Demand for Remicade biosimilars in the USA is expected to grow from USD 0.8 billion in 2025 to USD 1.4 billion by 2035, reflecting a compound annual growth rate (CAGR) of 5.1%. Increasing adoption of biosimilars drives this growth as more affordable alternatives to biologic therapies, particularly in the treatment of autoimmune diseases such as rheumatoid arthritis, Crohn’s disease, and ulcerative colitis. As healthcare costs continue to rise, the use of biosimilars is expected to increase, providing patients with more cost-effective treatment options while maintaining efficacy and safety profiles similar to those of the reference biologics.
The market is further supported by the increasing pressure on healthcare systems to reduce drug spending. Biosimilars, which offer the same therapeutic benefits as the reference biologics but at a lower cost, are becoming increasingly important in meeting the healthcare needs of a growing and aging population. As the patent exclusivity of original biologics continues to expire, the demand for biosimilars like Remicade is expected to grow steadily over the next decade, contributing to a more sustainable healthcare system.
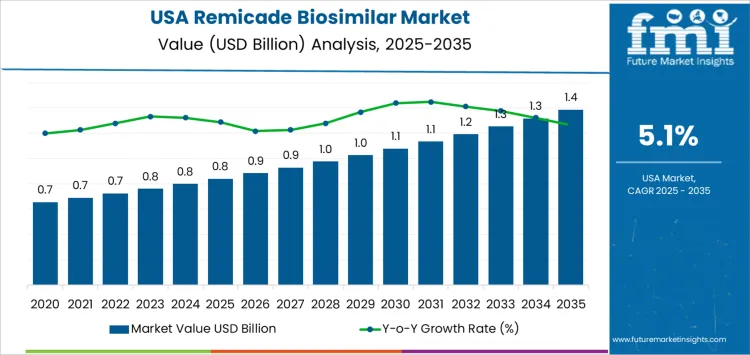
The 10-year growth comparison for the Remicade biosimilar market in the USA shows gradual, steady increases in demand over the forecast period. Starting at USD 0.8 billion in 2025, the market grows modestly, reaching USD 0.9 billion in 2026 and USD 0.9 billion in 2027. This early stage reflects the initial uptake of Remicade biosimilars, as patients and healthcare providers gradually become more familiar with their benefits and affordability.
The market continues to expand steadily, reaching USD 1.0 billion in 2028 and USD 1.0 billion in 2029, followed by USD 1.1 billion in 2030. As the market matures and more healthcare providers embrace biosimilars, the demand for Remicade biosimilars grows more quickly. By 2032, the market reaches USD 1.2 billion and USD 1.3 billion in 2033, reflecting increased adoption and expanded indications for biosimilars. The market reaches its peak of USD 1.4 billion by 2035, demonstrating consistent growth driven by cost-containment pressures, broader acceptance of biosimilars, and the expiration of patents for reference biologics. The steady rise in market demand throughout the decade highlights the growing role of biosimilars in the healthcare system.
| Metric | Value |
|---|---|
| Industry Sales Value (2025) | USD 0.8 billion |
| Industry Forecast Value (2035) | USD 1.4 billion |
| Industry Forecast CAGR (2025-2035) | 5.1% |
Demand for biosimilars of Remicade (infliximab) is increasing in the USA as healthcare providers and payers look to balance managing chronic autoimmune diseases with controlling drug costs. As the number of patients with conditions treated by infliximab - such as rheumatoid arthritis, Crohn’s disease, ulcerative colitis, and psoriasis - remains significant, biosimilar versions offer a lower-cost but therapeutically equivalent alternative. This helps expand patient access to biologic therapies while reducing the financial burden on both insurers and patients.
At the same time, advances in regulatory acceptance, growing clinician confidence, and increasing institutional adoption support broader uptake. Biosimilars are approved as having no clinically meaningful differences from the original biologic - meaning similar safety and efficacy. As more infliximab biosimilars enter the market and become accepted by specialty clinics, hospitals, and insurance formularies, switching from originator biologic to biosimilar becomes more common. This trend is reinforced by demonstrated cost-savings for the healthcare system, which in turn improves affordability and supports continued demand growth for Remicade biosimilars across the USA.
