Microarray Patch Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Microarray Patch Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Microarray Patch Market Forecast and Outlook 2026 to 2036
By 2036, microarray patch programs support USD 3,347.9 million in annual value, growing from a USD 1,117.2 million base in 2026 at an 11.6% CAGR. This delivery format moves dosing away from hypodermic injection toward controlled skin based administration. Development teams evaluate systems on dose uniformity, insertion reliability, and handling steps rather than on device appearance. Once a formulation and patch geometry complete clinical validation, the pair becomes locked inside the product file. Packaging format, training burden, and disposal rules shape rollout plans. Health systems review clinic throughput and storage needs before adoption.
Commercial progress in this field follows pharmaceutical pipeline decisions more than consumer device cycles. Manufacturing focuses on mold precision, coating accuracy, and sterility assurance across large lots. Release depends on mechanical insertion tests, content uniformity checks, and seal integrity. Fill finish partners influence schedules because patches ship with the drug product. Supply chains stage finished kits near distribution hubs with temperature control. For large immunization campaigns, vaccine shippers become part of deployment planning because temperature excursions directly impact usable inventory at the last mile. Regulatory teams manage combination product documentation and change control. Uptake depends on prescriber comfort and patient acceptance. Financial results track yield, scrap, and stable high volume production rather than frequent device redesign.
Quick Stats for Microarray Patch Market
- Microarray Patch Market Value (2026): USD 1,117.2 million
- Microarray Patch Market Forecast Value (2036): USD 3,347.9 million
- Microarray Patch Market Forecast CAGR (2026-2036): 11.6%
- Leading Product Type in Microarray Patch Market: Dissolving microneedle patches
- Leading Application in Microarray Patch Market: Vaccine delivery
- Key Growth Regions in Microarray Patch Market: Asia Pacific, North America, Europe
- Top Players in Microarray Patch Market: Vaxxas, 3M Drug Delivery, Zosano Pharma, Corium, Raphas
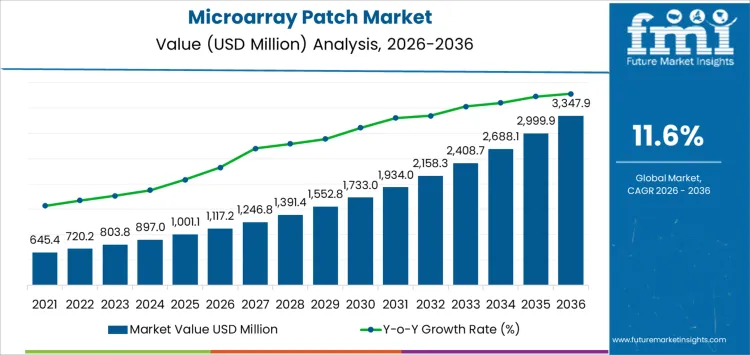
What Is the Growth Forecast for the Microarray Patch Market through 2036?
Demand for microarray patches is driven more by how quickly new formats are adopted and how delivery pathways are selected than by overall drug volume growth. In the near term, use is most visible in vaccination programs, dermatology treatments, and a limited set of chronic therapies where ease of administration and consistent dosing offer clear advantages. These patches are typically locked in during clinical development through device–drug integration work, so demand tends to follow program milestones and rollout timelines rather than short-term purchasing patterns. Uptake increases as more candidates transition from trial supply into routine distribution and as health systems expand patch-based delivery in settings where trained injection staff or cold-chain handling is limited. Growth reflects gradual inclusion in public health protocols and treatment guidelines, not rapid replacement of existing delivery methods.
In the later phase, market expansion depends heavily on manufacturing scalability and supply chain maturity. As adoption spreads, buyers focus on production yield, patch uniformity, and packaging stability for large-scale distribution. Manufacturers respond by investing in automated fabrication, inspection systems, and higher-capacity production lines that can support long-duration procurement contracts. Product ranges also widen to accommodate different dose strengths and active compounds. Over time, the category grows through accumulated approvals and broader geographic rollout, with companies winning long-term positions based on consistent output quality, dependable delivery schedules, and strong regulatory support.
Microarray Patch Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1,117.2 million |
| Forecast Value (2036) | USD 3,347.9 million |
| Forecast CAGR (2026-2036) | 11.6% |
How Is the Microarray Patch Market Changing Drug and Vaccine Delivery Methods?
