Mosquito-borne infections Testing Market
The Mosquito-borne Infections Testing Market is segmented by Test Type (Molecular Diagnostics, Immunoassay Systems, ELISA, Rapid Tests, Blot Systems and Evaluation Software, and Microspot Array Diagnostics), Indication (Malaria, Zika Virus, Chikungunya, Dengue, and Others), End User (Hospitals and Clinics, Private Commercial Laboratories, Academic and Research Centers, and Others), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Mosquito-borne Infections Testing Market Forecast and Outlook By FMI
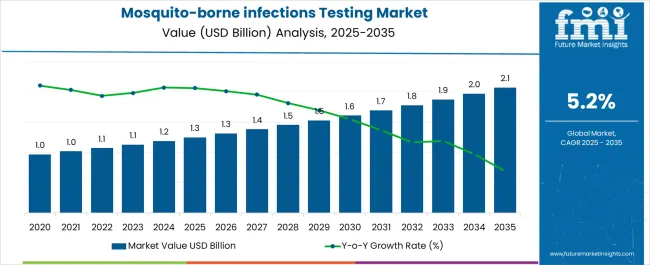
In 2025, the mosquito-borne infections testing market was valued at USD 1.3 billion. Based on Future Market Insights' analysis, demand for mosquito-borne infections testing is estimated to grow to USD 1.4 billion in 2026 and USD 2.1 billion by 2036. FMI projects a CAGR of 4.7% during the forecast period.
Absolute dollar growth of USD 0.8 billion over the decade signals steady incremental expansion driven by endemic region population growth and outbreak surveillance investments rather than transformational technology shifts. Despite public health funding volatility and pricing pressure in low-resource settings, major diagnostic manufacturers sustain profitability through high-volume rapid test sales and premium molecular diagnostic placements in reference laboratories.
Summary of Mosquito-borne Infections Testing Market
- Mosquito-borne Infections Testing Market Definition:
- The industry covers diagnostic assays and platforms for detecting mosquito-transmitted pathogens including malaria parasites and arboviruses (dengue, Zika, chikungunya).
- Demand Drivers in the Market:
- Climate change expands vector habitats into new geographic regions.
- Global travel trade increases imported case testing requirements.
- Elimination programs require widespread surveillance testing.
- Key Segments Analyzed in the FMI Report:
- Test Type: Molecular diagnostics lead with 28.0% share.
- Indication: Malaria dominates at 35.0% share.
- End User: Hospitals and clinics lead with 40.0% share.
- Analyst Opinion at FMI:
- Sabyasachi Ghosh, Principal Consultant for Healthcare at Future Market Insights, opines, "In the updated version of the Mosquito-borne Infections Testing Market Report for 2026 to 2036, public health officials and diagnostic manufacturers will find insights into how multiplex platform adoption is reshaping laboratory workflows and how climate change modeling is informing strategic capacity planning. Besides this, my findings point at pricing pressure in malaria-endemic markets directly impacting rapid test margins and the shift toward integrated surveillance platforms combining diagnostic data with epidemiological tracking."
- Strategic Implications/Executive Takeaways:
- Shift focus from single-pathogen tests to multiplex platforms.
- Treat pricing sustainability as market access requirement for endemic regions.
- Prioritize connectivity-enabled tests for real-time surveillance integration.
- Methodology:
- Validated through WHO procurement data and manufacturer shipment records.
- Zero reliance on speculative third-party market research reports.
- Based on verifiable disease burden statistics and national testing volume data.
As Peter Schultz, PhD, Chief Executive Officer of Calibr and Scripps Research, noted regarding the urgency of outbreak prevention, “Insect-borne infectious diseases remain primary causes of severe illnesses and fatalities worldwide, and new approaches to preventing outbreaks of these diseases are critically needed.” [1]
China (6.5% CAGR) and India (6.0% CAGR) drive new opportunities through expanding healthcare infrastructure and government surveillance programs. Brazil (5.0% CAGR) contributes via endemic disease burden and public health system investments. Mature testing markets such as Germany (5.5%), the United States (4.4%), the United Kingdom (3.9%), and Japan (3.4%) generate replacement demand through technology upgrades, with volume expansion constrained by low endemicity and reliance on travel-related testing.
