Demand for Endoluminal Suturing Device in UK
Endoluminal Suturing Devices Industry Analysis in UK Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Endoluminal Suturing Devices Industry Analysis in UK Forecast Outlook 2026 to 2036
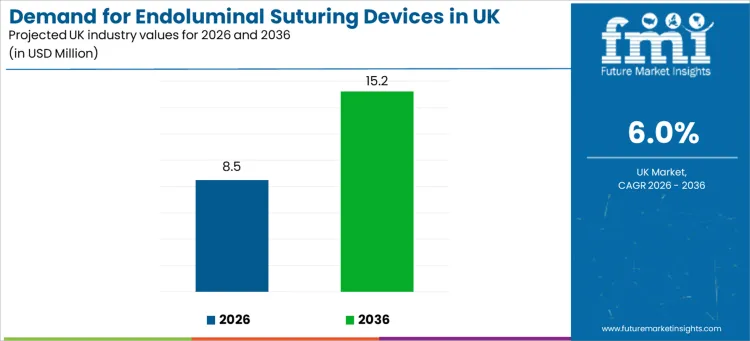
Demand for endoluminal suturing devices in the UK is projected at USD 8.5 million in 2026 and is expected to reach USD 15.2 million by 2036, expanding at a 6.0% CAGR. The day-to-day procedural planning decisions taken by hospital endoscopy units, bariatric service lines, gastroenterology teams, ambulatory surgery centers, and specialty clinics define this demand curve.
Endoluminal suturing sits inside clinical workflows where secure tissue approximation, closure repeatability, and predictable handling determine adoption. Buyers judge performance through closure integrity, procedural time impact, device usability in narrow anatomical spaces, and the ability to maintain consistent outcomes across operators. Clinical leaders prioritise systems that reduce unplanned follow-up interventions, support reliable closure in bariatric and gastrointestinal repairs, and fit smoothly into endoscopy room turnover expectations.
Quick Stats for Endoluminal Suturing Devices Demand in the UK
- Endoluminal Suturing Devices Valuation in the UK (2026): USD 8.5 million
- Endoluminal Suturing Devices Forecast Valuation in the UK (2036): USD 15.2 million
- Endoluminal Suturing Devices Forecast CAGR (2026-2036): 6.0%
- Leading Regional Growth: England (6.60%)
- Leading Product Type: Needle-based Suturing Devices (55.0% share)
- Leading Application: Bariatric (41.0% share)
- Leading End User: Hospitals (39.3% share)
- Key Companies Profiled: Boston Scientific Corporation; Medtronic; Olympus; Ovesco Endoscopy AG; Endo Robotics Co., LTD.
For health system decision-makers and technology service providers, the purchasing conversation rarely stays limited to the device alone. Choices connect to endoscopy tower readiness, accessory availability, staff training, and service support that keeps utilisation consistent. The ability to standardise technique and reduce variability across sites strengthens adoption across both large hospital networks and specialist centers.
Endoluminal Suturing Devices Industry in the UK Key Takeaways
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 8.5 million |
| Industry Forecast Value (2036) | USD 15.2 million |
| Forecast CAGR 2026 to 2036 | 6.0% |
Why is the UK Building Steady Demand for Endoluminal Suturing Devices?
Demand in the UK grows as clinical teams prioritise minimally invasive closure pathways that support faster recovery and reduce procedural trauma. Hospitals and specialty centers increasingly focus on tools that can strengthen safety confidence during bariatric interventions and gastrointestinal repairs, where closure stability directly influences patient outcomes.
Bariatric service activity remains an important anchor for adoption. NHS Digital publishes National Obesity Audit releases that include measures linked to NHS-funded bariatric surgical procedures delivered in England. This visibility reinforces the need for procedure-ready tools that clinicians can deploy with confidence, especially when teams want predictable closure performance and stable workflow execution.
Regulatory expectations also shape procurement discipline. UK government guidance notes strengthened post-market surveillance requirements for medical devices in Great Britain, with measures coming into force on 16 June 2025 to improve incident traceability and trend monitoring. This supports stronger attention on device documentation, training readiness, and supplier responsiveness.
Many endoscopy leadership teams align capability-building programs in endoluminal suturing devices, mapping closure strategy across adjacent therapeutic categories such as endoscopic closure systems when aligning clips and suturing approaches within the same procedural toolkit.
How is Endoluminal Suturing Devices Demand Segmented by Product Type, Application, and End User?
The segmentation of endoluminal suturing devices demand in the UK reflects how clinical teams assign closure needs, evaluate procedural reliability, and expand endoscopy capability across care settings.