Microarray patches use arrays of microscopic projections to deliver drugs or vaccines through the outer skin layers without conventional needles. The format targets consistent dosing, simplified training, and reduced sharps handling in clinics and field programs. Earlier delivery routes relied on syringes, which required cold chain discipline, skilled staff, and waste control procedures. Patch systems shift part of that burden into the device design through preloaded dose control and sealed application steps. Developers specify these products by insertion reliability, coating uniformity, dose recovery, and skin response across age groups. Use centers on vaccination programs, hormonal therapies, and selected biologics where adherence and access matter. Manufacturing focuses on microfabrication yield, coating repeatability, and packaging that protects sterility and mechanical integrity during transport and storage under varied conditions.
Commercial adoption depends on alignment between product sponsors, device makers, and regulators because the patch becomes part of the finished dosage form. Early programs concentrated on pilots and clinical supply, which limited scale and kept unit cost high. Current plans target automated molding, coating, and inspection to support larger campaigns. Suppliers compete on process control, lot traceability, and evidence that insertion and delivery remain consistent across batches. Buyers assess total cost through training needs, waste handling, and administration time, not device price alone. Cold chain requirements and shelf life claims influence deployment models. Over time, demand follows expansion of mass immunization and self-administration therapies, where simplified logistics and predictable dosing can change access economics while keeping safety and documentation within established regulatory expectations.
What Factors Are Shaping the Demand for Microarray Patch Market in 2026?
Adoption in Microarray Patch Market is determined by delivery route validation, manufacturing scale readiness, and regulatory classification rather than by formulation novelty alone. Once a product is approved around a specific patch design, the device becomes part of the finished dosage form and is controlled through change management procedures. This ties demand to pipeline progression, clinical conversion into commercial programs, and fill finish capacity planning. Segment structure reflects differences in material dissolution behavior, mechanical strength, and dose transfer efficiency. Buyers and developers prioritize reproducible dose delivery, shelf stability, and integration with existing packaging and distribution systems because device performance is inseparable from product approval and post market obligations.
How Does Product Type Selection Influence Manufacturing Control and Clinical Use in the Microarray Patch Market?
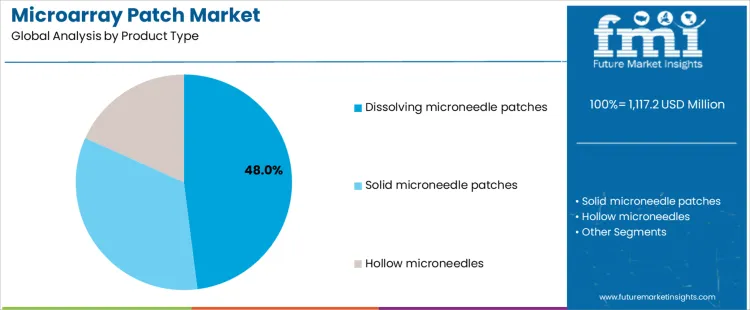
Dissolving microneedle patches represent about 48% of demand in the Microarray Patch Market because they eliminate sharps waste and simplify post use handling while providing reliable dose transfer. Their use aligns with programs that seek integrated drug and device formats. Solid microneedle patches account for about 34% of demand and are applied where skin pretreatment or surface delivery is required, which introduces additional handling steps and coordination with topical formulations. Hollow microneedles represent about 18% of demand and serve applications that require liquid delivery through the patch, increasing complexity in sealing, filling, and sterility assurance. Each type establishes different production controls, packaging requirements, and validation pathways.
From a manufacturing and regulatory perspective, product type choice fixes many downstream obligations. Dissolving systems require control of polymer composition, drying conditions, and mechanical integrity. Solid systems emphasize needle geometry consistency and coating uniformity. Hollow systems introduce requirements for lumen integrity, leak testing, and aseptic assembly. Once a clinical program is built around a specific design, changes are avoided because dose delivery, human factors, and stability data must be regenerated. Demand by product type therefore follows development pipeline decisions and scale up readiness rather than short term procurement considerations.
Why Does Application Mix Determine Volume Concentration and Evidence Threshold in the Microarray Patch Market?
