Demand for Remicade biosimilars in Japan is expected to grow from USD 119.8 million in 2025 to USD 178.2 million by 2035, reflecting a CAGR of 4.1%. In the early years, from 2025 to 2030, demand grows consistently year-on-year. Starting at USD 119.8 million in 2025, it increases to USD 124.6 million in 2026, representing a 4.0% growth rate. By 2027, demand reaches USD 129.7 million, followed by USD 134.9 million in 2028. Growing adoption of biosimilars drives this steady rise as healthcare providers and patients seek more cost-effective alternatives to the originator drug, Remicade. Ongoing improvements in the affordability and accessibility of biosimilars will continue to support this growth in the early years.
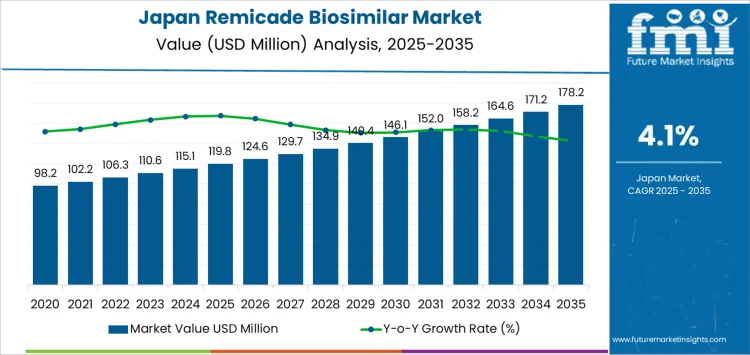
As demand progresses into the 2030s, growth accelerates slightly, reaching USD 178.2 million by 2035. From USD 140.4 million in 2030, it increases to USD 146.1 million in 2031, continuing its upward trend. This growth is attributed to the increased acceptance of biosimilars by healthcare professionals and patients, as well as the expansion of reimbursement schemes and greater industry penetration. By the mid-2030s, Remicade biosimilars will play a more significant role in Japan’s immunology treatment landscape, with continued improvements in efficacy, safety, and cost-effectiveness. The cumulative growth during this period reflects the growing confidence in biosimilars, driven by both cost-driven healthcare reforms and their success in clinical applications.
| Metric | Value |
|---|---|
| Industry Value (2025) | USD 119.8 million |
| Industry Forecast Value (2035) | USD 178.2 million |
| Forecast CAGR (2025-2035) | 4.1% |
Demand for Remicade biosimilars in Japan is experiencing significant growth as healthcare providers seek affordable alternatives to the original Remicade (infliximab) for managing autoimmune diseases such as rheumatoid arthritis, inflammatory bowel disease, and psoriasis. Biosimilars provide similar efficacy, safety, and quality to the reference product, but at a lower cost, making them an attractive option in Japan's cost-conscious healthcare environment. As healthcare systems face increasing financial pressure and the need for cost-reduction measures intensifies, Remicade biosimilars are seen as an effective way to maintain high-quality care while reducing overall treatment expenditures. This cost-saving potential makes them an essential part of healthcare strategies, benefiting both patients and the healthcare system.
Regulatory support for biosimilars in Japan has been a significant driver of growth. The Japanese government has introduced policies aimed at promoting the use of biosimilars to control healthcare costs, offering incentives for healthcare providers to adopt these alternatives. Clinical studies showing the equivalence of Remicade biosimilars in treating chronic conditions further support their growing use. As more biosimilar options become available, both physicians and patients are increasingly inclined to choose these cost-effective therapies, bolstering the demand for Remicade biosimilars in the country.
Despite the positive growth trajectory, challenges remain. Physician acceptance of biosimilars, patient concerns about switching from reference products, and competition from other biosimilar candidates may slow the adoption rate. Educating patients and healthcare providers about the safety and efficacy of biosimilars is crucial. The strong regulatory framework, coupled with growing awareness and the ongoing demand for affordable treatment options, is expected to continue driving the demand for Remicade biosimilars in Japan, supporting long-term growth in the sector.
Demand for remicade biosimilar in Japan is segmented by indication, distribution channel, and region. By indication, rheumatoid arthritis leads the demand at 43%, followed by Crohn’s disease, ulcerative colitis, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis. In terms of distribution channel, hospital pharmacies dominate, accounting for 62.1%, with retail pharmacies and online pharmacies also contributing to the overall demand. Regionally, demand spans across Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, and the rest of Japan, with varying levels of uptake based on regional healthcare infrastructure and disease prevalence.