Mosquito-borne Infections Testing Market Definition
Mosquito-borne infections testing comprises diagnostic assays and platforms designed to detect pathogens transmitted by mosquito vectors, including parasites (Plasmodium species causing malaria), viruses (dengue, Zika, chikungunya, West Nile, yellow fever), and associated immunological responses. These tests utilize molecular, serological, and rapid diagnostic technologies to identify active infections, support clinical management, enable outbreak surveillance, and guide public health interventions across endemic and non-endemic regions.
Mosquito-borne Infections Testing Market Inclusions
The report includes a comprehensive analysis of market dynamics, featuring Global and Regional Market Sizes (Volume and Value) and a 10-year Forecast (2026-2036). It covers segmental breakdowns by test type (Molecular Diagnostics, Immunoassay Systems, ELISA, Rapid Tests, Blot Systems, Microspot Array Diagnostics), indication (Malaria, Zika, Chikungunya, Dengue, Others), end user (Hospitals and Clinics, Private Commercial Laboratories, Academic and Research Centers), and geographic regions.
Mosquito-borne Infections Testing Market Exclusions
The scope excludes vector surveillance products not used for human diagnostics, insecticides and vector control products, and therapeutic pharmaceuticals for mosquito-borne infections. It also omits veterinary testing for animal arboviruses, focusing strictly on human diagnostic applications for mosquito-transmitted pathogens.
Mosquito-borne Infections Testing Market Research Methodology
- Primary Research: Interviews were conducted with diagnostic manufacturers, reference laboratory directors, hospital infectious disease specialists, and public health officials across major regions. Epidemiologists clarified disease surveillance requirements.
- Desk Research: WHO disease burden data, national surveillance statistics, and corporate financial filings supported volume benchmarking.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was developed. Demand was reconstructed from global malaria testing volumes, dengue incidence data, and outbreak surveillance testing requirements, then validated against manufacturer shipment data and procurement records.
- Data Validation and Update Cycle: Outputs undergo anomaly screening, variance checks across production and trade datasets, and structured peer review prior to release.
Segmental Analysis
Mosquito-borne Infections Testing Market Analysis by Test Type
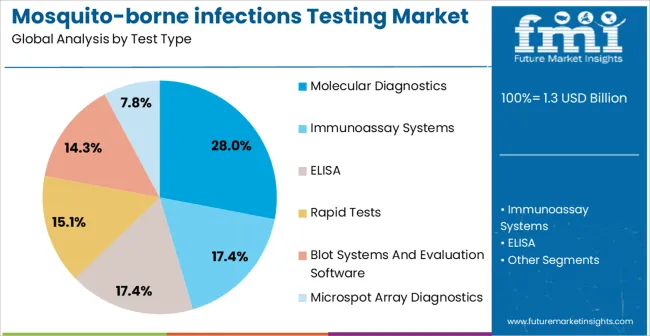
Based on FMI’s mosquito-borne infections testing market report, molecular diagnostics is estimated to hold a 28.0% share in 2026. The shift toward molecular methods is driven by the need for early detection during the viremic phase, where antibody tests often fail, pushing labs and frontline settings toward automated, high-throughput workflows and targeted multiplex capability.
- Point-of-Care Molecular Expansion: "In January 2026, Cepheid announced that it had received FDA clearance for its Xpert GI Panel, continuing its expansion of the GeneXpert system which supports broad detection of pathogens including high-priority infectious diseases." [2]
- Malaria Molecular Surveillance: "Abbott and its partner organizations are joining forces with local government to end malaria for good, with Abbott contributing to the partnership by helping to design community health worker training on the use of rapid diagnostic tests and the data collection from those tests." [3]
- Early-Window Advantage: Molecular assays are increasingly adopted where programs prioritize early rule-in confidence and faster escalation decisions, especially in outbreak-prone geographies with repeated seasonal exposure.
Mosquito-borne Infections Testing Market Analysis by Indication
Based on FMI’s mosquito-borne infections testing market report, malaria testing is estimated to hold a 35.0% share in 2026. While rapid tests provide the volume, value capture is shifting toward higher-information workflows and multiplexed kits that help differentiate co-endemic infections in clinically similar febrile presentations.
