Future Market Insights anticipates the global MRI-compatible guidewire market to grow from USD 190.0 million in 2026 to USD 480.0 million by 2036, reflecting a CAGR of 9.7% over the forecast period. Market expansion is driven by steady but continuous adoption of MRI-guided interventions and MRI-assisted interventions, mainly in vascular and neurovascular procedures where soft tissue visualization is valuable and there is a need to avoid ionizing radiation.
FMI identifies the structural features of this market as those involving material innovation and the compatibility of materials with MRI equipment field strengths. There is a rise in the adoption of guidewires that incorporate nitinol core materials, composites of polymers, and MRI-compatible wrought grades of stainless steels. The compatibility of guidewires with 1.5T and 3T MRI equipment field strengths continues to be a dominant attribute of decision-making during guidewire procurement. In this respect, hydrophilic and PTFE-based materials are also emerging as preferred materials for these guidewires.
Recent trends from leading companies demonstrate this shift from volume growth to focused capability development. Companies operating in the MRI-compatible guidewire market remain focused on material sciences, coating technologies, and MRI conditional labeling to ensure stringent standards for safe use within the market. Generally speaking, erstwhile leaders among interventional device companies always highlight the need to incorporate MRI-safe ecosystems to support the next generation of image-guided interventions, as hospitals consider alternatives to fluoroscopy-based procedures.
Rudolf Schulze Vohren, CEO of Nano4Imaging, stated, “This collaboration brings us closer to our USA customers at a critical time when there is a significant shortage of MRI-compatible medical devices. Partnering with Arrotek enables us to address this gap effectively, positioningus as a leader in the fast-growing interventional MRI market, which is expected to expand at a compound annual growth rate of 24%, reaching $2.3 billion in the next five years.”
The manufacturers are concerned with the long-term prospects of evolutionary changes in MRI-guided intervention platforms, rather than the short-term rate of growth in volume of procedures. To some extent, this emphasis of firms in the market on the importance of MR compatibility, coatings, and surgeon confidence does seem to have enabled them to carve out niches in the lucrative and differentiated space of healthcare. Guided by evolutions in technology, is likely to propel growth, albeit at a modest rate, up to 2036 in the domains of hospitals and specialty imaging facilities.
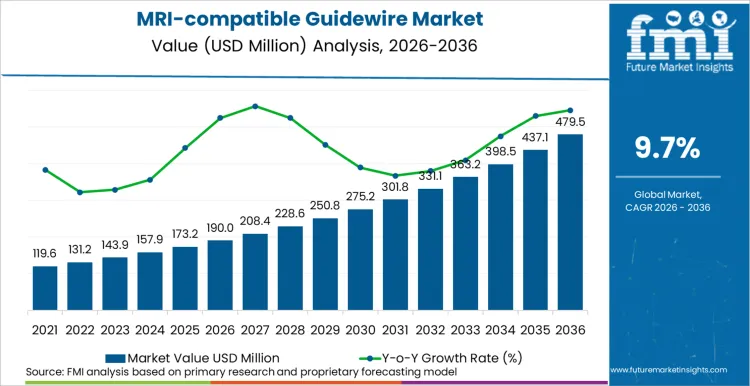
Future Market Insights projects the MRI-compatible guidewire market to expand at a CAGR of ~9.7% from 2026 to 2036, increasing from USD 190.0 Million in 2026 to USD 480.0 Million by 2036. Expansion is being supported by the growing clinical preference for MR-conditional tools in environments where image-guided interventions are being planned around MRI access, alongside rising demand for guidewires engineered to minimize heating risk and preserve handling performance under field constraints.
FMI Research Approach: Historical market size, MRI field-strength penetration, MR-conditional labeling patterns, guidewire material and coating adoption, and procedure-led utilization assumptions were used to model demand scenarios through 2036.
The demand is being shaped by material science choices and MR-environment labeling expectations, with stronger pull toward nitinol-based cores and hydrophilic coatings that preserve deliverability while meeting MR-conditional use conditions. Product design is being refined around torque response, visibility, and predictable tracking in peripheral and cardiac pathways, where MRI-enabled workflow expansion is being evaluated.