The remicade biosimilar market in the USA is segmented by indication and distribution channel. Rheumatoid arthritis is the leading indication, holding 43% of the market share. In terms of distribution channels, hospital pharmacies dominate, accounting for 62.1% of the market. The demand for Remicade biosimilars is driven by the increasing need for affordable treatment options for autoimmune diseases, as well as the growing shift towards biosimilar drugs in the healthcare system.
Rheumatoid arthritis is the dominant indication for Remicade biosimilars, accounting for 43% of the market share. This autoimmune disease, which causes inflammation in the joints, is one of the most common conditions treated with Remicade and its biosimilars. The efficacy of biosimilars in treating rheumatoid arthritis, coupled with their lower cost compared to the reference product, makes them an attractive option for patients and healthcare providers. With the increasing prevalence of rheumatoid arthritis in the USA, the demand for affordable, effective treatment options continues to grow. While other indications such as Crohn’s disease, ulcerative colitis, and ankylosing spondylitis also contribute to the market, rheumatoid arthritis remains the largest driver of biosimilar demand due to its high incidence and long-term treatment requirements.
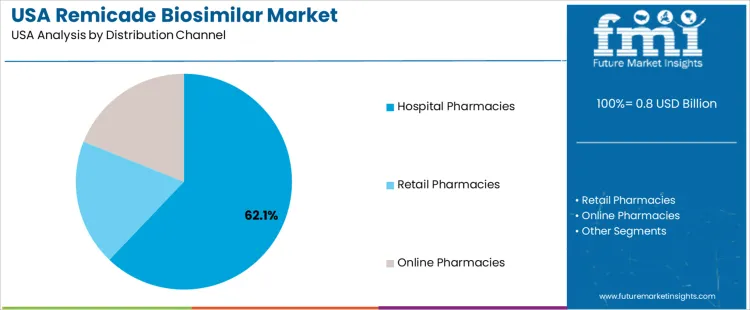
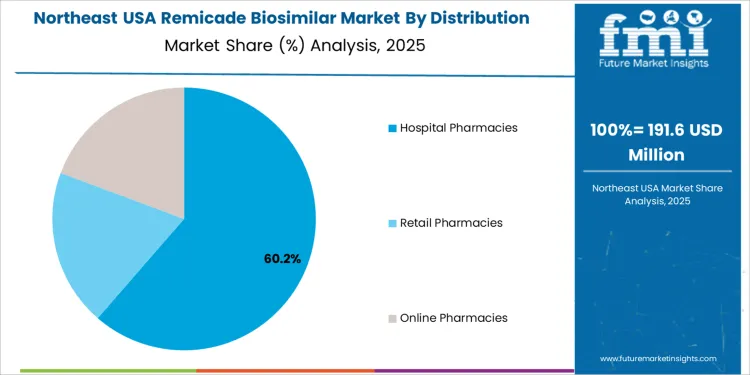
Hospital pharmacies lead the distribution channels for Remicade biosimilars, representing 62.1% of the market share. Hospital pharmacies are the primary point of distribution for biosimilars due to the need for specialized care and administration of these drugs, often through infusion in clinical settings. Hospital pharmacies also play a critical role in managing the treatment of chronic conditions like rheumatoid arthritis, where long-term care and close monitoring are required. Retail pharmacies and online pharmacies also contribute to the market, particularly for oral formulations of biosimilars and for patients receiving treatment through outpatient services. However, hospital pharmacies remain the largest distribution channel due to their essential role in delivering biologic treatments that require professional healthcare management and monitoring.
The demand for Remicade biosimilars in the USA is increasing as healthcare providers and patients seek cost-effective alternatives to the original Remicade (infliximab) for the treatment of autoimmune diseases such as rheumatoid arthritis, Crohn's disease, and psoriasis. Biosimilars, which are highly similar to the reference biologic but are typically offered at a lower cost, provide an opportunity to reduce healthcare spending while maintaining the efficacy and safety profile of the original drug. With the expiration of Remicade’s patents and the introduction of multiple biosimilar versions, the market for Remicade biosimilars is expanding, offering more treatment options for patients and increasing competition in the biologics market.