- Rapid Test Volume Leadership: "Investments in laboratories, oxygen provision and supply chains save lives and reinforce health security. The Results achieved demonstrate the strength of the Global Fund model, and underscore why investing in this unique partnership is one of the most effective ways to save lives." [4]
- Multiplex Innovation: "In April 2025, QIAGEN announced the launch of the QIAprep Plasmodium Kit and two companion assays to support malaria research and surveillance efforts, combining sample preparation and quantitative PCR (qPCR) into a single workflow." [5]
- Differentiation Premium: Buyers increasingly prioritize tests that reduce misclassification risk across co-circulating mosquito-borne diseases, lifting demand for higher-performance assays even when RDTs remain the throughput backbone.
Mosquito-borne Infections Testing Market Drivers, Restraints, and Opportunities
Future Market Insights analysis suggests that historical patterns point to a mature, public-health anchored diagnostics market where testing volume is structurally tied to seasonality, outbreak cycles, and government procurement rather than discretionary consumer demand. Estimated valuation in 2026 reflects an immediate "preparedness push" as health systems expand surveillance coverage and laboratory capacity to reduce time-to-detection and prevent downstream hospitalization costs during dengue, malaria, and other mosquito-borne outbreaks.
While rapid diagnostic tests continue to drive throughput, value growth increasingly comes from molecular and multiplex workflows that improve early-window detection and help clinicians differentiate co-endemic febrile illnesses. The forecast assumes a "new normal" where buyers prioritize total system reliability, data reporting, and supply continuity, with growth aligned to sustained vector control funding and routine screening programs.
- Automation and Multiplex Upgrading: Demand shifts toward high-throughput molecular platforms and multiplex panels that compress turnaround time and improve clinical confidence in the early phase of infection.
- Access and Compliance Constraints: Restraints come from limited lab infrastructure in high-burden regions, reimbursement variability, and regulatory requirements that slow adoption of new assays.
- Surveillance-Linked Expansion: Opportunity rises where testing integrates with digital reporting and community deployment models, enabling faster outbreak signals, targeted interventions, and repeat procurement tied to measurable public-health outcomes.
Regional Analysis
Based on the regional analysis, mosquito-borne infections testing market is segmented into Africa, South Asia, Southeast Asia, Latin America, Europe, and North America across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.

| Country | CAGR (2026 to 2036) |
|---|---|
| China | 6.5% |
| India | 6.0% |
| Germany | 5.5% |
| Brazil | 5.0% |
| United States | 4.4% |
| United Kingdom | 3.9% |
| Japan | 3.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Mosquito-borne Infections Testing Market Analysis
North America is characterized by travel medicine demand, imported case management, and rising alertness to localized transmission, which increases the need for rapid confirmation testing and public health surveillance support.
- United States: Demand for mosquito-borne infections testing in the United States is projected to rise at a 4.4% CAGR through 2036. Growth is increasingly tied to locally acquired cases, where local dengue transmission was reported in 2024 in California, Florida, and Texas, and Florida also reported local dengue transmission in 2025 [6]
FMI’s report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the USA and Canada. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
South Asia Mosquito-borne Infections Testing Market Analysis
South Asia, led by India, is characterized by a dual-burden dynamic where endemic malaria coincides with increasingly severe seasonal dengue outbreaks, raising the value of differentiated kits that help clinicians and surveillance teams separate co-circulating febrile illnesses.
- India: Domestic demand is accelerating at a 6.0% CAGR through 2036, supported by expanding surveillance and the rising threat of multiple arboviruses, where dengue and chikungunya incidence has been rising and dengue, chikungunya, and Japanese encephalitis are cited as emerging threats alongside malaria [7]
The full report analyze the mosquito-borne infections testing market across South Asia from 2021-2036, covering disease surveillance trends, testing adoption, and growth drivers in India and other South Asian countries. The assessment highlights trends that dictate test mix, procurement, and reporting practices.
Latin America Mosquito-borne Infections Testing Market Analysis
Latin America is a critical market for arbovirus diagnostics, where dengue volume and re-emerging outbreaks increase decentralized testing needs across public health networks and local laboratories.