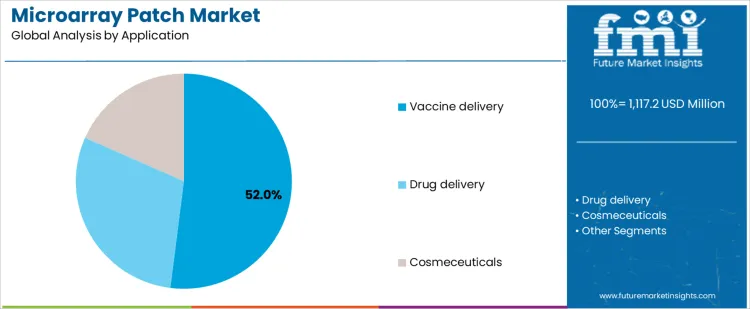
Vaccine delivery accounts for about 52% of demand in the Microarray Patch Market because mass immunization programs value simplified administration, reduced cold chain burden, and lower reliance on trained injectors. This creates a strong incentive to adopt patch based delivery once programs reach commercial scale. Drug delivery applications represent about 30% of demand and focus on chronic or specialty therapies, where dose precision and long term stability impose higher evidence requirements. Cosmeceutical applications account for about 18% of demand and operate under faster development cycles, though volumes are tied to consumer adoption and brand strategy.
Application mix also shapes development timelines and commercialization models. Vaccine programs proceed through large clinical and procurement frameworks, producing high volume but long approval cycles. Therapeutic drug programs move at smaller scale with more diverse indications and delivery needs. Cosmeceutical products enter markets faster but with narrower regulatory scope. The resulting demand structure concentrates future volume in vaccines while technical scrutiny and customization remain higher in therapeutic and cosmetic uses. Segment shares therefore move with clinical success rates, public health procurement programs, and regulatory acceptance of patch based delivery rather than with short term product launch activity.
How Is the Microarray Patch Market Being Defined by Delivery Reliability and Clinical Workflow Fit?
The Microarray Patch Market is shaped by how reliably patches can deliver a defined dose while fitting into vaccination and drug administration routines. Microarray patches must meet expectations for dose accuracy, skin penetration consistency, packaging integrity, and ease of use by clinical or non-clinical staff. Buyers focus on performance consistency, storage behavior, and integration with existing distribution and administration models. Selection decisions involve development teams, clinical operations, and supply chain planners. This positions microarray patches as part of the delivery system rather than as a simple device, making validation data, handling procedures, and operational compatibility central to adoption decisions.
How Is Program Standardization Changing Product Expectations in The Microarray Patch Market?
Many immunization and therapy programs aim to standardize delivery methods across regions and sites to simplify training and logistics. This is changing expectations in the Microarray Patch Market toward patches that can be used with the same procedures, packaging formats, and documentation everywhere. Program owners prefer devices with predictable application steps and clear confirmation of correct use. This reduces dependence on highly trained staff and lowers process variation. As a result, suppliers are evaluated on consistency of design, clarity of instructions, and long-term product stability rather than on narrowly optimized designs for single studies.
Where Is Long-Term Volume Being Built in The Microarray Patch Market?
Long-term volume is being built in vaccination programs and chronic therapy applications where repeated dosing or large population coverage is required. Once a microarray patch format is approved and integrated into a program, demand follows the scale of that program rather than individual procurement cycles. The Microarray Patch Market also benefits when organizations adopt the same delivery format across multiple products. There is opportunity for suppliers that can support scale manufacturing, packaging, and distribution planning. Being selected as a standard delivery platform creates recurring demand tied to program rollout schedules and patient population size.
Why Do Regulatory Pathways and Manufacturing Scale-Up Challenges Limit Faster Adoption in The Microarray Patch Market?
Microarray patches combine device and drug delivery functions, which places them under complex regulatory review. Any change in design, materials, or manufacturing process can trigger additional testing and approval work. The Microarray Patch Market also faces challenges in moving from pilot production to high-volume manufacturing while maintaining consistent quality. Buyers are cautious about relying on a format that is still building large-scale production track records. In addition, switching from established delivery methods requires changes in training and logistics. These factors slow adoption and keep deployment focused on programs with strong development and validation support.
What is the Demand for Microarray Patch by Country?