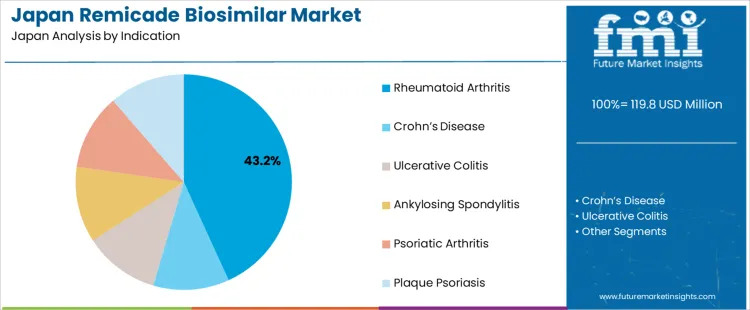
Rheumatoid arthritis accounts for 43% of the demand for remicade biosimilars in Japan, driven by its high prevalence and the increasing use of biosimilars as a cost-effective treatment. Remicade biosimilars offer a more affordable alternative to the original biologic, providing similar efficacy in managing joint inflammation and pain. The demand is particularly strong in regions like Kanto and Kansai, where access to healthcare and specialized treatments is widespread. As both patients and healthcare providers seek affordable treatment options, the adoption of biosimilars continues to rise. Japan’s aging population and the growing number of rheumatoid arthritis cases further fuel the demand for remicade biosimilars. The shift toward biosimilars is also encouraged by their proven safety and effectiveness, making them a preferred choice for treating rheumatoid arthritis. This trend ensures remicade biosimilars maintain strong demand in this indication.
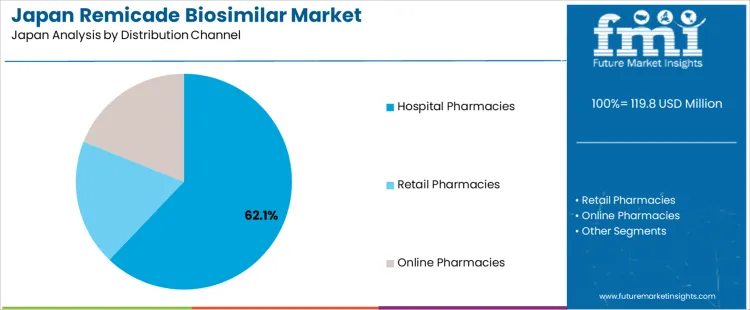
Hospital pharmacies account for 62.1% of the demand for remicade biosimilars in Japan, reflecting their role as the primary distribution channel for biologics and biosimilars. Hospitals are central in providing specialized care for complex conditions such as rheumatoid arthritis, Crohn’s disease, and ulcerative colitis. Biosimilars like remicade are preferred in these settings due to their cost-effectiveness, helping manage the rising healthcare costs associated with chronic diseases. Hospital pharmacies also provide more direct access to these treatments through prescriptions and inpatient care, ensuring timely administration. The demand is especially high in regions with advanced healthcare infrastructure, such as Kanto and Kansai, where hospitals have the capacity to deliver specialized biologic treatments. As hospital pharmacies continue to be a key provider for these therapies, their dominant role in the distribution of remicade biosimilars is expected to remain strong in Japan.
Demand for Remicade biosimilars in Japan is increasing as healthcare systems look for cost-effective alternatives to reference biologics. With the aging population and rising cases of autoimmune diseases, including rheumatoid arthritis and inflammatory bowel disease, the demand for biologics remains high. Biosimilars offer an affordable treatment option to meet this demand. Uptake is slow in Japan due to hesitancy among physicians and patients about the safety and efficacy of biosimilars compared to originator products. Limited substitution policies and modest price differences also hinder faster adoption. Regulatory hurdles and a need for more clinical data to support biosimilars’ safety and efficacy further challenge widespread adoption. While biosimilars hold promise, these barriers need to be addressed for them to reach their full potential in Japan's healthcare system.
Why is Demand for Remicade Biosimilar Growing in Japan?