- Brazil: A projected 5.0% CAGR through 2036 is supported by sustained dengue burden, where Brazil reported 1,660,190 probable dengue cases as of 26 Dec 2025 and accounted for approximately 87% of all reported cases in the Americas [8]
The report consists of a detailed analysis for the market in Brazil, Mexico, Argentina, Colombia, Peru and Rest of Latin America. Readers can find detailed information about several factors, such as surveillance intensity, testing access, and public health procurement trends impacting growth in Latin America.
Europe Mosquito-borne Infections Testing Market Analysis
Europe is driven by travel-linked case management and high-tech surveillance, where testing demand concentrates around returning travelers, imported case monitoring, and public health lab confirmation workflows.
- Germany: Demand is projected to rise at a 5.5% CAGR through 2036, where returning-traveler surveillance remains central and 2,079 German travelers with dengue infection returning from abroad were registered between September 2023 and December 2024 [9]
FMI’s analysis of the mosquito-borne infections testing market in Europe consists of country-wise assessment that includes Germany, the UK, France, Italy, Spain, Nordics, Benelux and Rest of Europe. Readers can know various surveillance practices, reporting structures, and evolving regional testing trends.
Competitive Aligners for Market Players
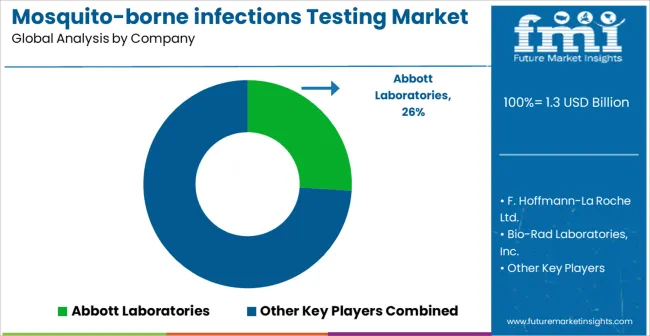
Market structure remains fragmented overall, yet practical competition is concentrated among a limited set of suppliers capable of meeting volume, regulatory, and operational expectations of national malaria programs, large hospital networks, and reference laboratories. A meaningful share of market value sits with manufacturers that can supply both rapid diagnostic tests and higher-complexity molecular workflows, while smaller players compete on single-disease kits with limited pricing leverage and higher demand volatility during non-outbreak periods. The primary competitive variable is not peak margin capture, but tender win-rate driven by proof of performance, quality consistency, and reliable delivery under surge conditions.
Producers with integrated manufacturing, validated quality systems, and multi-region regulatory clearances absorb raw material and logistics shocks more effectively and maintain continuity during outbreak spikes. Suppliers without scale rely on third-party manufacturing or constrained distribution, increasing exposure to stock-outs, batch variability, and requalification risk with public health buyers. Compliance capability further narrows the field. Ability to meet WHO-related procurement expectations, national regulatory requirements, and post-market surveillance obligations demands strong clinical evidence packages and traceable lot control, creating attrition among underinvested vendors.
Customer concentration reinforces buyer leverage. Large donors and ministries dual-source to avoid dependency, compressing pricing power and elevating service, training, connectivity, and reporting support as differentiators.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In October 2025, Roche received CE mark approval for its automated Elecsys Dengue Ag test, delivering results in 18 minutes and enabling faster, reliable dengue diagnosis during outbreaks worldwide [10].
- In June 2025, bioMérieux agreed to acquire Day Zero Diagnostics’ sequencing assets for under USD 25 million, strengthening its rapid infectious disease testing pipeline and advancing next-generation diagnostics for antibiotic-resistant infections [11].
Key Players in Mosquito-borne Infections Testing Market
- Abbott Laboratories
- F. Hoffmann-La Roche Ltd.
- Bio-Rad Laboratories, Inc.
- Siemens Healthineers AG
- Qiagen N.V.
- Thermo Fisher Scientific Inc.
- Cepheid (Danaher Corporation)
- DiaSorin S.p.A.
- Hologic, Inc.
- Meridian Bioscience, Inc.
