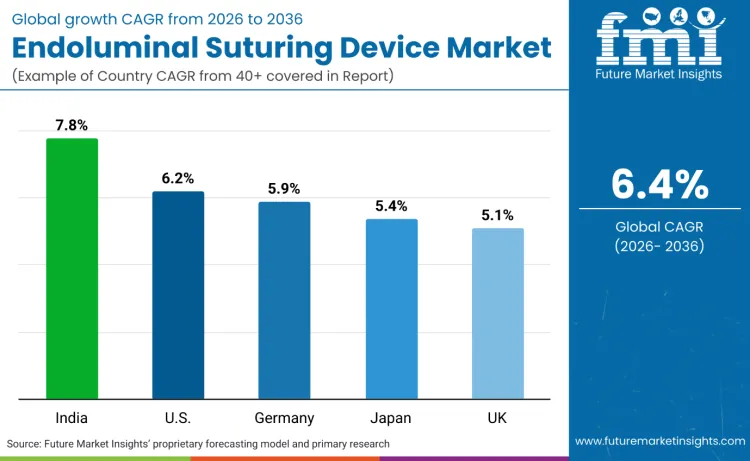
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 7.8% |
| USA | 6.2% |
| Germany | 5.9% |
| Japan | 5.4% |
| UK | 5.1% |
India is emerging as a global hub for cost-effective bariatric and gastrointestinal surgeries. A primary driver is the expansion of indigenous digital health initiatives in 2025 to 2026, which has facilitated the adoption of next-generation endoluminal tools in Tier-1 private hospital networks like Apollo and Manipal. These facilities are increasingly utilizing suturing devices for complex procedures such as Endoscopic Sleeve Gastroplasty (ESG) to attract international patients. Driven by this influx of medical tourism and rising domestic healthcare infrastructure, the Indian endoluminal suturing device market is set to grow at a 7.8% CAGR during the study period.
In the USA, the market is characterized by rapid technological convergence. A major driver is the 2025 FDA clearance of advanced robotic-assisted suturing modules that integrate with existing flexible endoscopes, allowing for higher precision in "hand-sewn" quality closures without open surgery. This is particularly relevant for the high volume of revision bariatric surgeries performed in the USA annually. Reflecting this shift toward high-tech, minimally invasive standards, the USA endoluminal suturing device market is projected to grow at a 6.2% CAGR during the study period.
Germany stands as Europe’s leader in endoscopic device manufacturing and adoption. A critical driver is the 2025 update to German surgical protocols by the DGVS (German Society for Digestive and Metabolic Diseases), which has increasingly recognized endoluminal suturing as a primary treatment for anastomotic leaks and fistulas. This has boosted the market for local innovators like Ovesco Endoscopy AG. Consequently, the German endoluminal suturing device market is expected to grow at a 5.9% CAGR during the study period, supported by a robust reimbursement framework (DRG system).
Japan’s market is driven by the unique needs of its geriatric population, where traditional open surgery poses high risks. A major development is the 2025 to 2026 surge in Endoscopic Submucosal Dissection (ESD) procedures for early-stage gastric cancer, which now utilize suturing devices for immediate defect closure to prevent post-operative bleeding. As the "Silver Economy" demands safer, faster-healing interventions, the Japanese endoluminal suturing device market is set to grow at a 5.4% CAGR during the study period.
The UK market is influenced by the urgent need to reduce the NHS surgical backlog. A key driver is the January 2026 NHS England "Day-Case" mandate, which encourages the use of endoluminal suturing for GERD and bariatric cases to move patients out of hospital beds faster. By shifting complex closures from the operating theater to the endoscopy suite, the NHS is significantly increasing procedural throughput. Supported by these public health mandates, the UK endoluminal suturing device market is set to grow at a 5.1% CAGR during the study period.
Quality assessment includes biocompatibility testing, mechanical performance, sterilization compatibility, and integration with existing surgical protocols. Procurement behavior reflects extensive clinical validation, supply reliability requirements, and reliance on suppliers offering technical support and consistent device specifications. Trends in the endoluminal suturing device market reflect emphasis on device quality and surgical efficiency in gastrointestinal procedures, bariatric surgery, and minimally invasive applications.
Apollo Endosurgery Inc. accounts for a leading position through integrated endoluminal device operations designed to deliver surgical precision, procedural reliability, and supply consistency capabilities. Johnson & Johnson (Ethicon) competes by combining advanced suturing technology with comprehensive surgical support systems supporting device performance across diverse surgical applications. Medtronic Plc supports demand through specialized surgical platforms aligned with precision and quality requirements. Cook Group Inc. maintains relevance by supplying advanced devices enhancing surgical protection and procedural performance potential. Boston Scientific Corporation participates with device solutions emphasizing cost efficiency and surgical integration. Competitive differentiation centers on device quality, supply reliability, clinical specifications, and depth of surgical collaboration.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Application Type | Gastrointestinal Surgery; Bariatric Surgery; GERD Surgery; Others |
| End-Use | Hospitals; Ambulatory Surgical Centers; Specialty Clinics; Others |
| Technology Type | Endoscopic Suturing Devices; Robotic-Assisted Systems; Manual Devices; Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, USA, Germany, Japan, UK, and 40+ countries |
| Key Companies Profiled | Apollo Endosurgery Inc.; Johnson & Johnson (Ethicon); Medtronic Plc; Cook Group Inc.; Boston Scientific Corporation; Others |
| Additional Attributes | Dollar sales by application type, end-use, and technology type; performance in surgical precision and cost efficiency across gastrointestinal procedures, bariatric surgery, and minimally invasive matrices; device quality improvement, surgical efficiency enhancement, and patient safety benefit under medical operations; impact on surgical outcomes, supply reliability, and cost management during clinical procedures; compatibility with surgical systems and quality control targets; procurement dynamics driven by medical facility expansion, quality improvement programs, and long-term device supply partnerships. |
How big is the endoluminal suturing device market in 2026?
The global endoluminal suturing device market is estimated to be valued at USD 85.2 million in 2026.
What will be the size of the endoluminal suturing device market in 2036?
The market size for the endoluminal suturing device market is projected to reach USD 158.7 million by 2036.
How much will the endoluminal suturing device market grow between 2026 and 2036?
The endoluminal suturing device market is expected to grow at a 6.4% CAGR between 2026 and 2036.
What are the key application types in the endoluminal suturing device market?
The key application types in the endoluminal suturing device market include gastrointestinal surgery, bariatric surgery, GERD surgery, and other specialized surgical applications.
Which application segment will contribute a significant share in the endoluminal suturing device market in 2026?
In terms of application type, the gastrointestinal surgery segment is set to command a 38.1% share in the endoluminal suturing device market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.