The growing demand for remicade biosimilars in Japan is driven by the rising need for affordable biologic treatments. The aging population and the increase in autoimmune diseases create a substantial demand for biologics, which biosimilars can meet. Biosimilars offer a more cost-effective alternative to reference biologics, helping reduce overall healthcare costs. Japan’s regulatory framework supports the adoption of biosimilars, encouraging their use in healthcare settings. With the increasing availability of clinical data showing the safety and efficacy of biosimilars, physicians and patients are becoming more confident in using them. Studies demonstrating that switching from originator infliximab to biosimilars does not compromise efficacy also support the growing demand. These factors are contributing to greater acceptance and use of remicade biosimilars in Japan.
How are Technological & Industry Innovations Driving Remicade Biosimilar Demand in Japan?
Technological advancements in the production of biosimilars are playing a key role in increasing their demand in Japan. Improvements in manufacturing processes, such as better protein expression systems and more efficient purification techniques, have made biosimilars more consistent and reliable. These innovations ensure that biosimilars can match the reference biologic in terms of quality and effectiveness. Enhanced regulatory standards and post-industry surveillance are boosting healthcare providers' confidence in the safety of biosimilars. The growing availability of comparative clinical data and real-world evidence also supports the growing adoption of biosimilars. As competition in the biosimilar industry increases, it is expected that prices will decrease, improving accessibility and further promoting the widespread use of remicade biosimilars in Japan.
What are the Key Challenges and Risks That Could Limit Remicade Biosimilar Demand in Japan?
Despite the growth potential for Remicade biosimilars in Japan, several challenges may limit adoption. Physician and patient hesitation over the safety and efficacy of biosimilars compared to the originator product remains a key barrier. Many healthcare providers require clear, robust clinical data before prescribing biosimilars. Japan’s lack of automatic substitution policies and limited incentives for prescribing biosimilars slow their uptake. Although biosimilars are priced lower than originator products, the price difference may not be significant enough to drive widespread adoption. Regulatory concerns, including approval processes and clinical evidence, may also hinder the growth of the biosimilar industry. Competition from other biologics and emerging therapies could further limit industry share for remicade biosimilars in Japan.
Demand for remicade biosimilar in Japan is growing steadily across all regions, with Kyushu & Okinawa leading at a 5.1% CAGR, driven by increasing adoption of cost-effective biosimilars in healthcare. Kanto follows with a 4.7% CAGR, supported by the region’s large healthcare infrastructure and high demand for affordable biologics. Kansai shows a 4.1% CAGR, driven by regional hospitals and healthcare providers adopting biosimilars for treatment of autoimmune diseases. Chubu experiences a 3.6% CAGR, as demand rises with growing awareness and accessibility of biosimilars in local healthcare settings. Tohoku and the Rest of Japan see more moderate growth at 3.2% and 3.0%, respectively, as the adoption of biosimilars gradually increases in smaller industries.
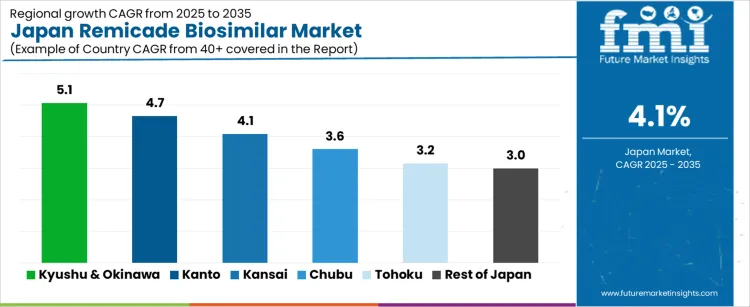
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 5.1% |
| Kanto | 4.7% |
| Kansai | 4.1% |
| Chubu | 3.6% |
| Tohoku | 3.2% |
| Rest of Japan | 3.0% |
Kyushu & Okinawa leads the demand for remicade biosimilar, growing at a 5.1% CAGR. The region’s focus on improving healthcare access, along with the increasing adoption of cost-effective biologics, has contributed to the rise in demand for remicade biosimilars. Okinawa’s growing healthcare infrastructure, combined with a strong emphasis on providing affordable treatments for autoimmune diseases such as rheumatoid arthritis, has driven the uptake of biosimilars in local hospitals and clinics. Kyushu & Okinawa’s expanding aging population is contributing to the demand for remicade biosimilars, as the incidence of autoimmune diseases rises with age. As healthcare providers in the region continue to embrace cost-effective treatment options, remicade biosimilars are expected to play an increasingly important role in providing accessible and affordable care for patients. The demand for remicade biosimilars in Kyushu & Okinawa is likely to remain strong, driven by continued healthcare modernization and patient-centered care.