- bioMérieux SA
- QuidelOrtho Corporation
- Beckman Coulter, Inc. (Danaher Corporation)
- BD (Becton, Dickinson and Company)
- PerkinElmer, Inc.
- Grifols S.A.
- Oxford Immunotec International
- Trinity Biotech plc
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 1.4 billion (2026) to USD 2.1 billion (2036), at a CAGR of 4.7% |
| Market Definition | The mosquito-borne infections testing market comprises diagnostic assays and platforms designed to detect mosquito-transmitted pathogens in human samples, including malaria parasites (Plasmodium species) and arboviruses such as dengue, Zika, chikungunya, West Nile, yellow fever, and Japanese encephalitis. Tests include molecular, serological, and rapid diagnostic technologies used for clinical diagnosis, treatment decisions, outbreak surveillance, and public health response across endemic and non-endemic regions. |
| Test Type Segmentation | Molecular Diagnostics, Immunoassay Systems, ELISA, Rapid Tests, Blot Systems and Evaluation Software, Microspot Array Diagnostics |
| Indication Segmentation | Malaria, Zika Virus, Chikungunya, Dengue, Others |
| End User Segmentation | Hospitals and Clinics, Private Commercial Laboratories, Academic and Research Centers, Others |
| Regions Covered | Africa, South Asia, Southeast Asia, Latin America, Europe, North America |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Vietnam, Thailand, Indonesia, Nigeria, Kenya and 40 plus countries |
| Key Companies Profiled | Abbott Laboratories, F. Hoffmann-La Roche Ltd., QIAGEN N.V., Thermo Fisher Scientific Inc., Cepheid (Danaher Corporation), bioMérieux SA, Siemens Healthineers AG, BD (Becton, Dickinson and Company), Bio-Rad Laboratories, Inc., DiaSorin S.p.A. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with diagnostic manufacturers, reference laboratory directors, hospital infectious disease specialists, and public health officials, supported by WHO disease burden statistics, national surveillance reporting, and procurement and shipment benchmarking. |
Mosquito-borne Infections Testing Market Analysis by Segments
Test Type:
- Molecular Diagnostics
- PCR (Polymerase Chain Reaction)
- RT-PCR (Real-Time PCR)
- Isothermal Amplification
- Next-Generation Sequencing
- Immunoassay Systems
- Chemiluminescence Immunoassay
- Fluorescence Immunoassay
- ELISA (Enzyme-Linked Immunosorbent Assay)
- Rapid Tests
- Rapid Diagnostic Tests (RDTs)
- Lateral Flow Assays
- Blot Systems and Evaluation Software
- Microspot Array Diagnostics
Indication:
- Malaria
- Plasmodium falciparum
- Plasmodium vivax
- Mixed/Other Species
- Zika Virus
- Chikungunya
- Dengue
- Dengue NS1 Antigen
- Dengue IgM/IgG Antibodies
- Others (West Nile, Yellow Fever, Japanese Encephalitis)
End User:
- Hospitals and Clinics
- Infectious Disease Departments
- Emergency Departments
- Outpatient Clinics
- Private Commercial Laboratories
- Reference Laboratories
- Regional Diagnostic Centers
- Academic and Research Centers
- Universities
- Research Institutes
- Others
- Blood Banks
- Public Health Laboratories
- Travel Medicine Centers
Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Colombia
- Peru
- Rest of Latin America
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- Bangladesh
- Sri Lanka
- Pakistan
- Nepal
- Rest of South Asia
- Southeast Asia
- Indonesia
- Thailand
- Vietnam
- Malaysia
- Philippines
- Rest of Southeast Asia
- Oceania
- Australia
- New Zealand
- Pacific Islands
- Middle East & Africa
- Nigeria
- Kenya
- Ghana
- Tanzania
- Uganda
- South Africa
- Saudi Arabia
- UAE
- Rest of Middle East & Africa
Bibliography
- [1] Lab Manager. (2025). Well-known animal health drug could stop outbreaks of malaria and Zika virus. Lab Manager.
- [2] Cepheid. (2026, January 20). Cepheid receives FDA clearance for Xpert® GI Panel to support broad detection of gastrointestinal pathogens. Cepheid Press Release.