Why do needle-based suturing devices lead adoption in endoscopic closure workflows?
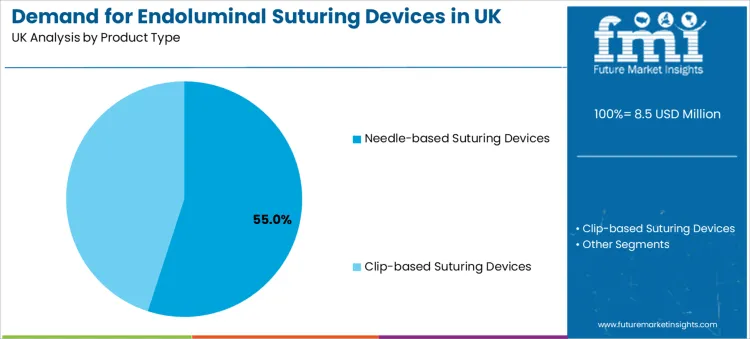
Needle-based suturing devices hold a 55.0% share, making them the leading product type. This reflects the preference for controlled tissue capture and predictable stitch placement in procedures where closure integrity is a primary success factor. Clinical teams selecting needle-based systems often prioritise handling stability, consistent deployment response, and closure repeatability across varied anatomical sites.
Hospitals also value these systems where training pathways can be standardised across multiple clinicians. Consistency becomes a practical advantage when service lines want fewer outcome variations between operators. Many endoscopy suites align this capability with platform readiness associated with flexible endoscopes, since stable access and visualisation support reliable execution.
What is positioning bariatric procedures as the leading application for endoluminal suturing devices?
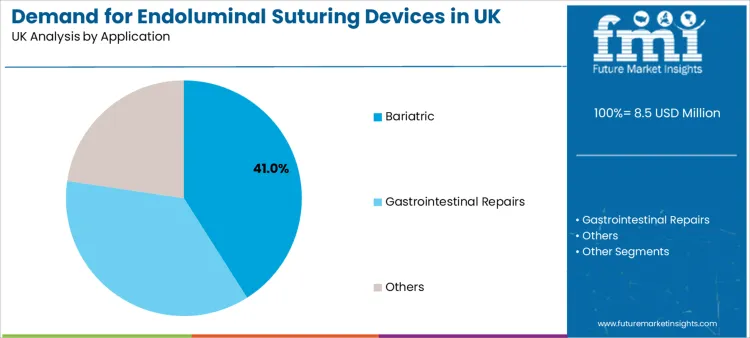
Bariatric procedures account for a 41.0% share, making them the largest application segment. This reflects rising focus on minimally invasive weight-management pathways and the operational need to maintain safety and procedural reliability. Closure performance matters significantly in bariatric interventions because it influences post-procedure stability, complication risk, and clinician confidence to scale adoption.
Clinical capacity planning in this area is supported by NHS audit reporting that tracks developing measures related to weight management services and bariatric procedures in England. Devices that reduce variability in closure outcomes gain stronger attention as teams work to protect patient outcomes while improving procedural throughput.
Why do hospitals remain the leading end-user setting despite outpatient expansion?
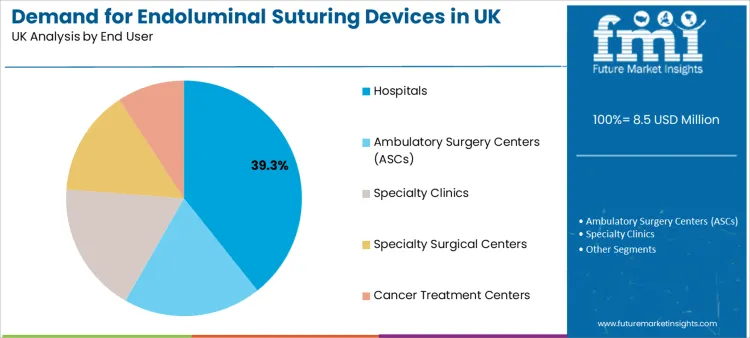
Hospitals represent a 39.3% share, making them the leading end user segment. Their lead position reflects higher case complexity, access to multidisciplinary resources, and stronger readiness for complications management when advanced endoscopic interventions are performed. Hospitals also operate structured governance for training and credentialing, supporting wider adoption once a device demonstrates reliable outcomes.
Ambulatory surgery centers (ASCs) and specialty clinics remain important adoption pathways where workflows support shorter stays and faster scheduling cycles. Specialty surgical centers and cancer treatment centers contribute through targeted intervention programs where minimally invasive approaches help limit trauma burden.