FMI Research Approach: Adoption trends were analyzed across MRI field strength bands (1.5T, 3T), core material selections, and coating preferences, aligned to application mix and end-user procurement behavior.
The United States holds the largest share in the global MRI-compatible guidewire market, supported by the depth of interventional infrastructure, high availability of advanced MRI systems, and stronger institutional emphasis on MR safety labeling and standardized device-use conditions. Demand is further reinforced by hospital-led procedure concentration and structured procurement pathways.
FMI Research Approach: Country-level inputs included MRI installed base indicators, procedure mix in vascular and cardiac interventions, MR safety labeling expectations, and hospital purchasing concentration.
The global MRI-compatible guidewire market is projected to reach USD 480.0 Million by 2036.
FMI Research Approach: Market size was calculated by modeling guidewire demand across the dominant field-strength band and leading material class, using weighted utilization factors by application and end user.
As per FMI, stricter expectations around MR safety testing and standardized MR labeling practices are accelerating demand for MR-conditional guidewires that can be used under defined field conditions. Adoption is being strengthened where hydrophilic-coated, low-friction designs reduce procedural variability and where hospitals are tightening compliance with device labeling, documentation, and operating-room safety protocols.
FMI Research Approach: Incorporated MR safety testing and labeling guidance, hospital compliance practices, and field-strength utilization trends into forecast modeling to refine demand estimates.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 190.0 Million |
| Industry Value (2036) | USD 480.0 Million |
| CAGR (2026-2036) | 9.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The demand for MRI-compatible guidewires growing as MR-conditional device use is being formalized through clearer testing and labeling expectations, and as MRI access is being evaluated for a wider set of image-guided interventional workflows. Utilization is being anchored in peripheral vascular applications and cardiology, where deliverability and tracking remain decisive while MR-environment constraints must be respected. Preference is being observed for nitinol-based cores and hydrophilic coatings, reflecting the need for consistent handling and low-friction navigation without compromising MR-conditional conditions of use. Demand is being led by hospitals, where MRI system availability, safety governance, and standardized documentation are more mature. As MR field use continues to broaden across 1.5T and 3T environments, adoption is being reinforced by procurement decisions favoring clearly labeled, protocol-ready guidewire platforms.
The MRI-compatible guidewire market is segmented by application, MRI field strength, material or core, coating, and end user. By application, the market includes peripheral vascular, cardiology, interventional radiology, neurovascular, urology, and structural heart procedures. Based on MRI field strength, guidewires are categorized into 1.5T, 3T, and ≤1.0T or low-field systems. By material or core, the market spans nitinol-based designs, polymer-jacketed composites, MRI-conditional stainless steel, and hybrid alloy constructions. By coating type, products include hydrophilic, PTFE or low-friction, hydrophobic, and uncoated variants. By end user, demand is segmented into hospitals, ambulatory surgical centers, and specialty imaging centers.
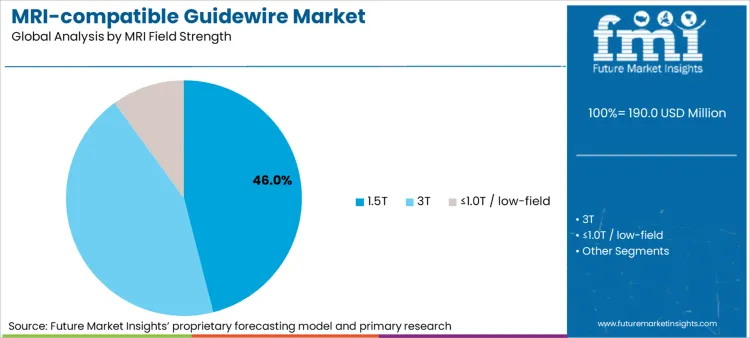
As per FMI, 1.5T MRI Guidewires are found to have the largest share in the MRI Guidewires market due to their compatibility with the largest number of MRI installations in interventional and hybrid imaging settings. The estimates and share of MRI Guidewires are found to be around 46.0% by market size for 1.5T MRI Guidewires.
From a clinical and operational viewpoint, the 1.5T devices strike an optimal chord between system safety, device usability, and image quality. The guidewires rated for 1.5T have a well-clarified testing regime, creating an overall landscape conducive to reducing onboarding challenges in hospitals. Furthermore, the vast majority of MRI-enabled interventional equipment uses a 1.5T system as a foundation, validating the basis on which 1.5T compatibility is considered a dominant driver in selecting MRI-Guided guidewires.