- [3] Abbott Laboratories. (2025). Joining together to end malaria. Abbott USA
- [4] [Corporate Author Unknown]. (2025). Core 2025 results report (PDF).
- [5] QIAGEN N.V. (2025). QIAGEN launches QIAprep® Plasmodium Kit to strengthen malaria research and surveillance efforts. QIAGEN Press Release.
- [6] Centers for Disease Control and Prevention (CDC). (2025). Current dengue outbreak. CDC.
- [7] National Health Mission (NHM) Mizoram. (2025). Official website. Government of Mizoram.
- [8] BEACON Consortium. (2025). BEACON initiative overview.
- [9] PubMed. (2024). Dengue virus infection in travellers after dengue vaccination, Germany 2023-24. PubMed.
- [10] Roche Diagnostics. (2025). Roche receives CE mark for novel automated high-throughput Elecsys Dengue Ag test to diagnose dengue. Roche Press Release.
- [11] bioMérieux. (2025). bioMérieux strengthens its next-generation sequencing capabilities with the acquisition of Day Zero Diagnostics solutions and technologies. bioMérieux Press Release.
This Report Addresses
- Market intelligence to enable structured strategic decision-making across endemic and non-endemic testing markets
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by validated disease burden data and testing volume statistics
- Growth opportunity mapping across test types, indications, and end-user segments with emphasis on molecular diagnostics and multiplex platforms
- Segment and regional revenue forecasts covering malaria, dengue, Zika, chikungunya, and other mosquito-borne infections
- Competition strategy assessment including product portfolio breadth, regulatory qualification status, and procurement channel access benchmarking
- Technology roadmap tracking including point-of-care molecular platforms, multiplex assay development, and connectivity-enabled diagnostics
- Regulatory impact analysis covering WHO prequalification, CE marking, FDA clearance, and country-level registration requirements
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for mosquito-borne infections testing in the global market in 2026?
Demand for mosquito-borne infections testing in the global market is estimated to be valued at USD 1.4 billion in 2026.
What will be the market size of mosquito-borne infections testing in the global market by 2036?
Market size for mosquito-borne infections testing is projected to reach USD 2.1 billion by 2036.
What is the expected demand growth for mosquito-borne infections testing in the global market between 2026 and 2036?
Demand for mosquito-borne infections testing in the global market is expected to grow at a CAGR of 4.7% between 2026 and 2036.
Which test type segment is poised to lead global sales by 2026?
Molecular diagnostics are expected to be the dominant test type, capturing approximately 28.0% of global market share in 2026 due to superior sensitivity and specificity enabling early detection and pathogen differentiation.
Which indication segment is projected to contribute significant share in the mosquito-borne infections testing market in 2026?
In terms of indication, the malaria segment is projected to command 35.0% share in the mosquito-borne infections testing market in 2026, driven by sustained high disease burden with 247 million annual cases and elimination program testing requirements.
What is driving mosquito-borne infections testing demand in China?
China's 6.5% CAGR is driven by expanding healthcare infrastructure, government surveillance programs for imported cases, and emerging autochthonous dengue transmission in southern provinces requiring enhanced diagnostic capacity.
What compliance standards are referenced for China?
China's NMPA medical device registration requirements and national surveillance reporting standards are referenced as key compliance benchmarks.
What is the United States growth outlook in this report?
The United States is projected to grow at a CAGR of 4.4% during 2026 to 2036, supported by CDC ArboNET surveillance system requirements, travel medicine practice demand, and emerging local transmission monitoring.
Why is Germany's growth rate significant in this report?
Germany's 5.5% CAGR reflects increasing travel to endemic regions, emerging autochthonous transmission in Southern Europe requiring surveillance, and strong public health laboratory capacity.
What type of demand dominates in endemic regions?
Volume demand through public health procurement for malaria rapid diagnostic tests and outbreak response testing dominates endemic regions, with pricing pressure from donor-funded mechanisms.
What is Japan's growth outlook in this report?
Japan is projected to grow at a CAGR of 3.4% during 2026 to 2036, characterized by imported case testing for travelers and expatriates returning from endemic regions.