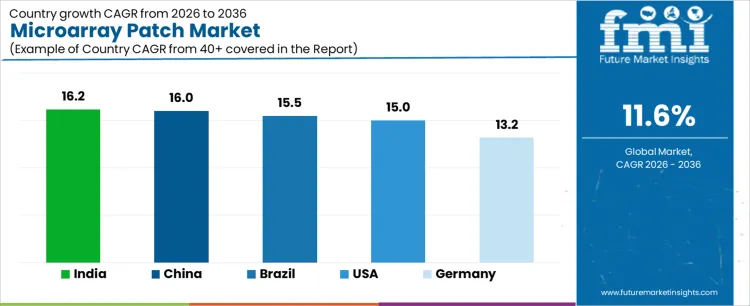
| Country | CAGR |
|---|---|
| USA | 15.0% |
| Brazil | 15.5% |
| China | 16.0% |
| India | 16.2% |
| Germany | 13.2% |
Demand for microarray patches is expanding as vaccine delivery, dermatology, and chronic therapy programs seek needle free, dose sparing administration methods with simpler logistics. India leads with a 16.2% CAGR, supported by public immunization programs, local manufacturing pilots, and interest in scalable delivery platforms. China follows at 16.0%, driven by large population health initiatives and investment in next generation drug delivery systems. Brazil records 15.5%, reflecting trials in vaccination and dermatology and growing public health adoption. The USA grows at 15.0%, shaped by clinical development pipelines and funding for alternative delivery technologies. Germany, at 13.2%, reflects structured clinical adoption within a regulated healthcare environment.
How Are Public Health Delivery Programs Shaping Demand in the Microarray Patch Market in the United States?
Public health delivery modernization keeps Microarray Patch Market demand in United States on a 15% CAGR path. Adoption centers on vaccination programs, chronic therapy pilots, and decentralized care models. Federal agencies, state health systems, and large provider networks guide procurement priorities. Device selection emphasizes dose accuracy, ease of use, cold chain relief, and waste reduction. Integration into existing immunization workflows shapes rollout speed. Domestic manufacturing capacity supports scale plans. Clinical validation and regulatory clearance affect deployment timing. Distribution partners handle training and field logistics. Hospital systems use pilot programs before wider adoption. Spending follows multi year public health funding cycles. Commercial success depends on inclusion in national rollout programs and large provider frameworks rather than individual clinic purchases today.
How Are Immunization Access Programs Influencing Demand in the Microarray Patch Market in Brazil?
Expansion of vaccination coverage and primary care access keeps Microarray Patch Market demand in Brazil on a 15.5% CAGR path. Public health authorities and large hospital groups lead adoption initiatives. Focus areas include rural delivery, school programs, and campaign based immunization drives. Device choice emphasizes ease of administration, reduced training burden, and logistics simplification. Import systems dominate early deployments. Local distribution partners support training and field execution. Procurement timing aligns with national immunization calendar planning. Pilot programs precede wider scale rollout. Budget allocation follows public health funding cycles. Private healthcare adoption remains limited. Commercial success depends on participation in government programs and reference use in national campaigns rather than fragmented sales to individual clinics or pharmacies.
What Institutional Scale Programs Are Driving Adoption in the Microarray Patch Market in China?
Large scale public health modernization and preventive care expansion keep Microarray Patch Market demand in China on a 16% growth track. Central health authorities and provincial systems guide technology selection. Deployment targets mass immunization, infectious disease control, and community health services. Domestic manufacturers compete in basic platforms while international suppliers retain roles in advanced designs. Procurement follows tender systems and multiyear rollout plans. Training simplicity and throughput capacity influence acceptance. Integration with national health programs shapes demand timing. Pilot zones precede national scale use. Data reporting requirements affect system design. Commercial success depends on inclusion in national health initiatives and provincial programs rather than pursuit of isolated hospital level projects or private clinic sales.
Why Is Primary Care Expansion Driving Growth in the Microarray Patch Market in India?
Expansion of preventive healthcare and vaccination coverage keeps Microarray Patch Market demand in India on a 16.2% CAGR path. Government health missions and large hospital groups guide early adoption. Use cases include mass immunization, maternal health programs, and chronic therapy pilots. Device choice focuses on ease of use, reduced dependence on trained staff, and logistics efficiency. Import systems serve initial deployments. Domestic manufacturing programs aim to localize supply. Procurement follows central and state health program schedules. Pilot projects validate workflows before broader rollout. Spending aligns with multiyear public health budgets. Commercial success depends on participation in national health initiatives and large scale campaigns rather than sales to individual clinics.
How Are Clinical Adoption and Quality Programs Shaping Demand in the Microarray Patch Market in Germany?
Preventive care modernization and structured clinical adoption programs keep Microarray Patch Market demand in Germany on a 13.2% CAGR path. University hospitals and public health agencies lead evaluation and deployment. Focus areas include vaccination clinics and outpatient therapy delivery. Device assessment emphasizes safety data, dosing consistency, and patient acceptance. Procurement follows health technology evaluation processes. Import suppliers dominate early adoption stages. Integration into regulated care pathways shapes rollout pace. Pilot programs operate before network wide deployment. Reimbursement alignment influences scale decisions. Spending follows public health and hospital investment plans. Commercial success depends on strong clinical evidence, inclusion in guideline based pathways, and access to national and regional health procurement frameworks.