According to Future Market Insights, peripheral vascular is considered the leading clinical segment in the market, accounting for 30.0% of the overall usage of MRI-compatible guidewires. Peripheral interventions are considered the most common clinical indication of MRI-guided wires, mainly used in complex vascular anatomy where there is an advantage of the procedure over using fluoroscopy alone.
This demand is further fueled by the increased reliance on MRI guidance in cases where repeated procedures are needed, as well as in cases that require a greater sensitivity to ionizing radiation. The use cases in peripheral vascular procedures take advantage of the torque control and malleability of materials used in making the nitinol composite and other guidewires, which have been optimized for MRI compatibility. As such, peripheral vascular procedures remain the leading source of demand in the market.
The regulatory and safety aspects are key drivers of the market for MRI-compatible guidewires. The selection of the appropriate device is becoming heavily dependent on factors like magnetic susceptibility, RF induced heat, and the prevention of image artifact creation. Safety labeling of the MRI equipment is of prime importance for the usage of the product.
At the same time, healthcare providers are focusing on guidewires with well-established MRI conditional profiles, which reduce procedural risk in an appropriate fashion. In this regard, with increasing MRI-guided interventions, regulatory-related scrutiny on device-scanner interactions is fueling the market for well-validated guidewires.
The growing interest in MRI-guided and MRI-assisted intervention procedures is radically changing the market requirements for guidewires used in a magnetic environment. The increase in the use of MRI in cardiovascular procedures, by improving visualization and limiting cumulative radiation dose, is leading to an increasing requirement for guidewires that retain mechanical performance while being MRI compatible.
With rise in procedural confidence levels, MRI-compatible guidewires are moving from specialty products to core components in specialty interventional programs. This notion is especially true in tertiary facilities and specialty imaging clinics where advanced imaging capabilities exist for high case volumes.
The driving forces behind innovation in the MRI compatible guidewire market include developments in the field of materials engineering, which have helped the prevalent use of core materials consisting of Nitinol, owing to their flexibility and non-ferromagnetic properties.
However, at the same time, improvements are being made in the hydrophilic materials that help in trackability as well as in reducing trauma during vessel navigation. Such advancements in materials and coatings are contributing towards a safer, efficient, and effective use of MRI-guided devices.
Future Market Insights identifies the United States (CAGR ~8.6%), Germany (~7.1%), France (~6.8%), the United Kingdom (~6.4%), Japan (~5.7%), China (~9.3%), and South Korea (~7.8%) as the core demand drivers shaping MRI-compatible guidewire adoption globally. The USA leads through earlier procedural readiness for MRI-guided interventions, stronger penetration of advanced imaging infrastructure, and higher institutional capacity to operationalize radiation-free navigation workflows. Germany, France, and the UK are bolstering the European region with safe procurement disciplines and interventional radiology services where MRI-guided interventions are being investigated for high-performing use cases.
The Japanese market is being solidified through clinical practices, methodical workflow validation, and investment in high-quality imaging equipment being implemented in the nation. The Chinese market is being accelerated through rapid expansion of advanced imaging procedures and increasing procedural interest in radiation-free navigation procedures adopted in high-volume tertiary centers. South Korea has been deemed to receive support due to the continued modernization of imaging infrastructure, improved institutional readiness to incorporate new image-guided procedure pathways, and FMI estimates an underlying strength to the long-term demand curves projected to run through 2036.