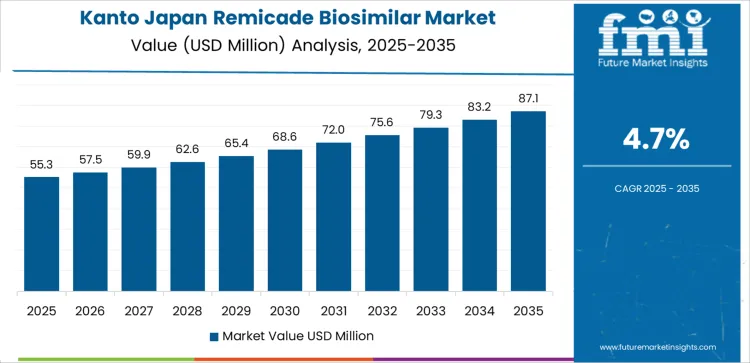
In Kanto, demand for remicade biosimilar is rising at a 4.7% CAGR. Kanto’s large and diverse healthcare industry, particularly in Tokyo, is a key driver behind the growing adoption of biosimilars. As healthcare providers and hospitals focus on reducing costs while maintaining high-quality patient care, the demand for biosimilars such as remicade is increasing. Kanto’s high population density, combined with a rising demand for biologics to treat autoimmune diseases, has contributed to the increased usage of remicade biosimilars in both public and private healthcare settings. The growing awareness of the affordability and effectiveness of biosimilars among both patients and healthcare providers has supported this trend. As Kanto continues to prioritize cost-effective healthcare solutions and increased access to biologic therapies, demand for remicade biosimilars is expected to grow steadily, driven by both consumer preferences and institutional adoption in the region.
Kansai shows steady demand for remicade biosimilar, growing at a 4.1% CAGR. The region’s strong healthcare infrastructure, particularly in cities like Osaka and Kyoto, has supported the adoption of biosimilars in the treatment of autoimmune diseases. Kansai’s healthcare providers are increasingly utilizing remicade biosimilars as a cost-effective alternative to reference biologics, addressing the growing need for affordable treatment options. The region’s focus on providing high-quality care to an aging population, with rising cases of autoimmune diseases, has contributed to the steady increase in demand for biosimilars. As the healthcare sector continues to prioritize efficient and sustainable treatment options, remicade biosimilars are becoming an essential part of the therapeutic landscape in Kansai. With increasing awareness of the benefits of biosimilars and government support for their adoption, demand for remicade biosimilars in Kansai is expected to continue to expand steadily over the coming years.
Chubu is experiencing steady demand for remicade biosimilar, growing at a 3.6% CAGR. The region’s healthcare sector, particularly in cities like Nagoya, is increasingly adopting biosimilars due to their cost-effectiveness and effectiveness in treating autoimmune diseases. Chubu’s healthcare providers are looking to biosimilars, such as remicade, to help reduce treatment costs while maintaining high-quality care. The growing interest in biologic treatments and the increasing incidence of autoimmune diseases in Chubu’s aging population are contributing to the rise in demand for biosimilars. As more hospitals and clinics in the region embrace these affordable alternatives, remicade biosimilars are expected to become a more integral part of the treatment landscape. With the continued push towards cost-saving and patient-centered healthcare, demand for remicade biosimilars in Chubu will continue to grow as the region modernizes its healthcare offerings.
Tohoku is seeing moderate growth in remicade biosimilar demand, with a 3.2% CAGR. While the region’s adoption of biosimilars is slower compared to larger urban areas, there is a growing recognition of the benefits of remicade biosimilars as a cost-effective alternative to reference biologics. Tohoku’s healthcare providers, especially in more rural areas, are increasingly turning to biosimilars to offer affordable treatment options for patients with autoimmune diseases. The region’s aging population, combined with a rising prevalence of autoimmune conditions, is contributing to the growing need for affordable biologic treatments. As Tohoku’s healthcare infrastructure continues to evolve and patients become more aware of the availability of biosimilars, demand is expected to rise steadily. While the growth rate may be more gradual in Tohoku, the increased adoption of remicade biosimilars in the region will continue to play a significant role in improving patient care and expanding access to treatment.