How Do Developers Compete for Platform Acceptance in the Microarray Patch Market?
Microarray patch adoption depends on delivery route strategy, dose constraints, and manufacturing feasibility rather than device novelty. Vaccine and drug developers define target molecule stability, required skin depth, application time, and packaging format before selecting platforms. Vaxxas, 3M Drug Delivery, Zosano, Corium, Raphas compete inside those development programs with different technical routes. Once a patch format enters clinical protocols, tooling, stability studies, and regulatory files anchor that choice. Vaxxas builds around high density microprojection arrays for vaccine delivery. 3M Drug Delivery brings coating and manufacturing scale experience. Zosano focuses on coated titanium microarrays for biologics. Corium applies transdermal formulation and patch engineering knowledge. Raphas develops laser formed microneedle arrays aimed at precise geometry control. Platform decisions lock in long before commercial launch timelines.
Competition reflects manufacturability, regulatory risk, and partner confidence rather than theoretical delivery efficiency. Development teams review coating uniformity, dose repeatability, skin response, wear time, and pack stability during evaluation. Scale up planning favors platforms that integrate with high volume coating and packaging lines. Supply groups value predictable yields and controllable scrap rates. Vaxxas benefits from vaccine focused partnerships and clinical data depth. 3M Drug Delivery secures programs that prioritize industrialization and supply chain robustness. Zosano competes where high payload delivery drives design choices. Corium holds positions in reformulation driven programs. Raphas attracts partners seeking precise microstructure control. Market positions vary by therapy class, regulatory pathway, and sponsor risk tolerance during development.
Key Players in the Microarray Patch Market
- Vaxxas
- 3M Drug Delivery
- Zosano Pharma
- Corium
- Raphas
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Million |
| Product Type | Dissolving microneedle patches; Solid microneedle patches; Hollow microneedles |
| Application | Vaccine delivery; Drug delivery; Cosmeceuticals |
| End User | Pharma companies; CMOs/CDMOs; Research institutes |
| Region | Asia Pacific; Europe; North America; Latin America; Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, Rest of regions |
| Key Companies Profiled | Vaxxas; 3M Drug Delivery; Zosano Pharma; Corium; Raphas |
| Additional Attributes | Dollar by sales by product type and application, pipeline stage distribution, manufacturing scale-up capacity, regulatory pathway mix, vaccine versus therapeutic demand split, platform adoption by region, yield and scrap rate impact, packaging and cold chain requirements |
Microarray Patch Market Segmentation
Product Type:
- Dissolving microneedle patches
- Solid microneedle patches
- Hollow microneedles
Application:
- Vaccine delivery
- Drug delivery
- Cosmeceuticals
End User:
- Pharma companies
- CMOs/CDMOs
- Research institutes
Region
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- World Health Organization. (2024). Measles–Rubella Microarray Patch Phase III clinical trial framework. World Health Organization / PubMed Central
- National Institutes of Health. (2023). Microneedle‑mediated transdermal delivery of biopharmaceuticals. Pharmaceutics, 15(1), 277.
- Centers for Disease Control and Prevention. (2023). Advances in vaccine delivery technologies: Microneedles and skin‑based administration. CDC.
Frequently Asked Questions
How big is the microarray patch market in 2026?
The global microarray patch market is estimated to be valued at USD 1,117.2 million in 2026.
What will be the size of microarray patch market in 2036?
The market size for the microarray patch market is projected to reach USD 3,347.9 million by 2036.
How much will be the microarray patch market growth between 2026 and 2036?
The microarray patch market is expected to grow at a 11.6% CAGR between 2026 and 2036.
What are the key product types in the microarray patch market?
The key product types in microarray patch market are dissolving microneedle patches, solid microneedle patches and hollow microneedles.
Which application segment to contribute significant share in the microarray patch market in 2026?
In terms of application, vaccine delivery segment to command 52.0% share in the microarray patch market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Dissolving microneedle patches
- Solid microneedle patches
- Hollow microneedles
- Dissolving microneedle patches
- Y-o-Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Vaccine delivery
- Drug delivery
- Cosmeceuticals
- Vaccine delivery
- Y-o-Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Pharma companies
- CMOs/CDMOs
- Research institutes
- Pharma companies
- Y-o-Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- 3M Drug Delivery
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Zosano Pharma
- Corium
- Raphas
- Others
- 3M Drug Delivery
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