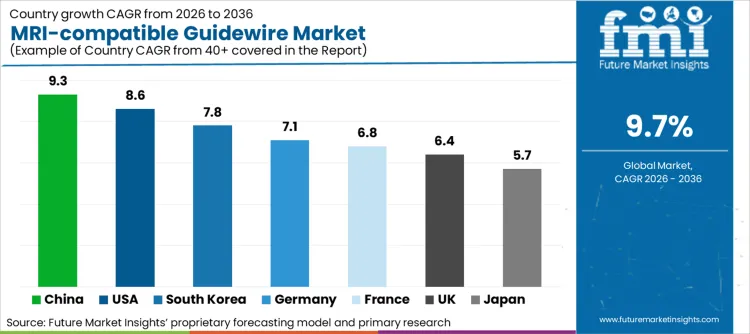
| Country | CAGR (2026-2036) |
|---|---|
| USA | 8.6% |
| Germany | 7.1% |
| France | 6.8% |
| UK | 6.4% |
| Japan | 5.7% |
| China | 9.3% |
| South Korea | 7.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
MRI equipment in the USA is expected to grow at an 8.6% CAGR, primarily fueled by a head start in terms of being ready for MRI-guided interventional procedures and more robust availability in terms of high-field MRI equipment, especially in tertiary centers and specialty imaging facilities. This trend is further fueled by an increase in procedure preference, particularly based on a radiation-free option, especially in more challenging peripheral vascular and interventional radiology procedures where soft tissue is prioritized. As stated by the analysts at Future Market Insights, this trend is further fueled as a growing number of facilities are investing in MRI-conditional accessories, ensuring that these procedures are integrated while keeping safety and precision at a high level.
According to Future Market Insights, Germany’s MRI-compatible guidewire market is projected to expand at a 7.1% CAGR, shaped by safety-led procurement norms and structured evaluation of advanced interventional technologies. Demand is being supported where MRI-conditional navigation is being assessed for selected procedures requiring improved visualization and minimized radiation exposure, particularly within rigorously controlled hospital environments. FMI analysts observe that adoption is being paced by performance assurance expectations, material reliability requirements, and workflow validation discipline. Growth is expected to remain steady as MRI suite integration improves and specialized interventional programs scale within high-capability centers.
MRI-compatible guidewire market, France is expected to experience a growth rate of 6.8% over the long term, the trend toward modernization of interventional imaging pathways and acceptance of radiation-avoidance techniques, which can reinforce demand, especially where MRI guidance is being assessed for its comparative advantage in tissue visualization and its ability to improve procedure confidence within certain clinical scenarios. According to FMI, growth in this industry will be steady and dependent on clinical validation.
The UK MRI-compatible guidewire market is expected to expand at a 6.4% CAGR, supported by controlled adoption within structured pathways where patient safety and radiation reduction objectives are being prioritized. Demand is being shaped by procurement discipline and operational feasibility assessments, resulting in stepwise integration rather than broad-based deployment. FMI analysts emphasize that uptake is being reinforced where MRI-assisted navigation is being positioned as a complementary capability for selected complex interventions. Growth is expected to remain stable as institutional familiarity improves and procedural protocols are refined around MRI-conditional device use.
As per FMI, Japan’s MRI-compatible guidewire market to grow at a 5.7% CAGR, shaped by conservative technology adoption patterns, high clinical rigor, and careful validation of procedural workflow changes. Demand is being supported where MRI-guided intervention capability is being integrated in a controlled manner, with strong emphasis being placed on reliability, repeatability, and compatibility assurance. FMI analysts note that adoption is being driven more by precision and safety goals than by aggressive expansion strategies. As interventional imaging continues to evolve, stable demand is expected to be sustained through disciplined implementation and long-cycle capital planning.
China’s MRI-compatible guidewire market is projected to expand at a 9.3% CAGR, underpinned by faster expansion of advanced imaging infrastructure and rising interest in MRI-guided interventional pathways within high-volume tertiary hospitals. Demand is being reinforced as radiation-free navigation advantages are being increasingly recognized in selected vascular and interventional radiology procedures, particularly where soft-tissue visualization requirements are high. The adoption is being supported by increasing clinician exposure to image-guided workflows and growing institutional investment in MRI suite capability. As procedural readiness improves, demand is expected to remain driven by performance assurance and workflow integration rather than by device volume alone.
MRI-compatible guidewire market in South Korea will see a 7.8% CAGR growth in the forecast period, underpinned by the rapid strengthening of advanced imaging capacity and stronger institutional willingness to operationalize next-generation minimally invasive workflows. The growth is further being complemented because hospitals aim for navigation capabilities free from radiation hazards and integrate image-guided procedure ecosystems requiring workable MRI-conditional accessories. Technology openness to uptake, training readiness, and quickening integration of specialized procedural programs have tended to assist the process. All structural factors point toward continued growth through 2036, as MRI-assisted interventional capability broadens further and procedural specialization deepens.