Procedure standardisation and turnover efficiency planning often aligns with supporting consumables and workflow tooling ushered in by endoscope procedure kits, especially when facilities optimise setup consistency across multiple procedure types.
What are the Dynamics, Restraints, Opportunities, and Threats Shaping this Space?
What dynamics keep adoption active across multiple care settings?
Minimally invasive care pathways drive demand, particularly where clinical teams want reliable closure outcomes without shifting procedures into higher-intensity surgical routes. Bariatric interventions and gastrointestinal repairs strengthen the use case because closure stability influences downstream safety and patient recovery expectations.
Post-market surveillance expectations also influence confidence and procurement discipline. Government and MHRA guidance highlights strengthened surveillance requirements coming into force in June 2025, improving traceability and enabling faster response when safety concerns emerge.
What restraints slow uptake even when procedural value is clear?
Training and skill standardisation remain key restraints. Endoluminal suturing relies on technique discipline, and outcomes are sensitive to operator familiarity and workflow setup. Budget prioritisation can also slow purchasing decisions when facilities are balancing multiple endoscopy investments, including imaging stack upgrades and service-line expansion programs.
Where are the strongest opportunities for suppliers and healthcare providers?
Structured onboarding and training support creates an adoption advantage. Providers respond well when suppliers deliver technique guidance, clear troubleshooting pathways, and service responsiveness that protects uptime.
Integration planning expands where facilities align suturing capabilities with a broader endoscopic closure strategy, ensuring that technical requirements and system efficiencies are cross-referenced to support long-term clinical goals
What threats can disrupt demand expectations?
Inconsistent closure outcomes can slow expansion beyond specialist users. Supply continuity for accessories also matters, since procedural schedules depend on predictable availability. Changing regulatory documentation expectations can influence vendor selection when buyers prioritise traceability and compliance readiness.
How is Endoluminal Suturing Devices Demand Evolving across Key Regions in the UK?
Regional growth varies based on specialist center density, procedural throughput, training availability, and endoscopy suite capability.
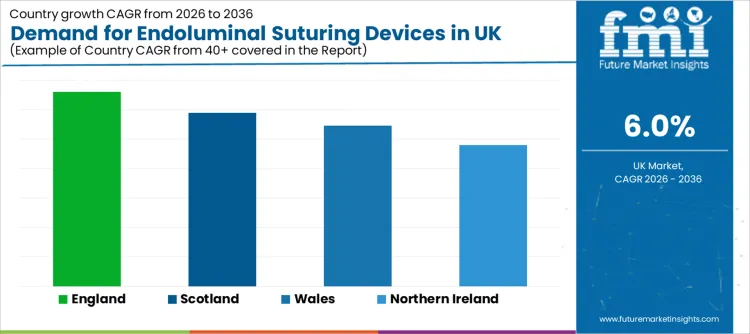
| Region | CAGR (2026-2036) |
|---|---|
| England | 6.6% |
| Scotland | 5.9% |
| Wales | 5.5% |
| Northern Ireland | 4.8% |
Why is England leading growth through higher procedural throughput and wider capability coverage?
England grows at 6.6%, supported by larger hospital networks and higher endoscopy volumes. Adoption expands faster when services can standardise technique across sites and maintain structured training pathways. England also benefits from higher concentration of specialist programs where bariatric and gastrointestinal repair workflows support consistent device utilisation.
How is Scotland sustaining uptake through focused program development and reliability-led procurement?
Scotland expands at 5.9%, shaped by careful adoption where clinical leaders prioritise dependable outcomes and disciplined procedural integration. Facilities tend to value predictable handling, consistent closure performance, and service support that limits downtime risk.
What is shaping Wales as it scales demand through targeted clinical adoption and workflow readiness?
Wales grows at 5.5%, driven by selective uptake in programs where endoluminal suturing provides clear clinical and operational value. Adoption strengthens when teams can demonstrate stable closure outcomes and build repeatable workflows that reduce procedural variability.
Why is Northern Ireland progressing through measured expansion of advanced endoscopy capability?
Northern Ireland rises at 4.8%, reflecting value-driven investment decisions and phased adoption. Facilities often expand usage once early procedural results establish confidence and when supporting training and device access remain stable.
What Defines the Competitive Landscape for Endoluminal Suturing Devices in the UK?
Competition is shaped by clinician usability, closure reliability, training support, and supplier responsiveness. Hospitals and specialty centers evaluate offerings based on handling predictability, ability to deliver consistent closure strength, and time efficiency in real procedure settings. Vendor support also plays a major role, especially when organisations want to expand adoption beyond a small group of expert users.