Several factors are driving the demand for Remicade biosimilars in the USA. First, the rising cost of biologic therapies, including the original Remicade, has prompted healthcare providers to seek more affordable alternatives. Biosimilars offer the same therapeutic benefits at a lower price, helping to reduce the financial burden on both patients and the healthcare system. Second, the increasing prevalence of autoimmune diseases and inflammatory conditions, which require long-term treatment with drugs like Remicade, fuels demand for more accessible treatment options.
Third, growing acceptance of biosimilars by physicians, patients, and regulatory agencies has led to wider adoption of Remicade biosimilars, as their safety and efficacy are demonstrated through clinical trials and real-world data. Finally, the introduction of multiple Remicade biosimilars from different manufacturers has created more competition in the market, further driving affordability and accessibility.
Despite the growing demand for Remicade biosimilars, several factors may limit their broader adoption in the USA. One significant restraint is the reluctance of some physicians and patients to switch from the original Remicade to a biosimilar, often due to concerns about the potential for slight differences in the formulations or the long-term effects of the biosimilar. This hesitancy can slow the uptake of biosimilars in clinical practice.
Another challenge is the complex regulatory environment surrounding the approval and reimbursement of biosimilars, which can create barriers to market entry and limit their availability in certain regions or healthcare settings. Additionally, the presence of exclusive contracts between manufacturers of the original biologic and healthcare providers can restrict the availability of biosimilars. Finally, insurance coverage and reimbursement policies may vary, and some patients may face higher out-of-pocket costs for biosimilars compared to the original biologic, affecting demand.
Several key trends are influencing the demand for Remicade biosimilars in the USA. One trend is the increasing focus on cost containment in the healthcare industry, which has driven the adoption of biosimilars as a way to reduce overall spending on biologic therapies. Another trend is the growing body of real-world evidence supporting the safety, efficacy, and interchangeability of Remicade biosimilars with the reference product, which has helped build confidence among healthcare providers and patients.
The expansion of biosimilar acceptance in both private and public insurance plans, including Medicare and Medicaid, is also contributing to increased adoption. Additionally, as the patents for more biologic drugs expire, the market for biosimilars is expected to continue to grow, providing patients with more affordable treatment options. Finally, the rising emphasis on patient-centered care and personalized medicine is encouraging healthcare providers to explore cost-effective alternatives that can offer the same therapeutic benefits as the original biologics.
The Remicade Biosimilar market in the USA is expected to grow steadily, with the West USA leading the way at a projected CAGR of 5.9%. South USA follows closely with a growth rate of 5.3%, while the Northeast USA is expected to grow at 4.7%. The Midwest USA shows moderate growth with a CAGR of 4.1%. Remicade biosimilars, which offer a more affordable alternative to the reference drug Remicade for the treatment of inflammatory diseases like rheumatoid arthritis and Crohn's disease, are gaining traction as healthcare providers and patients look for cost-effective therapies. As biosimilar adoption increases across the USA, the market for Remicade biosimilars is expected to expand.
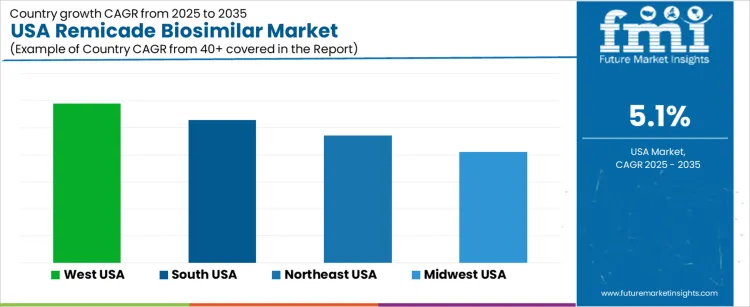
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 5.9% |
| South USA | 5.3% |
| Northeast USA | 4.7% |
| Midwest USA | 4.1% |
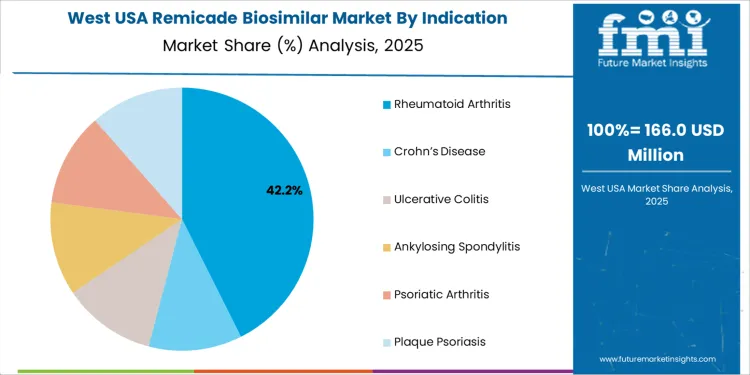
The West USA is projected to experience the highest growth in the Remicade Biosimilar market, with a projected CAGR of 5.9%. The region’s focus on cost-effective healthcare solutions, combined with its large population and healthcare infrastructure, is driving the adoption of biosimilars like Remicade. The West’s progressive healthcare policies and the push for affordable treatment options are key drivers of this growth.