The competition for market players within the MRI-compatible guidewire domain has begun to shift away from more conventional measures of guidewire performance, particularly towards material science advancements, MRI safety data, and procedure versatility within various imaging disciplines, including multi-modality environments.
The second strategic move that can further define the market are the guidewires made as per the needs of various requirements of MRI field strengths, particularly the1.5T and 3T machines, which are dominant across the installed base of MRI equipment across the globe. Terumo Corporation and Boston Scientific are attempting to design these guidewires for application-specific requirements, which fit into the needs of peripheral vascular, cardiology, interventional radiology, and neurovascular procedures all under the auspices of MRI procedures.
FMI also highlights continued differentiation with respect to coating and engineering strategies such as hydrophilic, low friction PTFE, and hybrid coatings optimized to address trackability, visibility, and procedural control. Emphasis is also given to the adoption of guidewires in hospitals, followed by ASC and specialty imaging center market growth strategies. Overall, these strategies are changing the landscape of competition and favoring guidewire manufacturers with respect to regulatory and design expertise utilizing MRI technology through 2036.
Recent Developments:
The MRI compatible guidewire market comprises the global industry of designing, developing, and applying guidewires designed to be used in an MRI or magnetic resonance imaging related procedure in a safe manner. System design is carried out in such an efficient way that it does not show significant effects of the MRI forces of attraction, heating, and distortion, keeping this market in millions of USD and forecasting from 2026 to 2036.
The market also includes MRI-compatible guidewires used in various Applications such as Peripheral Vascular, Cardiology, Interventional Radiology, Neurovascular, Urology, and Structural Heart. The products included in the MRI guidewire market can be classified based on MRI Field Strength, Material/Core, and Coating and are used in Hospitals, ASCs, and Specialty Imaging Centers.
The market does not include conventional fluoroscopy-only guidewires, diagnostic wires not suited for intravascular guidance, non-guidewire catheters, or accessories not suited for use within an MRI setting. Excluded are guidewires employed solely in non-clinical research studies, veterinary applications, or those not performed under MRI guidance.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 190.0 Million |
| Application | Peripheral vascular, Cardiology, Interventional radiology, Neurovascular, Urology, Structural heart |
| MRI Field Strength | 1.5T, 3T, ≤1.0T / low-field |
| Material / Core | Nitinol-based, Polymer-jacketed composites, Stainless steel (MR-conditional), Hybrid / other alloys |
| Coating | Hydrophilic, PTFE low-friction, Hydrophobic, Uncoated |
| End User | Hospitals, ASCs, Specialty imaging centers |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | USA, Germany, France, UK, Japan, China, South Korea, and 40+ countries |
| Key Companies Profiled | Nano4Imaging, EPflex Feinwerktechnik GmbH and MaRVis Interventional GmbH |
| Additional Attributes | Revenue analysis by application and MRI field strength, assessment of material and coating innovation, evaluation of adoption trends in MRI-guided interventions, competitive positioning based on safety compliance and procedural performance |
What is the current global market size for MRI-compatible guidewires?
The global MRI-compatible guidewire market is valued at USD 190.0 million in 2026, reflecting early but increasing adoption of MR-guided interventional workflows across vascular and specialty procedures.
What is the projected CAGR for the market over the next 10 years?
The market is projected to grow at a 9.7% CAGR from 2026 to 2036, supported by expanding use of high-field MRI systems and growing clinical interest in radiation-free image-guided interventions.
Which regions are experiencing the fastest expansion?
The United States, South Korea, and Germany are experiencing comparatively faster expansion, driven by higher MRI penetration, advanced interventional radiology infrastructure, and earlier adoption of MR-conditional devices.
What are the primary market drivers?
Growth is being driven by demand for radiation-free guidance, increasing complexity of peripheral and cardiovascular interventions, advancements in nitinol-based and polymer-jacketed materials, and rising availability of MR-conditional procedural suites.
Who are the leading manufacturers in the market?
Nano4Imaging, EPflex Feinwerktechnik GmbH and MaRVis Interventional GmbH are among the leading manufacturers, differentiated by proprietary materials engineering, MR-conditional labeling breadth, and established interventional portfolios.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.