Boston Scientific Corporation competes through its endoscopy-focused portfolio presence. Medtronic holds strong positioning through broad surgical technology coverage and established hospital procurement relationships. Olympus remains closely connected to endoscopy suite infrastructure and procedural workflow alignment. Ovesco Endoscopy AG is recognised for specialised endoscopic therapy tools aligned with advanced closure needs. EndoRobotics Co., LTD. adds competitive pressure through innovation-led offerings and technology differentiation.
Key Industry Participants
- Boston Scientific Corporation
- Medtronic
- Olympus
- Ovesco Endoscopy AG
- EndoRobotics Co., LTD.USD 8.5 million
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| Product Type | Needle-based Suturing Devices; Clip-based Suturing Devices |
| Application | Bariatric; Gastrointestinal Repairs; Others |
| End User | Hospitals; Ambulatory Surgery Centers (ASCs); Specialty Clinics; Specialty Surgical Centers; Cancer Treatment Centers |
| Regions Covered | England; Scotland; Wales; Northern Ireland |
| Key Companies Profiled | Boston Scientific Corporation; Medtronic; Olympus; Ovesco Endoscopy AG; EndoRobotics Co., LTD. |
Endoluminal Suturing Devices Demand in the UK by Segment
By Product Type:
- Needle-based Suturing Devices
- Clip-based Suturing Devices
By Application:
- Bariatric
- Gastrointestinal Repairs
- Others
By End User:
- Hospitals
- Ambulatory Surgery Centers (ASCs)
- Specialty Clinics
- Specialty Surgical Centers
- Cancer Treatment Centers
By Region:
- England
- Scotland
- Wales
- Northern Ireland
Bibliography
- Medicines and Healthcare products Regulatory Agency. (2025). MHRA guidance on new medical devices post-market surveillance requirements.
- NHS Digital. (2025). National Obesity Audit: April 2025 - June 2025.
- NHS Digital. (2026). National Obesity Audit: July 2025 - September 2025.
- UK Government. (2025). Regulating medical devices in the UK.
Frequently Asked Questions
What is the outlook for the Endoluminal Suturing Devices Industry in the UK for 2026?
The endoluminal suturing devices industry in the UK is expected to total USD 8.5 million in 2026.
What value is expected for the Endoluminal Suturing Devices Industry in the UK for 2036?
In 2036, demand for endoluminal suturing devices in the UK will likely be valued at USD 15.2 million.
At what rate will the Endoluminal Suturing Devices Industry in the UK progress from 2026 to 2036?
The endoluminal suturing devices landscape in the UK is anticipated to advance at a 6.0% CAGR from 2026 to 2036.
Which application will constitute the bulk of all endoluminal suturing devices deployment in the UK?
Bariatric procedures are likely to be the dominant application, capturing 41.0% of the total revenue in 2026.
Which product type is expected to be most preferred in the UK for endoluminal suturing devices?
By product type, needle-based suturing devices will account for 55.0% of all uses by key healthcare providers.
Table of Content
- Executive Summary
- UK Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- UK Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- UK Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- UK Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Needle-based Suturing Devices
- Clip-based Suturing Devices
- Needle-based Suturing Devices
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- UK Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Bariatric
- Gastrointestinal Repairs
- Others
- Bariatric
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- UK Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Ambulatory Surgery Centers (ASCs)
- Specialty Clinics
- Specialty Surgical Centers
- Cancer Treatment Centers
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Boston Scientific Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Medtronic
- Olympus
- Ovesco Endoscopy AG
- EndoRobotics Co., LTD.
- Boston Scientific Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: UK Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: UK Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: UK Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: UK Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: UK Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: UK Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: UK Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: UK Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: UK Market Pricing Analysis
- Figure 2: UK Market Value (USD Million) Forecast 2021-2036
- Figure 3: UK Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: UK Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: UK Market Attractiveness Analysis by Product Type
- Figure 6: UK Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: UK Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: UK Market Attractiveness Analysis by Application
- Figure 9: UK Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: UK Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: UK Market Attractiveness Analysis by End User
- Figure 12: UK Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: UK Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: UK Market Attractiveness Analysis by Region
- Figure 15: UK Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: UK Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 17: UK Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 18: UK Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 19: UK Market Attractiveness Analysis by Product Type
- Figure 20: UK Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 21: UK Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 22: UK Market Attractiveness Analysis by Application
- Figure 23: UK Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 24: UK Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 25: UK Market Attractiveness Analysis by End User
- Figure 26: UK Market - Tier Structure Analysis
- Figure 27: UK Market - Company Share Analysis