Does the report cover Australia in its regional analysis?
Australia is included within Oceania under the regional coverage framework, recognizing its imported case testing requirements and emerging local transmission risk in northern Queensland.
What are the sources referred to for analyzing the market in Africa?
WHO Global Malaria Programme reports, The Global Fund procurement data, and national malaria control program statistics are cited as primary reference sources.
What is the main demand theme linked to Southeast Asia coverage?
Southeast Asia demand centers on high dengue burden, seasonal outbreak response requirements, and tourism sector travel medicine testing.
Which end-user segment is strategically important for volume growth?
Hospitals and clinics remain the dominant end-user segment at approximately 40% market share, serving as first point of care for symptomatic patients and outbreak surveillance reporting hubs.
What are mosquito-borne infections tests and what are they mainly used for?
Mosquito-borne infections tests are diagnostic assays detecting pathogens transmitted by mosquitoes, primarily used for clinical diagnosis of malaria, dengue, Zika, and chikungunya, and for public health surveillance and outbreak monitoring.
What does the mosquito-borne infections testing market mean in this report?
The mosquito-borne infections testing market refers to global production and supply of diagnostic assays and platforms for detecting mosquito-transmitted pathogens in human samples for clinical and surveillance applications.
What is included in the scope of this mosquito-borne infections testing market report?
Scope covers mosquito-borne infections testing by test type (molecular diagnostics, rapid tests, immunoassays), indication (malaria, dengue, Zika, chikungunya), end user (hospitals, laboratories), and geographic regions.
What is excluded from the scope of this report?
Vector surveillance products, insecticides, therapeutic pharmaceuticals, and veterinary testing are excluded.
What does market forecast mean on this page?
Market forecast represents a model-based projection built on defined assumptions for strategic planning purposes.
How does FMI build and validate the mosquito-borne infections testing market forecast?
Forecast is developed using hybrid top-down and bottom-up modeling validated through WHO disease burden data, national testing volume statistics, and manufacturer shipment records.
What does zero reliance on speculative third-party market research mean here?
Primary interviews and verifiable public datasets including WHO procurement data, national surveillance reports, and corporate financial filings are used instead of unverified syndicated market estimates.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Type , 2026 to 2036
- Molecular Diagnostics
- Immunoassay Systems
- ELISA (Enzyme-Linked Immunosorbent Assay)
- Rapid Tests
- Molecular Diagnostics
- Y to o to Y Growth Trend Analysis By Test Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Test Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Test Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Test Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test Type
- Competition Analysis
- Competition Deep Dive
- Abbott Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- F. Hoffmann-La Roche Ltd.
- Bio-Rad Laboratories, Inc.
- Siemens Healthineers AG
- Qiagen N.V.
- Thermo Fisher Scientific Inc.
- Cepheid (Danaher Corporation)
- DiaSorin S.p.A.
- Abbott Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 3: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 5: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: Latin America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 7: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Western Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 9: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Eastern Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 11: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: East Asia Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 13: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: South Asia and Pacific Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 15: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Middle East & Africa Market Value (USD Million) Forecast by Test Type , 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Test Type
- Figure 6: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Region
- Figure 9: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 10: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 11: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 12: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 17: North America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 18: North America Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 19: North America Market Attractiveness Analysis by Test Type
- Figure 20: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 21: Latin America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 22: Latin America Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 23: Latin America Market Attractiveness Analysis by Test Type
- Figure 24: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 25: Western Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 26: Western Europe Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 27: Western Europe Market Attractiveness Analysis by Test Type
- Figure 28: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: Eastern Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 30: Eastern Europe Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 31: Eastern Europe Market Attractiveness Analysis by Test Type
- Figure 32: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: East Asia Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 34: East Asia Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 35: East Asia Market Attractiveness Analysis by Test Type
- Figure 36: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 37: South Asia and Pacific Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 38: South Asia and Pacific Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 39: South Asia and Pacific Market Attractiveness Analysis by Test Type
- Figure 40: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Middle East & Africa Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 42: Middle East & Africa Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 43: Middle East & Africa Market Attractiveness Analysis by Test Type
- Figure 44: Global Market - Tier Structure Analysis
- Figure 45: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