The Rest of Japan is experiencing steady demand for remicade biosimilar, with a 3.0% CAGR. Although the adoption rate is slower compared to urban centers, there is growing interest in biosimilars as more healthcare providers in rural areas adopt these cost-effective solutions. As the healthcare sector in smaller cities and rural regions continues to modernize, there is a gradual shift toward the use of remicade biosimilars to treat autoimmune diseases such as rheumatoid arthritis and Crohn’s disease. The increasing focus on healthcare accessibility and cost reduction is driving the adoption of biosimilars, which provide effective treatment options at a lower price point. As the government continues to promote the use of biosimilars and their benefits in terms of cost-saving, the demand for remicade biosimilars is expected to grow steadily across the Rest of Japan, supporting the overall expansion of biosimilars in the Japanese healthcare system.
The demand for remicade biosimilars in Japan is growing because there is a rising need for affordable biologic therapies to treat chronic autoimmune and inflammatory diseases such as rheumatoid arthritis, Crohn’s disease, ulcerative colitis, and psoriasis. With the increasing prevalence of such long‑term conditions and concerns about healthcare costs and accessibility, biosimilar versions of Infliximab, the active ingredient in Remicade, offer a lower-cost alternative while maintaining efficacy and safety comparable to the original drug. As treatment paradigms shift toward long-term disease management and cost containment becomes more important, biosimilars are becoming more attractive to both medical institutions and patients.
Major suppliers of Remicade biosimilars in Japan include Celltrion Healthcare, Pfizer Inc., Samsung Bioepis, Merck & Co., Inc., and Amgen Inc. Celltrion Healthcare leads, with a strong footprint and early entry into infliximab biosimilars. These companies supply a mix of biosimilar infliximab products that align with Japan’s regulatory standards, providing clinicians with infusion therapy options and contributing to the broader availability of biologic treatments at more accessible costs.
Shifts driving uptake in Japan include increasing regulatory support for biosimilars, efforts to lower treatment costs across the healthcare system, and growing clinical acceptance of biosimilar infliximab, especially as real‑world data from Japanese studies support its safety and effectiveness when switching from the original biologic. As patients, physicians, and payers become more comfortable with biosimilars and as cost pressures rise, usage of Remicade biosimilars is likely to expand further, offering wider access to effective biologic therapy for patients in need.
| Items | Values |
|---|---|
| Quantitative Units (2025) | USD million |
| Indication | Rheumatoid Arthritis, Crohn’s Disease, Ulcerative Colitis, Ankylosing Spondylitis, Psoriatic Arthritis, Plaque Psoriasis |
| Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies |
| Region | Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan |
| Countries Covered | Japan |
| Key Companies Profiled | Celltrion Healthcare, Pfizer Inc., Samsung Bioepis, Merck & Co., Inc., Amgen Inc. |
| Additional Attributes | Dollar sales by indication and distribution channel; regional CAGR and growth trends; increasing adoption of biosimilars in inflammatory conditions; innovations in biosimilar formulations and manufacturing |
How big is the demand for remicade biosimilar in Japan in 2025?
The demand for remicade biosimilar in Japan is estimated to be valued at USD 119.8 million in 2025.
What will be the size of remicade biosimilar in Japan in 2035?
The market size for the remicade biosimilar in Japan is projected to reach USD 178.2 million by 2035.
How much will be the demand for remicade biosimilar in Japan growth between 2025 and 2035?
The demand for remicade biosimilar in Japan is expected to grow at a 4.1% CAGR between 2025 and 2035.
What are the key product types in the remicade biosimilar in Japan?
The key product types in remicade biosimilar in Japan are rheumatoid arthritis, crohn’s disease, ulcerative colitis, ankylosing spondylitis, psoriatic arthritis and plaque psoriasis.
Which distribution channel segment is expected to contribute significant share in the remicade biosimilar in Japan in 2025?
In terms of distribution channel, hospital pharmacies segment is expected to command 62.1% share in the remicade biosimilar in Japan in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.