States like California and Washington are at the forefront of healthcare innovation, and biosimilars are becoming an increasingly popular choice among providers looking to manage treatment costs without compromising patient care. Additionally, the high demand for autoimmune disease treatments and the growing acceptance of biosimilars by both healthcare providers and patients further supports the market expansion in the West USA.
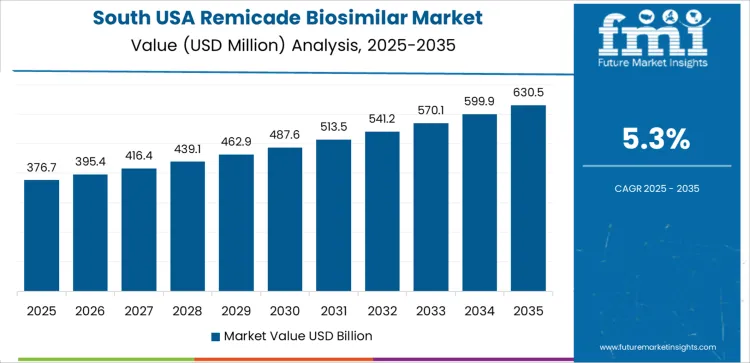
In South USA, the demand for Remicade Biosimilars is projected to grow at a CAGR of 5.3%. The region’s healthcare industry, with its growing number of patients diagnosed with autoimmune diseases, is increasingly turning to affordable alternatives like Remicade biosimilars. States like Texas, Florida, and Georgia have large populations with a significant number of patients requiring treatments for conditions like rheumatoid arthritis and Crohn's disease.
The rising healthcare costs and the push for more affordable treatment options are encouraging the use of biosimilars. As more healthcare providers in the South USA become familiar with the efficacy and cost benefits of biosimilars, the market for Remicade biosimilars is expected to grow. Additionally, the increasing acceptance of biosimilars by patients and providers, as well as expanding Medicaid and insurance coverage, further supports the growth of the market.
In Northeast USA, the Remicade Biosimilar market is projected to grow at a CAGR of 4.7%. The Northeast’s strong healthcare infrastructure, particularly in cities like New York, Boston, and Philadelphia, is contributing to the adoption of biosimilars in the treatment of autoimmune diseases. As healthcare providers in the region seek cost-effective alternatives to expensive biologic drugs, Remicade biosimilars are becoming a viable option.
The region’s increasing focus on controlling healthcare costs, along with the growing acceptance of biosimilars in clinical settings, is driving market growth. The Northeast’s large population of aging adults, who are more likely to suffer from conditions like rheumatoid arthritis and inflammatory bowel diseases, further supports the demand for Remicade biosimilars. As insurance coverage for biosimilars continues to expand, the adoption of Remicade biosimilars is expected to increase in the Northeast.
In Midwest USA, the Remicade Biosimilar market is expected to grow at a more moderate rate of 4.1%. While the Midwest has traditionally been slower to adopt biosimilars compared to other regions, the increasing pressure to lower healthcare costs and improve access to treatments is driving growth in the market. The region’s healthcare providers are increasingly turning to biosimilars like Remicade to provide affordable treatments for autoimmune diseases.
Additionally, the Midwest’s strong healthcare institutions and growing recognition of the effectiveness of biosimilars are contributing to the expansion of the market. States like Illinois, Michigan, and Ohio are seeing rising demand for cost-effective alternatives to traditional biologic drugs, and as patient awareness and provider confidence in biosimilars grow, the market for Remicade biosimilars is expected to increase gradually.
Demand for Remicade biosimilars in the USA has increased as patients and healthcare providers seek more affordable treatment options for chronic autoimmune diseases such as rheumatoid arthritis, Crohn’s disease, ulcerative colitis, and psoriasis. As the original biologic Infliximab (Remicade) lost its exclusivity, biosimilar versions became more accessible. The shift toward biosimilars has been encouraged by rising healthcare costs and growing insurance and payer interest in cost-effective biologic therapies. Acceptance of biosimilar infliximab by clinicians and infusion centers, combined with the proven clinical equivalence to the reference product, has helped increase uptake across hospitals and specialty clinics.
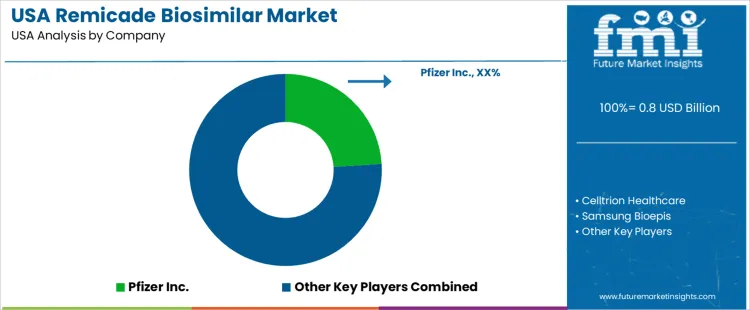
On the supply side a group of pharmaceutical and biotech firms provide biosimilar infliximab products competing in the USA market. Leading suppliers include companies such as Pfizer, Amgen, and other major biosimilar manufacturers. Those firms supply infliximab biosimilars through standardized regulatory-approved pathways as alternatives to the reference biologic. Competition among them centres on price, supply reliability, regulatory compliance, and support services for clinics and hospitals. As of recent years biosimilar infliximab has captured a growing share of the market, reflecting cost savings and increased confidence among prescribers and payers. Healthcare providers and patients who prioritize affordability, steady supply, and therapeutic equivalence are more likely to shift toward biosimilar use.
Companies that combine competitive pricing, quality manufacturing, strong distribution networks, regulatory compliance, and effective stakeholder education are best positioned to capture growth in the USA Remicade biosimilar market as demand continues to rise for affordable, long-term biologic therapies.
| Items | Details |
|---|---|
| Quantitative Units | USD Billion |
| Regions Covered | USA |
| Indication | Rheumatoid Arthritis, Crohn’s Disease, Ulcerative Colitis, Ankylosing Spondylitis, Psoriatic Arthritis, Plaque Psoriasis |
| Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies |
| Key Companies Profiled | Pfizer Inc., Celltrion Healthcare, Samsung Bioepis, Merck & Co., Inc., Amgen Inc. |
| Additional Attributes | Dollar sales by indication and channel show accelerating adoption of infliximab biosimilars in the USA, particularly for conditions like ulcerative colitis, rheumatoid arthritis, and Crohn’s disease. Biosimilars are increasingly preferred due to cost-effectiveness, and hospital + retail pharmacies are the main distribution points. |
How big is the demand for remicade biosimilar in USA in 2025?
The demand for remicade biosimilar in USA is estimated to be valued at USD 0.8 billion in 2025.
What will be the size of remicade biosimilar in USA in 2035?
The market size for the remicade biosimilar in USA is projected to reach USD 1.4 billion by 2035.
How much will be the demand for remicade biosimilar in USA growth between 2025 and 2035?
The demand for remicade biosimilar in USA is expected to grow at a 5.1% CAGR between 2025 and 2035.
What are the key product types in the remicade biosimilar in USA?
The key product types in remicade biosimilar in USA are rheumatoid arthritis, crohn’s disease, ulcerative colitis, ankylosing spondylitis, psoriatic arthritis and plaque psoriasis.
Which distribution channel segment is expected to contribute significant share in the remicade biosimilar in USA in 2025?
In terms of distribution channel, hospital pharmacies segment is expected to command 62.1% share in the remicade biosimilar in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.